Back to Journals » Infection and Drug Resistance » Volume 13
COVID-19 (Coronavirus Disease 2019): A New Coronavirus Disease
Authors Sadeghi Dousari A , Taati Moghadam M, Satarzadeh N
Received 21 April 2020
Accepted for publication 28 July 2020
Published 12 August 2020 Volume 2020:13 Pages 2819—2828
DOI https://doi.org/10.2147/IDR.S259279
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Amin Sadeghi Dousari,1 Majid Taati Moghadam,2,3 Naghmeh Satarzadeh4,5
1Department of Microbiology, School of Medicine, Jiroft University of Medical Sciences, Jiroft, Iran; 2Department of Microbiology, School of Medicine, Iran University of Medical Sciences, Tehran, Iran; 3Student Research Committee, Iran University of Medical Sciences, Tehran, Iran; 4Student Research Committee, Faculty of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran; 5Department of Pharmaceutical Biotechnology, Faculty of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
Correspondence: Naghmeh Satarzadeh
Student Research Committee, Faculty of Pharmacy, Kerman University of Medical Sciences, Kerman, Iran
Tel +989363699045
Email [email protected]
Abstract: Coronavirus disease 2019 (COVID-19) is a type of viral pneumonia with an uncommon outbreak in Wuhan, China, in December 2019, which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV2). SARS-CoV-2 is extremely contagious and has resulted in a fast pandemic of COVID-19. Currently, COVID-19 is on the rise around the world, and it poses a severe threat to public health around the world. This review provides an overview about the COVID-19 virus to increase public awareness and understanding of the virus and its consequences in terms of history, epidemiology, structure, genome, clinical symptoms, diagnosis, prevention, and treatment.
Keywords: novel coronavirus, coronavirus disease 2019, COVID-19, SARS-CoV-2
History
The human coronavirus was first diagnosed in 1965 by Tyrrell and Bynoe from the respiratory tract sample of an adult with a common cold cultured on human embryonic trachea.1 Naming the virus is based on its crown-like appearance on its surface.2 Coronaviruses (CoVs) are a large family of viruses belonging to the Nidovirales order, which includes Coronaviridae, Arteriviridae, and Roniviridae families.3 Coronavirus contains an RNA genome and belongs to the Coronaviridae family.4 This virus is further subdivided into four groups, ie, the α, β, γ, and δ coronaviruses.5 α- and β-coronavirus can infect mammals, while γ- and δ- coronavirus tend to infect birds.6 Coronavirus in humans causes a range of disorders, from mild respiratory tract infections, such as the common cold to lethal infections, such as the severe acute respiratory syndrome (SARS), Middle East respiratory syndrome (MERS) and Coronavirus disease 2019 (COVID-19). The coronavirus first appeared in the form of severe acute respiratory syndrome coronavirus (SARS-CoV) in Guangdong province, China, in 20027 followed by Middle East respiratory syndrome coronavirus (MERS-CoV) isolated from the sputum of a 60-year-old man who presented symptoms of acute pneumonia and subsequent renal failure in Saudi Arabia in 2012.8 In December 2019, a β-coronavirus was discovered in Wuhan, China. The World Health Organization (WHO) has named the new disease as Coronavirus disease 2019 (COVID-19), and Coronavirus Study Group (CSG) of the International Committee has named it as SARS-CoV-2.9,10 Based on the results of sequencing and evolutionary analysis of the viral genome, bats appear to be responsible for transmitting the virus to humans11 (Figure 1).
 |
Figure 1 History of harmful human coronaviruses. |
Virion Structure and Structural Proteins
Coronavirus is a medium-sized spherical RNA virus, with a diameter of about 80–160 nm, which can be observed using an electron microscope.12 This virus has the largest viral RNA (30 kb in length), which is single-stranded, positive in sense, and polyadenylated.13 The coronavirus genome encodes five protein structures, including the spike (S), membrane (M), envelope (E) glycoproteins, hemagglutinin esterase (HE), and nucleocapsid (N) (Figure 2). N, M, S and E proteins are present in all virions, while, HE is only present in some β-coronaviruses.14 The S glycoproteins outside of the virions are homotrimers with crown-like structures, accounting for the name of the virus.15 These homotrimeric glycoproteins belong to class 1 fusion proteins16 that mediate binding host cell receptors.17 S protein of betacoronavirus (β-CoV) is critical for cross-species transmission as it mediates virus-receptor recognition and viral pathogenesis. The receptor-binding domain is located on the N-terminal of S protein and is necessary for the penetration of β-CoV into the host cells. Moreover, cross-species transmission and severity of the infection are affected via the mutations in CoV RBD.18 M glycoproteins have three transmembrane regions19 leading to the fusion of the virion into the cell and the emergence of antigenic protein that is involved in shaping the viral structure.20 The N protein is the only protein present in nucleocapsid,21 which contributes to the formation of a complex structure by binding genomic RNA.22 E glycoproteins are small proteins comprised of about 76 to 109 amino acids.23 These glycoproteins play an essential role in the assembly and morphogenesis of virions within the cell.23 E glycoproteins can act as ion channels which are considered necessary for pathogenesis.24 The HE acts as a hemagglutinin, binds sialic acids on surface glycoproteins, and has an acetyl-esterase activity.25 Apparently, these activities enhance S protein-mediated cell entry and virus extension through the mucosa.26
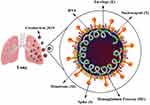 |
Figure 2 SARS-CoV-2 virus structure. |
Using genomic analysis of coronavirus 2019 isolates from patients COVID-19 in China and the USA, Malik et al27 reported that the length of the S protein sequence in this new virus was different from that of other β-coronaviruses. The major differences of S protein in novel Coronavirus (nCoV) compared with SARS-CoV include three short insertions at the N-terminal region, and four changes in the receptor-binding motif within the receptor-binding domain.28 Recent studies have reported that the binding capacity of n-CoV S protein with human angiotensin-converting enzyme 2 (ACE2) is as efficient as SARS-CoV, which further raises the possibility of human-to-human transmission.27,28
Epidemiology
Coronaviruses are disseminated widely among humans, other mammals, and birds, further causing acute respiratory diseases.29 The epidemics of unknown acute respiratory tract infection at the first time on December 29th 2019, in Wuhan, China, was very likely associated with a seafood market.30,31 Many studies have reported that bats may be the primary reservoirs of the disease.29,31-33 Currently, COVID-19 is a pandemic that according to the latest WHO report, until July 08, 2020, a total of 11,635,939 cases were infected in more than 216 countries around the world, and 539,026 cases died.34
Respiratory droplets are the major transmission route of this disease,31 and transmission among people occurs through direct (cough, sneeze, and droplet inhalation) and indirect (contact with oral, nasal, and eye mucous membranes) routes.35 Human-to-human transmission of this infection occurs mostly among family members, including relatives and friends who are in close contact with patients or carriers.36,37 Furthermore, occupational exposure to health-care workers is another concern.38
Older people with underlying diseases such as asthma, heart failure, diabetes, and Immunodeficiency diseases are at a higher risk of a more severe COVID-19 disease.39
New evidence demonstrated that the SARS-CoV-2 may also be an enteric virus that can spread through the fecal-oral route. According to initial papers from Wuhan, 2–10% of patients with COVID-19 showed gastrointestinal (GI) symptoms, such as nausea, abdominal pain, vomiting, and diarrhea. A current meta-analysis article on >4000 East Asian COVID-19 cases illustrated that up to 20% of cases showed GI signs, and viral RNA was detected in the stool of approximately 50% of cases.40
Dental patients and professionals are at a high risk of contracting COVID-19 infection due to exposure to blood and saliva as well as face-to-face interaction, and handling of sharp tools.41
Smoking is harmful to the immune system and causes smokers to be more vulnerable to infectious diseases.42 Verdavas et al43 reported that the smokers are 1.4 times more at risk of showing severe signs of COVID-19, and are almost 2.4 times more likely to be admitted to an ICU, require mechanical ventilation or die compared with non-smokers.
Diverse maternal physiological adaptations in the immune (immunosuppression) and cardiorespiratory (such as diaphragm elevation and physiologic anemia) systems are observed in pregnant women leading to poor outcomes of viral respiratory infections.44 It is unknown whether the COVID-19 can be transferred by breast milk, but it is clear that an infected mother can transfer the COVID-19 by respiratory droplets to her child.45,46 Wang et al47 reported a neonatal case with COVID-19 36 hours after birth whose pharyngeal swabs tested positive with real-time reverse-transcription polymerase chain reaction assay in China. However, whether this was a vertical transmission case from mother to neonate remains to be confirmed47 (Figure 3).
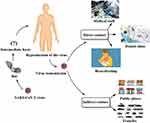 |
Figure 3 SARS-CoV-2 virus transmission. |
Clinical Symptoms and Diagnosis
Clinical signs of COVID-19 disease appear after an incubation period of about 5.2 days48 which takes about 6–41 days (average of 14 days) until death depending on the age of the patient or the patient’s immune status.49,50 Generally, the clinical symptoms of COVID-19 are nonspecific and common symptoms include fever, cough, and fatigue.51 Fever is considered as the dominant symptom, however, it is not always the initial symptom of infection.52 Other symptoms include headache, sputum production, diarrhoea, dyspnoea, and lymphopenia.53,54 At the beginning, infection rapidly advances to organs resulting in symptoms such as acute respiratory distress syndrome (ARDS), shock, acute kidney failure, acute cardiac failure, and even death in severe cases. Patients may demonstrate normal or lowered white blood cell counts, lymphopenia, thrombocytopenia with greater thromboplastin activation time, and raised C-reactive protein level55 (Table 1).
 |
Table 1 Difference Between Clinical Symptoms of the Common Cold and Flu Diseases with COVID-19 |
Fever, reduction in lymphocytes and white blood cells (WBC), new pulmonary infiltrates on chest radiography, and no improvement in symptoms after three days of antibiotics treatment are common ways of diagnosing patients with COVID-19 infection.48 For patients suspected with COVID-19, real-time PCR on nasopharyngeal and/or pharyngeal swabs, sputum, and secretions of the lower respiratory tract samples has been suggested for the detection of COVID-19 DNA.56,57
Testing for antibody response to infection is usually based on measuring immunoglobulin M (IgM) and immunoglobulin G (IgG). In serological studies immunoglobulin A (IgA) and total antibodies may also be measured. Most tests evaluate both IgM and IgG. IgM usually increases rapidly with infection and decreases soon after the clearance of infection. IgG is thought to increase more slowly and may even remain stable thereby representing longer-term immunity. Antibody assays are likely to be available in laboratories in the form of enzyme-linked immunosorbent assay (ELISA), or point-of-care tests by a disposable device using one or two blood drops from thumb prick upon a testing strip, which takes about 10 minutes.58
The sensitivity of chest radiography or chest X-ray (CXR) in the primary stages can be low as ground glass lesions are not observed, however, in severe cases, bilateral multifocal consolidation and even advancement to the white lung can be seen.59
Recent chest CT scan findings of COVID-19 pneumonia show as multifocal unilateral or bilateral ground glass opacity (GGO) or mixed GGO and consolidation mostly peripherally located. GGO is generally seen in the primary days and advance to the consolidation in the following days. Generally, lymphadenopathy is not seen, and pleural effusion is rare and mild. Generally, a normal chest CT scan may be useful in ruling out COVID-19.60,61
In the complete blood count-differential (CBC-Diff) test shows leukopenia or lymphocytopenia less than normal rates based on age (an absolute lymphocyte count <3000/microliter in infants (1 month to 12 months), <2000/microliters in Childs 1–5 years of age and <1100/microliters are older than 5 years).62,63
Among four structural proteins (S, M, E, and N) in the coronavirus family, S and M proteins seem to contain antigenic sites for the development of serological tests for COVID-19 detection. Serological tests have focused on detecting serum antibodies for S proteins of the coronavirus spike. S proteins are determined with the S gene and are functionally divided into S1 and S2 subunits. Another protein that seems to be a significant antigenic site for the development of serological tests for COVID-19 detection is the N protein, which is a structural constituent of the helical nucleocapsid.64
The commonly used nucleic acid diagnosis technique for this disease is real-time semi-quantitative polymerase chain reaction (RT-qPCR). The authoritative identification approach for SARS-CoV-2 is virus blood culture and high-throughput whole genome sequencing. Nonetheless, the use of high-throughput sequencing technology in clinical diagnosis is limited due to high price and equipment dependency.65 Thus, RT-qPCR is the most common and efficient method for detecting pathogenic viruses in respiratory secretions and blood.66,67
Treatment and Prevention
To date, there have been no specific antiviral drugs or vaccines to combat COVID-19, and treatment is merely provided as supportive and palliative approaches, as well as several antiviral and anti-inflammatory drugs care at the clinic. However, WHO has announced that the COVID-19 vaccine is expected to be launched in the next 18 months.68 Supportive therapy for patients with coronavirus is one of the most important principles along with other treatments for patient management, including oxygen therapy, fluid retention, and the use of antibiotics to avoid secondary bacterial infections.65 The use of antiviral drugs is another treatment for COVID-19. In one study, remdesivir has been used to treat COVID-19 patients and favorable results have been achieved in vitro.69 It is also possible that antiviral agents such as ribavirin, galidesivir, and favipiravir, which are nucleoside analogues, could be clinically used to treat coronaviruses.70,71 EIDD-2801 (β -D-N4-hydroxycytidine-5′-isopropyl ester) can be considered as a drug for the treatment of COVID-19 infection due to its high efficacy properties against respiratory infections such as seasonal influenza virus and pandemic influenza.72 Arbidol is another antiviral drug used against some viruses such as influenza A and B, hepatitis C and SARS-CoV viruses. For this reason, in a study, Arbidol and Kaletra (lopinavir/ritonavir) were used against COVID-19, and according to the results, Arbidol had a better therapeutic effect and was able to reduce the prevalence of severe cases. Oral administration of ritonavir, lopinavir, oseltamivir, and intravenous administration of ganciclovir for 3–14 days were also helpful in 75 patients.73 In addition, chloroquine has been shown to have an immune-suppressive activity and is effective in vitro and in the treatment of patients with COVID-19.74,75 In Chinese medicine, it has been suggested that the use of a decoction such as Shuanghuanglian oral liquid (Sol) can inhibit COVID-19 by lung detoxification and clearance.76 Sol contains active ingredients such as forsythin, baicalin, and chlorogenic acid with inhibitory properties against a variety of viruses and bacteria by reducing inflammatory responses.77,78 Natural herbal medicine Lianhuaqingwen capsule has also antiviral properties, regulates immune responses, and reduces inflammatory factors in the early stages of infection.79 Natural compounds such as flavonoids, Glycyrrhizin, diarylheptanoids, and cinanserin, which inhibit main coronavirus enzymes involved in replication, can be considered as attractive options for COVID-19 treatment.80,81 Immunoenhancement therapy is one of the treatment methods that employs recombinant interferon, thymosin alpha-1 (Ta1), Thymopentin, Levamisole, Cyclosporine A, and intravenous immunoglobulin against viral infections.82 Previous studies have shown that recombinant interferon-alpha affects MERS, suggesting its possible effectiveness against COVID-19.83 Intravenous immunoglobulin is an immunomodulator used for all ages that enhances the production of anti-inflammatory mediators and inhibits the production of pro-inflammatory cytokines.84 Ta1 is also an immune system booster that has been effectively used in SARS outbreaks.85 Intravenous immunoglobulin and Ta1 may be considered as an effective treatment for COVID-19.82 Combining thalidomide (an immunomodulatory and anti-inflammatory agent) with a low-dose of glucocorticoids in the treatment of COVID-19 pneumonia showed immune-regulating effects, inhibited inflammatory cytokines, relieved vomiting, and reduced anxiety, oxygen consumption, as well as lung exudation.86 In the lack of specific vaccines and antiviral drugs for COVID-19, convalescent plasma therapy is an appropriate way to reduce the severe form of the disease.87 In previous studies, plasma therapy against SARS and influenza resulted in reduced mortality and shorter hospital stay.88,89 Most patients recovered from COVID-19 produce specific antibodies against the virus that can limit COVID-19 reinfection. Therefore, plasma of recovered patients can be considered as a potential treatment option.82 Another treatment option is extracorporeal blood purification, which can be used to treat severe n-CoV pneumonia cases,90 which can eliminate cytokine storms as well as inflammatory agents, and maintain acid-base balance in patients with COVID-19.91 The recombinant monoclonal antibody is another important treatment option against respiratory viruses such as SARS-CoV. Various recombinant monoclonal antibodies, including CR3022, m396, and CR3014, have been used against human coronavirus that can be considered as therapeutic candidates for SARS-CoV-2 infection.65 The WHO recommends that steroids and methylprednisolone can be used for patients with COVID-19 only in the acute stage of respiratory distress syndrome as they prolong viral shedding in patients.92
Although there is no vaccine available yet, COVID-19 is an RNA virus, and present RNA virus vaccines (measles, polio, influenza, and Japanese encephalitis virus) can offer a promising option to prevent person-to-person transmission, immunization of health-care workers and non-infected people.68 The use of decoctions for one week (not long) is one of the principles of COVID-19 prevention by fortifying the body’s strength.93 Previous studies have suggested the use of vitamins such as vitamins A, B, C, D, and E and the use of Omega-3 polyunsaturated fatty acids, Zinc, Selenium, and iron supplementation as a good solution to prevent and control low respiratory tract infections such coronavirus infection, especially in people with underlying conditions (diabetes, high blood pressure, tumors, and heart disease) as they have low systemic immunity.94,95 As a result, strengthening the immune system and creating individual resistance is very important to combat COVID-19 and the most important approach for boosting personal immunity include maintaining personal health, following a healthy lifestyle, and maintaining adequate food intake.96 Also, taking protective measures such as improving personal hygiene, washing hands frequently, keeping a distance from others (at least one meter), avoiding unnecessary gatherings, wearing masks, getting enough rest, covering the face when sneezing and coughing, avoiding touching the face with contaminated hands and proper ventilation can prevent COVID-19.97
Conclusion
According to the pandemic, COVID-19 is a life-threatening infectious disease, especially in immunocompromised individuals. The main symptoms of this respiratory disease are fever, cough and fatigue. The primary source of these infections is not yet known, but the disease can be transmitted through droplets and close contact. After several months of the spread of this dangerous virus, there are still no specific drugs and definitive treatments for the virus. Definitive and timely diagnosis of this disease and isolating patients can be helpful in treating patients and preventing the spread of the disease. The onset and progression of COVID-19 depend on the interaction between the virus and the immune system. Therefore, the best way to control this disease is disruption of the transmission route, using decoction and vitamins to strengthen the immune system and using drugs to control the progression of the disease. Following recommendations of WHO should be a priority for everyone.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kahn JS, McIntosh K. History and Recent Advances in Coronavirus Discovery. Pediatr Infect Dis J. 2005;24(11):S223–S227. doi:10.1097/01.inf.0000188166.17324.60
2. Tyrrell DAJ, Almeida JD, Cunningham CH, et al. Coronaviridae. Intervirology. 1975;5(1–2):76–82. doi:10.1159/000149883
3. Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi:10.1007/978-1-4939-2438-7_1
4. Gorbalenya AE, Baker SC, Baric RS, et al. The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020;5(4):536–544. doi:10.1038/s41564-020-0695-z
5. Fan Y, Zhao K, Shi Z, Zhou P. Bat Coronaviruses in China. Viruses. 2019;11(3):210. doi:10.3390/v11030210
6. Yin Y, Wunderink R. MERS, SARS and other coronaviruses as causes of pneumonia: MERS, SARS and coronaviruses. Respirology. 2017;23. doi:10.1111/resp.13196
7. Peiris JSM, Lai ST, Poon LLM, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361(9366):1319–1325. doi:10.1016/S0140-6736(03)13077-2
8. Sexton NR, Smith EC, Blanc H, Vignuzzi M, Peersen OB, Denison MR. Homology-based identification of a mutation in the coronavirus RNA-Dependent RNA polymerase that confers resistance to multiple mutagens. J Virol. 2016;90(16):7415–7428. doi:10.1128/JVI.00080-16
9. Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395(10224):565–574. doi:10.1016/S0140-6736(20)30251-8
10. Guo Y-R, Cao Q-D, Hong Z-S, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak – an update on the status. Mil Med Res. 2020;7(1):11. doi:10.1186/s40779-020-00240-0
11. Zhou P, Yang X-L, Wang X-G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. doi:10.1038/s41586-020-2012-7
12. Bárcena M, Oostergetel GT, Bartelink W, et al. Cryo-electron tomography of mouse hepatitis virus: insights into the structure of the coronavirion. Proc Natl Acad Sci U S A. 2009;106(2):582–587. doi:10.1073/pnas.0805270106
13. Brian DA, Baric RS. Coronavirus genome structure and replication. Curr Top Microbiol Immunol. 2005;287:1-30. doi:10.1007/3-540-26765-4_1
14. Tatar G, Taskin Tok T. Structures and Functions of Coronavirus Proteins: molecular Modeling of Viral Nucleoprotein. Int J Virol Infect Dis. 2017;2:1.
15. Graham RL, Baric RS. Recombination, reservoirs, and the modular spike: mechanisms of coronavirus cross-species transmission. J Virol. 2010;84(7):3134–3146. doi:10.1128/JVI.01394-09
16. Bosch BJ, van der Zee R, de Haan CAM, Rottier PJM. The coronavirus spike protein is a class I virus fusion protein: structural and functional characterization of the fusion core complex. J Virol. 2003;77(16):8801–8811. doi:10.1128/jvi.77.16.8801-8811.2003
17. Collins AR, Knobler RL, Powell H, Buchmeier MJ. Monoclonal antibodies to murine hepatitis virus-4 (strain JHM) define the viral glycoprotein responsible for attachment and cell-cell fusion. Virology. 1982;119(2):358–371. doi:10.1016/0042-6822(82)90095-2
18. Ramadan N, Shaib H. Middle East respiratory syndrome coronavirus (MERS-CoV): A review. Germs. 2019;9(1):35–42. doi:10.18683/germs.2019.1155
19. de Haan CA, Kuo L, Masters PS, Vennema H, Rottier PJ. Coronavirus particle assembly: primary structure requirements of the membrane protein. J Virol. 1998;72(8):6838–6850. doi:10.1128/JVI.72.8.6838-6850.1998
20. de Haan CAM, de Wit M, Kuo L, et al. The glycosylation status of the murine hepatitis coronavirus M protein affects the interferogenic capacity of the virus in vitro and its ability to replicate in the liver but not the brain. Virology. 2003;312(2):395–406. doi:10.1016/s0042-6822(03)00235-6
21. Hurst KR, Koetzner CA, Masters PS. Identification of in vivo-interacting domains of the murine coronavirus nucleocapsid protein. J Virol. 2009;83(14):7221–7234. doi:10.1128/JVI.00440-09
22. Escors D, Ortego J, Laude H, Enjuanes L. The membrane M protein carboxy terminus binds to transmissible gastroenteritis coronavirus core and contributes to core stability. J Virol. 2001;75(3):1312–1324. doi:10.1128/JVI.75.3.1312-1324.2001
23. Raamsman MJ, Locker JK, de Hooge A, et al. Characterization of the coronavirus mouse hepatitis virus strain A59 small membrane protein E. J Virol. 2000;74(5):2333–2342. doi:10.1128/jvi.74.5.2333-2342.2000
24. Nieto-Torres JL, DeDiego ML, Verdiá-Báguena C, et al. Severe acute respiratory syndrome coronavirus envelope protein ion channel activity promotes virus fitness and pathogenesis. PLoS Pathog. 2014;10(5):e1004077. doi:10.1371/journal.ppat.1004077
25. Klausegger A, Strobl B, Regl G, Kaser A, Luytjes W, Vlasak R. Identification of a coronavirus hemagglutinin-esterase with a substrate specificity different from those of influenza C virus and bovine coronavirus. J Virol. 1999;73(5):3737–3743. doi:10.1128/JVI.73.5.3737-3743.1999
26. Cornelissen LA, Wierda CM, van der Meer FJ, et al. Hemagglutinin-esterase, a novel structural protein of torovirus. J Virol. 1997;71(7):5277–5286. doi:10.1128/JVI.71.7.5277-5286.1997
27. Malik YS, Sircar S, Bhat S, et al. Emerging novel coronavirus (2019-nCoV)—current scenario, evolutionary perspective based on genome analysis and recent developments. Vet Q. 2020;40(1):68–76. doi:10.1080/01652176.2020.1727993
28. Zhou P, Yang X-L, Wang X-G, et al. Discovery of a novel coronavirus associated with the recent pneumonia outbreak in humans and its potential bat origin. bioRxiv;2020.
29. She J, Jiang J, Ye L, Hu L, Bai C, Song Y. 2019 novel coronavirus of pneumonia in Wuhan, China: emerging attack and management strategies. Clin Transl Med. 2020;9(1):19. doi:10.1186/s40169-020-00271-z
30. Chan JF-W, Yuan S, Kok K-H, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514–523. doi:10.1016/S0140-6736(20)30154-9
31. Jin Y-H, Cai L, Cheng Z-S, et al. A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version). Mil Med Res. 2020;7(1):4. doi:10.1186/s40779-020-0233-6
32. Giovanetti M, Benvenuto D, Angeletti S, Ciccozzi M. The first two cases of 2019-nCoV in Italy: where they come from? J Med Virol. 2020;92(5):518–521. doi:10.1002/jmv.25699
33. Paraskevis D, Kostaki EG, Magiorkinis G, Panayiotakopoulos G, Sourvinos G, Tsiodras S. Full-genome evolutionary analysis of the novel corona virus (2019-nCoV) rejects the hypothesis of emergence as a result of a recent recombination event. Infect Genet Evol. 2020;79:104212. doi:10.1016/j.meegid.2020.104212
34. Organization World Health.Coronavirus Disease (COVID-19) Outbreak Situation. WHO; 2020. https://www.who.int/emergencies/diseases/novel-coronavirus-2019.
35. Lu C, Liu X, Jia Z. 2019-nCoV transmission through the ocular surface must not be ignored. Lancet. 2020:395. doi:10.1016/S0140-6736(20)30313-5
36. Phan LT, Nguyen TV, Luong QC, et al. Importation and Human-to-Human Transmission of a Novel Coronavirus in Vietnam. N Engl J Med. 2020;382(9):872–874. doi:10.1056/NEJMc2001272
37. Guan W, Ni Z, Hu Y, et al. Clinical characteristics of 2019 novel coronavirus infection in China. medRxiv. 2020:25458. doi:10.1101/2020.02.06.20020974
38. Hoe GAN W, Wah LIM J, Koh D. Preventing intra-hospital infection and transmission of COVID-19 in healthcare workers. Saf Health Work. 2020. doi:10.1016/j.shaw.2020.03.001
39. Lai -C-C, Shih T-P, Ko W-C, Tang H-J, Hsueh P-R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020;55(3):105924. doi:10.1016/j.ijantimicag.2020.105924
40. Ding S, Liang T. Is SARS-CoV-2 also an enteric pathogen with potential Fecal-Oral transmission: a COVID-19 virological and clinical review. Gastroenterology. 2020;159(1):53–61. doi:10.1053/j.gastro.2020.04.052
41. Peng X, Xu X, Li Y, Cheng L, Zhou X, Ren B. Transmission routes of 2019-nCoV and controls in dental practice. Int J Oral Sci. 2020;12(1):9. doi:10.1038/s41368-020-0075-9
42. Zhou Z, Chen P, Peng H. Are healthy smokers really healthy? Tob Induc Dis. 2016;14(1). doi:10.1186/s12971-016-0101-z
43. Vardavas CI, Nikitara K. COVID-19 and smoking: A systematic review of the evidence. Tob Induc Dis. 2020;18(March). doi:10.18332/tid/119324
44. Teles Abrao Trad A, Ibirogba ER, Elrefaei A, et al. Complications and outcomes of SARS-CoV-2 in pregnancy: where and what is the evidence? Hypertens Pregnancy. 2020:1–9. doi:10.1080/10641955.2020.1769645.
45. Rasmussen SA, Smulian JC, Lednicky JA, Wen TS, Jamieson DJ. Coronavirus Disease 2019 (COVID-19) and Pregnancy: what obstetricians need to know. Am J Obstet Gynecol. 2020: doi:10.1016/j.ajog.2020.02.017
46. Karimi-Zarchi M, Neamatzadeh H, Dastgheib SA, et al. Vertical Transmission of Coronavirus Disease 19 (COVID-19) from infected pregnant mothers to neonates: a review. Fetal Pediatr Pathol. 2020:1–5. doi:10.1080/15513815.2020.1747120.
47. Wang S, Guo L, Chen L, et al. A case report of neonatal 2019 coronavirus disease in China. Clin Infect Dis. 2020. doi:10.1093/cid/ciaa225
48. Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus–infected pneumonia. N Engl J Med. 2020;382(13):1199–1207. doi:10.1056/NEJMoa2001316
49. Wang W, Tang J, Wei F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China. J Med Virol. 2020;92(4):441–447. doi:10.1002/jmv.25689
50. Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. 2020;109:102433. doi:10.1016/j.jaut.2020.102433
51. Wang Y, Kang H, Liu X, Tong Z. Combination of RT-qPCR testing and clinical features for diagnosis of COVID-19 facilitates management of SARS-CoV-2 outbreak. J Med Virol. 2020. doi:10.1002/jmv.25721
52. Lai -C-C, Liu YH, Wang C-Y, et al. Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): facts and myths. J Microbiol Immunol Infect. 2020;53(3):404–412. doi:10.1016/j.jmii.2020.02.012
53. Carlos WG, Dela Cruz CS, Cao B, Pasnick S, Jamil S. Novel Wuhan (2019-nCoV) Coronavirus. Am J Respir Crit Care Med. 2020;201(4):P7–P8. doi:10.1164/rccm.2014P7
54. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi:10.1016/S0140-6736(20)30183-5
55. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. doi:10.1001/jama.2020.1585
56. Hassan SA, Sheikh FN, Jamal S, Ezeh JK, Akhtar A. Coronavirus (COVID-19): A Review of Clinical Features, Diagnosis, and Treatment. Cureus. 2020;12(3):e7355. doi:10.7759/cureus.7355
57. Adhikari SP, Meng S, Wu Y-J, et al. Epidemiology, causes, clinical manifestation and diagnosis, prevention and control of coronavirus disease (COVID-19) during the early outbreak period: a scoping review. Infect Dis Poverty. 2020;9(1):29. doi:10.1186/s40249-020-00646-x
58. Deeks JJ, Dinnes J, Takwoingi Y, et al. Diagnosis of SARS‐CoV‐2 infection and COVID‐19: accuracy of signs and symptoms; molecular, antigen, and antibody tests; and routine laboratory markers. Cochrane Database Syst Rev. 2020;4.
59. Wilder-Smith A, Low J. Risk of respiratory infections in health care workers: lessons on infection control emerge from the SARS outbreak. Southeast Asian J Trop Med Public Health. 2005;36(2):481–488.
60. Ai T, Yang Z, Hou H, et al. Correlation of Chest CT and RT-PCR Testing in Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020:200642. doi:10.1148/radiol.2020200642.
61. Mahdavi A, Khalili N, Davarpanah AH, et al. Radiologic Management of COVID-19: preliminary Experience of the Iranian Society of Radiology COVID-19 Consultant Group (ISRCC). Iran J Radiol. 2020:e102324; doi:10.5812/iranjradiol.102324
62. Karimi A, Rajabnejad SRTM, Rahimi ZPH, et al. An Algorithmic Approach to Diagnosis and Treatment of Coronavirus Disease 2019 (COVID-19) in Children: iranian Expert’s Consensus Statement. Arch Pediatr Infect Dis. 2020;8(2):e102400. doi:10.5812/pedinfect.102400
63. Bradley JS, Byington CL, Shah SS, et al. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the pediatric infectious diseases society and the infectious diseases society of America. Clin Infect Dis. 2011;53(7):e25–e76. doi:10.1093/cid/cir531
64. Tang Y-W, Schmitz JE, Persing DH, Stratton CW. Laboratory Diagnosis of COVID-19: current Issues and Challenges. McAdam AJ, ed. J Clin Microbiol. 2020;58(6):e00512–200520. doi:10.1128/JCM.00512-20
65. Li X, Geng M, Peng Y, Meng L, Lu S. Molecular immune pathogenesis and diagnosis of COVID-19. J Pharm Anal. 2020;10(2):102–108. doi:10.1016/j.jpha.2020.03.001
66. Yang T, Wang Y-C, Shen C-F, Cheng C-M. Point-of-Care RNA-Based Diagnostic Device for COVID-19. Diagnostics. 2020;10(3):165. doi:10.3390/diagnostics10030165
67. Corman VM, Landt O, Kaiser M, et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 2020;25(3):2000045. doi:10.2807/1560-7917.ES.2020.25.3.2000045
68. Zhang L, Liu Y. Potential interventions for novel coronavirus in China: A systematic review. J Med Virol. 2020;92(5):479–490. doi:10.1002/jmv.25707
69. Holshue ML, DeBolt C, Lindquist S, et al. First Case of 2019 Novel Coronavirus in the United States. N Engl J Med. 2020;382(10):929–936. doi:10.1056/NEJMoa2001191
70. De Clercq E. New nucleoside analogues for the treatment of hemorrhagic fever virus infections. Chem an Asian J. 2019;14(22):3962–3968. doi:10.1002/asia.201900841
71. Zumla A, Chan JFW, Azhar EI, Hui DSC, Yuen K-Y. Coronaviruses - drug discovery and therapeutic options. Nat Rev Drug Discov. 2016;15(5):327–347. doi:10.1038/nrd.2015.37
72. Toots M, Yoon -J-J, Cox RM, et al. Characterization of orally efficacious influenza drug with high resistance barrier in ferrets and human airway epithelia. Sci Transl Med. 2019;11(515):eaax5866. doi:10.1126/scitranslmed.aax5866
73. Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi:10.1016/S0140-6736(20)30211-7
74. Gao J, Tian Z, Yang X. Breakthrough: chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. 2020;14(1):72–73. doi:10.5582/bst.2020.01047
75. Wang M, Cao R, Zhang L, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30(3):269–271. doi:10.1038/s41422-020-0282-0
76. Li W. [The curative effect observation of shuanghuanglian and penicillin on acute tonsillitis]. Lin Chuang Er Bi Yan Hou Ke Za Zhi. 2002;16:475–476. Chinese.
77. Chen X, Howard OM, Yang X, Wang L, Oppenheim J, Krakauer T. Effects of Shuanghuanglian and Qingkailing, two multi-components of traditional Chinese medicinal preparations, on human leukocyte function. Life Sci. 2002;70(24):2897–2913. doi:10.1016/S0024-3205(02)01541-2
78. Lu HT, Yang JC, Yuan ZC, Sheng WH, Yan WH. [Effect of combined treatment of Shuanghuanglian and recombinant interferon alpha 2a on coxsackievirus B3 replication in vitro]. Zhongguo Zhong Yao Za Zhi. 2000;25(11):682–684.Chinese.
79. Ding Y, Zeng L, Li R, et al. The Chinese prescription lianhuaqingwen capsule exerts anti-influenza activity through the inhibition of viral propagation and impacts immune function. BMC Complement Altern Med. 2017;17(1):130. doi:10.1186/s12906-017-1585-7
80. Jo S, Kim S, Shin DH, Kim M-S. Inhibition of SARS-CoV 3CL protease by flavonoids. J Enzyme Inhib Med Chem. 2020;35(1):145–151. doi:10.1080/14756366.2019.1690480
81. Chen L, Gui C, Luo X, et al. Cinanserin Is an Inhibitor of the 3C-like proteinase of severe acute respiratory syndrome coronavirus and strongly reduces virus replication in vitro. J Virol. 2005;79(11):7095–7103. doi:10.1128/JVI.79.11.7095-7103.2005
82. Wang L, Wang Y, Ye D, Liu Q. A review of the 2019 Novel Coronavirus (COVID-19) based on current evidence. Int J Antimicrob Agents. 2020;55(6):105948. doi:10.1016/j.ijantimicag.2020.105948
83. Mustafa S, Balkhy H, Gabere MN. Current treatment options and the role of peptides as potential therapeutic components for Middle East Respiratory Syndrome (MERS): A review. J Infect Public Health. 2018;11(1):9–17. doi:10.1016/j.jiph.2017.08.009
84. Gilardin L, Bayry J, Kaveri SV. Intravenous immunoglobulin as clinical immune-modulating therapy. CMAJ. 2015;187(4):257–264. doi:10.1503/cmaj.130375
85. Kumar V, Jung Y-S, Liang P-H. Anti-SARS coronavirus agents: a patent review (2008 – present). Expert Opin Ther Pat. 2013;23(10):1337–1348. doi:10.1517/13543776.2013.823159
86. Chen C, Qi F, Shi K, et al. Thalidomide combined with low-dose glucocorticoid in the treatment of COVID-19 pneumonia. 2020.
87. Mair-Jenkins J, Saavedra-Campos M, Baillie JK, et al. The effectiveness of convalescent plasma and hyperimmune immunoglobulin for the treatment of severe acute respiratory infections of viral etiology: a systematic review and exploratory meta-analysis. J Infect Dis. 2015;211(1):80–90. doi:10.1093/infdis/jiu396
88. Soo YOY, Cheng Y, Wong R, et al. Retrospective comparison of convalescent plasma with continuing high-dose methylprednisolone treatment in SARS patients. Clin Microbiol Infect. 2004;10(7):676–678. doi:10.1111/j.1469-0691.2004.00956.x
89. Hung IFN, To KKW, Lee C-K, et al. Convalescent plasma treatment reduced mortality in patients with severe pandemic influenza A (H1N1) 2009 Virus Infection. Clin Infect Dis. 2011;52(4):447–456. doi:10.1093/cid/ciq106
90. Monteil V, Kwon H, Prado P, Hagelkrüys A, Wimmer RA. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell Press. 2020. doi:10.1016/j.cell.2020.04.004
91. Zarbock A, Kellum JA, Schmidt C, et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. J Am Med Assoc. 2016;315(20):2190–2199. doi:10.1001/jama.2016.5828
92. Vetter P, Eckerle I, Kaiser L. Covid-19: a puzzle with many missing pieces. BMJ. 2020;368:m627. doi:10.1136/bmj.m627
93. Luo H, Tang Q, Shang Y, et al. Can Chinese medicine be used for prevention of corona virus disease 2019 (COVID-19)? A review of historical classics, research evidence and current prevention programs. Chin J Integr Med. 2020;26(4):243–250. doi:10.1007/s11655-020-3192-6
94. Nonnecke BJ, McGill JL, Ridpath JF, Sacco RE, Lippolis JD, Reinhardt TA. Acute phase response elicited by experimental bovine diarrhea virus (BVDV) infection is associated with decreased vitamin D and E status of vitamin-replete preruminant calves1. J Dairy Sci. 2014;97(9):5566–5579. doi:10.3168/jds.2014-8293
95. Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21(3):335–337. doi:10.1016/S1470-2045(20)30096-6
96. High KP. Nutritional Strategies to Boost Immunity and Prevent Infection in Elderly Individuals. Clin Infect Dis. 2001;33(11):1892–1900. doi:10.1086/324509
97. Abdi M. Coronavirus disease 2019 (COVID-19) outbreak in Iran; actions and problems. Infect Control Hosp Epidemiol. 2020;1–5.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
