Back to Journals » OncoTargets and Therapy » Volume 9
Correlation of PDK1 expression with clinicopathologic features and prognosis of hepatocellular carcinoma
Authors Wang J, Liu F, Ao P, Li X, Zheng H, Wu D, Zhang N, She J, Yuan J, Wu X
Received 15 April 2016
Accepted for publication 22 June 2016
Published 9 September 2016 Volume 2016:9 Pages 5597—5602
DOI https://doi.org/10.2147/OTT.S110646
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Min Li
Junrong Wang, Fenqin Liu, Peiran Ao, Xianneng Li, Haixiao Zheng, Di Wu, Nina Zhang, Junping She, Junhui Yuan, Xiuying Wu
Department of Laboratory Medicine, Wenling Maternal and Child Health Care Hospital, Wenling, Zhejiang Province, People’s Republic of China
Objective: To explore the clinical significance of 3-phosphoinositide-dependent protein kinase-1 (PDK1) expression in hepatocellular carcinoma (HCC) and its association with clinicopathologic features and prognosis in HCC patients.
Materials and methods: A total of 128 HCC patients who received radical resection were enrolled from Wenling Maternal and Child Health Care Hospital between May 2005 and December 2008, and tumor and adjacent tissue samples were collected. Expression of PDK1 was detected by immunohistochemistry method. Correlation of PDK1 expression with clinicopathological features and prognosis was determined by Spearman’s correlation analysis. Impact of expression of PDK1 on overall survival and recurrence was determined by Kaplan–Meier analysis.
Results: Immunohistochemistry results showed that PDK1 expression in HCC tissues was significantly higher than that in the corresponding adjacent cancer tissues. Univariate analysis showed that PDK1 messenger RNA expression can predict time to recurrence with diagnostic significance (P=0.001). Univariate analysis showed that alpha-fetoprotein level, tumor number, tumor encapsulation, microvascular invasion, and tumor–node–metastasis stage were also unfavorable prognostic variables for recurrence (P<0.05). Kaplan–Meier analysis showed that overexpression of PDK1 correlates with significantly shorter postoperative overall survival and higher recurrence rates (hazard ratio =2.68; 95% confidence interval: 2.46–4.42, P=0.001) in HCC patients after curative resection.
Conclusion: Our study indicated that PDK1 may serve as a candidate pro-oncogene and a potential prognostic biomarker for HCC.
Keywords: hepatocellular carcinoma, HCC, 3-phosphoinositide-dependent protein kinase-1, PDK1, prognosis, alpha-fetoprotein, AFP, microvascular invasion, tumor–node–metastasis, TNM stage
Introduction
Hepatocellular carcinoma (HCC) is the most common type of liver cancer, which accounts for the fifth global incidence of malignancy with an increasing prevalence all over the world.1,2 The incidence of HCC is severe in the People’s Republic of China. More than 50% of patients worldwide who died from malignant hepatoma were from the People’s Republic of China. Although surgical resection and liver transplantation are the solutions for HCC treatment, the prognosis of HCC is still poor for its high rates of recurrence and metastasis.3 Therefore, it is critical to find the molecular markers associated with the prognosis and therapy of the disease. 3-Phosphoinositide-dependent kinase-1 (PDK1) is a 63 kD serine/threonine kinase, which can be divided into the kinase domain and the PH domain.4,5 PDK1 is reported to be abnormally expressed and activated in a variety of tumors and participate in the regulation of cell proliferation, tumor invasiveness, angiogenesis, and progression.6 Herein, we observed the relationship between the expression of PDK1 in tumor tissues of 128 HCC patients with radical operation and the clinical characteristics of these patients to investigate the feasibility of PDK1 expression differences as the prediction index of postoperation of HCC.
Materials and methods
Patients and sample collection
Carcinoma and adjacent tissue samples from 128 patients with primary HCC who received radical resection of liver tumor at Wenling Maternal and Child Health Care Hospital from May 2005 to December 2008 were collected. All the samples were confirmed by definitive pathological diagnosis. All patients had not received any radiotherapy and chemotherapy treatment before the operation. Moreover, the patients had no distant metastasis and liver function was A Child-Pugh score. The HCC staging was made according to the tumor–node–metastasis (TNM) classification of malignant tumors used by the International Union against Cancer.7 The protocols were approved by the Ethics Committee of Wenling Maternal and Child Health Care Hospital, and informed consent was obtained from participants prior to their participation in this study.
Follow-up data
All patients had complete follow-up data. Every patient was followed up at 3, 6, 9, 12, 18, and 24 months postoperation, and subsequent follow-up was done once a year from the third year after operation. Postoperative overall survival was defined as time from surgery to death or last follow-up visit. Time to recurrence (TTR) was defined as the interval between HCC resection and tumor recurrence or metastasis by imaging confirmed. In this study, the final follow-ups were at October 2014, and the median follow-up time was 26.0 (5.0–138.0) months.
Immunohistochemistry
Carcinoma and adjacent tissue samples from 128 HCC patients were collected, and immunohistochemical staining was performed using VECTOR® M.O.M.™ Immunodetection Kit (Vector Laboratories Inc., Burlingame, CA, USA). Xylene dewaxing and gradient ethanol hydration of the histological sections were performed before soaking in 3% hydrogen peroxide solution for 10 minutes at room temperature. Then, the sections were put into citric acid solution (pH 6.0) and boiled. After cooling to room temperature, each of the sections was washed three times for 3 minutes using phosphate-buffered saline and blocked in 5% fetal bovine serum for 15 minutes at room temperature. Anti-PDK1 was added to the sections and then incubated at 4°C overnight. Then, each of the sections was washed three times for 3 minutes, and streptavidin biotin peroxidase solution (D agent) was added to the samples. After incubating for 10 minutes at room temperature, the samples were again washed three times. Freshly prepared 3,3′-diaminobenzidine (DAB) was added, followed by water immersion, hematoxylin counterstaining for 2 minutes, water immersion, hydrochloric acid alcohol differentiation for 5 seconds, and water rinse for 20 minutes that were carried out in sequence. Finally, gradient ethanol hydration, xylene transparent neutral balata, and camera observation were performed. The median integrated optical density (mIOD) was set as the cutoff point, and all samples were divided into high-expression group (integrated optical density [IOD] > mIOD) and low-expression group (IOD < mIOD).
Statistical analysis
Statistical analyses were performed using SPSS 17.0 software (SPSS Inc., Chicago, IL, USA). The correlations between PDK1 expression and the clinical characteristics were evaluated using Spearman’s rank correlation. Kaplan–Meier survival curve was drawn and compared with log-rank test. Univariate and multivariate analyses were used to estimate the effect of different factors on the prognosis. P<0.05 was considered statistically significant for all tests.
Results
The expression of PDK1 is upregulated in HCC
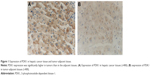
Carcinoma and adjacent tissue samples from 128 HCC patients with radical resection of liver tumor were collected. Then, immunohistochemistry was performed to investigate PDK1 expression. Expression of PDK1 was found in the nucleus of tumors and adjacent tissues, and all of them could be divided into high-expression group (47.7%, 61/128) and low-expression group (52.3%, 67/128) based on staining intensity. PDK1 expression was significantly higher in tumors than in adjacent tissues (Figure 1).
The connection between PDK1 expression and clinical characteristics
In this study, we observed PDK1 expression in tumors and adjacent tissues as well as the clinical characteristics of HCC patients. As shown in Table 1, PDK1 expression was positively correlated with clinical stage (P=0.008), microvascular invasion (P=0.021), and alpha-fetoprotein (AFP) (P=0.003). No significant differences were found in PDK1 expression with regard to age, gender, liver cirrhosis, tumor size, and capsule.
The relationship between PDK1 overexpression and postoperative prognosis
In this study, 74 out of 128 cases showed reoccurrence within the follow-up time of 38.8±4.3 months (median, 26 months; range, 5.0–138.0 months), sixty one patients with intrahepatic metastasis, five patients with pulmonary metastasis, and eight patients with both pulmonary and intrahepatic metastasis. Messenger RNA expression of PDK1 in postoperative HCC patients could predict TTR effectively (P=0.001; Table 2). Compared with PDK1 low-expression group, PDK1 high-expression group had a significantly shorter TTR median (9.0 months vs not reached) and a remarkably higher recurrence rate (81.0% vs 37.1%) (P<0.001 in Table 1). As shown in Table 2, the level of AFP (P=0.024), the amount of tumors (P=0.022), tumor encapsulation (P=0.001), microvascular invasion (P=0.003), and TNM stage (P=0.005) were the probable risk factors for recurrence by univariate analysis. In addition, multivariate analysis indicated that the most forceful prognostic factor for TTR was high mRNA expression of PDK1 (hazard ratio =2.68; 95% confidence interval: 2.46–4.42, P=0.001; Table 2). Moreover, patients in the high-expression group had a higher recurrence probability but a shorter overall survival time, compared with those in the low-expression group (Figure 2). This finding illustrated that PDK1 might be a potential therapeutic target for HCC therapy.
Discussion
Primary HCC is one of the malignant tumors associated with high mortality. It is the third cause of cancer-related deaths globally and the second reason for death from cancer in the People’s Republic of China.8 Early diagnosis of HCC is difficult due to its high-grade malignancy and tumor invasion, and the poor prognosis is expected because of high postoperative recurrence.9 Currently, gene therapy and molecularly targeted therapy are used in clinical research; various genes for targeted therapy of HCC are effective than the traditional forms of treatments. It is important to find the molecular markers associated with the prognosis and therapy of this disease.
PDK1 is a member of serine/threonine kinases family whose catalytic domain is homologous to cyclic AMP-dependent protein kinase and protein kinase C.10 Researches indicated that PDK1 is abnormally expressed and activated in tumors and may be a major regulatory factor associated with tumorigenesis and metastasis.6 Recently, Bai et al11 found that PDK1 could be an indicator for long-term survival of gastric cancer. Lohneis et al12 also pointed that PDK1 was an independent prognostic factor that is significantly negatively correlated to tumor grade in ovarian serous carcinoma. Moreover, other studies proved that PDK1 was correlated to prognosis of malignant gliomas,13 breast cancer,14,15 gall bladder cancer,16 colorectal cancer,17 and pancreatic cancer.18 However, the relationship between PDK1 expression and prognosis of HCC has not yet been explored. The transcriptional regulation activity can be affected by the content of PDK1 in the nucleus, posttranslational modification, and other various factors.19
In this study, analyses of PDK1 expression in HCC tissues and tumor-adjacent tissues by immunohistochemical staining were performed. The results showed that PDK1 expression was high in HCC tumors but low in adjacent tissues. This indicated that PDK1 performed as a regulator in tumorigenesis, metastasis, and recurrence.
Multivariate analysis was carried out to investigate liver tumors and adjacent tissues in HCC patients. We found that the prognosis of HCC patients correlated with liver cirrhosis, tumor size, microvascular invasion, and TNM stage. Furthermore, there were no significant differences in PDK1 expression with regard to age, gender, liver cirrhosis, serum AFP, tumor size, or capsule. On the contrary, overexpression of PDK1 was related closely to microvascular invasion. These results suggested that high expression of PDK1 occurred frequently in tumors, and PDK1 might have an effect on angiogenesis. This corresponded to the conclusion of our previous studies that the specific binding of PDK1 and the promoter of vascular endothelial growth factor was the most critical step to regulate angiogenesis, metastasis, and recurrence of HCC.20 We inferred that high expression of PDK1 in HCC might upregulate vascular endothelial growth factor expression to promote the angiogenesis of HCC. Furthermore, angiogenesis of HCC was an important trigger for tumor metastasis and recurrence and related to HCC prognosis. As a result, PDK1, as a regulator of angiogenesis, is probably related to HCC prognosis. In our results, the expression of PDK1 was negatively correlated with overall survival rate but was positively correlated with postoperative recurrence rate. Both the correlations were statistically significant. Univariate analysis showed that the level of AFP, the amount of tumors, tumor encapsulation, microvascular invasion, and TNM stage were probably unfavorable prognostic variables for recurrence (P<0.05). In addition, PDK1 expression was closely related to postoperative recurrence to indicate PDK1 as an independent prognostic factor in HCC. This might be a new perspective for exploration of the correlation between high expression of PDK1 and unfavorable prognosis of HCC patients. Moreover, we demonstrated that PDK1 had an effect on HCC progression through MAPK1 pathway dependent on epithelial mesenchymal transition activation.
Conclusion
Our results showed that PDK1 could be used as a potential marker and might be helpful for finding an effective treatment for HCC.
Acknowledgments
The authors thank all the enrolled patients who supported and contributed to our study and Ning Yang and Yuandan Ying for sample collection and technical support for this research. This work was supported by grants from the Science and Technology Foundation of Wenling City of Zhejiang Province, China (2016C311).
Disclosure
The authors report no conflicts of interest in this work.
References
Chan SL, Wong AM, Lee K, Wong N, Chan AK. Personalized therapy for hepatocellular carcinoma: where are we now? Cancer Treat Rev. 2016;45:77–86. | ||
Singh S, Singh PP, Roberts LR, Sanchez W. Chemopreventive strategies in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2014;11(1):45–54. | ||
Sieghart W, Hucke F, Peck-Radosavljevic M. Transarterial chemoembolization: modalities, indication, and patient selection. J Hepatol. 2015;62(5):1187–1195. | ||
Gagliardi PA, di Blasio L, Primo L. PDK1: a signaling hub for cell migration and tumor invasion. Biochim Biophys Acta. 2015;1856(2):178–188. | ||
Calleja V, Laguerre M, de Las Heras-Martinez G, Parker PJ, Requejo-Isidro J, Larijani B. Acute regulation of PDK1 by a complex interplay of molecular switches. Biochem Soc Trans. 2014;42(5):1435–1440. | ||
Raimondi C, Falasca M. Targeting PDK1 in cancer. Curr Med Chem. 2011;18(18):2763–2769. | ||
Kudo M. Real practice of hepatocellular carcinoma in Japan: conclusions of the Japan Society of Hepatology 2009 Kobe Congress. Oncology. 2010;78(suppl 1):180–188. | ||
Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol. 2013;47(suppl):S2–S6. | ||
Bolondi L. Screening for hepatocellular carcinoma in cirrhosis. J Hepatol. 2003;39(6):1076–1084. | ||
Galgano MT, Conaway M, Spencer AM, Paschal BM, Frierson HF Jr. PRK1 distribution in normal tissues and carcinomas: overexpression and activation in ovarian serous carcinoma. Hum Pathol. 2009;40(10):1434–1440. | ||
Bai X, Li P, Xie Y, et al. Overexpression of 3-phosphoinositide-dependent protein kinase-1 is associated with prognosis of gastric carcinoma. Tumour Biol. 2016;37(2):2333–2339. | ||
Lohneis P, Darb-Esfahani S, Dietel M, Braicu I, Sehouli J, Arsenic R. PDK1 is expressed in ovarian serous carcinoma and correlates with improved survival in high-grade tumors. Anticancer Res. 2015;35(11):6329–6334. | ||
Huang N, Cheng S, Mi X, et al. Downregulation of nitrogen permease regulator like-2 activates PDK1-AKT1 and contributes to the malignant growth of glioma cells. Mol Carcinog. Epub 2015 Oct 12. | ||
Du J, Yang M, Chen S, Li D, Chang Z, Dong Z. PDK1 promotes tumor growth and metastasis in a spontaneous breast cancer model. Oncogene. 2016;35(25):3314–3323. | ||
Dupuy F, Tabariès S, Andrzejewski S, et al. PDK1-dependent metabolic reprogramming dictates metastatic potential in breast cancer. Cell Metab. 2015;22(4):577–589. | ||
Lian S, Shao Y, Liu H, et al. PDK1 induces JunB, EMT, cell migration and invasion in human gallbladder cancer. Oncotarget. 2015;6(30):29076–29086. | ||
Yoon S, Kim JG, Seo AN, et al. Clinical implication of serine metabolism-associated enzymes in colon cancer. Oncology. 2015;89(6):351–359. | ||
Ohtsubo K, Yamada T, Zhao L, et al. Expression of Akt kinase-interacting protein 1, a scaffold protein of the PI3K/PDK1/Akt pathway, in pancreatic cancer. Pancreas. 2014;43(7):1093–1100. | ||
Liu H, Song G, Zhou L, et al. Compared analysis of LncRNA expression profiling in pdk1 gene knockout mice at two time points. Cell Physiol Biochem. 2013;32(5):1497–1508. | ||
Yamazaki T, Akada T, Niizeki O, Suzuki T, Miyashita H, Sato Y. Puromycin-insensitive leucyl-specific aminopeptidase (PILSAP) binds and catalyzes PDK1, allowingVEGF-stimulated activation of S6K for endothelial cell proliferation and angiogenesis. Blood. 2004;104(8):2345–2352. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.