Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 16
Correlation of Luminal Mucus Score in Large Airways with Lung Function and Quality of Life in Severe Acute Exacerbation of COPD: A Cross-Sectional Study
Authors Yang C , Zeng HH, Du YJ, Huang J, Zhang QY, Lin K
Received 22 March 2021
Accepted for publication 9 May 2021
Published 28 May 2021 Volume 2021:16 Pages 1449—1459
DOI https://doi.org/10.2147/COPD.S311659
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Cheng Yang,1 Han-Hua Zeng,1 Yan-Jia Du,1 Juan Huang,1 Qian-Yun Zhang,1 Kun Lin2
1Department of Pulmonary and Critical Care Medicine, Meizhou People’s Hospital, Meizhou, 5143031, Guangdong, People’s Republic of China; 2Department of Preventive Medicine, Shantou University Medical College, Shantou, 515041, Guangdong, People’s Republic of China
Correspondence: Qian-Yun Zhang
Department of Pulmonary and Critical Care Medicine, Meizhou People’s Hospital, No. 63 Huangtang Road, Meijiang District, Meizhou, 514031, People’s Republic of China
Tel +86 07532202723
Fax +86 07532204840
Email [email protected]
Kun Lin
Department of Preventive Medicine, Shantou University Medical College, No. 22 Xinling Road, Jinping District, Shantou, 515041, People’s Republic of China
Tel +86 075488900445
Fax +86 075482903637
Email [email protected]
Background and Objective: Luminal mucus plugging in small airways is associated with lung function decline and death of patients with chronic obstructive pulmonary disease (COPD). However, little attention has been paid to the possible role of mucus in large airways in acute exacerbation of COPD (AECOPD). Therefore, this study aimed to explore the relationship between the luminal mucus score of large airways and other physiological parameters of severe AECOPD.
Subjects and Methods: A total of 74 AECOPD inpatients were enrolled in this cross-sectional study. All patients underwent lung function tests and bronchoscopy, and their luminal mucus was observed and scored through bronchoscopy. Four questionnaires, including the St. George Respiratory Questionnaire (SGRQ), modified Medical Research Council dyspnea scale (mMRC), COPD Assessment Test (CAT) and Exacerbation of Chronic pulmonary disease Tool (EXACT), were used to assess health-related quality of life (HRQoL).
Results: The luminal mucus score of large airways was significantly correlated with spirometry parameters and HRQoL score. Both mMRC grade and SGRQ score were significantly positively correlated with luminal mucus score (ρ=0.527, P< 0.001; ρ=0.441, P< 0.001, respectively). Forced expiratory flow at 25% to 75% of the FVC (FEF25%-75%) and FEV1% predicted, as functional measures reflecting small airway disease, were significantly negatively correlated with luminal mucus score (ρ=− 0.518, P< 0.001; ρ=− 0.498, P< 0.001, respectively). The stepwise multiple linear regression model suggested that mMRC grade and FEV1% predicted could predict luminal mucus score (R2=0.348, F=18.960, P< 0.001).
Conclusion: For severe acute exacerbation of COPD, bronchoscopy-identified luminal mucus in large airways is associated with reduced lung function and worse health-related quality of life.
Keywords: chronic obstructive pulmonary disease, acute exacerbation, luminal, mucus, bronchoscopy
Introduction
Mucus dysfunction is a major pathophysiological feature of chronic obstructive pulmonary disease (COPD), and is manifested by excessive production of mucus and luminal occlusion.1 Mucous exudates in small airways are related to reduced lung function and increased mortality in COPD patients, as confirmed by pathological examination of isolated lung tissue.2 In large airways (> 2 mm in diameter), it has been observed by CT that the luminal plugging is related to airflow obstruction, poor health-related quality of life (HRQoL) and emphysema phenotype.3 However, whether the luminal plugging identified by CT is caused by mucus or not remains to be confirmed by bronchoscopy.
Acute exacerbation of COPD (AECOPD), which is characterized by a worsening of respiratory symptoms such as dyspnea, increased sputum volume, and purulence, is the leading cause of hospitalization and death from the disease.4 However, little attention has been paid to the possible role of the mucus in large airways in exacerbations. It was found that a bronchitis index score, determined by secretions, erythema, edema, and friability via bronchoscopy, was significantly higher in patients with chronic bronchitis than in normal subjects.5 Therefore, we hypothesized that the luminal mucus in large airways may also be strongly correlated with traditional indices of AECOPD.
The main purpose of the present study was to explore the relationship of bronchoscopy-identified luminal mucus score (LMS) in large airways of inpatients with functional parameters which reflected the severity of AECOPD, including lung function, HRQoL score, and laboratory findings.4 In addition, clinical indicators that could be more easily measured were tested to predict LMS in large airways, because bronchoscopy is an invasive procedure.
Subjects and Methods
Subjects
This cross-sectional study was conducted in Meizhou People’s Hospital in Guangdong, China, from May 2020 to January 2021. A total of 90 inpatients clinically diagnosed with AECOPD were screened consecutively, and 74 of them were enrolled into the study. The screening process for participants is shown in Figure 1.
 |
Figure 1 Flowchart of participant inclusion in the study. Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; LFT, lung function test. |
The inclusion criteria were as follows: (1) a postbronchodilator FEV1/FVC ratio less than 0.7; (2) diagnosis of AECOPD, defined as:6 two major symptoms (dyspnea, increased sputum volume or purulent sputum), or one major symptom, combined with at least one secondary symptom (wheezing, chills, fever, sore throat or cough); (3) written informed consent obtained before participating in the study. Exclusion criteria were as follows: (1) contraindication(s) existing for bronchoscopy or lung function test; (2) patients with known respiratory diseases other than COPD, including sarcoidosis, active tuberculosis, pulmonary fibrosis, cystic fibrosis, or diseases that could significantly affect clinical evaluation according to the judgment of investigators; (3) diagnosis of lung cancer, including current and recent 5-year diagnoses; (4) presence of nervous system diseases affecting the ability to expectorate; (5) lack of the ability to read and understand Mandarin.
Protocol
On the first day of admission, complete blood count, C-reactive protein (CRP), four items of coagulation (prothrombin time, activated partial thromboplastin time, thrombin time, and fibrinogen), and an electrocardiogram were completed. The HRQoL assessment,7 including the modified Medical Research Council dyspnea scale (mMRC), St. George Respiratory Questionnaire (SGRQ), COPD Assessment Test (CAT), and EXAcerbation of Chronic pulmonary disease Tool (EXACT) questionnaires, was completed on the second day. A bronchoscopy was performed immediately after the HRQoL assessment, and a lung function test was performed within 12 to 48 hours after the bronchoscopy. The research process did not interfere with patient-management decisions. This study was registered with the Chinese Clinical Trial Registry (Registration Number: ChiCTR2000033101).
Bronchoscopy and Luminal Mucus Scoring
The bronchoscopy was performed by Yang, according to the guidance of the Chinese Medical Association.8 The entire examination under the bronchoscope was recorded by a video processor (CV-290 EVIS LUCERA ELITE, Olympus, Tokyo, Japan). All patients were examined using the same bronchoscope (BF-Q290, Olympus, Tokyo, Japan). Patients without contraindications received intravenous midazolam for conscious sedation 10 minutes before the operation, and the total dose did not exceed 10 mg. Before the bronchoscope passed the vocal cords and was introduced to the airways, 2% lidocaine was sprayed through the working channel, and the total dose did not exceed 5mg/kg. The mucus observed under the bronchoscope was removed. Based on the clinical needs, we performed lavage and/or biopsy, and collected specimens for microbiological and/or cytological examinations. The patients’ blood pressure, pulse oxygen saturation, heart rate and respiratory rate were monitored throughout operations as appropriate.
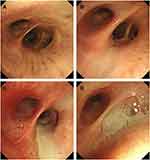
The video file was named with the corresponding subject number instead of the patient’s name or other information. Zeng who was blinded to the other clinical data of the patient scored the luminal mucus based on the video. Regarding the scoring system, Thompson et al5 proposed one involving six sites, including five lobes and the lingula. We used this scoring system with the addition of another four center airways which were involved, including the trachea, right main bronchus, left main bronchus, and right middle bronchus. The luminal mucus was scored from 0 to 3 points (0=normal, 3=severely abnormal), and thus the total LMS ranged from 0 to 30 points (Figure 2). The mucus was scored based on the most severe segment in each lobe and the lingula, and based on the most severe section in the center airway, regardless of the airway length involved.
Lung Function Test
The lung function test was performed using an electronic spirometer (microQuark PFT, Cosmed, Rome, Italy), according to the guidance published by the American Thoracic Society and European Respiratory Society.9 After four to six restful breaths, the patients were asked to inhale as much air as possible to total lung capacity (TLC), and then breathe out with the maximum effort for at least six seconds to the residual volume (RV); the volume of exhalation was the forced vital capacity (FVC). After inhaling 400 μg of Ventolin and then waiting for 20 minutes, spirometry was repeated. Only postbronchodilator spirometric parameters were adopted in this analysis.
Chronic Bronchitis
Chronic bronchitis was defined as productive cough present during most days or nights for at least 3 months of the year for ≥2 consecutive years. Chronic bronchitis was considered to be present when patients answered the following question positively: “Did you cough up phlegm (during most days or nights) for at least 3 months each year during the past 2 years?”10
Health-Related Quality of Life
Health-related quality of life (HRQoL) questionnaires were used to assess the quality of life.7 Altogether we used four different questionnaires, including mMRC, SGRQ, CAT, and EXACT. The mMRC score is used to assess the degree of dyspnea in COPD patients, which ranged from 0 to 4 points.11 The SGRQ score, ranging from 0 to 100 points, is widely applied to evaluate airway diseases resulting in a decline in HRQoL.12 CAT is a validated questionnaire containing eight items to evaluate and quantify the impact of symptoms on the health status of COPD patients.4 EXACT is used to directly measure the patient-reported symptoms of exacerbations and to conduct standardized assessment of patients’ condition.13 Subjects were asked to choose the description that best matched their symptoms, or their degree of agreement with each statement. Yang was available to provide explanations if required, but he did not give any suggestions or answers. For all four questionnaires, a higher grade or score indicated a worse quality of life.
Study Size
We estimated a sample of 72 participants, calculated based on the reported prevalence of 25% of luminal mucus plug in large airways,3 with 10% precision (95% CI). We also estimated that 20% of patients would not be able to complete bronchoscopy or lung function tests, and we therefore planned to recruit 90 subjects.
Statistical Analyses
All results were presented with descriptive data. Results were expressed by mean ± SD or median (IQR 25–75) as appropriate. Based on the data distribution, differences in parameters between the in- and excluded patients were tested by the t-test or Wilcoxon rank sum test. Spearman’s rank correlation coefficient was used to evaluate the correlation of LMS with lung function parameters, HRQoL scores, and laboratory findings; the correlation coefficient was expressed as ρ. Indicators that showed significantly correlation were used as potential covariates in the later multiple linear regression analysis. The stepwise regression algorithm was used for multiple linear regression analysis to further test the relationship between the total LMS in large airways and many other variables. Body mass index (BMI), partial pressure of carbon dioxide (PaCO2), FVC, FVC % predicted, FEV1, FEV1% predicted, FEV1/FVC, peak expiratory flow (PEF), forced expiratory flow at 25% to 75% of the FVC (FEF25%-75%), mMRC grade, SGRQ score, CAT score, and EXACT score were included in the model as covariates. A two-sided P-value less than 0.05 was considered statistically significant. In addition, in the correlation analysis, Bonferroni correction was applied to re-determine the α value. IBM SPSS Statistics 13.0 (IBM Corporation, Armonk, NY, USA) was used to process and analyze all data.
Ethics Statement
We carried out this study in accordance with the Declaration of Helsinki and with approval from the Ethics Committee of Meizhou People’s Hospital (approval number: 2020-C-07). Written informed consent was obtained from all participants.
Results
Clinical characteristics of patients are summarized in Table 1, and show that there was no statistically significant difference between the included and excluded patients (P>0.05). Of the 74 included patients, 73 (98.7%) were male. The average age was 68.2±6.3 years. Bacterial culture of the bronchoalveolar lavage fluid (BALF) and/or sputum revealed positive findings in four patients; two patients tested positive for Pseudomonas aeruginosa, while the remaining two patients tested positive for Klebsiella pneumoniae. The serum procalcitonin (PCT) level was >0.25 μg/L in two patients. During hospitalization, none of patients died or received invasive mechanical ventilation, one patient was admitted to the ICU, and five patients received noninvasive positive pressure ventilation (NPPV). The median duration of hospital stay was 5.0 (IQR 25–75, 5.0–7.0).
 |
Table 1 Comparison Between Included and Excluded Patients |
The median total LMS was 15.0 (IQR 25–75, 12.0–21.0). In at least one site, 58 (78.4%) patients had luminal mucus plugging — score of 3 for the corresponding site. Results of postbronchodilator lung function are presented in Table 2. The median FEF25%-75% and FEV1% predicted, reflecting small airway disease, were 0.42 L/sec (IQR 25–75, 0.30–0.62 L/sec) and 39.6 (IQR 25–75, 29.8–52.4), respectively.
 |
Table 2 Postbronchodilator Lung Function of 74 COPD Patients |
Relationship Between Total LMS and Other Parameters
Table 3 demonstrates results of the correlation analysis of total LMS with lung function parameters and HRQoL scores (CAT, mMRC, EXACT, SGRQ). Among demographic characteristics, BMI, hospital stay, and presence of chronic bronchitis were weakly correlated with total LMS (ρ=−0.269, P=0.020; ρ=0.257, P=0.027; and ρ=0.300, P=0.009, respectively), while age and amount of smoking were not significantly correlated with total LMS.
 |
Table 3 Relationship Between Total Luminal Mucus Score and Other Parameters |
Scores of all four questionnaires (CAT, mMRC, EXACT and SGRQ) were significantly positively correlated with total LMS, among which correlation coefficients between total LMS and scores of mMRC and SGRQ were relatively high (ρ=0.527, P<0.001; ρ=0.441, P<0.001, respectively) (Figure 3). Parameters of postbronchodilator lung function were significantly negatively correlated with total LMS, and among them, FEF25%-75% and FEV1% predicted showed strong correlation with total LMS (ρ=−0.518, P<0.001; ρ=−0.498, P<0.001, respectively) (Figure 3). Of these laboratory findings, only PaCO2 was weakly positively correlated with total LMS (ρ=0.273, P=0.018), while serum CRP levels and counts of neutrophils, lymphocytes, eosinophils showed no correlation with total LMS.
In order to reduce the risk of type I errors in the correlation analysis of multiple indicators, Bonferroni correction was used to determine a stricter α level of 0.0023 (0.05/22), which was adopted to reassess the correlations between total LMS and other parameters. At the new test level, total LMS showed significant correlation with mMRC, EXACT, SGRQ and spirometric parameters.
Relationship of LMS in Various Lobes with FEV1% Predicted and mMRC Grade
The relationship of LMS in various lobes with FEV1% predicted and mMRC grade is presented in Table 4. Nine lobes were examined, including the right upper lobe bronchus (RULB), right middle lobe bronchus (RMLB), right lower lobe bronchus (RLLB), left upper division bronchus (LUDB), lingular bronchus, left lower lobe bronchus (LLLB), bilateral upper lobe bronchi (RULB and LUDB), RMLB and lingular bronchus, and bilateral lower lobe bronchi (RLLB and LLLB). Except LMS in the RLLB and RULB, LMS in all other lobes was significantly and positively correlated with FEV1% predicted. With regard to the mMRC grade, with which LMS in the all other lobes except the RLLB showed a significant correlation. At the stricter α level of 0.0027 (0.05/18), which was redetermined by Bonferroni correction, LMS in the LUDB and bilateral ULB was significantly correlated with the FEV1% predicted; LMS in the RULB, RMLB, LUDB, lingular bronchus, RMLB and lingular bronchus, and bilateral ULB was significantly correlated with mMRC grade.
 |
Table 4 Relationship of Luminal Mucus Score in Various Lobes with FEV1% Predicted and mMRC Grade |
Stepwise Multiple Regression Analysis
Stepwise multiple linear regression was performed to identify factors that could be used to predict luminal mucus. In the final regression model, mMRC score and FEV1% predicted were significant factors (R2=0.348, F=18.960, P<0.001). Results of multiple linear regression are shown in Table 5.
 |
Table 5 Stepwise Multiple Linear Regression Analysis for Luminal Mucus Scorea |
Discussion
The current study examined the relationship between bronchoscopy-identified LMS of inpatients and indicators reflecting the severity of AECOPD, including lung function, HRQoL assessment, and laboratory findings. Our results show that LMS was associated with reduced lung function and worse HRQoL of patients with severe AECOPD. Thus, this study provides a new perspective of luminal mucus in large airways of the disease.
There are several advantages of our research. First, previous studies mainly focused on small airways rather than large ones. Secondly, previous studies paid more attention to the airway mucus in stable COPD, while we focused on acute exacerbation, characterized by significant increased volume in sputum expectoration. Thirdly, it is generally believed that CT signs of “luminal plug” reflects “mucus plug” in large airways, but this remains to be confirmed. We took advantage of bronchoscopy to score luminal mucus and obtain direct evidence regarding this question. Last but not least, numerous clinical parameters of AECOPD, such as lung function, four different questionnaires for HRQoL, and laboratory indicators, were included in order to obtain more representative and convincing data.
We found that 78.4% of subjects with AECOPD had mucus plugging in at least one site, a higher percentage than for patients with stable COPD (25%),3 or severe asthma (58–67%).14,15 Besides, in patients with AECOPD, their airway mucus increased with reduced lung function, which is consistent with results of recent studies. In patients with stable COPD3 or severe asthma,14 CT scan-identified luminal plugging in large airways (diameter > 2 mm) was associated with lower FEV1% predicted. It is widely acknowledged that the accumulation of mucus in small airways is closely related to the severity of COPD.2 As a matter of fact, mucus-producing glands are mainly distributed in large airways,1 and storage of mucin is significantly increased in the epithelial cells of large airways in smokers with airflow limitation.16 In addition, some studies suggest that the mucus in some small airways contains relatively fewer cells. Therefore, this mucus could have been produced in large airways and aspirated into small airways.17 Patients with COPD often experienced skeletal muscle dysfunction, diaphragmatic dysfunction, and impaired ciliary function,18,19 which frequently worsen during acute exacerbations. Under combined effects of these factors, luminal mucus cannot be effectively cleared, and stays in large airways which served as a common channel for expulsion of mucus. These factors may partially explain the relationship between the LMS in large airways and the airflow obstruction of AECOPD, but the exact mechanism remains to be elucidated.
Chronic cough and sputum production, also known as chronic bronchitis, are common symptoms and key components of COPD.18 Studies that have focused on the potential impact of chronic bronchitis in COPD called “chronic mucus hypersecretion.”20,21 Our results showed that chronic bronchitis was weakly positively correlated with total LMS. However, in two previous studies, luminal plugging was not related to chronic cough and phlegm in stable COPD and severe asthma.3,14 A possible explanation for these inconsistent results is that larger airways that can be observed by bronchoscopy may have more cough receptors.22
COPD patients with a higher luminal mucus score in large airways has a higher SGRQ score that represented a worse HRQoL.3 In our study, not only the SGRQ score, but also the CAT score, mMRC score and EXACT score were all significantly correlated with total LMS, further demonstrating the relationship between the mucus in large airways and HRQoL.
The correlation analysis of LMS in various lobes and the FEV1% predicted and mMRC grade indicated that LMS in the upper lobe bronchi had a greater correlation than that in the lower lobe bronchi (Table 4). Our study cannot explain the results because numerous factors and mechanisms affected the regional distribution of ventilation and perfusion in the lung.23 Well-designed studies are needed to explore potential effects of luminal mucus in different lobes on lung function.
Patients with exacerbation had a significantly higher CRP level and leukocyte count in the sputum than when in a stable state, and serum CRP is positively correlated with sputum leukocyte count, which suggests that the systemic inflammation of AECOPD is correlated with lower airway inflammation.24 Nevertheless, our findings indicate that total LMS in large airways does not show significant correlation with counts of neutrophil, lymphocyte, eosinophil, fibrinogen, or CRP, and this is puzzling. Therefore, in order to reach more definitive conclusions, it will be necessary to further study the relationship between systemic inflammation, lower airway inflammation, and luminal mucus in large airways in the future.
EXACT is widely used to clinically assess the severity and duration of AECOPD,25 and also is a sensitive, reliable tool for measuring changes in the exacerbation condition during recovery.26 CAT scores increased significantly at exacerbation compared to those at baseline and reflected exacerbation severity as determined by lung function and exacerbation length, which indicated that the CAT provides a reliable score of exacerbation severity.4 In our study, total LMS was significantly correlated with the CAT score and EXACT score, which suggests that total LMS may partially reflect severity of AECOPD.
Our results may have therapeutic implications. For AECOPD27 patients undergoing tracheal intubation and children suffering acute asthma28 with respiratory failure, bronchoscopic sputum suction showed clinical benefits. Furthermore, for the COPD patients with acute respiratory failure who were candidates for conventional mechanical ventilation because of hypercapnic encephalopathy and inability to clear copious secretions, bronchoscopic sputum suction combined with non-invasive positive pressure ventilation enabled 80% (12/15) of them to avoid tracheal intubation.29 We found that 78.4% of inpatients had luminal mucus plugging in at least one site and also that total LMS was proportional to PaCO2, which indicates that early application of airway clearance techniques, including bronchoscopic sputum suction, might be effective for AECOPD inpatients.
Limitations
There are some limitations in our study. First of all, this was a cross-sectional study without follow-up; therefore, causal relationships could not be established and changes in parameters in stable COPD are not clear. Therefore, further cohort studies are required. Secondly, all subjects in this study were inpatients who were within the “severe” classification of acute exacerbation according to GOLD 2021,18 thus the conclusion of this study still need to be verified in various grades of the disease. Lastly, bronchoscopy is invasive, which limits its widely clinical use for luminal mucus scoring.
Conclusion
In AECOPD inpatients, mucus frequently occludes the lumen of large airways, and LMS is significantly associated with reduced lung function and worse HRQoL. If replicated, our results suggest that LMS may be a direct indicator for evaluating the severity of AECOPD.
Abbreviations
COPD, chronic obstructive pulmonary disease; AECOPD, acute exacerbation of COPD; SGRQ, St. George Respiratory Questionnaire; mMRC, modified Medical Research Council dyspnea scale; CAT, COPD Assessment Test; EXACT, EXAcerbation of Chronic pulmonary disease Tool; HRQoL, health-related quality of life; FEF25%–75%, Forced expiratory flow at 25% to 75% of the FVC; FEV1, forced expiratory volume in 1 second; LMS, luminal mucus score; FVC, forced vital capacity; CRP, C-reactive protein; LFT, lung function test; BMI, Body mass index; PaCO2, partial pressure of carbon dioxide; PEF, peak expiratory flow; RULB, right upper lobe bronchus; RMLB, right middle lobe bronchus; RLLB, right lower lobe bronchus; LUDB, left upper division bronchus; LLLB, left lower lobe bronchus; ULB, upper lobe bronchi; LLB, lower lobe bronchi.
Data Sharing Statement
Datasets used and/or analyzed in the current study are available from the corresponding author (Qian-Yun Zhang) upon reasonable request.
Acknowledgments
Authors would like to thank the participants and their families involved in this study. We would like to thank staff of the Department of Pulmonary and Critical Care Medicine, Meizhou People’s Hospital for their support.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; had drafted or written, or substantially revised or critically reviewed the article; had agreed on the journal to which the article will be submitted; gave final approval of the version to be published; and agree to take responsibility and be accountable for the contents of the article.
Disclosure
Dr Yang report grants from Health Commission of Guangdong Province, during the conduct of the study. Dr Yang declared that he has received Medical Scientific Research Foundation of Guangdong Province, China (No.A2021410). The other authors report no conflicts of interest in this work.
References
1. Fahy JV, Dickey BF. Airway mucus function and dysfunction. N Engl J Med. 2010;363(23):2233–2247. doi:10.1056/NEJMra0910061
2. Hogg JC, Chu F, Utokaparch S, et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(26):2645–2653. doi:10.1056/NEJMoa032158
3. Okajima Y, Come CE, Nardelli P, et al. Luminal plugging on chest CT scan: association with lung function, quality of life, and COPD clinical phenotypes. Chest. 2020;158(1):121–130. doi:10.1016/j.chest.2019.12.046
4. Mackay AJ, Donaldson GC, Patel AR, Jones PW, Hurst JR, Wedzicha JA. Usefulness of the chronic obstructive pulmonary disease assessment test to evaluate severity of COPD exacerbations. Am J Respir Crit Care Med. 2012;185(11):1218–1224. doi:10.1164/rccm.201110-1843OC
5. Thompson AB, Daughton D, Robbins RA, Ghafouri MA, Oehlerking M, Rennard SI. Intraluminal airway inflammation in chronic bronchitis. Characterization and correlation with clinical parameters. Am Rev Respir Dis. 1989;140(6):1527–1537. doi:10.1164/ajrccm/140.6.1527
6. Rabe KF, Martinez FJ, Ferguson GT, et al. Triple inhaled therapy at two glucocorticoid doses in moderate-to-very-severe COPD. N Engl J Med. 2020;383(1):35–48. doi:10.1056/NEJMoa1916046
7. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for prevention, diagnosis and management of COPD. 2020. Available from: https://goldcopd.org/gold-reports/.
8. Interventional Pulmonology Group of The Chinese Thoracic Society CMA. [Guideline for diagnostic flexible bronchoscopy in adults (2019)]. Chin J Tuberculosis Respir Dis. 2019;42(8):573–590. doi:10.3760/cma.j.issn.1001-0939.2019.08.005
9. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. doi:10.1183/09031936.05.00034805
10. Burgel PR, Nesme-Meyer P, Chanez P, et al. Cough and sputum production are associated with frequent exacerbations and hospitalizations in COPD subjects. Chest. 2009;135(4):975–982. doi:10.1378/chest.08-2062
11. Han MK, Muellerova H, Curran-Everett D, et al. GOLD 2011 disease severity classification in COPDGene: a prospective cohort study. Lancet Respir Med. 2013;1(1):43–50. doi:10.1016/S2213-2600(12)70044-9
12. Weatherall M, Marsh S, Shirtcliffe P, Williams M, Travers J, Beasley R. Quality of life measured by the St George’s Respiratory Questionnaire and spirometry. Eur Respir J. 2009;33(5):1025–1030. doi:10.1183/09031936.00116808
13. Mackay AJ, Donaldson GC, Patel AR, Singh R, Kowlessar B, Wedzicha JA. Detection and severity grading of COPD exacerbations using the exacerbations of chronic pulmonary disease tool (EXACT). Eur Respir J. 2014;43(3):735–744. doi:10.1183/09031936.00110913
14. Dunican EM, Elicker BM, Gierada DS, et al. Mucus plugs in patients with asthma linked to eosinophilia and airflow obstruction. J Clin Invest. 2018;128(3):997–1009. doi:10.1172/JCI95693
15. Svenningsen S, Haider E, Boylan C, et al. CT and functional MRI to evaluate airway mucus in severe asthma. Chest. 2019;155(6):1178–1189. doi:10.1016/j.chest.2019.02.403
16. Innes AL, Woodruff PG, Ferrando RE, et al. Epithelial mucin stores are increased in the large airways of smokers with airflow obstruction. Chest. 2006;130(4):1102–1108. doi:10.1378/chest.130.4.1102
17. Hogg JC. Pathophysiology of airflow limitation in chronic obstructive pulmonary disease. The Lancet. 2004;364(9435):709–721. doi:10.1016/S0140-6736(04)16900-6
18. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for prevention, diagnosis and management of COPD. 2021. Available from: https://goldcopd.org/gold-reports/.
19. Kim V, Evans CM, Dickey BF. Dawn of a new era in the diagnosis and treatment of airway mucus dysfunction. Am J Respir Crit Care Med. 2019;199(2):133–134. doi:10.1164/rccm.201808-1444ED
20. Wedzicha JA. Airway mucins in chronic obstructive pulmonary disease. N Engl J Med. 2017;377(10):986–987. doi:10.1056/NEJMe1707210
21. Shen Y, Huang S, Kang J, et al. Management of airway mucus hypersecretion in chronic airway inflammatory disease: chinese expert consensus (English edition). Int J Chron Obstruct Pulmon Dis. 2018;13:399–407. doi:10.2147/COPD.S144312
22. Burgel PR, Martin C. Mucus hypersecretion in COPD: should we only rely on symptoms? Eur Respir Rev. 2010;19(116):94–96. doi:10.1183/09059180.00004410
23. Bryan AC, Bentivoglio LG, Beerel F, Macleish H, Zidulka A, Bates DV. Factors affecting regional distribution of ventilation and perfusion in the lung. J Appl Physiol. 1964;19:395–402. doi:10.1152/jappl.1964.19.3.395
24. Hurst JR, Perera WR, Wilkinson TM, Donaldson GC, Wedzicha JA. Systemic and upper and lower airway inflammation at exacerbation of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2006;173(1):71–78. doi:10.1164/rccm.200505-704OC
25. Leidy NK, Wilcox TK, Jones PW, et al. Development of the EXAcerbations of Chronic Obstructive Pulmonary Disease Tool (EXACT): a patient-reported outcome (PRO) measure. Value Health. 2010;13(8):965–975. doi:10.1111/j.1524-4733.2010.00772.x
26. Leidy NK, Wilcox TK, Jones PW, Roberts L, Powers JH, Sethi S. Standardizing measurement of chronic obstructive pulmonary disease exacerbations. Reliability and validity of a patient-reported diary. Am J Respir Crit Care Med. 2011;183(3):323–329. doi:10.1164/rccm.201005-0762OC
27. Qiao Z, Yu J, Yu K, Zhang M. The benefit of daily sputum suction via bronchoscopy in patients of chronic obstructive pulmonary disease with ventilators: a randomized controlled trial. Medicine. 2018;97(31):e11631. doi:10.1097/MD.0000000000011631
28. Maggi JC, Nussbaum E, Babbitt C, Maggi FE, Randhawa I. Pediatric fiberoptic bronchoscopy as adjunctive therapy in acute asthma with respiratory failure. Pediatr Pulmonol. 2012;47(12):1180–1184. doi:10.1002/ppul.22591
29. Scala R, Naldi M, Maccari U. Early fiberoptic bronchoscopy during non-invasive ventilation in patients with decompensated chronic obstructive pulmonary disease due to community-acquired-pneumonia. Crit Care. 2010;14(2):R80. doi:10.1186/cc8993
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.