Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 11 » Issue 1
COPD assessment test score and serum C-reactive protein level in stable COPD patients
Authors Kang HK, Kim K, Lee H , Jeong BH, Koh WJ, Park HY
Received 26 July 2016
Accepted for publication 25 October 2016
Published 8 December 2016 Volume 2016:11(1) Pages 3137—3143
DOI https://doi.org/10.2147/COPD.S118153
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Hyung Koo Kang,1,2,* Kang Kim,1,* Hyun Lee,1 Byeong-Ho Jeong,1 Won-Jung Koh,1 Hye Yun Park1
1Division of Pulmonary and Critical Care Medicine, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, 2Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, Ilsan Paik Hospital, Inje University School of Medicine, Goyang, Gyeonggi, South Korea
*These authors contributed equally to this work
Background: An eight-item questionnaire of the COPD assessment test (CAT) is widely used to quantify the impact of COPD on the patient’s health status. C-reactive protein (CRP) is associated with disease severity and adverse health outcomes of patients with COPD. This study aimed to evaluate the relationship between CAT score and serum CRP levels in stable COPD patients.
Methods: We evaluated the medical records of 226 patients with CAT and serum CRP measured within a week at Samsung Medical Center between October 2013 and October 2015.
Results: Serum CRP levels had a significantly positive relationship with CAT score (Spearman’s r=0.20, P=0.003). Patients with elevated serum CRP levels (>0.3 mg/dL) were significantly more likely to have CAT scores of ≥14. The adjusted odds ratio for elevated serum CRP levels in total CAT score was 1.06 (95% confidence interval, 1.02–1.09). Among CAT components, cough (adjusted P=0.005), phlegm (adjusted P=0.001), breathlessness going up hills/stairs (adjusted P=0.005), low confidence leaving home (adjusted P=0.002), and feeling low in energy (adjusted P=0.019) were independently associated with elevated serum CRP levels.
Conclusion: In stable COPD patients, serum CRP levels were independently associated with total CAT score and CAT components related to respiratory symptoms, confidence leaving home, and energy.
Keywords: chronic obstructive pulmonary disease assessment test, C-reactive protein, quality of life, inflammation
Background
COPD is a chronic inflammatory lung disease characterized by progressive airflow limitation and parenchymal destruction.1 It often causes remarkable symptom burden, including chronic cough, sputum production, breathlessness, and exercise intolerance.1–3 The Global Initiative for Chronic Obstructive Lung Disease guideline recommends multiple symptomatic assessments rather than just measuring breathlessness with lung function.1 Among several questionnaires measuring health-related quality of life, the COPD assessment test (CAT) is widely used in routine practice.4–6 It is an easy and simple measurement with an eight-item questionnaire, which is designed to quantify the impact of COPD symptoms on the patient health status with scores of 0–40.4 In clinically stable COPD patients, the CAT is also closely related to the St George’s Respiratory Questionnaire.7,8
C-reactive protein (CRP) is a representative systemic biomarker reflecting the total systemic burden of inflammation in individuals.9 Serum CRP is elevated in stable COPD, and it correlates with disease severity and adverse health outcomes of patients with mild-to-moderate COPD.10 The Copenhagen City Heart Study and Copenhagen General Population Study showed that increased levels of CRP and other systemic biomarkers were associated with increased risk of COPD exacerbations.11 CAT score is also associated with patients at high risk of exacerbation and changes significantly during and after exacerbations.6,12–14 Moreover, an elevated CAT score at exacerbation reflects exacerbation severity.6 Regarding the relationship between CAT score and CRP, change in CAT score from baseline to exacerbation onset was positively correlated to change in serum CRP.6,13 However, the relationship between the CAT score and the serum CRP levels has not been adequately investigated in stable COPD patients. Thus, we sought to determine the association between CAT score and serum CRP levels in stable COPD patients.
Methods
Subjects
The medical records of 246 COPD patients who took the CAT and had serum CRP levels within 1 week from CAT, at the Samsung Medical Center outpatient clinic from October 2013 to October 2015, were retrospectively reviewed. Among them, 20 patients with moderate or severe exacerbation at CAT and serum CRP measurements were excluded, and a total of 226 stable COPD patients were included in the present study (Figure 1). The Institutional Review Board of Samsung Medical Center approved this study and waived the requirement for informed consent due to the retrospective nature of the study (IRB No. 2015-12-098).
  | Figure 1 Flow chart for study enrollment. |
Measurements
Demographic features, smoking history, comorbidities, and current medications, including inhalers, were obtained by reviewing available medical records on the day patients performed the CAT. Comorbidities including diabetes mellitus, hypertension, cardiovascular disease (CVD), asthma, tuberculosis destroyed lung, and bronchiectasis were reviewed. Patients with CVD were defined as patients who had been diagnosed with myocardial infarction, angina pectoris, stroke, and congestive heart failure, including cor pulmonale. Patients with stable COPD were defined as those without moderate or severe exacerbation when CAT score and serum CRP measurements were performed. The moderate or severe exacerbations were defined as a worsening of symptoms that required oral corticosteroids and/or antibiotics and/or hospitalization.15
Spirometry was performed according to the joint American Thoracic Society and European Respiratory Society guidelines.16 Absolute values of forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) were obtained, and the percentage of predicted values (% pred) for FEV1 and FVC were calculated from equations obtained in a representative Korean sample.17
The CAT consists of eight questions for cough, phlegm, chest tightness, breathlessness going up hills/stairs, activity limitations at home, confidence leaving home, sleep and energy.7 Each factor is presented on a semantic 6-point (0–5) differential scale, providing a total score of 40.4
The value of serum CRP concentration measured within 1 week before or after CAT was selected for analysis. Serum CRP levels were measured using immunoturbidimetric assays (CRPL3, Roche Diagnostics, Indianapolis, IN, USA), and the analytical measurement range was 0.03–35 mg/dL. The elevated serum CRP levels were defined when serum CRP levels were >0.3 mg/dL,18 which is associated with increased risk of CVD.19,20
When subjects underwent more than one CAT and CRP during the study period, data from only the first measurement were used in this analysis.
Statistical analysis
Baseline characteristics are presented as medians and interquartile ranges (IQRs) for continuous variables and as numbers (%) for categorical variables. Spearman’s correlation and a linear model were used to assess the relationship between CAT score and serum CRP levels. Serum CRP values were dichotomized based on the upper limits of normal range (0.3 mg/dL) to evaluate the relationship between CAT score and increased serum CRP levels. The multiple logistic regression analyses were performed on age, sex, variables with P<0.20 based on univariate results (Table S1) or variables that were generally considered to be associated with CAT or elevated serum CRP levels. The number of comorbidities, including underlying lung disease, was also included as serum CRP levels can be elevated in chronic inflammation caused by noninfectious inflammation disorders. To determine the CAT score associated with elevated serum CRP levels (>0.3 mg/dL), CAT scores were dichotomized from 6 points to 24 points with 2-point intervals. Pearson’s chi-square test was used for these dichotomous variables. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA).
Results
Clinical characteristics of subjects
The clinical characteristics of study participants are summarized in Table 1. The study group included 198 males (87.6%) and 28 females (12.4%), with a median age of 70 years (IQR, 63–75 years). The median body mass index (BMI) was 22.9 kg/m2 (IQR, 20.3–25.0 kg/m2). Thirty-three patients (14.6%) were lifetime never smokers. Median serum CRP level was 0.18 mg/dL (IQR, 0.07–0.57 mg/dL), and 88 patients (38.9%) had serum CRP levels >0.3 mg/dL. The median FEV1 (% pred) was 56% (IQR, 41%–69%), and 93 patients (41.2%) had FEV1 (% pred) <50%. The median number of comorbidities was 1 (IQR, 0–2). Median CAT score was 16 points (IQR, 9–22). The median scales of CAT components are shown in Figure 2.
Relationship between CAT score and serum CRP levels
Correlations between CAT component and serum CRP levels are shown in Table 2. The serum CRP levels had a significant positive relationship with total CAT score (Spearman’s r=0.199, P=0.003). Among CAT components, serum CRP levels were significantly correlated with cough (Spearman’s r=0.153, P=0.021), phlegm (Spearman’s r=0.184, P=0.006), breathlessness going up hills/stairs (Spearman’s r=0.161, P=0.015), low confidence leaving home (Spearman’s r=0.254, P<0.001), and feeling low in energy (Spearman’s r=0.180, P=0.007). There were no significant relationships between serum CRP levels and chest tightness, activity limitations at home, or sleep (Table 3).
Relationship between CAT score and elevated serum CRP levels
As shown in Table 3, total CAT score was an independent factor associated with having elevated serum CRP levels after adjustment for covariates, including age, sex, BMI, smoking status, FEV1 (% pred), and number of comorbidities (adjusted odds ratio [aOR], 1.06; 95% confidence interval [CI], 1.02–1.09). The aORs of cough, phlegm, breathlessness, low confidence leaving home, and feeling low in energy for elevated serum CRP levels were 1.38 (95% CI, 1.10–1.72), 1.38 (95% CI, 1.13–1.68), 1.35 (95% CI, 1.09–1.66), 1.29 (95% CI, 1.09–1.52), and 1.25 (95% CI, 1.04–1.50), respectively. Regarding relationship of elevated serum CRP levels with CAT component, there were no significant relationships between elevated serum CRP levels and chest tightness, activity limitations at home, or sleep.
The points of total CAT score associated with elevated serum CRP levels
The CAT scores were dichotomized from 6 points to 24 points with 2-point intervals. Patients with elevated serum CRP levels significantly increased from 14 points to 24 points compared with the other group (serum CRP level ≤0.3 mg/dL). The P-values for elevated serum CRP levels with 14, 16, 18, 20, 22, and 24 points were 0.012, 0.004, 0.003, 0.034, 0.003, and <0.001, respectively. However, there were no significant relationships of elevated serum CRP levels with CAT scores of 6, 8, 10, and 12 points (Figure 3).
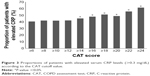  | Figure 3 Proportions of patients with elevated serum CRP levels (>0.3 mg/dL) according to the CAT cutoff value. |
Discussion
In the present study, we found that serum CRP levels were independently associated with total CAT score in stable COPD patients. Among CAT components, this positive relationship was shown with cough, phlegm, breathlessness going up hills/stairs, low confidence leaving home, and feeling low in energy. The proportion of patients with elevated serum CRP levels significantly increased with 14 points of total CAT, suggesting that CAT score is a useful indicator to predict systemic inflammation in stable COPD patients.
CAT is a validated health status questionnaire designed to quantify the impact of COPD symptoms.4 Previous studies showed that this instrument provides reliable scores for predicting COPD exacerbation and severity, suggesting the CAT as a potential tool for the stratification of patients who are at risk for exacerbations.6,13,14 Systemic inflammation is measured by plasma fibrinogen and serum CRP, which are increased in COPD and aggravated during exacerbations.11 Accordingly, several studies demonstrated that CRP is elevated corresponding to increased CAT score during exacerbations.6,13 In stable COPD, our study showed that CAT score is also independently associated with elevated serum CRP levels even after adjusting for confounding factors, although this statistical relationship is weak. Elevated CRP levels can occur in association with chronic inflammation caused by noninfectious inflammation disorders, such as CVD,9,18,21 and patients with COPD often have significant extrapulmonary comorbidities contributing to symptoms.1 Thus, the number of comorbidities was additionally adjusted, and we found that the positive relationship between CAT score and serum CRP levels was preserved in stable COPD patients.
In particular, there are significantly more patients with elevated serum CRP levels, when CAT scores are ≥14. According to the guidelines, a CAT score of ≥10 or modified Medical Research Council (mMRC) grade ≥2 is categorized as the “more symptomatic group”.1 However, there is increasing recognition that a CAT cutoff value of 10 could overestimate the symptom severity compared with an mMRC cutoff value of 2.22,23 Recent data using the Korean COPD Subgroup Study (KOCOSS) cohort also reported that a CAT score of 15 is concordant with mMRC grade ≥2.24 This is in line with our findings showing a significant relationship between elevated serum CRP levels and CAT scores of ≥14.
In addition to total CAT score, we showed that certain CAT components related to respiratory symptoms (cough, phlegm, breathlessness going up hills/stairs), confidence leaving home, and energy were associated with increased levels of serum CRP. This is consistent with the characteristics of COPD, which is associated with an enhanced chronic inflammatory response in the airways, parenchyma, and pulmonary vasculature,25,26 and with low-grade systemic inflammation with increased levels of systemic inflammatory markers, such as CRP, fibrinogen, leukocytes, and TNF-α. With interlinking relationships, the present study suggests that increased CAT score can reflect systemic inflammation as well as inflamed respiratory symptoms in stable COPD.27
Our study has several limitations. First, given the observational nature of the study, there is always the possibility that selection bias or confounding might have influenced our findings. Second, as blood samples were not collected for this study, we could not evaluate other systemic biomarkers, such as fibrinogen, interleukin-6, and TNF-α. Further studies investigating the relationships of those systemic biomarkers with CAT scores would be informative to reinforce the relationship between inflammation and CAT score in stable COPD patients. Third, because this is a cross-sectional study, further investigation is needed to determine intraindividual variation of the association between CAT scores and serum CRP levels over time.
Conclusion
In conclusion, this study demonstrated an independent association between CAT score and serum CRP levels in stable COPD patients. This relationship was most obvious in CAT components related to respiratory symptoms, confidence leaving home, and energy. The results suggest that CAT score can be a reliable indicator that incorporate both lung and systemic inflammation in stable COPD patients.
Acknowledgments
The authors would like to thank Soohyun Kim for electronic medical record development for this study. This study was supported by the Samsung Medical Center Foundation for Medical Research (SMO1140211).
Disclosure
The authors report no conflicts of interest in this work.
References
Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease; 2016. Available from: http://www.goldcopd.org. Accessed May 1, 2016. | ||
Kim V, Criner GJ. The chronic bronchitis phenotype in chronic obstructive pulmonary disease: features and implications. Curr Opin Pulm Med. 2015;21(2):133–141. | ||
O’Donnell DE, Lam M, Webb KA. Measurement of symptoms, lung hyperinflation, and endurance during exercise in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;158(5 Pt 1):1557–1565. | ||
Jones PW, Harding G, Berry P, Wiklund I, Chen WH, Kline Leidy N. Development and first validation of the COPD assessment test. Eur Respir J. 2009;34(3):648–654. | ||
Jones PW. COPD assessment test – rationale, development, validation and performance. COPD. 2013;10(2):269–271. | ||
Mackay AJ, Donaldson GC, Patel AR, Jones PW, Hurst JR, Wedzicha JA. Usefulness of the Chronic Obstructive Pulmonary Disease Assessment Test to evaluate severity of COPD exacerbations. Am J Respir Crit Care Med. 2012;185(11):1218–1224. | ||
Jones PW, Brusselle G, Dal Negro RW, et al. Properties of the COPD assessment test in a cross-sectional European study. Eur Respir J. 2011;38(1):29–35. | ||
Hwang YI, Jung KS, Lim SY, Lee YS, Kwon NH. A validation study for the Korean version of chronic obstructive pulmonary disease assessment test (CAT). Tuberc Respir Dis (Seoul). 2013;74(6):256–263. | ||
Black S, Kushner I, Samols D. C-reactive protein. J Biol Chem. 2004;279(47):48487–48490. | ||
Man SF, Connett JE, Anthonisen NR, Wise RA, Tashkin DP, Sin DD. C-reactive protein and mortality in mild to moderate chronic obstructive pulmonary disease. Thorax. 2006;61(10):849–853. | ||
Thomsen M, Ingebrigtsen TS, Marott JL, et al. Inflammatory biomarkers and exacerbations in chronic obstructive pulmonary disease. JAMA. 2013;309(22):2353–2361. | ||
Jones PW, Harding G, Wiklund I, et al. Tests of the responsiveness of the COPD assessment test following acute exacerbation and pulmonary rehabilitation. Chest. 2012;142(1):134–140. | ||
Tu YH, Zhang Y, Fei GH. Utility of the CAT in the therapy assessment of COPD exacerbations in China. BMC Pulm Med. 2014;14:42. | ||
Lee SD, Huang MS, Kang J, et al. The COPD assessment test (CAT) assists prediction of COPD exacerbations in high-risk patients. Respir Med. 2014;108(4):600–608. | ||
Rodriguez-Roisin R. Toward a consensus definition for COPD exacerbations. Chest. 2000;117(5 Suppl 2):398S–401S. | ||
Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. | ||
Choi JK, Paek DM, Lee JO. Normal predictive values of spirometry in Korean population. Tuberc Respir Dis (Seoul). 2005;58:230–242. | ||
Kushner I, Rzewnicki D, Samols D. What does minor elevation of C-reactive protein signify? Am J Med. 2006;119(2):166.e17–e28. | ||
Pearson TA, Mensah GA, Alexander RW, et al. Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation. 2003;107(3):499–511. | ||
Cushman M, Arnold AM, Psaty BM, et al. C-reactive protein and the 10-year incidence of coronary heart disease in older men and women: the cardiovascular health study. Circulation. 2005;112(1):25–31. | ||
Libby P, Ridker PM. Inflammation and atherosclerosis: role of C-reactive protein in risk assessment. Am J Med. 2004;116(Suppl 6A):9S–16S. | ||
Jones PW, Adamek L, Nadeau G, Banik N. Comparisons of health status scores with MRC grades in COPD: implications for the GOLD 2011 classification. Eur Respir J. 2013;42(3):647–654. | ||
Lee CH, Lee J, Park YS, et al. Chronic obstructive pulmonary disease (COPD) assessment test scores corresponding to modified Medical Research Council grades among COPD patients. Korean J Intern Med. 2015;30(5):629–637. | ||
Rhee CK, Kim JW, Hwang YI, et al. Discrepancies between modified Medical Research Council dyspnea score and COPD assessment test score in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2015;10:1623–1631. | ||
Cosio MG, Saetta M, Agusti A. Immunologic aspects of chronic obstructive pulmonary disease. N Engl J Med. 2009;360(23):2445–2454. | ||
Hogg JC, Chu F, Utokaparch S, et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(26):2645–2653. | ||
Gan WQ, Man SF, Senthilselvan A, Sin DD. Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax. 2004;59(7):574–580. |
Supplementary material
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





