Back to Journals » Clinical Ophthalmology » Volume 17
Continuous Eplerenone Treatment in Chronic Central Serous Chorioretinopathy: Long-Term Results from a Pilot Study
Authors Boscia G, Viggiano P , Marzulli F, Grassi MO, Puzo P , Dore S, Pinna A , Alessio G , Boscia F
Received 17 March 2023
Accepted for publication 1 June 2023
Published 17 July 2023 Volume 2023:17 Pages 2003—2012
DOI https://doi.org/10.2147/OPTH.S411094
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Giacomo Boscia,1,* Pasquale Viggiano,1,* Federica Marzulli,1 Maria Oliva Grassi,1 Pasquale Puzo,1 Stefano Dore,2 Antonio Pinna,2 Giovanni Alessio,1 Francesco Boscia1
1Department of Translational Biomedicine Neuroscience, University of Bari “Aldo Moro”, Bari, Italy; 2Department of Medicine, Surgery and Pharmacy, University of Sassari, Sassari, Italy
*These authors contributed equally to this work
Correspondence: Pasquale Viggiano, Department of Translational Biomedicine Neuroscience, University of Bari “Aldo Moro”, Piazza Giulio Cesare, 11, Bari, Italy, Tel +390805593728, Email [email protected]
Purpose: To assess the long-term morpho-functional retinal and choroidal changes in chronic central serous chorioretinopathy (cCSC) pachychoroid eyes in response to continuous oral eplerenone (EPL) treatment.
Methods: This pilot study was conducted on patients with unilateral exudative cCSC. We enrolled a total of 17 exudative cCSC and 17 non-exudative fellow eyes of 17 patients. Baseline best-corrected visual acuity (BCVA) and anatomical (structural optical coherence tomography [OCT] and OCT angiography) parameters in both eyes were collected at baseline. Follow-up data were collected at 6, 12, and 48 months after initiation of EPL treatment.
Results: (i) Exudative cCSC eyes: Compared with baseline (0.34± 0.13 LogMAR), BCVA significantly improved at follow-up examinations (6 months: 0.28± 0.13 LogMAR, p=0.039; 12 months: 0.22± 0.11 LogMAR, p=0.025; 48 months: 0.21± 0.08 LogMAR, p=0.028). Furthermore, there was a significant reduction from baseline in all structural OCT parameters (subretinal fluid and subfoveal choroidal thickness [SFCT]; p< 0.05). (ii) Non-exudative fellow eyes: There was no significant change in BCVA. There was a significant reduction from baseline in SFCT and choriocapillaris flow deficit percentage (p< 0.05).
Conclusion: In this pilot study, continuous oral EPL therapy in cCSC pachychoroid eyes resulted in long-term morpho-functional improvement. The beneficial effect of EPL occurred within the first year and was maintained after four years. Based on these preliminary observations, EPL may be effective in the exudative forms of CSC.
Keywords: central serous chorioretinopathy, choriocapillaris reperfusion, OCT and OCTA
Introduction
Central serous chorioretinopathy (CSC) is a significant cause of central vision loss among individuals of working age.1 It is characterized by the accumulation of subretinal fluid (SRF) accompanied by detachment of the neuroretina.1,2 Several risk factors have been identified, including a “type A” personality cluster, corticosteroid use, stress, and pregnancy.3,4 CSC can be classified into two distinct forms: acute CSC (aCSC) and chronic CSC (cCSC).5 Acute CSC typically resolves on its own within a period of 2 to 3 months but can lead to progressive vision impairment if SRF persists. On the other hand, cCSC frequently affects both eyes (approximately 20% of cases) and often results in permanent damage to ganglion cells and the chorio-retinal layer.5,6 A revised classification for central serous chorioretinopathy (CSCR) has been proposed by Chhablani et al.7 In this updated classification, CSCR is further divided into two subcategories: simple and complex. The categorization is based on the presence of serous retinal detachment and alterations in the retinal pigment epithelium (RPE).
Despite limited high-quality evidence supporting their effectiveness, various treatments have been utilized for the management of Central Serous Chorioretinopathy (CSCR). One such treatment is Verteporfin Photodynamic Laser Therapy (vPDT), which is employed for certain patients with CSCR. Randomized controlled trials investigating the use of half-dose vPDT have demonstrated promising results in the short term.8
Although the pathogenesis of CSC is poorly understood, the majority of authors speculate that choroidal abnormalities are the primary underlying mechanism of this disease.9 The typical anatomical pattern of CSC is characterized by dilated choroidal vessels in the Haller’s layer, with a concomitant thinning of Sattler’s layer and choriocapillaris.10 The latter phenotype is referred to as “pachychoroid”, and is suggested to exert pressure on the choriocapillaris, with a consequent breakdown of the RPE-Bruch membrane complex.11 Importantly, the choroidal layer of CSC eyes is characterized by enhanced vessels permeability.2 Several authors correlated this abnormality to overstimulation of choroidal mineralocorticoids receptor (MR).5 Thus, an antagonist of MR such as Eplerenone (EPL), has been widely proposed in the treatment of CSC.12 Although its effectiveness in clinical practice is still controversial,9,13 several researchers have reported a resolution of SRF and a reduction in subfoveal choroidal thickness (SFCT).14
With the recent advent of novel techniques of ophthalmic imaging, such as optical coherence tomography (OCT) angiography (OCTA), several researchers have reported changes in the retinal and choroidal layers in different eye disorders.15 Recently, various papers have focused on the evaluation of choriocapillaris (CC) characteristics in CSC, suggesting an impaired CC flow, which might be linked to focal ischemia.16,17 For instance, Yun et al11 have suggested a potential relationship between CC flow alteration and the underlying dilatation of choroidal vessels in the fellow eyes of CSC patients.
While multiple studies have focused on choroidal morphology changes of unilateral CSC following different therapies,18,19 a recent paper by Toto et al5 has showed choroidal morphology recovery in both eyes of patients undergoing oral EPL for cCSC with monolateral SRF. However, the current literature lacks evidence regarding long-term choroidal modifications of the fellow eye in unilateral exudative cCSC in response to oral EPL therapy. Considering this, it would be important to characterize the long-term impact of MR antagonists on the choroidal status.
The purpose of this pilot study was to assess the long-term morpho-functional retinal and choroidal changes in cCSC eyes associated with pachychoroid spectrum. Structural OCT and OCTA was used to investigate the affected and non-exudative fellow eyes during continuous EPL treatment.
Methods
Study Design
This was a retrospective observational pilot study including patients with bilateral cCSC who had subretinal fluid in 1 eye. We registered a total of 17 patients with unilateral exudative chronic central serous chorioretinopathy (cCSC) and their corresponding non-exudative fellow eyes, totaling 17 eyes in each group, at the Department of Translational Biomedicine Neuroscience, University of Bari “Aldo Moro”, Italy, between August 2017 and November 2022. The current investigation was performed in compliance with the tenets of the Declaration of Helsinki for research involving human subjects and approved by the Ethical Committee of the Department of Translational Biomedicine Neuroscience, University of Bari “Aldo Moro”.
Written Informed Consent to Participate Was Signed by Each Participant
Unilateral exudative cCSC was considered as the presence of SRF for at least 6 months before therapy initiation. Diagnostic criteria for cCSC were fluorescein angiography, fundus autofluorescence, ICGA (Heidelberg Spectralis; Heidelberg Engineering, Germany) and structural OCT (DRI OCT, Triton, Topcon Corp., Tokyo, Japan). Successful treatment was considered as a complete SRF resorption. All patients were treated with 50 mg daily of eplerenone. EPL was started after approval by the patient’s general practitioner, and throughout the study duration, we closely monitored the potassium levels of the participants. While we were prepared to exclude patients if significant adverse events occurred, all patients were able to continue the EPL therapy as the observed adverse events were not deemed significant. Each patient underwent a complete ophthalmological evaluation, including measurement of best correct visual acuity (BCVA), intraocular pressure (IOP), and a dilated fundus examination. Additionally, we utilized a questionnaire on general information to investigate the presence of potential systemic risk factors. The questionnaire gathered data on patient demographics, including age and gender, as well as information on possible risk factors such as hypertension, steroid use, cigarette use, and the presence of stress. To evaluate stress levels, we inquired about stressful life events experienced by the patients within the past 12 months. The imaging assessment was performed with a swept-source OCTA (SS-OCTA) (DRI OCT, Triton, Topcon Corp., Tokyo, Japan). We analyzed data at EPL initiation, and then after 6, 12, and 48 months’ follow-up.
It is significant to note that the fellow eyes without exudative episodes and subretinal fluid (SRF) in the past have remained non-exudative throughout the study period.
The following exclusion criteria were adopted: (i) bilateral exudative CSC, (ii) other choroidal or retinal diseases, (ii) previous ophthalmological surgery or laser therapy, (iii) use of systemic steroids, (iv) previous photodynamic therapy, (v) presence of other systemic contraindications to the administration of mineralocorticoids, and (vi) presence of MNV evaluated with OCTA and ICGA.
Outcomes Measures
The main outcomes measured were: (i) best-corrected visual acuity (converted into LogMAR scale) in both eyes; (ii) subretinal fluid (SRF) height in exudative cCSC eyes; (iii) sub-foveal choroidal thickness (SFCT) in both eyes; (iv) choriocapillaris flow deficit percentage (CC FD %) in non-exudative fellow eyes.
OCT and OCTA Imaging Analysis
OCT images of the macular area (volume scan of 12×9 mm) were obtained by SS-OCTA, which uses an A-scan rate of 100,000 A-scans per second and a light source centered at 1050 nm. En face OCTA 4.5×4.5 mm volume scans (500 × 500 pixels resolution) were acquired using the same SS-OCTA. The manufacturer’s semi-automated segmentation tool was applied to delimitate retinal layers. Such a tool was used to obtain choriocapillaris en face OCTA images (slabs 30 μm thick, starting 31 μm posterior to the RPE–Bruch’s membrane complex). The automatic segmentation was subsequently rechecked and, in case of errors, manual segmentation was performed by two investigators (P.V. and G.B.). Choriocapillaris assessment was performed by analyzing SS-OCTA en face images choriocapillaris slabs (10 μm thick, 21 and 31 μm beneath Bruch’s membrane).20 Choriocapillaris areas located under retinal vessels were excluded in order to avoid artifacts and potential shadows.21 Only high-quality images with a strength index greater than 60 were included in the analysis. Any acquisitions that exhibited a shadowing effect or significant artifacts were excluded from the analysis to ensure the reliability and accuracy of the results.
Imaging Processing
Subretinal Fluid Height (SRF)
SRF was assessed by manually measuring the distance between the external limiting membrane and RPE at the fovea (Figure 1A).22
Sub-Foveal Choroidal Thickness (SFCT)
SFCT was measured with the caliper function of structural OCT. The SFCT was measured from Bruch’s membrane to the chorio-retinal interface perpendicularly in the center of the fovea (Figure 1A).22
Choriocapillaris Flow Deficits Percentage (CC FD %)
Each CC en face OCTA image was exported and analyzed with Fiji ImageJ (software version 2.0.0; National Institute of Health, Bethesda, MD). Then, CC images were binarized by the Phansalkar method using a radius of 15 pixels, in agreement with previous studies.23 To calculate CC FD %, the “analyze particles” command was used (Figure 1B). We did not analyze the choriocapillaris (CC) flow in the subretinal fluid (SRF) of the eyes with exudative chronic central serous chorioretinopathy (cCSC). This decision was made to avoid potential artifacts from SRF and any masking effects that may occur during OCTA analysis. To assess reproducibility of our measurements, all patients underwent second OCTA measurements on a different day.
Statistics Analysis
All quantitative variables were reported as mean ± standard deviation (SD). Data were assessed for normality with Shapiro–Wilk test for all variables. Pairwise comparisons were performed with a paired t-test to compare BCVA, OCT and OCTA metrics between follow-up examinations. The analysis of variance was used to assess the reproducibility between the 2 measurements. Statistical calculations were performed using Statistical Package for Social Sciences (version 20.0; SPSS Inc., Chicago, IL). The chosen level of statistical significance was P<0.05.
Results
Characteristics of Patients Included in the Analysis
Seventeen Caucasian patients (12 men, 5 women; mean age: 46.3 ± 2.1 years) with unilateral exudative cCSC were included. The duration of cCSC was 6.5 ± 0.7 months. No patients were under concurrent steroid therapy. Characteristics of subjects included in the analysis are shown in Table 1. The baseline characteristics of the analyzed cohort using multimodal imaging are shown in Supplementary Material 1.
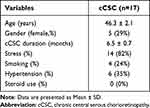 |
Table 1 The Clinical and Medical Characteristics of Subjects Included in the Analysis |
BCVA in Exudative cCSC Eyes
Mean BCVA was 0.34±0.13 LogMAR at baseline. Compared with baseline, BCVA significantly improved at follow-up examinations (6 months: 0.28±0.13 logMAR, p=0.039; 12 months: 0.22±0.11 logMAR, p=0.025; 48 months: 0.21±0.08 logMAR, p=0.028) (Table 2). However, we found a not statistically significant improvement at the last follow-up compared with the examination at 12 months (see Supplemental Material 2).
 |
Table 2 Functional and Anatomical (OCT) Analysis in cCSC Affected Eyes. Data and Comparisons |
BCVA in Non-Exudative Fellow Eyes
We did not find any statistically significant difference between the various follow-up examinations (Table 3).
 |
Table 3 Functional and Anatomical (OCT and OCTA) Analysis in cCSC Fellow Eyes. Data and Comparisons |
OCT Parameters Analysis in Exudative cCSC Eyes
When compared to the baseline measurements, there was a substantial decrease in subretinal fluid (SRF) at 6 months, with a mean value dropping from 208.3±155.3 μm to 25.6±44.1 μm (p<0.0001). The SRF resolution continued to improve, reaching complete resolution at the 12-month follow-up (p<0.0001). This notable improvement from the baseline was sustained even at the 48-month follow-up assessment (p<0.0001). Similarly, as illustrated in Figure 2, the subfoveal choroidal thickness (SFCT) demonstrated a significant decrease throughout the study period. The SFCT measurement decreased from 639±199.3 μm at baseline to 547±188.9 μm at 6 months (p=0.001), 520.4±170.6 μm at 12 months (p=0.001), and 507±164.6 μm at 48 months (p=0.001), as indicated in Table 2. These findings highlight the sustained reduction in SFCT over the course of the study.
 |
Figure 2 Graph showing mean sub-foveal choroidal thickness changes in affected and unaffected eyes throughout the follow-up. (*) Comparison with baseline values. |
OCT and OCTA Parameters Analysis in Non-Exudative Fellow Eyes
Similarly to exudative cCSC eyes, SFCT showed a significant reduction from 594±234.4 μm at baseline to 533±203.2 μm at 6 months (p=0.001), 506±184.2 μm at 12 months (p=0.011) and 491.3±179.5 μm at 48 months (p=0.001) (Table 3). However, we did not find a statistically significant SFCT reduction at the last follow-up compared with the results at 12 months (see Supplemental Material 3).
Regarding OCTA parameters, CC FD % analysis showed a statistically significant reduction after continuous EPL treatment (Figure 3). SS-OCTA analysis disclosed a significant FD % decrease from 28.9±1.96% at baseline to 26.8±1.61% at 6 months (p= 0.022), 26.6±1.42% at 12 months (p=0.001), and 26.4±1.44% at 48 months (p<0.001) (Table 3 and Figure 4). Conversely, we failed to find any statistically significant CC FD % reduction between follow-up examinations (see Supplemental Material 2).
 |
Figure 4 Graph showing mean choriocapillaris FD% modifications in unaffected eyes throughout the follow-up. (*) Comparison with baseline values. |
Safety Analysis
There were only mild adverse events (dry mouth and dizziness) as a result of EPL treatment. Overall, EPL was well tolerated and did not lead to potassium level changes or low creatinine clearance. All the patients continued EPL treatment because adverse events were not significant.
Discussion
In this pilot study, we evaluated the long-term effects of mineralocorticoid antagonists on eyes with chronic CSC associated with pachychoroid spectrum disease. Using structural optical coherence tomography (OCT) and OCT angiography (OCTA), we assessed the impact of eplerenone (EPL) on both quantitative and qualitative parameters in both the exudative chronic CSC eyes and non-exudative fellow eyes. In the exudative chronic CSC eyes, we observed a statistically significant improvement in mean best-corrected visual acuity (BCVA) and a decrease in mean subretinal fluid (SRF) height and choroidal thickness during the follow-up examinations at 6, 12, and 48 months. This indicates that EPL treatment led to improved visual acuity and reduced fluid accumulation and choroidal thickness in these eyes. In the non-exudative fellow eyes, we analyzed choroidal thickness (CT) and choriocapillaris (CC) flow deficits percentage before and during EPL treatment. After just 6 months of EPL treatment, we found a significant reduction in CT and CC ischemia. Importantly, EPL treatment was well tolerated with minimal side effects, and none of the observed side effects required discontinuation of therapy. This indicates a favorable safety profile for EPL in the treatment of chronic CSC.
Verteporfin photodynamic therapy (PDT) is currently the most extensively studied treatment option for both acute and chronic central serous chorioretinopathy (CSC).24–26 However, it is important to note that PDT can be associated with adverse events such as retinal pigment epithelium (RPE) atrophy, the development of choroidal neovascularization (CNV), and choroidal ischemia.27,28 Additionally, treatment success may be limited in cases with pre-existing RPE atrophy.27,28 Another potential treatment option for both acute and chronic CSC is subthreshold microsecond pulse laser (SML),29 but its anatomical and functional outcomes can vary significantly.30,31 In light of these considerations, oral mineralocorticoid receptor (MR) antagonists may offer a promising alternative for the treatment of chronic CSC.
MR antagonists are well-known drugs in the treatment of cardiovascular diseases.32 EPL is primarily used to treat heart failure and systemic hypertension through its competitive aldosterone-blocking abilities.33 Endogenous mineralocorticoid dysfunction has been described in cCSC patients.34 Moreover, prolonged exposure to increased levels of exogenous and endogenous glucocorticoids plays a role in the pathogenesis of CSC.35 In rats, the administration of corticosteroids causes choroidal thickening and increased expression of MRs, thus suggesting that MR antagonists may be used to treat cCSC.36
Recent reports have described groups of CSC patients treated with EPL.4,22 These studies have frequently been limited in size and follow-up duration. Using MR antagonists (EPL and spironolactone), Bousquets et al37 have yielded promising results during the 3 months of treatment in terms of BCVA, central macular thickness, and SRF. In a study assessing 28 eyes followed up for 6 months, Gergely et al38 concluded that patients with cCSC can safely be treated with EPL, because it reduces choroidal permeability with an accompanying SRF resolution and BCVA improvement. In a larger retrospective study analyzing 100 eyes treated with EPL for a mean follow-up of 20.6 months,39 the authors found complete SRF resolution within the first year of treatment, which appeared to be maintained over subsequent years. In agreement with the latter investigation, our study found similar improvements in BCVA and SRF resorption within the first year of follow-up. In the patients with total SRF resolution, this effect was maintained at 4 years. Likewise, SFCT did show significant anatomical improvement over 12 months of follow-up. Overall, these findings suggest that maintenance of continuous therapy may be beneficial on anatomical outcomes.
Based on the recent vision of pachychoroid pigment epitheliopathy,40 we also evaluated the non-exudative fellow eye of cCSC patients to assess the effects of EPL therapy on CC flow and SFCT. Results on choroidal thickness of non-exudative fellow eyes of cCSC patients are still controversial. Ghadialy et al41 did not find significant SFCT changes in patients treated with MR antagonists; however, only two patients were exclusively treated with EPL. By contrast, Gergely et al38 showed significantly SFCT reduction in non-exudative fellow eyes during the treatment that returned to initial values after therapy discontinuation. Our findings disclosed a significant decrease in mean SFCT as early as 6 months of EPL treatment. Importantly, the significant SFCT decrease was also persisted during the follow-up period. Furthermore, following the theory that the abnormally dilated choroidal vessels cause direct compression of the overlying choriocapillaris, we also analyzed the CC FD % changes in non-exudative fellow eyes. We demonstrated that CC perfusion remodels after 6 months of EPL treatment, and it was maintained in the follow up period. Previous papers have focused on the signal changes in CC flow in patients with unilateral CSC following different therapies. Ho et al42 suggested that both PDT and MLT promote CC flow recovery, confirming that the barrier function of the CC-RPE complex can be impaired, leading to fluid accumulation in CSC cases. Nevertheless, we did not analyze the CC flow of the exudative cCSC eyes to avoid possible OCTA artifacts from SRF and masking effects.
Taking these factors into consideration, we can speculate that the significant improvement observed in OCT and OCTA parameters, as well as the absence of subretinal fluid (SRF) development during EPL treatment, may be attributed to the reverse effect induced by EPL on choroidal vasodilatation in patients with chronic central serous chorioretinopathy (cCSC). The continuous administration of eplerenone seems to consistently attenuate the activation of mineralocorticoid receptors, which play a role in regulating ion and fluid balance in the retinal pigment epithelium (RPE) through sodium channels (ENaC-α), potassium channels (Kir4.1), and aquaporin channels. Additionally, we hypothesize that the resolution of SRF and potentially the normalization of choroidal circulation may have a beneficial impact on photoreceptor regeneration, leading to an improvement in best-corrected visual acuity (BCVA). Based on our long-term findings, continuous administration of EPL may serve as a valid approach to prevent further functional and anatomical damage in patients with cCSC.
As this was a pilot study, the patient numbers were small and sample size planning to ensure an adequate power was not necessary. The primary drawback of the current study is that it lacked a randomized prospective design, where patients would have been randomly assigned to either a study group receiving EPL or a control group not receiving EPL. The inclusion of a control group would have been crucial to distinguish the specific impact of EPL on the progression of the disease and determine the optimal treatment approach. Additionally, it is worth noting that it would have been ethically challenging to have a control group without any treatment for central serous chorioretinopathy (cCSC) over a span of 48 months.
In conclusion, our study focused on evaluating the long-term anatomical and visual outcomes in the eyes of patients who received continuous eplerenone (EPL) treatment for unilateral exudative chronic central serous chorioretinopathy (cCSC). The results obtained from OCT assessments of the exudative eyes and OCTA evaluations of the non-exudative eyes indicate that the beneficial effects of EPL on cCSC are primarily observed within the first year of treatment. Furthermore, in patients who achieved complete resolution of subretinal fluid (SRF), this positive effect was maintained even after 4 years. However, further confirmation of these findings is necessary through future randomized prospective case-control trials. Such studies will provide a more comprehensive understanding of the efficacy of EPL in the long-term management of cCSC.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kitzmann AS, Pulido JS, Diehl NN, Hodge DO, Burke JP. The incidence of central serous chorioretinopathy in Olmsted County, Minnesota, 1980-2002. Ophthalmology. 2008;115(1):169–173. doi:10.1016/j.ophtha.2007.02.032
2. Rijssen van TJ, Dijk van EHC, Yzer S, et al. Progress in Retinal and Eye Research Central serous chorioretinopathy: towards an evidence-based treatment guideline. Prog Retin Eye Res. 2019;73(July):100770. doi:10.1016/j.preteyeres.2019.07.003
3. Wong KH, Lau KP, Chhablani J, Tao Y, Li Q, Wong IY. Central serous chorioretinopathy: what we have learnt so far. Acta Ophthalmol. 2016;94(4):321–325. doi:10.1111/aos.12779
4. Daruich A, Matet A, Dirani A, et al. Oral Mineralocorticoid-Receptor Antagonists: real-Life Experience in Clinical Subtypes of Nonresolving Central Serous Chorioretinopathy With Chronic Epitheliopathy. Transl. Vis. Sci. Technol. 2016;5(2):2. doi:10.1167/tvst.5.2.2
5. Toto L, Ruggeri ML, Evangelista F, et al. Choroidal modifications assessed by means of choroidal vascularity index after oral eplerenone treatment in chronic central serous chorioretinopathy. Eye. 2022;37(6):1214–1218. doi:10.1038/s41433-022-02091-6
6. Gawęcki M, Grzybowski A. Ganglion Cell Loss in the Course of Central Serous Chorioretinopathy. Ophthalmol. Ther. 2023;12(1):517–533. doi:10.1007/s40123-022-00625-5
7. Chhablani J, Cohen FB, Aymard P, et al. Multimodal Imaging-Based Central Serous Chorioretinopathy Classification. Ophthalmol Retin. 2020;4(11):1043–1046. doi:10.1016/j.oret.2020.07.026
8. van Dijk EHC, Fauser S, Breukink MB, et al. Half-Dose Photodynamic Therapy versus High-Density Subthreshold Micropulse Laser Treatment in Patients with Chronic Central Serous Chorioretinopathy: the PLACE Trial. Ophthalmology. 2018;125(10):1547–1555. doi:10.1016/j.ophtha.2018.04.021
9. Sacconi R, Borrelli E, Querques G. Eplerenone for chronic central serous chorioretinopathy. Lancet. 2020;396(10262):1556. doi:10.1016/S0140-6736(20)31610-X
10. Dansingani KK, Balaratnasingam C, Naysan J, Freund KB. EN FACE IMAGING OF PACHYCHOROID SPECTRUM DISORDERS WITH SWEPT-SOURCE OPTICAL COHERENCE TOMOGRAPHY. Retina. 2016;36(3):499–516. doi:10.1097/IAE.0000000000000742
11. Yun C, Huh J, Ahn SM, et al. Choriocapillaris flow features and choroidal vasculature in the fellow eyes of patients with acute central serous chorioretinopathy. Graefe’s Arch Clin Exp Ophthalmol. 2019;257(1):57–70. doi:10.1007/s00417-018-4179-2
12. Iacono P, Toto L, Costanzo E, Varano M, Parravano MC. Pharmacotherapy of Central Serous Chorioretinopathy: a Review of the Current Treatments. Curr. Pharm. Des. 2018;24(41):4864–4873. doi:10.2174/1381612825666190123165914
13. Lotery A, Sivaprasad S, O’Connell A, et al. Eplerenone for chronic central serous chorioretinopathy in patients with active, previously untreated disease for more than 4 months (VICI): a randomised, double-blind, placebo-controlled trial. Lancet. 2020;395(10220):294–303. doi:10.1016/S0140-6736(19)32981-2
14. Zola M, Daruich A, Matet A, Mantel I, Behar-Cohen F. Two-year follow-up of mineralocorticoid receptor antagonists for chronic central serous chorioretinopathy. Br. J. Ophthalmol. 2019;103(8):1184–1189. doi:10.1136/bjophthalmol-2018-312892
15. Gao SS, Jia Y, Zhang M, et al. Optical Coherence Tomography Angiography. Investigative Opthalmology & Visual Science. 2016;57(9):OCT27. doi:10.1167/iovs.15-19043
16. Shinojima A, Kawamura A, Mori R, Fujita K, Yuzawa M. Findings of Optical Coherence Tomographic Angiography at the Choriocapillaris Level in Central Serous Chorioretinopathy. Int. J. Ophthalmol. 2016;236(2):108–113.
17. Teussink MM, Breukink MB, van Grinsven MJJP, et al. OCT Angiography Compared to Fluorescein and Indocyanine Green Angiography in Chronic Central Serous Chorioretinopathy. Invest. Ophthalmol. Vis. Sci. 2015;56(9):5229–5237. doi:10.1167/iovs.15-17140
18. Iovino C, Pellegrini M, Bernabei F, et al. Choroidal Vascularity Index: an In-Depth Analysis of This Novel Optical Coherence Tomography Parameter. J. Clin. Med. 2020;9(2):595. doi:10.3390/jcm9020595
19. van Rijssen TJ, Singh SR, van Dijk EHC, et al. Prospective evaluation of changes in choroidal vascularity index after half-dose photodynamic therapy versus micropulse laser treatment in chronic central serous chorioretinopathy. Graefe’s Arch Clin Exp Ophthalmol. 2020;258(6):1191–1197. doi:10.1007/s00417-020-04619-6
20. Byon I, Nassisi M, Borrelli E, Sadda SR. Impact of Slab Selection on Quantification of Choriocapillaris Flow Deficits by Optical Coherence Tomography Angiography. Am J Ophthalmol. 2019;208:397–405. doi:10.1016/j.ajo.2019.08.026
21. Garrity ST, Iafe NA, Phasukkijwatana N, Chen X, Sarraf D. Quantitative Analysis of Three Distinct Retinal Capillary Plexuses in Healthy Eyes Using Optical Coherence Tomography Angiography. Invest. Ophthalmol. Vis. Sci. 2017;58(12):5548–5555. doi:10.1167/iovs.17-22036
22. Bousquet E, Dhundass M, Lejoyeux R, et al. Predictive Factors of Response to Mineralocorticoid Receptor Antagonists in Nonresolving Central Serous Chorioretinopathy. Am J Ophthalmol. 2019;198:80–87. doi:10.1016/j.ajo.2018.09.034
23. Viggiano P, Grassi MO, Pignataro M, et al. Topographical Analysis of the Choriocapillaris Reperfusion After Loading Anti-VEGF Therapy in Neovascular AMD. Transl. Vis. Sci. Technol. 2022;11(9):18. doi:10.1167/tvst.11.9.18
24. Ober MD, Yannuzzi LA, Do DV, et al. Photodynamic therapy for focal retinal pigment epithelial leaks secondary to central serous chorioretinopathy. Ophthalmology. 2005;112(12):2088–2094. doi:10.1016/j.ophtha.2005.06.026
25. Canakis C, Livir-Rallatos C, Panayiotis Z, et al. Ocular photodynamic therapy for serous macular detachment in the diffuse retinal pigment epitheliopathy variant of idiopathic central serous chorioretinopathy. Am J Ophthalmol. 2003;136(4):750–752. doi:10.1016/S0002-9394(03)00397-0
26. Koh AHC. Photodynamic Therapy for Focal RPE Leaks. Ophthalmology. 2006;113(11):
27. Fujita K, Imamura Y, Shinoda K, et al. Quantification of metamorphopsia in chronic central serous chorioretinopathy after half-dose verteporfin photodynamic therapy. Retina. 2014;34(5):964–970. doi:10.1097/IAE.0000000000000027
28. Koytak A, Erol K, Coskun E, Asik N, Öztürk H, Özertürk Y. Fluorescein angiography-guided photodynamic therapy with half-dose verteporfin for chronic central serous chorioretinopathy. Retina. 2010;30(10):1698–1703. doi:10.1097/IAE.0b013e3181da4354
29. Sun Z, Huang Y, Nie C, et al. Efficacy and safety of subthreshold micropulse laser compared with threshold conventional laser in central serous chorioretinopathy. Eye. 2020;34(9):1592–1599. doi:10.1038/s41433-019-0692-8
30. Bartollino S, Semeraro F, Morescalchi F, et al. Central Serous Chorioretinopathy: pathogenesis and Management. Clin Ophthalmol. 2019;13:2341–2352. doi:10.2147/OPTH.S220845
31. Kim JY, Park HS, Kim SY. Short-term efficacy of subthreshold micropulse yellow laser (577-nm) photocoagulation for chronic central serous chorioretinopathy. Graefes Arch. Clin. Exp. Ophthalmol. 2015;253(12):2129–2135. doi:10.1007/s00417-015-2965-7
32. Struthers A, Krum H, Williams GH. A comparison of the aldosterone-blocking agents eplerenone and spironolactone. Clin. Cardiol. 2008;31(4):153–158. doi:10.1002/clc.20324
33. FDA, CDER. Highlights of prescribing information. Available from: www.fda.gov/medwatch.
34. Haimovici R, Rumelt S, Melby J. Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology. 2003;110(4):698–703. doi:10.1016/S0161-6420(02)01975-9
35. Daruich A, Matet A, Dirani A, et al. Central serous chorioretinopathy: recent findings and new physiopathology hypothesis. Prog Retin Eye Res. 2015;48:82–118. doi:10.1016/j.preteyeres.2015.05.003
36. Zhao M, Célérier I, Bousquet E, et al. Mineralocorticoid receptor is involved in rat and human ocular chorioretinopathy. J. Clin. Invest. 2012;122(7):2672–2679. doi:10.1172/JCI61427
37. Bousquet E, Beydoun T, Zhao M, Hassan L, Offret O, Behar-Cohen F. Mineralocorticoid receptor antagonism in the treatment of chronic central serous chorioretinopathy: a pilot study. Retina. 2013;33(10):2096–2102. doi:10.1097/IAE.0b013e318297a07a
38. Gergely R, Kovács I, Schneider M, et al. Mineralocorticoid receptor antagonist treatment in bilateral chronic central serous chorioretinopathy: a comparative study of exudative and nonexudative fellow eyes. Retina. 2017;37(6):1084–1091. doi:10.1097/IAE.0000000000001303
39. Petkovsek DS, Cherfan DG, Conti FF, et al. Eplerenone for the treatment of chronic central serous chorioretinopathy: 3-year clinical experience. Br. J. Ophthalmol. 2020;104(2):182–187. doi:10.1136/bjophthalmol-2019-314047
40. Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013;33(8):1659–1672. doi:10.1097/IAE.0b013e3182953df4
41. Ghadiali Q, Jung JJ, Yu S, Patel SN, Yannuzzi LA. Central serous chorioretinopathy treated with mineralocorticoid antagonists: a one-year pilot study. Retina. 2016;36(3):611–618. doi:10.1097/IAE.0000000000000748
42. Ho M, Lai FHP, Ng DSC, et al. Analysis of choriocapillaris perfusion and choroidal layer changes in patients with chronic central serous chorioretinopathy randomised to micropulse laser or photodynamic therapy. Br. J. Ophthalmol. 2021;105(4):555–560. doi:10.1136/bjophthalmol-2020-316076
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


