Back to Journals » Cancer Management and Research » Volume 13
Comparison of Hematological and Biochemical Profile Changes in Pre- and Post-Chemotherapy Treatment of Cancer Patients Attended at Ayder Comprehensive Specialized Hospital, Mekelle, Northern Ethiopia 2019: A Retrospective Cohort Study
Authors Wondimneh B , Anekere Dasappa Setty S, Gebregzabher Asfeha G, Belay E , Gebremeskel G, Baye G
Received 30 July 2020
Accepted for publication 31 December 2020
Published 22 January 2021 Volume 2021:13 Pages 625—632
DOI https://doi.org/10.2147/CMAR.S274821
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Antonella D'Anneo
Bayu Wondimneh,1 Sathisha Anekere Dasappa Setty,2 Gebrekidan Gebregzabher Asfeha,2 Ezra Belay,2 Gidey Gebremeskel,2 Gelagay Baye1
1Department of Biomedical Sciences, Debre Markos University, Debre Markos, Ethiopia; 2Departments of Biochemistry and Molecular Biology, Mekelle University, Mekelle, Ethiopia
Correspondence: Bayu Wondimneh
Department of Biomedical Sciences, Debre Markos University, P.O. Box: 269, Debre Markos, Ethiopia
Tel +251 920031998
Fax +251 587780673
Email [email protected]
Purpose: This study aims to compare hematological and biochemical profile changes in pre- and post-chemotherapy among cancer patients admitted at the Oncology Unit of Ayder Comprehensive Specialized Hospital (ACSH), Mekelle, Northern Ethiopia.
Patients and Methods: A retrospective cohort study was conducted in 376 cancer patients admitted in the Oncology Unit at ACSH. Demographic data, hematological and biochemical profiles were collected from smart care and patient cards. The data were analyzed using SPSS version 20 statistical package. Descriptive statistics and paired sample students T-test statistical methods were used.
Results: From 376 study subjects, 228 (60.6%) were females. All the hematological profiles, except lymphocyte (LYM) (P > 0.05), showed significant decrement in post-chemotherapy compared to pre-chemotherapy; white blood cell (WBC) (P < 0.01), red blood cell (RBC) (P < 0.01), hemoglobin (Hb) (P< 0.001), hematocrit (HCT) (P < 0.05), platelet (PLT) (P < 0.001) and neutrophil (NUT) (P < 0.05). The biochemical profiles showed that blood urea nitrogen and creatinine levels were non-significantly decreased, urea (P > 0.05) and creatinine (CR) (P > 0.05), in post-chemotherapy compared to pre-chemotherapy whereas alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels were non significantly increased, ALT (P > 0.05) and AST (P> 0. 05), in post-chemotherapy compared to pre-chemotherapy.
Conclusion: Hematological profiles, except lymphocytes, were found significantly decreased whereas biochemical profiles, urea, and creatinine were decreased non-significantly, while AST and ALT showed non-significant increments in post-chemotherapy compared to pre-chemotherapy.
Keywords: cancer, chemotherapy, hematological profiles, biochemical profiles, Ethiopia
Introduction
Cancer is a disease involving abnormal cell growth with the potential to invade and spread to other parts of the body with many possible causes such as genetic, environmental, or constitutional characteristics of the individual.1
Cancer is the 21st century most dangerous and severe disease in the world and it is the second leading cause of death globally.2 The cancer treatment that a patient receives is determined by the stage of cancer at diagnosis, the type, and location of cancer, the standard medical practices and treatment guidelines in the patient’s country. Several options are available for cancer treatment such as surgery, chemotherapy, and radiation therapy.4 However, these treatments are not usually curative and put the patients at the risk of several side effects. Consequently, several side effects occur with any cancer drug treatment like anemia, infection, bleeding problem, nausea, and vomiting, allergic reactions, pain or tenderness, constipation or diarrhea, hair loss, sore mouth, increased energy, and difficulty of sleeping. If this situation is not monitored properly, this may lead to treatment failure.6
Chemotherapies have various effects on hematological and biochemical profiles. Chemotherapies act as alkalizing agents and lead to progressive depletion of hematopoietic stem cells in the bone marrow.7 Chemotherapy causes cell death or prevents cell growth, generally through inhibiting microtubule function, protein function, and deoxyribonucleic acid (DNA) synthesis.5 Chemotherapeutic agents bind covalently with bone marrow cells’ DNA to form intra- and inter-strand cross-links of DNA that cause DNA damage during replication. This situation results in a decrease of hemoglobin, platelets, and leukocytes levels.9
Immune alteration and anemia have been reported in cancer patients, who receive chemotherapy. It has been reported that frequent intensive chemotherapy suppresses immune cell that causes opportunistic infections.4 Side-effects from chemotherapy are evident on bone-marrow which is a major site for major pluripotent hematopoietic stem cells.8
Furthermore, in the complex world of cancer therapy, the administration of medications intentionally designed to be cytotoxic inevitably causes negative consequences on biochemical profiles. The liver is the primary site of metabolism for many of these drugs, and these liver-drug interactions must be accounted for while dosing chemotherapy.11 The administration of chemotherapy is a challenge for the tight regulation and balance of liver function. Most chemotherapy drugs are readily taken up by the liver and up to 85% of patients develop liver steatosis.12 Repeated chemotherapy induces irreversible hepatocellular damage through the recruitment of inflammatory cells and leads to alteration of biochemical profiles like; ALT, AST, alkaline phosphatase (ALP), and lactate dehydrogenase (LDH).13 Similarly, it has been shown that chemotherapy can induce apoptosis and necrosis of kidney cells. This damage is clinically manifested as an increase in urea and creatinine levels.14
Despite several studies showed that enormous problems related to chemotherapy especially on hematological and biochemical profile; there are limited reports regarding hematological and biochemical profile changes in pre- and post-chemotherapy of cancer patients in the study area.
Patients and Methods
Study Area
The study was conducted at Ayder Comprehensive Specialized Hospital, Mekelle, Tigray region, Northern part of Ethiopia. It is located around 780 kilometers from Addis Ababa, the capital city of Ethiopia. The oncology units in Ayder Comprehensive Specialized Hospital have two sub-units, adult and infant oncology.
Study Design and Period
A retrospective cohort study was conducted from March 2019 to July 2019 to compare hematological and biochemical profile changes in pre- and post-chemotherapy treatments of cancer patients admitted at the Oncology unit of ACSH.
Source Population
All cancer patients attended at the Oncology unit of Ayder Comprehensive Specialized Hospital; Mekelle, Northern Ethiopia.
Study Population
Cancer patients started chemotherapy at the Oncology Unit of ACSH from January 2014 to December 2018.
Inclusion Criteria
All cancer patients with complete demographic records such as age, sex, diagnosis, stage, treatment and medical records such as type and stage of cancer, hematological and biochemical profiles in pre- and post-chemotherapy treatments.
Exclusion Criteria
- Cancer patients who had incomplete medical records.
- A cancer patient who stopped their follow-up before completing the treatment for a cycle- IV.
- Patients who had other diseases and conditions that affect hematological and biochemical profiles.
Sampling Method and Sample Size Determination
A non-probability convenient sampling technique was implemented. Required data were collected from 376 cancer patients admitted in the Oncology unit of ACSH between January 2014 and December 2018.
Measurements
Data Collection Tools/Techniques
The case files of the cancer patients were reviewed by using the data collection form. Data were extracted from the case files including demographic, clinical characteristics, hematologic, and biochemical profiles. To get the data we used the following steps: Firstly, we reviewed the registration book of the patients and took their medical record number (MRN). Secondly, we filtered the patient’s MRN on excel and removed repetition. Finally, after getting access to smart care, the patients’ data were collected from both smart care and cards.
Data Processing and Analysis
Data were cleaned, entered, and analyzed using SPSS version 20 statistical package. Descriptive statistics were used to analyze and present the demographic and clinical characteristics of the cancer patient’s data. The hematological and biochemical profile changes in pre- and post-chemotherapy were compared by using the paired t-test.
Study Variables
Dependent Variable
- White blood cells, red blood cells, hemoglobin, hematocrit, platelet, neutrophil, lymphocyte, urea, creatinine, alanine aminotransferase, and aspartate aminotransferase.
Independent Variable
- Chemotherapy.
Data Management and Quality Control
- The data collection form was prepared and all variables were carefully filled daily on it.
- To improve the data collection form (if any) and to check the availability of enough sample size, an inspection was carried out on 50 cancer patients.
- Two data collectors were engaged; one from the smart care administration and one from the card room. Both of them were trained about how to count the cycle-IV and how to fill the format by checking MRN with smart care and the patient’s card.
Operational Terms
- Hematological profiles of pre-chemotherapy: laboratory results of hematological profiles at the time of cancer diagnosis.
- Hematological profiles of post-chemotherapy: laboratory result of hematological profiles after finishing the Phase four of chemotherapy treatments.
Results
Demographic Characteristics of the Patients
The general study subjects’ characteristics are summarized in Table 1. Among the total number of cancer patients treated with chemotherapy from January 2014 to December 2018 in the oncology unit, 376 subjects were included in this study. There were 148 (39.4%) males and 228 (60.6%) females (Table 1). The average age of respondents is 41.34 (Figure 1).
 |
Table 1 Age versus Sex of Respondents, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
 |
Figure 1 Age of respondents, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019. |
Clinical Characteristics of the Patient
Common types of cancer in this study were breast cancer, lymphoma and sarcoma which account 37.5%, 24.73% and 10.37%, respectively (Table 2). In the first diagnosis, 8.25% study subjects were stage II, 61.44% were stage III, and 30.32% were stage IV, but there was no stage I at all (Table 3).
 |
Table 2 Type of Cancer versus Sex of Respondents, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
 |
Table 3 Stage of Cancer versus Sex of Respondents, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
Commonly encountered anti-cancer drugs in Northern Ethiopia include: - Methotrexate, Cisplatin, Doxorubicin, Paclitaxel, Gemcitabine, Levoleucovorin, Vincristine, 5-FU/5- fluorouracil/, Oxaliplatin and Cyclophosphamide. It is given to cancer patients with a combination of two or more drugs once a time. If the patient took two drugs in the first phase, in the second phase the drug regimen combination was changed, and the same as phase three and forth. Through the course of chemotherapy, the patient took more than four chemotherapy drugs in the last phase four. It is not specific to the types of cancer. Because of this, it’s difficult to know the effect of each chemotropic regimen on hematological and biochemical parameters (Table 4).
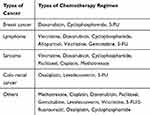 |
Table 4 Types of Cancer versus Chemotherapy Regimen, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
Comparison of Hematological Profiles in Pre- and Post-Chemotherapy of Study Subjects
As shown in Table 5, the hematological profile parameters were white blood cells, red blood cells, hemoglobin, hematocrit, platelets, neutrophils, and lymphocytes. The mean of all hematological profiles was lower in post-chemotherapy than pre-chemotherapy. The mean differences of hematological profiles between pre-chemotherapy and post-chemotherapy were noted to be WBC (2.44×103 cell/ml3), RBC (0.6×106 cell/ml3), Hb (0.63 g/dl), HCT (2.12%), PLT (22.2×105 cell/mm3), NUT (2.19%) and LYM (2.88%). Except lymphocytes (P > 0.05), all hematological profiles showed statistically significant decrements in post-chemotherapy compared to pre-chemotherapy, WBC (P < 0.01), RBC (P < 0.01), Hb (P <0.001), HCT (P < 0.05), PLT (P < 0.001), and NUT (P < 0.05) (Table 5).
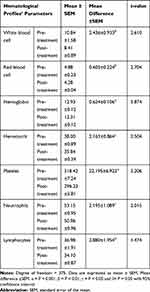 |
Table 5 Comparison of Hematological Profiles in Pre- and Post-Chemotherapy, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
Comparison of Biochemical Profiles in Pre- and Post-Chemotherapy of Cancer Patients
As can be seen in Table 6, the biochemical parameters were urea, creatinine, alanine transaminase, and aspartate transaminase. The mean differences of biochemical profiles between pre- and post- chemotherapies were noted to be, urea (0.59 mg/dl), CRT (0.04 mg/dl), ALT (−7.04 U/L), and AST (−3.23 U/L). Non-significant alterations were observed in all biochemical profiles. Urea and creatinine showed a non-significant decrement in post-chemotherapy compared to pre-chemotherapy, whereas alanine transaminase and aspartate transaminase showed a non-significant increment in post-chemotherapy compared to pre-chemotherapy (P > 0.05) (Table 6).
 |
Table 6 Comparison of Biochemical Profiles in Pre- and Post-Chemotherapies, Ayder Comprehensive Specialized Hospital, Oncology Unit, Northern Ethiopia, 2019 |
Discussion
Chemotherapy is a cytotoxic drug that mainly works on the active cell. Active cells are cells that are growing and replicating into the same type of cell. Cancer cells are active, but so are some healthy cells, including cells in the blood, and other organs. Side effects happen when chemotherapy damages these healthy cells.3
Cancer patients who receive chemotherapy commonly suffer from hematological and biochemical profile alteration, as a result, it leads to the potentially life-threatening condition due to severe anemia and infections.16 The study has reported that different biochemical and hematological profiles may get alteration due to toxicities induced by chemotherapy.15
The current study showed that statistically significant decrement of WBC count in post-chemotherapy compared to pre-chemotherapy (P < 0.01). The WBCs count decrement in post-treatment might be due to the suppressed hematopoietic stem-cells by chemotherapy which are important for WBC proliferation. This result is consistent with a recent study reported that the WBC count significantly decreased post-chemotherapy compared to pre-chemotherapy (P < 0.001) due to the side effect of chemotherapy on WBC production.10 In contrast with our findings, a previous study reported that a non-significant decrement of WBC in post-chemotherapy compared to pre-chemotherapy (P > 0.05).8 Consequently, cancer patients develop an infection after taking chemotherapy, because of the reduction of white blood cells that are important for fighting against infection.17
Similarly, RBC count revealed significant decrement in post-chemotherapy compared to pre-chemotherapy (P < 0.01). The reduction of RBC count in post-chemotherapy might be due to ineffective erythropoiesis. In line with this finding, a previous study also stated that RBC count decreased significantly in post-chemotherapy compared to pre-chemotherapy (P < 0.001) because of decreased new RBC production.11 After taking chemotherapy, the reduction of RBCs was also reported in cancer patients.12
Moreover, hemoglobin and hematocrit levels showed a statistically significant decrement in post-chemotherapy compared to pre-chemotherapy (P < 0.001 and P<0.05) respectively. Chemotherapy induces nephrotoxicity that led to anemia through decreased renal production of erythropoietin. Erythropoietin is a cytokine produced in the kidneys that stimulate erythropoiesis in patients. Impaired erythropoiesis will decrease the production of hemoglobin levels and hematocrit levels.13 In our case, the remarkable decrement of hemoglobin and hematocrit levels might be possible because chemotherapy can lead to the reduction of the production of new RBCs, or/and the increased elimination of RBCs or/and suppression of functional RBCs. These findings are in accord with the results from a previous study which reported that hemoglobin and hematocrit levels decrease significantly in post-chemotherapy compared to pre-chemotherapy (p < 0.001) because of impaired hematopoiesis in the bone marrow.12 As noted by another similar study, the lower levels of Hb and HCT are the manifestations of anemia, where its incidence increases with chemotherapy.11 Moreover, a finding suggested that more than 82% of cancer patients developed anemia in post-chemotherapy.17
In this study, platelet count showed a significant decrement in post-chemotherapy compared to pre-chemotherapy treatment (P < 0.001). The decrement of platelet count in post-treatment might be due to destroyed megakaryocytic progenitors at the early stages of differentiation by chemotherapy. Inconsistent to our study, it is stated that thrombocytopenia is a common problem in cancer patients who received chemotherapy treatment. 40 That is, chemotherapy causes deterioration of the bone marrow’s function to regenerate; as a result, new platelets are not produced any longer. Chemotherapy treatment cause impaired hepatic functioning and inhibits the production of clotting factors in the liver that contributes to easy bleeding.14 Moreover, our finding is in agreement with a study carried out on buccal mucosa carcinoma in India reported that platelet count was significantly decreased in post-chemotherapy compared to pre-chemotherapy (P < 0.001).18
Our study also showed that neutrophil levels were significantly declined after chemotherapy (P< 0.05). This result might be possibly due to the fact that the myelosuppressive effect of chemotherapy. Drug-induced neutropenia is a potentially serious and life-threatening adverse event that may occur secondary to chemotherapy. Cytotoxic chemotherapy can cause a predictable and dose-related decrease in neutrophil count. Neutropenia secondary to chemotherapy medications tends to be a particular reaction either as an immune-mediated reaction or because of direct myeloid cell line damage.15 Additionally, in agreement with our result, a previous study reported that the neutrophil level significantly declined in post-chemotherapy compared to pre-chemotherapy (P < 0.001).9 It was also reported that neutropenia can happen after the use of chemotherapeutic drugs.16
The current study showed a non-significant decrement of lymphocyte levels in post-chemotherapy compared to pre-chemotherapy (P > 0.05). It is revealed that chemotherapy reduces circulating lymphocyte levels, where T and B cells significantly decreased after completion of chemotherapy. Results of the current study are in contrast with previous studies, which might be due to the full extent of lymphocyte depletion that may not have been visible until a substantial portion of the chemotherapy was completed. In contrast to this finding, a previous study reported that lymphocyte levels significantly decreased in post-chemotherapy compared to pre-chemotherapy.15
Urea levels showed a non-significant decrement in post-chemotherapy compared to pre-chemotherapy (P > 0.05). The decrement in post-chemotherapy might be due to the low-protein diet intake of the patients since urea is a waste product of protein catabolism in the liver. In contrast with our findings, previous studies reported that urea levels significantly decreased in post-chemotherapy compared to pre-chemotherapy.8 On the other hand, a study reported those urea levels significantly increased in post-chemotherapy compared to pre-chemotherapy due to elevated cortisol and stress.15
Our study showed that a non-significant decrement of creatinine levels in post-chemotherapy than pre-chemotherapy (P > 0.05). The non-significant decrement of creatinine levels might be due to low muscle mass or malnutrition. It has been shown that chemotherapy can induce necrosis of kidney cells. This damage is clinically manifested as increased levels of creatinine.14 On the other hand, previous studies showed that creatinine levels significantly increased in post-chemotherapy than pre-chemotherapy due to impaired formation or impaired excretion of urine.16
The current study showed that non-significant increments of alanine aminotransferase levels in post-chemotherapy compared to pre-chemotherapy (P > 0.05). The non-significant increments of ALT levels might be possibly due to an inflamed condition of the liver, severe scarring of the liver, or death of liver tissue caused due to chemotherapy. Therefore, close liver function test monitoring is advised for patients starting a new chemotherapy regimen. On the contrary, previous studies reported that the serum levels of ALT significantly increased in post-chemotherapy than pre-chemotherapy.13 Moreover, results of the present study showed that non-significant increments of AST levels in post-chemotherapy compared to pre-chemotherapy (P > 0.05). This result can be due to the damage of the tissues and cells of the liver, skeletal muscle, heart, and brain, which resulted due to chemotherapy. On the other hand, a study reported that significant increment of AST levels in post-chemotherapy compared to pre-chemotherapy because of the toxic effect of chemotherapy on liver cells and other organs that leads to necrosis.17
Conclusion
Hematological profiles were found significantly decreased, except lymphocytes, in post-chemotherapy compared to pre-chemotherapy. Biochemical profiles such as urea and creatinine were decreased insignificantly, whereas AST and ALT levels showed non-significant increments in post-chemotherapy compared to pre-chemotherapy. Further studies could be conducted with a wide range of sample sizes. In our study there are contrary results happen to the other studies such as lymphocyte; the further study should be done for a consistent result.
Abbreviations
ACSH, Ayder Comprehensive Specialized Hospital; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CRT, creatinine; DNA, deoxyribonucleic acid; Hb, hemoglobin; HCT, hematocrit; LDH, lactate dehydrogenase; LYM, lymphocytes; MRN, medical record number; NUT, neutrophil; PLTs, platelets; RBCs, red blood cells; WBCs, white blood cells.
Data Sharing Statement
The dataset supporting the conclusion of this article is available from the authors on request.
Ethics Approval
The study was conducted per the Declaration of Helsinki. The ethical clearance was taken from Mekelle University, College of health science, Ethical review committee. The letter was formally submitted to Ayder Comprehensive Specialized hospital and got permission. To keep the confidentiality of the patients’ data recorded, the data collection procedure was anonymous.
Consent for Publication
Not Applicable.
Acknowledgments
We would like to start with a heartfelt THANK YOU to everyone who shared a kind word and gentle nudge to keep us moving forward toward completing our research. We would like to thank and express our deepest gratitude to Mekelle University which sponsored the research, Ayder Comprehensive Specialized Hospital, Oncology Unit professionals.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
Mekelle University research and community service directorate supported this research for data collection.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Lodish H, Arnold BS, Lawrence Z, Paul M, David BJE. Molecular Cell Biology.
2. Sobin LH, Wittekind C. TNM Classification of Malignant Tumors.
3. Noviyani R, Indrayathi PA, Budiana ING, Niruri R, Tunas K, Adnyani NMDD. Effect of paclitaxel-cisplatin chemotherapy towards hemoglobin, platelet, and leukocyte levels in epithelial ovarian cancer patients. Appl Pharm Sci. 2019;9(01):104–107.
4. Shahid S. Critical reviews in oncology/hematology review of hematological indices of cancer patients receiving combined chemotherapy & radiotherapy or receiving radiotherapy alone. Crit Rev Oncol Hematol. 2016;105:145–155. doi:10.1016/j.critrevonc.2016.06.001
5. Aziz HA, Habeeb JM. Study the effect of chemotherapy on some hematological and biochemical parameters of cancer patients in AL-Muthanna Province. Indian J Public Health Res Dev. 2019;10(2):813–816. doi:10.5958/0976-5506.2019.00395.4
6. Sheikh N, Masood MNN. Hematological and serological changes. J Soc Dev New Net Env. 2014;5:6.
7. Devi LI, Ralte L, Ma A. Serum biochemical profile of breast cancer patients. Eur J Pharm Med Res. 2006;2(6):210–214.
8. Mughal TI. Current and future use of hematopoietic growth factors in cancer medicine. Hematol Oncol. 2004;22(3):121–134. doi:10.1002/hon.736
9. Ramasamy P, Sundaresan S. Haematological and biochemical changes in pre and post; 2016. August 2017.
10. UK CR. Side effects of chemotherapy; 2018.
11. Steele M, Narendran A. Mechanisms of defective erythropoiesis and anemia in pediatric acute lymphoblastic leukemia (ALL). Ann Hematol. 2012;91(10):1513–1518. doi:10.1007/s00277-012-1475-5
12. Network NCC. Cancer- and Induced Anemia. In: NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). 2018:1–51.
13. HYOGO UO, Science GS of NA and. Disaster Nursing in a Ubiquitous Society Care Package for Cancer Patients In Times of Handbook of Self-Managing. Side Effects of Chemotherapy in Times of Disaster. In: Handbook of Self-Managing Side Effects of Chemotherapy in Times of Disaster; 2006;1–26.
14. C D. Drug-induced neutropenia. Pharmacovigil Forum. 2016;41(12):765–768.
15. Xuequan M. 2016:27
16. Siena S, Giannetta C. Optimizing management of neutropenia and anemia in cancer chemotherapy. Hematol. 2003;48:39–S47.
17. Kaplan BW, Ph D. Priority medicines for Europe and the world “ A public health approach to innovation “ update on 2004 background paper written by Warren Kaplan background paper 6. Cancer Ther. 2013;(April):5–62.
18. Padma R, Sundaresan S. Haematological and biochemical changes in pre and post-treatment of buccal mucosa carcinoma patients. Int J PharmSci Res. 2016;7(7):3002–3006.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
