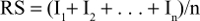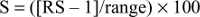Back to Journals » Cancer Management and Research » Volume 11
Comparison of analgesia, adverse effects, and quality of life in cancer patients during treatment of procedural pain with intravenous morphine, fentanyl nasal spray, and fentanyl buccal tablets
Authors Piotrowska W , Leppert W , Majkowicz M
Received 3 July 2018
Accepted for publication 16 November 2018
Published 18 February 2019 Volume 2019:11 Pages 1587—1600
DOI https://doi.org/10.2147/CMAR.S179012
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Rituraj Purohit
Wieslawa Piotrowska,1 Wojciech Leppert,1 Mikolaj Majkowicz2
1Laboratory of Quality of Life Research, Chair and Department of Palliative Medicine, Poznan University of Medical Sciences, Poznan, Poland; 2Institute of Health Sciences, Pomeranian Academy, Słupsk, Poland
Aim: Comparison of analgesia, adverse effects, and quality of life (QoL) of cancer patients in the treatment of procedural pain induced by nursing procedures with the use of intravenous morphine, fentanyl nasal spray, and fentanyl buccal tablets.
Methods: In adults with cancer with opioid tolerance and suffering procedural pain, intravenous morphine was used at an inpatient palliative medicine unit (20 patients) and fentanyl by intranasal (15 patients) and buccal routes (nine patients) at home. Five procedural pain episodes were examined: the Mini-Mental State Examination was used to assess cognitive function, the Brief Pain Inventory – short form (BPI-SF) to assess intensity and impact of pain on daily activities, a pain and adverse-effect questionnaire to assess the intensity of pain and adverse effects, and the European Organisation for Research and Treatment of Cancer QLQ-C15-PAL to assess QoL.
Results: All five procedural pain episodes were completed by 32 patients. Twelve patients stopped treatment due to death or referral to the hospital (four patients in each group), changes in the treatment of background pain (three patients), and intense drowsiness (one patient). Similar beneficial analgesic effects were observed in all patient groups. During fentanyl therapy, a smaller negative effect of pain on patients’ activity, walking, and work (BPI-SF) was observed. Among adverse effects, fewer breaths (10–14 per minute) were observed in 17 patients and slight disturbances of consciousness in seven. For QoL, an improvement in emotional functioning, overall QoL, and fatigue was observed. Patients treated with intranasal and buccal fentanyl had higher physical functioning and were more active.
Conclusion: In the treatment of procedural pain induced by nursing procedures in cancer patients, intravenous morphine and rapid-onset fentanyl show similarly high analgesic efficacy, with good tolerance of treatment and improvement in QoL.
Keywords: analgesia, cancer, procedural pain, adverse effects, quality of life
Introduction
Pain is a frequent symptom among cancer patients that significantly reduces quality of life (QoL) in both patients and caregivers, contributing to greater suffering of patients and their families. Pain can occur at any stage of cancer development, and is present in 50% of patients during anticancer treatment and 60%–70% in the advanced period of the disease.
Particularly difficult in treatment is breakthrough cancer pain, defined as short-term pain attacks, “overlapping” or “breaking through” by properly treated (usually with the use of opioids) basal pain.1,2 Breakthrough pain is characterized by a sudden-onset, rapid increase in intensity (usually after 3–5 minutes, the pain reaches maximum intensity), strong intensity according to the numeric rating scale (NRS; 0 – no pain, 10 – the most severe pain intensity, usually >5), short duration (usually up to 30 minutes), and varied pathophysiology.3 Most often, breakthrough pain occurs several times a day, is associated with higher frequency of anxiety and depression, reduces activity, significantly impairs the QoL of patients and caregivers, and considerably increases the cost of treatment.4
In recent years, the definition of breakthrough pain has been substantially extended by defining each significant, transient increase in pain intensity as episodic pain. The term includes not only patients with effectively treated background pain (one of the criteria for the diagnosis of breakthrough pain) but also patients in whom the background pain is not effectively treated, patients not treated with opioids, and patients in whom there is no background pain.5
Breakthrough pain can be classified as spontaneous pain (or idiopathic pain) and incident pain. Previously, end-of-dose pain was considered breakthrough pain, which occurs in 15%–30% of patients before administration of another dose of analgesics. Recently, end-of-dose pain was defined as pain caused by inadequate dosing of regular analgesics and an indication for correction of originally recommended treatment for background pain.
Spontaneous pain (or idiopathic pain) appears suddenly, and it is usually impossible to determine its cause or predictive factors.6 Incident pain is associated with a specific cause, and classified as:
- voluntary (volitional), dependent on will of patient, present during movement, swallowing, touching, and during urination and defecation;
- involuntary (non-volitional, independent of will), occurring during activities not involving the will of patient, eg, coughing, spasms, or stretching of the bladder and intestines;
- procedural, occurring during diagnostic, treatment, and nursing procedures.7,8
Incident pain associated with movement is more difficult to treat when compared to pain at rest, as most of the pain impulses at rest are carried by group C nerve fibers, when transmission of pain associated with movement involves also Aδ fibers, which (contrary to C fibers) do not bear opioid receptors.9
In the treatment of spontaneous breakthrough pain, medications are usually administered in the event of pain attacks: doses of analgesics administered in such cases are defined as rescue doses. In addition to traditional immediate-release opioids (eg, morphine, oxycodone) with oral or parenteral (subcutaneous and intravenous) administration, transmucosal fentanyl products with rapid onset of analgesic effect – water or pectin spray administered via the nasal route or tablets applied buccally and sublingually – are recommended.4 The aim of this study was a comparison of analgesic effects, adverse effects, and QoL during treatment of procedural pain induced by nursing procedures with intravenous morphine, water-based fentanyl nasal spray, and fentanyl buccal tablets in cancer patients.
Methods
The research protocol of the study was approved by the Bioethics Committee of Poznan University of Medical Sciences (resolution 579/13 on June 13, 2013), and the study was conducted in accordance with the Declaration of Helsinki. It was conducted from June 2013 to July 2016 in the Clinical Hospital of Lord’s Transfiguration in Poznan, and involved adult patients of both sexes diagnosed with cancer and severe procedural pain (NRS >6) due to different nursing procedures: changing position in bed, walking to bathroom, putting on clothes and napkin, whole-body toilet in bed, showers, and changing dressings. Patients had to show opioid tolerance in treatment of background pain for at least 7 days with morphine administered by the oral route at a daily dose of at least 60 mg or an equivalent dose of the drug administered by other routes, or transdermal fentanyl at a dose of at least 25 µg/h, buprenorphine applied by the transdermal route at a dose of at least 35 µg/h, or oxycodone administered orally at a daily dose of at least 30 mg. Patients who met these criteria were recommended intravenous morphine or fentanyl spray by nasal route or fentanyl tablets by buccal route in the treatment of pain caused by nursing procedures, based on individual clinical evaluation performed by the physician. The patients were divided into three groups: group 1 – 20 patients treated with intravenous morphine in the inpatient Palliative Medicine Department; group 2 – 15 patients treated with fentanyl in the form of a nasal spray; and group 3 – 9 patients treated with fentanyl in the form of tablets administered by the buccal route.
All patients from groups 2 and 3 were treated at home (the so-called home hospice). Patient groups did not differ regarding age and sex. After explanation of the study objectives to and written consent from the patients, they were interviewed about pain characteristics, indicating type of pain with respect to pathophysiology, occurrence, and intensity of procedural pain when performing nursing procedures assessed with the NRS, location of pain, and analgesics and their doses. Afterward, physical examinations were performed, as well as measurement of vital signs: blood pressure, pulse, and oxygen saturation.
All patients were diagnosed with different types of pain: bone, neuropathic, visceral, and somatic. In a group of 20 patients treated with intravenous morphine, one type of pain appeared in one patient, two types in 18, and three types in one. In the group of 15 patients treated with nasal fentanyl, one type of pain occurred in five patients and two types in ten. In the group of patients treated with buccal fentanyl, one type of pain was present in two patients and two types in seven.
Assessments was repeated during five subsequent nursing procedures inducing procedural pain episodes, carried out over 3–4 days. Each time, pain intensity in the previous 24 hours was assessed using the Brief Pain Inventory – Short Form (BPI-SF) questionnaire, and pain intensity and adverse effects of the investigated drugs used in the treatment of procedural pain were evaluated according to a pain and adverse-effect questionnaire (PAEQ) devised by the authors. The study monitored the vital parameters of patients and possible adverse effects, in particular drowsiness, disturbances in consciousness, and respiratory depression. In addition, other side effects, such as nausea, vomiting, constipation, and pruritus, were evaluated. The intensity of adverse reactions was assessed with the PAEQ.
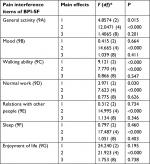
The intensity of background pain before administration of the investigated opioids before nursing-procedure commencement was assessed according to the BPI-SF, which was also used to assess pain occurring in the previous 24 hours. The BPI-SF allows assessment of pain location, pain intensity by NRS (pain at its worst, pain at its least, pain on average, pain right now), pain relief in percentage (0 – no pain relief, 100% –complete pain relief), and the negative impact of pain on the patient’s life dimensions (0 – no pain interference, 10 – complete pain interference): general activity, walking ability, normal work (at home and outside), relationships with other people and enjoyment of life.10 The European Organisation for Research and Treatment of Cancer (EORTC) QLQ-C15-PAL pain scale was used to assess background pain.11 QoL of patients was assessed using the EORTC QLQ-C15-PAL, which comprises 15 items on functioning scales – physical (3 items), emotional (2 items), and a single item of global QoL; symptom scales – pain (2) and fatigue (2); and single-symptom items – dyspnea, insomnia, appetite loss, nausea, and constipation. Both the BPI-SF and EORTC QLQ-C15-PAL have been adapted for a Polish population of cancer patients.10,11
To assess pain intensity before, during, and after nursing procedures, the PAEQ was used. This tool was also used to assess adverse effects. It comprises information regarding day and hour of nursing procedure, sex, age, education status, primary tumor location, type and location of pain, analgesics, and other drugs used. Pain intensity was assessed before nursing procedures that induced procedural pain and efficacy of drugs studied during nursing procedures within 60 minutes (every 5 minutes till 20 minutes after drug administration, and subsequently every 10 minutes till 60 minutes after drug administration), and also after a completion of nursing procedure by the NRS. Adverse-effect evaluation of drugs studied comprised drowsiness, nausea, vomiting, consciousness disturbances, respiratory disturbances, and skin pruritus, all assessed by a 5-point verbal scale: 0 – no, 1 – mild, 2 – moderate, 3 – strong, and 4 – very strong. Before drug administration and after completion of a nursing procedure (after approximately 60 minutes), vital signs, such as blood pressure, heart rate, and oxygen saturation, were measured. These assessments were repeated in each of five procedural pain episodes. Cognitive functions of patients were evaluated before the first procedural pain episode using the Mini-Mental State Examination (MMSE).12
Analgesic efficacy and adverse effects were assessed for 60 minutes after administration of drugs according to the PAEQ. Intensity of pain was assessed before administration of the opioid, followed by administration every 5 minutes to the 20th minute and every 10 minutes to the 60th minute after drug administration, using the PAEQ. Intensity of pain was assessed before opioid administration of prior to nursing procedures causing pain during the first and fifth procedural pain episodes, according to the pain scale in the EORTC QLQ-C15-PAL.
Before nursing procedures one of three opioids was administered according to the physician’s recommendation. Doses of the opioids investigated were chosen individually, wherein they had been titrated previously in the treatment of spontaneous breakthrough pain episodes in patients receiving nasal and buccal fentanyl. Patients treated with intravenous morphine received individually titrated doses, based on the effectiveness of the drug in the treatment of spontaneous breakthrough pain. In 15 patients in group 1, 1–3 mg midazolam was administered intravenously along with morphine before nursing procedures to induce sedation during procedural pain. Background pain in the majority of patients was treated with one opioid: morphine, fentanyl, oxycodone, or buprenorphine in 31 (70.45%) patients. The remaining 13 patients (29.55%) were treated with two or three opioids at the same time: morphine with fentanyl, fentanyl with oxycodone, morphine with buprenorphine, morphine with oxycodone and fentanyl, and morphine with oxycodone and buprenorphine. Application of research tools is presented in Table 1.
Statistical analysis
Statistical analysis was performed with the use of Statistica (StatSoft version 12). Results are presented as mean ± SD and ranges. EORTC QLQ-C15-PAL results were calculated in accordance with EORTC recommendations. Raw results were calculated for individual scales and symptoms:
|
where I1+ I2 + . . . + In indicates results for individual questions, with n indicating the number of questions in a particular scale. Then, a linear transformation of the results was carried out, ranging from 0 to 100. Results for the unchanged scale of pain and individual symptoms (dyspnea, insomnia, loss of appetite, and constipation) were calculated according to recommendations for calculating the results for the EORTC QLQ-C30:13
|
where S indicates the result, RS the raw result, and range the following values: for questions 1–14 it is 3, and for question 15 it is 6. Questions concerning overall QoL in EORTC QLQ-C15-PAL were calculated as autonomous: result = ([raw result – 1]/6) × 100. Results of shortened functioning scales EORTC QLQ-C15-PAL – physical and emotional functioning, fatigue scale, and a single question on nausea – were calculated according to the table prepared by EORTC.14
Age and QoL were continuous quantitative variables. Comparison of ages in individual groups was performed with ANOVA. Sex comparison between groups was carried out with χ2 independence tests. Data on pain intensity and adverse effects of opioids studied were discrete quantitative variables described on a 0–10 scale and semiquantitative ones described on a 0–4 scale, respectively. Distributions of these data were close to normal distribution (Shapiro–Wilk test), and the condition of homogeneity of variance was met in the vast majority of the tested parameters (Levene’s test).For comparative analysis of questionnaire results (BPI-SF, EORTC QLQ-C15-PAL, PAEQ, and Karnofsky performance status), repeated-measure two-way ANOVA was applied. Two main effects were examined: the effect of the type of treatment (intravenous morphine, nasal fentanyl spray, and fentanyl buccal tablets) and time of treatment and their interaction. As the condition of normality of distribution and homogeneity of variance was not met, the Kruskal–Wallis test was used to compare MMSE among the examined groups. Statistically significant results were assumed for P<0.05.
Results
The age (mean ± SD) of all patients was 65.7±14.4 years. The age of patients treated with intravenous morphine (n=20) was 63.6±15.2 years, fentanyl nasal spray (n=15) 70.1±13.9 years, and fentanyl buccal tablets (n=9) 63.2±13.1 years, and did not differ (F458, 2=1.055, P=0.357). Within the total group of 44 patients, there were 18 (40.9%) men and 26 (59.1%) women. Those treated with intravenous morphine, numbered seven (35.0%) and 13 (65.0%), those treated with fentanyl nasal spray six (40.0%) and nine (60.0%), and those treated with fentanyl buccal tablets 5 (55.6%) and 4 (44.4%) men and women, respectively. Patient sex did not differ (χ2=1.092, df 2; P=0.579). Patients differed on MMSE scores at baseline. The highest scores achieved were by patients treated with buccal fentanyl (28.6±2.7, range 23–30), lower scores those treated with nasal fentanyl (25.1±4.5, range 16–30), and lowest scores patients treated with intravenous morphine (24.0±5.1, 16–30; P=0.04).
Analgesic effects
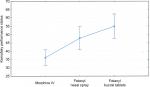
In patients treated at the inpatient Palliative Medicine Department with morphine administered intravenously, as well as in patients treated at home (home hospice) with fentanyl nasal spray and fentanyl buccal tablets administered before the start of nursing procedures causing severe pain, the reduction in background-pain intensity (assessed before nursing procedures) was obtained, which was expressed by a decrease in mean values of pain intensity on the NRS (statistically significant differences) for pain at its worst, pain at its least, and average pain at the time of questionnaire completion and by an increase in BPI-SF pain relief in all five procedural pain episodes. Significantly lower background pain intensity was shown on the EORTC QLQ-C15-PAL in all three investigated groups of patients between first and fifth examination. Intravenous morphine, fentanyl nasal spray, and fentanyl buccal tablets reduced the intensity of procedural pain induced by nursing procedures in a similar way, which was observed on the PAEQ (NRS). Initial mean pain intensity was assessed by patients between 3 and 5 (NRS), and within 15 minutes after administration of the opioid, had decreased to an average value <1 (Figure 1).
During the time from the start of opioid administration to achieving effective analgesia, it can be concluded that all three drugs induced a significant reduction in the initial intensity of pain (NRS), which correlated with a significant improvement in overall QoL. No statistically significant differences in degree or speed of analgesic effects of the investigated drugs were found, which was observed 5 minutes after administration, with a further downward trend, to <1 in NRS observed within 30 minutes, which remained on a constant level throughout the entire time of nursing procedures up to 60 minutes. Similar analgesic effects were observed in all five procedural pain episodes. In all five, after each drug administration and after completion of nursing procedures, a decrease in pain intensity was found – an effect of time (P<0.000). No differences were found regarding drug effect or drug–time interaction effect (Table 2).
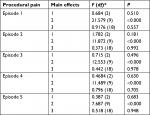
BPI-SF scales enable assessment of negative pain interference with a patient’s life: general activity, walking ability, normal work (at home and outside), relationships with other people, and enjoyment of their lives. Significant improvement was observed in all these dimensions during the five examinations: mostly similar during the use of the three investigated drugs. However, a smaller negative effect of pain on overall activity, walking ability, and normal work was seen in patients treated with fentanyl nasal spray and fentanyl buccal tablets compared to patients treated with intravenous morphine (Table 3). Interactions of drug type and treatment time in terms of pain interference with overall activity of patients are presented in Figure 2.
The interference of pain with walking ability depending on the opioid used is shown in Figure 3.
Adverse effects
Adverse effects observed in patients were usually of mild intensity. One patient in whom fentanyl was administered by the intranasal route at a dose of 50 µg before the nursing procedure was discontinued due to intense drowsiness. In terms of the assessment of the number of breaths per minute performed using the PAEQ, reductions in the number of breaths to ten to eleven per minute were observed: in the first procedural pain episode in four (9.09%), in the second in seven (16.28%), in the third in two (4.88%), in the forth in three (8.11%), and in the fifth in two (6.25%) patients (Table 4).
  | Table 4 Evaluation of number of breaths per minute on the pain and adverse-effect questionnaire |
There was no case of decrease in number of breaths below ten breaths per minute, which is a symptom of respiratory depression. Based on analysis of drowsiness intensity on the PAEQ, it can be concluded that the administered drugs did not cause increased drowsiness. After administration of morphine, a slight increase was initially observed, but was followed by a constant level of drowsiness. After intranasal administration of fentanyl, a gradual decrease in drowsiness occurred up to 30 minutes after drug administration to the level of drowsiness observed prior to drug administration. However, after administration of buccal fentanyl, a gradual decrease in drowsiness was observed. Interactions of the effect of treatment type and time on drowsiness according to the PAEQ are shown in Figure 4.
In the first procedural pain episode after administration of opioids before nursing procedures, a slight increase in consciousness disorders (1 on a 5-point scale) was observed in two (4.55%) patients, in the second procedural pain in three (6.98%) patients, and during the third and fourth procedural pain in one (2.70%) patient. For the last procedural pain episode, no disturbances of consciousness were observed. The intensity of disturbances in consciousness is presented in Table 5.
  | Table 5 Intensity of consciousness disturbances (pain and adverse-effect questionnaire) Note: Consciousness disturbances: 0 – none, 1 – mild, 2 – moderate, 3 – strong, 4 – very strong. |
The intensity of gastrointestinal (GI) symptoms was assessed with the PAEQ and EORTC QLQ-C15-PAL. Nausea and vomiting according to the PAEQ were characterized as small intensity (Table 6). Nausea of mild intensity occurred during the first examination in one (2.27%) patient and in two (4.65%) patients during the second and third examinations. No symptoms were observed during the remaining examinations.
  | Table 6 Assessment of nausea and vomiting intensity (pain and adverse-effect questionnaire) Note: Nausea and vomiting intensity: 0 – none, 1 – mild, 2 – moderate, 3 – strong, 4 – very strong. |
Nausea assessed with the EORTC QLQ-C15-PAL showed a downward trend during treatment with intravenous morphine and nasal fentanyl, remaining constant when using fentanyl buccal tablets. Vomiting assessed according to the PAEQ (Table 4) occurred in one (2.33%) patient during the second examination and in two (4.88%) patients in the third examination. According to the EORTC QLQ-C15-PAL, constipation was reduced to a similar extent as in patients treated with the three opioids tested. According to the PAEQ, pruritus of low intensity appeared in one (2.33%) of the patient during the second examination. Selected vital parameters (blood pressure, pulse, and oxygen saturation) were assessed before the administration of opioids and after the procedure. The values of all tested parameters were within normal limits and did not change significantly.
Quality of life
QoL was evaluated with the EORTC QLQ-C15-PAL questionnaire, assessing physical and emotional functioning and overall QoL. In terms of physical functioning, differences were observed depending on the type of treatment: higher level of physical functioning characterized patients treated with nasal and buccal fentanyl compared to patients treated with intravenous morphine (drug effect, P<0.0001). At the same time, the level of physical function remained constant (Figure 5).
On scales of emotional functioning (Figure 6) and overall QoL (Figure 7), an improvement was observed in all the examined groups of patients (time effect, both P<0.0001).
With the use of Karnofsky performance status, the effect of the drug was observed. Significantly lower activity was demonstrated by patients treated with morphine, while higher activity characterized patients receiving the fentanyl products (drug effect, P=0.0003). However, no effect of time or interaction was found (Figure 8).
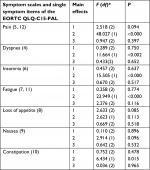
ANOVA results of symptom scales of the EORTC QLQ-C15-PAL are shown in Table 7. An effect of time was found in pain, dyspnea, insomnia, fatigue, and constipation: all these symptoms improved. However, neither drug effect nor interaction of drug and time effect was found in any symptom scales or single items.
Discussion
This clinical research concerned the comparison of analgesic and adverse effects and QoL of cancer patients treated in the inpatient Palliative Medicine Department and at home (home hospice), who due to severe procedural pain (NRS >6) required the use of opioid analgesics: intravenous morphine, most often in combination with midazolam (patients treated in the inpatient Palliative Medicine Department), fentanyl spray administered by nasal route and fentanyl tablets administered by buccal route, and both fentanyl products used in patients treated at home (home hospice). In the majority of patients, there occurred mixed pain with the receptor and neuropathic component (70% of patients experienced at least two types of pain), and classification in subgroups for type of pain was based on the predominant type of pain in a given patient.
Considering the fact that at the time of procedures before opioid treatment administered previously, patients assessed pain intensity as 6–10 on the NRS (before opioid administration, intensity of pain was 3–5 on the NRS), lowering pain intensity to <1 can be considered a very good analgesic effect, comparable for all three investigated drugs. Analysis of time from administration of opioids to the achievement of effective analgesia leads to the conclusion that all three drugs caused significant reductions in initial pain intensity assessed by the NRS, which correlated with a significant improvement in overall QoL. During the study, no significant differences in degree or speed of analgesic effect of the studied drugs were observed. This was monitored 5 minutes (till 20 minutes) after administration, with a further downward trend to <1 on the NRS scale observed after 30 minutes, which remained constant throughout nursing procedures, up to 60 minutes. Similar analgesic effects were observed in the next five examinations. On the one hand, the obtained results show that the treatment of background pain was more effective, and on the other hand, the increase in effectiveness of spontaneous breakthrough pain and procedural pain therapy, which was the subject of the study.
The BPI-SF questionnaire allows assessment of the negative impact of pain on patients’ life dimensions. On all pain-interference scales, significant improvements were obtained in all five nursing procedures inducing procedural pain, and were mostly similar across the three investigated drugs. However, a smaller negative effect of pain interference on overall activity, walking ability, and normal work was observed in patients treated with fentanyl nasal spray and fentanyl buccal tablets compared to patients treated with intravenous morphine. However, these observations should be interpreted with caution, due to the fact that patients treated with morphine in the inpatient Palliative Medicine Department required more intensive treatment of background pain and were less active than those treated with fentanyl at home. A similar analgesic effect of the studied drugs was indicated by improvement in performance of normal work and reduction in the negative impact of pain on enjoyment of life. Negative effects of pain on general activity, work at home and outside, and ability to walk after intranasal and buccal administration of fentanyl were smaller compared to intravenous morphine. The use of fentanyl administered with transmucosal route is also supported by a convenient route of administration, as well as no need for central or peripheral intravenous contact. A beneficial analgesic effect was assessed by the patients, with no significant differences between the studied drugs. There was no significant difference in the start of the analgesic effect among the investigated drugs.
Comparison of analgesic effects obtained in the treatment of severe procedural pain caused by nursing procedures in the literature is difficult, due to the lack of reports comparing intravenous morphine therapy in those treated as inpatients with rapid-onset fentanyl products administered intranasally and buccally at home. Available publications concern the use of tested drugs in the treatment of spontaneous breakthrough pain. The European Society for Medical Oncology recommends the use of transmucosal (nasal, buccal, sublingual, oral) fentanyl products, which provide fast onset of analgesia in the treatment of unpredictable spontaneous breakthrough-pain episodes with rapid-onset and immediate-release opioids before predictable pain episodes.15 Sevarino et al pointed out that fentanyl citrate administered via the transmucosal route provides relief from pain over a period comparable to that obtained after administration of intravenous morphine sulfate (approximately 5–10 minutes).16 Zeppetella et al found that the average time to a significant analgesic effect in the treatment of breakthrough pain after oral administration of morphine, methadone, oxycodone, and hydromorphone and fentanyl tablets used buccally was within 5–75 minutes (35 minutes on average). No differences were found between morphine, oxycodone, and hydromorphone. Methadone acted more quickly than morphine, but no more quickly than oxycodone or hydromorphone, while fentanyl provided faster analgesia compared to other opioids.17
The literature lacks comparisons of the analgesic effects of intravenous morphine and fentanyl administered by the transmucosal route used to treat predictable episodes of incident and procedural pain. In publications comparing the analgesic efficacy of oral morphine and transmucosal fentanyl, greater analgesic effectiveness of fentanyl products was demonstrated in a study conducted in 134 patients.18 A similar study, in which a more beneficial analgesic effect was provided by transmucosal fentanyl compared to morphine, was carried out by Hanks et al, indicating the side effects of fentanyl: nausea, mucositis, and dizziness, but no life-threatening adverse reactions were found.19
Adverse effects observed in the examined patients were usually of mild severity on the PAEQ. The tested drugs did not cause increased drowsiness. After administration of morphine, a slight increase was initially observed, followed by a constant level of drowsiness. After intranasal administration of fentanyl, a gradual decrease in drowsiness was observed up to 30 minutes after drug administration, followed by a return to the level of drowsiness experienced prior to drug administration. However, after the use of fentanyl by the buccal route, a reduction in drowsiness was observed in the fifth examination. Only one patient in whom fentanyl was administered via the intranasal route at a dose of 50 µg discontinued the treatment due to intense drowsiness. The patient again used fentanyl by the nasal route with very good analgesic effect after the end of the study, when he received higher doses of opioids in the treatment of background pain.
There was no decrease in the number of breaths fewer than ten per minute, which is a symptom of respiratory depression, from which it can be concluded that the treatment of procedural pain with nasal and buccal fentanyl is safe in patients at home. However, for the safety of therapy, it is recommended to have naloxone and knowledge of the proper use of the drug in the event of symptoms of respiratory depression, especially at home. Clinical studies confirm that with the appropriate opioid dosing, the risk of respiratory depression in cancer patients is low, which is associated with stimulation of the respiratory center by pain, provided that the opioid dose is carefully titrated and clinical observation accurate.20 Kongsgaard et al conducted a study of 107 patients who were treated with intranasal fentanyl, showing good analgesic effects and no life-threatening adverse reactions in the treatment of breakthrough pain.21
A prerequisite for safe and effective therapy of severe procedural pain induced by nursing procedures is the selection of patients for such treatment who regularly receive opioids for background pain (opioid-tolerant patients) and a careful titration of study drugs in the treatment of spontaneous breakthrough pain. The principle of titration of the opioid dose used in the treatment of spontaneous breakthrough pain and procedural pain induced by nursing procedures concerns especially fentanyl products with a rapid onset of analgesic action. Such a procedure also applies to patients who have been previously treated with another opioid, another route of administration, or different fentanyl product administered through the same route, eg, intranasal fentanyl (water aerosol and pectin spray) and sublingual fentanyl (sublingual tablets and buccal tablets). What is more, the principle of titration of fentanyl (prior to nursing procedures) in the treatment of spontaneous and procedural breakthrough pain is also applicable to patients with significant changes in the treatment of background pain (significant opioid-dose change, opioid rotation).
Opioids may affect cognitive functions and cause disturbances in consciousness. In the first procedural pain episode after administration of opioids before the nursing procedures (intravenous morphine, fentanyl administered by nasal route, and fentanyl by the buccal route), a slight increase of consciousness disturbances (1 on a 5-point scale) was observed in two (4.55%) patients, in three (6.98%) patients in the second nursing procedure, and in one (2.70%) patient during the third and fourth procedures. In the last procedural pain episode, no disturbances of consciousness were observed. Impaired consciousness may be a symptom of neurotoxicity induced by the accumulation of morphine metabolites, especially in patients with renal and hepatic impairment and those >70 years of age, hence the precise observation of patients and the correct titration of the dose is very important.22
GI side effects, ie, nausea and vomiting, were of minor intensity. According to the PAEQ, nausea of low intensity occurred during the first examination in one (2.27%) patients during the second and third examinations in two (4.65%) patients, and during the remaining tests, the symptom was not observed. Nausea assessed with the EORTC QLQ-C15-PAL showed a downward trend during morphine and fentanyl treatment administered by the intranasal route, remaining at a constant level when using fentanyl by the buccal route. Vomiting investigated according to the PAEQ was observed during the second study in one (2.33%) patient and in the third study in two (4.88%) patients. Constipation according to the EORTC QLQ-C15-PAL was reduced to a similar level as in patients treated with the three study drugs. According to the PAEQ, low-intensity pruritus appeared in the second study in one (2.33%) patient. The prevention of constipation, as well as the frequent use of antiemetics in patients treated with opioids, may have contributed to the low or absent symptoms.
Prior to administration of opioids and after procedure, selected vital parameters (blood pressure, heart rate, and oxygen saturation) were within normal range and did not show significant changes. The rare occurrence and low intensity of side effects were probably related to the proper titration and prevention of adverse effects of opioids. These observations suggest that treatment with intravenous morphine and intranasal and buccal fentanyl prior to nursing procedures inducing severe procedural pain is safe and does not cause any disturbing symptoms in the GI tract.
The observed improvement in QoL (assessed according to the EORTC QLQ-C15-PAL) in all three groups of patients is difficult to interpret, due to the lack of reports comparing QoL in patients treated with intravenous morphine and fentanyl products used before nursing procedures inducing severe procedural pain. The improvement in analgesia observed in the study during the nursing procedures probably had a positive effect on improvements in emotional functioning, overall QoL, and quality of sleep to a similar degree in the three groups of patients. Positive influence on the QoL of patients could be affected by more effective treatment of background and procedural pain, as well as reduced intensity of such symptoms as dyspnea, insomnia, fatigue, and constipation. Improved mood and less severe sleep disorders are the result of more effective treatment of pain and other symptoms, which is reflected in improvement in overall QoL. It should be noted that in this dimension of symptom treatment, slightly better effects were demonstrated by fentanyl administered by intranasal and buccal routes, and slightly less impact on improvement in overall QoL was observed in patients treated with intravenous morphine. A beneficial effect of symptom treatment on QoL was demonstrated by Cleeland.23 Obtained results confirm the legitimacy of applying tools assessing the effectiveness of analgesic therapy and monitoring QoL, which is significantly related to the intensity of pain and other symptoms in cancer patients.
Worse physical outcomes in patients treated with morphine probably resulted from the lower level of physical functioning of patients who demonstrated lower Karnofsky performance status and required more intense pain treatment compared to patients treated with both fentanyl products at home. According to Karnofsky performance status, the effect of the drug was observed, whereas significantly lower activity was demonstrated by patients treated with morphine, while higher activity was observed in patients receiving the investigated fentanyl products. However, it needs to be emphasized that the aforementioned differences did not result from the type of the opioids used, but from more the severe general condition of inpatients treated with morphine due to pain, which was more difficult to treat. This thesis can be confirmed with the fact that in this group of patients, morphine treatment of background pain was the most frequently used in constant parenteral infusions, a concurrent administration of two opioids, usually with adjuvant analgesics. There was no effect of time or interaction, which indicates that in the advanced phase of cancer, it is usually very difficult or impossible to improve activity of patients.
Limitations of the study
A significant limitation of the study is the small number of patients included, which resulted from considerable difficulties in recruiting patients, due to, among other factors, excluding patients due to their severe general condition and the need to fill several research tools. Other limitations include lack of randomization and blinding. Another potential source of bias may be a comparison of patients treated at home and at the inpatient Palliative Medicine Department, which may represent different populations, ie, more intense pain, lower activity, and worse cognitive function in the latter compared to the former. Despite these limitations, this study compared drugs for analgesia during nursing procedures inducing severe procedural pain, adverse effects, and cancer patient QoL. The comparison was performed during five procedural pain episodes with the use of several methods of pain, adverse-effect, and QoL assessment.
Recommendations for clinical practice
To ensure safe treatment and reduce the risk of adverse effects, we recommend careful titration of investigated opioids, particularly intranasal and buccal fentanyl, during the treatment of spontaneous breakthrough-pain episodes in opioid-tolerant patients. Fentanyl products administered by intranasal and buccal routes are characterized by high analgesic efficacy and rapid onset of analgesia, as well as a noninvasive route of administration, which suggest their usefulness in the treatment of severe procedural pain induced by nursing procedures, especially at home.
Conclusion
To our knowledge, this is the first study to demonstrate that intravenous morphine and nasal and buccal fentanyl display similar, satisfactory analgesic efficacy in the treatment of severe procedural pain induced by nursing procedures in cancer patients. During treatment with nasal and buccal fentanyl, less negative influence of pain was observed on patient overall activity, walking ability, and normal work compared to morphine therapy. Morphine administered intravenously and fentanyl by the intranasal and buccal routes show a similar profile, usually a low intensity of adverse effects, and improved patient QoL.
Disclosure
The authors report no conflicts of interest in this work.
References
Haugen DF, Hjermstand MJ, Hagen N, Caraceni A, Kassa S; European Palliative Care Research Collaborative (EPCRC). Assessment and classification of cancer breakthrough pain: a systematic literature review. Pain. 2010;149:476–482. | ||
Manchikonti L, Singh V, Carawaj DL. Breakthrough pain in chronic non–cancer pain: fact, fiction or abuse. Pain Physican. 2011;14:103–117. | ||
Portenoy RK, Hagen NA. Breakthrough pain: definition, prevalence and characteristics. Pain. 1990;41(3):273–281. | ||
Davies AN, Dickman A, Reid C, Stevens AM, Zeppetella G; Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. The management of cancer-related breakthrough pain: recommendations of a task group of the Science Committee of the Association for Palliative Medicine of Great Britain and Ireland. Eur J Pain. 2009;13(4):331–338. | ||
Løhre ET, Klepstad P, Bennett MI, et al. From “breakthrough” to “episodic” cancer pain? A European Association for Palliative Care Research Network Expert Delphi Survey toward a common terminology and classification of transient cancer pain exacerbations. J Pain Symptom Manage. 2016;51(6):1013–1019. | ||
Bobb BT, Coyne PJ. When cancer pain breaks through, what can you do? Oncology. 2010;24(Issue 10). Available from: http://www.cancernetwork.com/oncology-nursing/when-cancer-pain-breaks-through- what-can-you-do. Accessed October 1, 2018. | ||
Bennett D, Burton AW, Fishman S. Consensus panel recommendations for the assessment and management of breakthrough pain. Part I assessment. P&T. 2005;30:296–301. | ||
Vellucci R, Mediati RD, Gasperoni S, Mammucari M, Marinangeli M, Romualdi P. Assessment and treatment of breakthrough cancer pain: from theory to clinical practice. J Pain Res. 2017;10:2147–2155. | ||
Schmidt R, Schmelz M, Ringkamp M, Handwerker HO, Torebjörk HE. Innervation territories of mechanically activated C nociceptor units in human skin. J Neurophysiol. 1997;78(5):2641–2648. | ||
Leppert W, Majkowicz M. Polish brief pain inventory for pain assessment and monitoring of pain treatment in patients with cancer. J Palliat Med. 2010;13(6):663–668. | ||
Leppert W, Majkowicz M. Validation of the polish version of the European organization for research and treatment of cancer quality of life questionnaire – core 15 – palliative care in patients with advanced cancer. Palliat Med. 2013;27(5):470–477. | ||
Folstein M, Folstein SE, McHugh PR. “Mini-Mental State” a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198. | ||
Fayers PM, Aaronson N, Bjordal K, Groenvold M, Curran D, Bottomley A. The EORTC QLQ-C30 Scoring Manual. 3rd ed. Brussels: European Organization for Research and Treatment of Cancer; 2001. | ||
Groenvold M, Petersen MA; On Behalf of the EORTC Quality of Life Group. Addendum to the EORTC QLQ-C30 scoring manual: scoring of the EORTC QLQ-C15-PAL. Brussels, Belgium: EORTC Quality of Life Unit; 2006. | ||
Fallon M, Giusti R, Aielli F; On Behalf of the ESMO Guidelines Committee. Management of cancer pain in adult patients: ESMO clinical practice guidelines. Ann Oncol. 2018;29(suppl. 4):iv149–iv174. | ||
Sevarino FB, Ginsberg B, Lichtor JL, et al. Oral Transmucosal Fentanyl Citrate (OTFC) compared with IV morphine for acute pain in patients following abdominal surgery. Anesth Analg. 1997;84:S330. | ||
Zeppetella G. Opioids for cancer breakthrough pain: a pilot study reporting patient assessment of time to meaningful pain relief. J Pain Symptom Manage. 2008;35(5):563–567. | ||
Coluzzi PH, Schwartzberg L, Conroy JD, et al. Breakthrough cancer pain: a randomized trial comparing oral transmucosal fentanyl citrate (OTFC) and morphine sulfate immediate release (MSIR). Pain. 2001;91:123–130. | ||
Hanks GW, Nugent M, Higgs CMB, Busch MA; OTFC Multicentre Study Group. Oral transmucosal fentanyl citrate in the management of breakthrough pain in cancer: an open, multicentre, dose-titration and long-term use study. Palliat Med. 2004;18(8):698–704. | ||
Osborne R, Joel S, Trew D, Slevin M. Morphine and metabolite behavior after different routes of morphine administration: demonstration of the importance of the active metabolite morphine-6-glucuronide. Clin Pharmacol Ther. 1990;47(1):12–19. | ||
Kongsgaard UE, Eeg M, Greisen H. The use of Instanyl in the treatment of breakthrough pain in cancer patients: a 3-month observational, prospective, cohort study. Support Care Cancer. 2014;22(6):1655–1662. | ||
Pergolizzi J, Böger RH, Budd K, et al. Opioids and the management of chronic severe pain in the elderly: consensus statement of an International Expert Panel with focus on the six clinically most often used World Health Organization step III opioids (buprenorphine, fentanyl, hydromorphone, methadone, morphine, oxycodone). Pain Pract. 2008;8(4):287–313. | ||
Cleeland CS. The impact of pain on the patient with cancer. Cancer. 1984;54(11 Suppl):2635–2641. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.