Back to Journals » Drug Design, Development and Therapy » Volume 16
Comparison of 3 Rates for the Continuous Infusion of Mivacurium During Ambulatory Vitreoretinal Surgery Under General Anesthesia: A Prospective, Randomized, Controlled Clinical Trial
Authors Zhang Y , Xi C, Yue J, Zhao M, Wang G
Received 16 April 2022
Accepted for publication 5 September 2022
Published 16 September 2022 Volume 2022:16 Pages 3133—3143
DOI https://doi.org/10.2147/DDDT.S370978
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Tin Wui Wong
Yi Zhang,1,* Chunhua Xi,1,* Jianying Yue,1 Mengmeng Zhao,2 Guyan Wang1
1Department of Anesthesiology, Beijing Tongren Hospital, Capital Medical University, Beijing, People’s Republic of China; 2Operation Center, Beijing Tongren Hospital, Capital Medical University, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Guyan Wang, Department of Anesthesiology, Beijing Tongren Hospital, Capital Medical University, 1 Dongjiaominxiang Road, Dongcheng District, Beijing, 100730, People’s Republic of China, Tel +86-10-58268101, Email [email protected]
Purpose: Mivacurium, the shortest-acting benzylisoquinoline nondepolarizing neuromuscular blocker used in clinical practice, is suitable for short-term ambulatory operations under general anesthesia. We investigated the neuromuscular blockade effect of different maintenance doses of mivacurium during ambulatory vitreoretinal surgery under general anesthesia and tried to determine the appropriate maintenance dose.
Patients and Methods: Ninety-nine patients undergoing general anesthesia for elective ambulatory vitreoretinal surgery were randomly divided into three groups using the random number table method. Patients received three maintenance doses of mivacurium during surgery as follows: 3 μg/(kg·min) in group M1 (n = 33), 6 μg/(kg·min) in group M2 (n = 33), and 9 μg/(kg·min) in group M3 (n = 33). The primary outcome was the time from mivacurium withdrawal to a train-of-four stimulation ratio (TOFr) ≥ 0.9, and the secondary outcomes were the time from mivacurium withdrawal to TOFr ≥ 0.7, extubation time, incidence of TOFr < 0.9 after surgery and neuromuscular block effect.
Results: The time from mivacurium withdrawal to TOFr ≥ 0.9 and to TOFr ≥ 0.7 was significantly longer in group M3 than in groups M1 and M2 (25.6± 7.2 min vs 16.4± 5.9 min and 18.6± 5.3 min, P < 0.001; 22.1± 6.3 min vs 13.6 ± 5.8 min and 15.5 ± 4.8 min; P < 0.001, respectively). There was a significant difference in the extubation time, the incidence of TOFr < 0.9 during extubation and upon leaving the operating room between group M3 and group M1 (all P < 0.05), but there was no such significant difference between group M2 and group M1 (all P > 0.05). The intraoperative depth of neuromuscular blockade in the three groups was significantly different, with 69.7% shallow block in group M1, 75.8% moderate block in group M2 and 63.6% deep block in group M3 (P < 0.001). One patient in group M1 experienced slight body movement during the operation.
Conclusion: An intraoperative continuous infusion of 6 μg/(kg·min) mivacurium can not only achieve good postoperative recovery but also provide a satisfactory neuromuscular blockade effect during surgery, and this maintenance dose is suitable for neuromuscular blockade during ambulatory vitreoretinal surgery.
Keywords: mivacurium, vitreoretinal surgery, neuromuscular blocking agents, neuromuscular monitoring, postoperative period
Introduction
Ambulatory surgery is a surgical mode in which the patient is admitted, operated on and discharged within one working day (24 hours).1 Ambulatory surgery is suitable for operations with less interference to physiological function, a shorter operation time, relatively less risk and fewer postoperative complications.2 Ophthalmic surgery usually lasts a short time, with a low risk of intraoperative and postoperative bleeding, and postoperative complications are easy to detect,3 making these procedures suitable for ambulatory surgery. With the development of general anesthesia techniques and drugs as well as the refinement of surgical techniques, ambulatory ophthalmic general anesthesia is becoming increasingly popular.4
Compared with other ophthalmic operations, vitreoretinal surgery takes longer with a relatively variable operation time according to the complexity of the operation. Vitreoretinal surgery requires the patient to remain completely immobile during surgery because instruments are near or in the eye, and any unexpected body movement may lead to surgical failure and ocular injury disasters.5,6 During ambulatory vitreoretinal surgery, adequate neuromuscular blockade is helpful for maintaining eyeball position, intraocular pressure and body immobility and avoiding using large doses of sedative and analgesic drugs.7 Meanwhile, ambulatory surgery requires fast physical functional recovery. A method for balancing the effective depth of muscle relaxation and avoiding postoperative residual neuromuscular blockage (rNMB) is worthy of exploration during ambulatory vitreoretinal surgery. Nondepolarizing intermediate-acting drugs such as rocuronium and cisatracurium have a long action time and often cause rNMB when applied during short ophthalmic surgeries, which is not conducive to the postoperative recovery of patients.8,9
Mivacurium, which was first synthesized in 1981, is currently the shortest-acting benzylisoquinoline nondepolarizing neuromuscular blocking agent (NMBA) used in clinical practice and is mainly hydrolyzed by plasma cholinesterase.10 Mivacurium has a short elimination half-life of 2.6–3 min, does not accumulate in the body, and does not produce rapid tolerance in the body after prolonged infusion in the absence of butyrylcholinesterase deficiency.11 The aforementioned advantages indicate the beneficial use of mivacurium as an ideal NMBA in ambulatory surgery anesthesia,12 such as in laparoscopic cholecystectomy, the removal of tracheal foreign bodies, the removal of vocal cord polyps, and pediatric surgery.13–15 However, the clinical use of mivacurium is mainly applicable to the tracheal intubation phase of anesthesia induction,16 and few clinical reports have described the application of mivacurium and the proper dose in ambulatory ophthalmic surgery. Therefore, we compared the neuromuscular blockade effect of three different maintenance doses of mivacurium to investigate its feasibility and the appropriate dose in ambulatory vitreoretinal surgery under general anesthesia.
Materials and Methods
Ethics
This prospective randomized study was conducted in accordance with the Declaration of Helsinki. Ethical approval for this study (No. TRECKY2021-138) was provided by the ethics committee of Beijing Tongren Hospital, Capital Medical University on September 15, 2021. Written informed consent was obtained from all participants. This trial was registered in the China Clinical Trial Registry (No. ChiCTR2100052351) before patient enrollment.
Participants
This study recruited eligible patients who underwent ambulatory vitreoretinal surgery under general anesthesia in the day surgery ward of Beijing Tongren Hospital, Capital Medical University, from November 1, 2021, to January 31, 2022. The inclusion criteria were patients aged 18 to 65 years; American Society of Anesthesiologists (ASA) class I and II; body mass index (BMI) of 18.5 to 30 kg/m2; and an operation time of less than 2 hours. The exclusion criteria included patients who refused to participate in the study; ASA class III or higher; patients with severe organ dysfunction, a significantly difficult airway, or neuromuscular system diseases; patients taking drugs that might affect neuromuscular conduction function or affect neuromuscular monitoring; patients who were allergic to the test drug or had other contraindications; and pregnant and lactating women.
Randomization and Blinding
Patients were randomly divided into 3 groups using a computer-generated random number list (IBM SPSS Statistics 26.0, IBM Corp, Armonk, NY, USA) and received one of the following 3 infusion doses of mivacurium to maintain neuromuscular blockade during surgery: 3 μg/(kg·min) in the M1 group, 6 μg/(kg·min) in the M2 group, and 9 μg/(kg·min) in the M3 group. The group assignment for each patient was sealed in an opaque envelope by an assistant who was not involved in the clinical anesthesia, data collection and analysis. Two anesthesiologists (Dr. Xi and Dr. Yue) administered mivacurium to the different groups according to the envelope contents and did not know the results of the neuromuscular monitoring throughout the study. The data collector (Dr. Zhang) and postoperative evaluator (Miss Zhao) were blinded to the treatment allocation.
Procedure and Intervention
The general anesthesia protocol was the same for all patients, except for the intraoperative administration of different doses of mivacurium for maintenance. Patients were routinely fasted from food and water for 8 hours before surgery. After arriving at the operating room, the patient’s venous access was opened, and routine monitoring, including blood pressure (BP), heart rate (HR), pulse oxygen saturation (SpO2), and bispectral index (BIS), was conducted.
Neuromuscular monitoring was performed with an acceleromyography (AMG) neuromuscular monitor (JS-100, Beijing Silugao Medical Technology Co., Beijing, China) to stimulate the right ulnar nerve and monitor contraction of the adductor pollicis muscle as previously described.17 After midazolam 0.05 mg/kg and sufentanil 0.1 μg/kg were administered to sedate the patients, neuromuscular monitor calibration was performed, and train-of-four stimulations (TOF) were selected to start monitoring until the postoperative train-of-four stimulation ratio (TOFr) ≥ 0.9. A dedicated researcher (Dr. Zhang) who did not participate in clinical anesthesia observed and recorded the data.
The anesthetic induction drugs were 2 mg/kg propofol, 0.25 μg/kg sufentanil, and 0.25 mg/kg mivacurium. After 4 min of assisted ventilation, a flexible laryngeal mask (LMA) was placed to establish the airway, and the tidal volume was set at 6~8 mL/kg to maintain the partial pressure of end-tidal carbon dioxide (EtCO2) at 35~40 mmHg. During the operation, total intravenous anesthesia was performed, with a continuous infusion of 0.1~0.2 μg/(kg·min) remifentanil and 4~10 mg/(kg·h) propofol to maintain the BIS value between 40~60. After induction, the itinerant nurse handed the opaque envelope containing the random number to the anesthesiologist, and 3 μg/(kg·min), 6 μg/(kg·min), or 9 μg/(kg·min) mivacurium was continuously infused to maintain neuromuscular blockade according to the group assignment. In cases of unexpected movement or airway pressure increases of more than 3 cmH2O during the operation, an additional 0.1 mg/kg of mivacurium was administered.18 All drug infusions were stopped at the end of surgery. Neuromuscular blockade antagonists were routinely not used after surgery to observe rNMB, and the anesthesiologist judged the time of extubation according to the clinical criterion.5 After the LMA was removed, the patient was transferred to the postanesthesia care unit (PACU), and the data collector and postoperative evaluator continued to monitor the TOFr value until TOFr ≥ 0.9.
Measurements and Data Collection
The primary outcome of this study was the time from withdrawal of mivacurium to TOFr ≥ 0.9, and the secondary outcome measures were the time from withdrawal of mivacurium to TOFr ≥ 0.7, extubation time, and incidence of TOFr < 0.9 at recovery. Other data related to neuromuscular blockade in the three groups were recorded, including the onset time (the time from administration to the time when T1 disappeared), clinical action time (the time from withdrawal to 25% T1 recovery) and recovery index (the time from 25% to 75% T1 recovery). Furthermore, the T1 value, TOF count (TOFc) and TOFr value were also recorded to evaluate the depth of intraoperative and postoperative neuromuscular blockade and the distribution of TOFr, and they were classified as a shallow block (TOF count = 4), moderate block (TOF count = 1–3) and deep block (TOF count = 0).19–21
The baseline characteristics of the patients were recorded. During general anesthesia, the intraoperative dose of anesthetic drugs, LMA placement by the fiberoptic bronchoscopy (FOB) Campbell score, oropharyngeal leak pressure (OLP), and peak airway pressure (PIP) were recorded. Hemodynamic data, including the mean arterial pressure (MAP), HR, SpO2, and BIS were recorded at 6 specific time points and every 15 min during the operation. The quality of postoperative recovery including the PACU score, length of PACU stay, postanesthesia discharge scoring system (PADSS) value, length of hospital stay and discharge time, was recorded. Adverse effects associated with the use of mivacurium, such as allergic reactions, including unstable hemodynamics and skin flushing, were recorded. Other adverse events, such as intraoperative limb movement, and adverse respiratory events (hypoxemia, upper airway obstruction, bronchospasm, reflux and aspiration) were also recorded.
Sample Size
The primary outcome measure in this study was the time from withdrawal of mivacurium to TOFr ≥ 0.9. Based on the results from the preliminary experiment (16.3±4.6 min in group M1, 19.3±6.4 min in group M2, and 21.4±5.3 min in group M3, 8 patients in each group) and based on a significance level of 0.05 and a power of 0.9, the sample size was calculated using PASS 11.0 (NCSS, LLC. Kaysville, UT, USA) according to a 1:1:1 parallel control study. Thirty patients in each group were required for this clinical trial. Considering loss to follow-up, the sample size was expanded to 34 patients in each group and 102 cases in all patients.
Statistical Analysis
IBM SPSS Statistics 26.0 was used for statistical analyses. The Shapiro–Wilk test was used to determine the normality of the data distribution, and the Levene method was used to test the homogeneity of variance. Normally distributed data are presented as the means ± standard deviations (x ± s), and one-way ANOVA and Tukey’s post hoc test for pairwise comparisons were used. Nonnormally distributed data are presented as the medians (interquartile ranges), and the Kruskal–Wallis test and Dunn’s post hoc test for pairwise comparisons were used. The enumerated data are reported as numbers or frequencies (%) and were compared using the chi-square test or Fisher’s exact test. P > 0.05 was considered statistically significant.
Results
One hundred twenty patients were recruited for this study. Nine patients were excluded due to a refusal to participate, 5 patients were excluded due to a BMI > 30 kg/m2, and 4 patients were excluded due to temporary cancellation of surgery. The remaining 102 patients were enrolled and randomized to receive different maintenance doses of mivacurium, which were 3 μg/(kg·min) in group M1, 6 μg/(kg·min) in group M2, and 9 μg/(kg·min) in group M3. One patient in each of the three groups dropped out of the study due to incomplete intraoperative data collection, an operation time longer than 2 hours, and a change in the surgical approach respectively. The final analysis was performed based on the data from 99 patients (Figure 1).
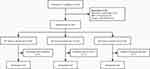 |
Figure 1 CONSORT flowchart of the study. |
The baseline characteristics of the patients, including their age, sex, BMI, ASA grade, and comorbidity and intraoperative characteristics, are presented in Table 1. No significant differences in the baseline characteristics were observed among the three groups (P > 0.05).
 |
Table 1 Patients and Anesthetic Characteristics |
Table 2 shows the primary and secondary outcomes. The time to TOFr ≥ 0.9 after discontinuation was significantly longer in the M3 group than in the M1 and M2 groups but was similar between the M1 and M2 groups (25.6 ± 7.2 min vs 16.4 ± 5.9 min and 18.6 ± 5.3 min, respectively; P < 0.001). The time to TOFr ≥ 0.7 was also significantly longer in the M3 group than in the M1 and M2 groups but was similar between the M1 and M2 groups (22.1±6.3 min vs 13.6 ± 5.8 min and 15.5 ± 4.8 min, respectively; P < 0.001). The time to extubation was significantly longer in the M3 group than in the M1 group (16.3 ± 4.6 min vs 13.0 ± 3.3 min, P = 0.009), but a significant difference was not observed between the M2 and M1 groups (15.2 ± 4.5 min vs 13.0 ± 3.3 min, P > 0.05) or the M2 and M3 groups (15.2 ± 4.5 min vs 16.3 ± 4.6 min). The M3 group had a significantly higher incidence of rNMB than the M1 group at extubation (90.9% vs 63.6%, P = 0.033), a higher incidence of rNMB than the M1 and M2 groups upon leaving the operating room (72.7% vs 33.3% and 39.4%, respectively; P = 0.003), and a higher incidence of rNMB when arriving at the PACU that was not significantly different from the M1 and M2 groups (33.3% vs 12.1% and 15.2%, respectively; P > 0.05).
 |
Table 2 Time to TOFr ≥ 0.9, TOFr ≥ 0.7, Extubation and rNMB During Recovery |
Other effects of mivacurium on neuromuscular blockade during surgery are shown in Table 3. Significant differences in the onset time of neuromuscular blockade or the recovery index during surgery were not observed among the three groups (P > 0.05). The clinical action time was longer in the M3 group than in the M1 group [11.0 (9.0–15.0) min vs 9.0 (8.4–10.3) min, P = 0.003], but a significant difference was not observed between the M2 group and the other two groups. The intraoperative depth of neuromuscular blockade in the three groups, namely, the distribution of shallow, moderate and deep neuromuscular blockade in the three groups, was significantly different (P < 0.001), with more shallow block detected in the M1 group (69.7%); moderate block was predominant in the M2 group (75.8%), and the majority of patients in the M3 group had a deep block (63.6%). Regarding the distribution of postoperative TOFr, significant differences were detected among the three groups (P < 0.05). In the M1 group, 15.2% of patients had already recovered to TOFr ≥ 0.9 at the end of surgery, and most patients had recovered to TOFr ≥ 0.9 when leaving the operating room (66.7%); 3% of patients in the M2 group recovered to TOFr ≥ 0.9 at the end of surgery, and 60.6% of patients recovered to TOFr ≥ 0.9 when leaving the operating room, similar to the M1 group. However, no patient in the M3 group recovered to TOFr ≥ 0.9 at the end of surgery, and only 27.3% recovered to TOFr ≥ 0.9 when leaving the operating room.
 |
Table 3 Other Effects of Neuromuscular Block of Mivacurium During Vitreoretinal Surgery |
The intraoperative hemodynamic data of MAP, HR and BIS values in the three groups are compared in Figure 2. Although significant differences were observed in the intragroup comparisons before and after induction (P < 0.05), the MAP, HR and BIS values of each group were similar, with no significant differences among the three groups at each recording time point during the operation (P > 0.05).
The postoperative recovery results are shown in Table 4. Among them, the scores of patients admitted and transferred out of the PACU and the incidence of hypoxemia during the PACU stay were similar in all three groups, with no significant differences (P > 0.05). In addition, significant differences in the length of PACU stay, length of hospital stay, time of discharge, or PADSS scores were not observed (P > 0.05).
 |
Table 4 Postoperative Recovery Quality |
All patients in the three groups completed the surgery. Among them, one patient each in the M1 and M2 groups exhibited transient skin flushing, and 1 patient in the M3 group showed transiently decreased BP and accelerated HR during induction. The three adverse events may be related to transient allergic reactions related to mivacurium. Notably, 1 patient in the M1 group experienced increased airway pressure and slight body movement during the operation, which was controlled by adding 0.1 mg/kg mivacurium, while the other two groups had no similar situation. The incidence of hypoxemia in the PACU, which was defined as fingertip pulse oxygen saturation SpO2 ≤ 90% under air inhalation, was 3.0% (1 patient), 9.0% (3 patients), and 9.0% (3 patients) in the M1, M2, and M3 groups, respectively, but a significant difference was not observed between the three groups (P > 0.05). Only 1 patient in the M3 group had a TOF ratio of 62% when arriving at the PACU, not excluding hypoxemia related to rNMB. The remaining 6 patients with hypoxemia had TOFr ≥ 0.9 when arriving in the PACU. Their hypoxemia was potentially associated with an insufficient respiratory rate due to drug residue from general anesthesia. Hypoxemia in these 7 patients was corrected by oxygen inhalation through the facial mask. Otherwise, none of the patients in the three groups had postoperative adverse events.
Discussion
This randomized clinical trial compared three maintenance doses of mivacurium during ambulatory vitreoretinal surgery. We found that intraoperative infusion of 6 μg/(kg·min) mivacurium can provide not only a lower incidence of rNMB compared with 9 μg/(kg·min) but also a more satisfactory moderate neuromuscular blockade for vitreoretinal surgery compared with 3 μg/(kg·min). We propose that maintenance anesthesia with 6 μg/(kg·min) mivacurium is more suitable for ambulatory vitreoretinal surgery than 3 μg/(kg·min) and 9 μg/(kg·min). Our study provides the basis for mivacurium application in ambulatory anesthesia.
Postoperative rNMB not only causes physical impairment, such as difficulty in respiration, swallowing and vision but also places patients at increased respiratory risk, such as hypoxemia, upper airway obstruction requiring intervention and postoperative pulmonary complications.22 TOFr < 0.9 indicates rNMB, and some studies considered a TOFr < 0.7 in the early stage.23,24 However, although many patients can perform powerful movements at a TOFr of 0.7, they still feel difficulty swallowing and completing the movements, and there is still a risk of aspiration.25 Studies in recent years utilizing the TOFr < 0.9 threshold have estimated that the proportion of patients with rNMB ranges from 4% to 88% at extubation, which has been confirmed in many countries.26,27 TOFr ≥ 0.9 is an indicator of satisfactory recovery of neuromuscular function, but some patients with TOFr ≥ 0.9 might be unable to tolerate intubation or swallow, resulting in displacement of the LMA during the recovery stage. In the absence of acceleromyography for various reasons, anesthesiologists determine rNMB by referring to clinical criteria (sustained head lift or hand grip for more than 5 s, the ability to follow simple commands, a stable ventilatory pattern with SpO2 greater than 95%). Extubation followed by close observation is clinically acceptable in these patients when they meet the clinical criteria for extubation. In our study, after comparing 3 rates of continuous infusion of mivacurium, we found that the time from discontinuation of the 6 μg/(kg·min) to TOFr ≥ 0.9 and to TOFr ≥ 0.7 was not longer than that of the 3 μg/(kg·min) dose but was significantly shorter than that of the 9 μg/(kg·min) dose. The incidence of rNMB at the time of extubation, upon leaving the operating room and upon arriving in the PACU was similar between the 3 μg/(kg·min) group and 6 μg/(kg·min) but was much higher in the 9 μg/(kg·min) group under the same criteria for clinical extubation in our study. In addition, the time from discontinuation to TOFr ≥ 0.7 was similar to the time of extubation in the 3 μg/(kg·min) group and 6 μg/(kg·min) group but was approximately 6 minutes longer than the time of extubation in the 9 μg/(kg·min) group, which means that when we followed the clinical indications to remove LMA, the TOFr of patients in the low-dose and medium-dose mivacurium maintenance groups was approximately 0.7, while those in the high-dose group were significantly lower than 0.7. Our results indicate that severe rNMB is more likely to occur in the high-dose group even if the clinical extubation criteria are met. Although significant differences in the PACU recovery time or other adverse events were not observed among the three groups, the high incidence of rNMB when arriving in the PACU in the 9 μg/(kg·min) group increased the potential risks in the PACU. It should be emphasized that our clinical study involved small samples, and all patients were closely observed for rNMB. We found that most patients who met the clinical indications for LMA removal reaching TOFr ≥0.7 but not ≥ 0.9. We suggest that if adequate equipment for rNMB monitoring is available, every patient undergoing general anesthesia and LMA with NMBAs should receive rNMB monitoring and meet the indicator of TOFr ≥ 0.9 to ensure safe and satisfactory recovery of neuromuscular function.
Few articles describe the depth of neuromuscular block required for eye surgery, as ophthalmic manipulation is less irritating and the eye muscles are slender. Therefore, the primary purpose of muscle relaxant application during vitreoretinal surgery is to keep the eyeball in the center position and tolerate endotracheal intubation ventilation. Deep neuromuscular block with the absence of a TOF response (TOFc = 0) is an appropriate depth of neuromuscular block in the cases of endotracheal intubation.19 With the use of supraglottic ventilation devices, NMBAs can be reduced or canceled by increasing the dosage of sedatives or analgesics. However, large doses of anesthetic drugs increase intraoperative hemodynamic instability, retard patient turnover in ambulatory surgery, and affect the quality of recovery. Recently, moderate neuromuscular block (TOFc = 1–2) has been reported to improve surgical conditions and reduce the incidence of adverse events in short-duration pediatric laparoscopic surgery with LMA.28 Whether LMA ventilation and moderate muscle relaxation are suitable for vitreoretinal surgery has not been reported.
Short-acting mivacurium was reported to decrease waiting time after deep neuromuscular blockage compared with intermediate-acting atracurium, rocuronium, or vecuronium.7 Early studies suggested that the infusion dose of mivacurium to maintain a 90%~99% muscle fibrillation inhibition effect should be 3~15 μg/(kg·min),29,30 but most of these studies were focused on tracheal intubation, and the recommended dose range is more extensive. During this study, we observed significant differences in the depth of neuromuscular blockade among the three different dose groups during the operation. The distribution of shallow neuromuscular blockade was greater in the low-dose group, resulting in one patient experiencing elevated airway pressure and slight body movement, with T1 reaching 64% and TOFr at 0.38. This result indicated that patients are in a state of minimal to shallow neuromuscular blockade and are at risk of intraoperative body movement when they receive an infusion of mivacurium at a 3 μg/(kg·min) dose. Therefore, we deduce that continuous infusion of mivacurium at 3 μg/(kg·min) may not be appropriate during vitreoretinal surgery, and when shallow block occurs (TOFc = 4), an additional muscular relaxant should be added. Mivacurium at 6 μg/(kg·min) provided moderate neuromuscular blockade in most patients, and no shallow neuromuscular blockade occurred in our study; it maintained a 90%~99% muscle fibrillation inhibition effect and was consistent with previous studies.31–33 Therefore, we believe that 6 μg/(kg·min) mivacurium provides a satisfactory neuromuscular blockade effect during vitreoretinal surgery.
Limitations
This study has some limitations. First, although TOFr ≥ 0.9 is strongly recommended for extubation, we still chose the clinical indications as the criteria for extubation because neuromuscular blockade monitoring is not used worldwide.19 However, we found that most of the patients could not reach TOFr ≥ 0.9 when the criteria for clinical removal of the LMA were met in our study. We suggest that NMB monitoring should be used as much as possible and the removal of LMA should meet the indicator of TOFr ≥ 0.9 if the device is available. Second, mivacurium is hydrolyzed by plasma cholinesterases,34,35 but we did not measure preoperative butyrylcholinesterase (BCHE) activity and phenotype, as it is not a routine perioperative test. BCHE deficiency prolongs the degradation of mivacurium or suxamethonium and results in a prolonged duration of action and mechanical ventilation, which is more likely to occur in patients with increasing age, pregnancy, severe liver disease, burn injuries and drug interactions.36,37 Although no severe rNMB and prolonged mechanical ventilation occurred in our study, BCHE activity should be screened when mivacurium or suxamethonium is intended to be applied in patients with the aforementioned risk factors. Once abnormal rNMB occurs when mivacurium is applied, BCHE abnormalities should be considered. In addition, controversy exists as to whether the cholinesterase inhibitor neostigmine accelerates the recovery of mivacurium.38 We did not use an antagonist to investigate muscular recovery from mivacurium at an infusion rate of 3–9 μg/(kg·min), and further studies should be performed to investigate whether the cholinesterase inhibitor neostigmine is effective at our recommended dose of mivacurium. Finally, the aim of our study was to determine a suitable dose of mivacurium infusion, and we did not compare the effect and recovery of mivacurium with those of other NMBAs, such as rocuronium and atracurium. Further studies are needed to determine whether mivacurium has an advantage over other NMBAs during ambulatory vitreoretinal surgery.
Conclusion
In summary, a continuous infusion of 6 μg/(kg·min) mivacurium not only achieves a lower incidence of postoperative rNMB than 9 μg/(kg·min) mivacurium but also provides an appropriate depth of neuromuscular blockade during the operation compared with 3 μg/(kg·min) mivacurium and thus is a suitable maintenance dose for ambulatory vitreoretinal surgery.
Data Sharing Statement
The raw data supporting the conclusions of this article will be available by the authors without undue reservation. After the article is published, readers can contact the corresponding author to obtain data by email.
Acknowledgments
Assistance with the study: We gratefully acknowledge Shaofei Su, PhD (Department of Epidemiology and Health Statistics, Beijing Obstetrics and Gynecology Hospital, Capital Medical University) for statistical consultation and the editors at American Journal Experts for their assistance in improving the English language herein.
Financial support and sponsorship: This study was supported by Beijing Hospitals Authority Clinical Medicine Development of Special Funding Support (ZYLX202103) and Beijing Hospitals Authority’s Ascent Plan (DFL20220203). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bailey CR, Ahuja M, Bartholomew K, et al. Guidelines for day-case surgery 2019: guidelines from the Association of Anaesthetists and the British Association of Day Surgery. Anaesthesia. 2019;74(6):778–792. doi:10.1111/anae.14639
2. Thevathasan T, Shih SL, Safavi KC, et al. Association between intraoperative non-depolarising neuromuscular blocking agent dose and 30-day readmission after abdominal surgery. Br J Anaesth. 2017;119(4):595–605. doi:10.1093/bja/aex240
3. Stagg BC, Talwar N, Mattox C, Lee PP, Stein JD. Trends in Use of Ambulatory Surgery Centers for Cataract Surgery in the United States, 2001-2014. JAMA Ophthalmol. 2018;136(1):53–60. doi:10.1001/jamaophthalmol.2017.5101
4. McGoldrick KE, Foldes PJ. General anesthesia for ophthalmic surgery. Ophthalmol Clin North Am. 2006;19(2):179–191. doi:10.1016/j.ohc.2006.02.005
5. Gropper MA, Miller RD, Eriksson LI, et al. Miller’s Anesthesia. Vol. 2. Set E-Book; 2019.
6. Goldberg M. Complications of anesthesia for ocular surgery. Ophthalmol Clin North Am. 2006;19(2):293–307. doi:10.1016/j.ohc.2006.02.018
7. Alfille PH, Merritt C, Chamberlin NL, Eikermann M. Control of perioperative muscle strength during ambulatory surgery. Curr Opin Anaesthesiol. 2009;22(6):730–737. doi:10.1097/ACO.0b013e328331d545
8. Nemes R, Fulesdi B, Pongracz A, et al. Impact of reversal strategies on the incidence of postoperative residual paralysis after rocuronium relaxation without neuromuscular monitoring: a partially randomised placebo controlled trial. Eur J Anaesthesiol. 2017;34(9):609–616. doi:10.1097/EJA.0000000000000585
9. Li S, Hu X, Tan F, Li W. Effects of Cisatracurium, Rocuronium, and Mivacurium on Intraocular Pressure During Induction of General Anesthesia in Ophthalmic Surgery. Drug Des Devel Ther. 2020;14:1203–1208. doi:10.2147/DDDT.S224544
10. Ostergaard D, Viby-Mogensen J, Rasmussen SN, Gatke MR, Varin F. Pharmacokinetics and pharmacodynamics of mivacurium in patients phenotypically homozygous for the atypical plasma cholinesterase variant: effect of injection of human cholinesterase. Anesthesiology. 2005;102(6):1124–1132. doi:10.1097/00000542-200506000-00011
11. Frampton JE, McTavish D. Mivacurium. A review of its pharmacology and therapeutic potential in general anaesthesia. Drugs. 1993;45(6):1066–1089. doi:10.2165/00003495-199345060-00009
12. Pendeville PE, Lois F, Scholtes JL. A comparison of intubation conditions and time-course of action with rocuronium and mivacurium for day case anaesthesia. Eur J Anaesthesiol. 2007;24(6):546–550. doi:10.1017/S0265021506002341
13. Weber F, Kriek N. The effects of mivacurium-induced neuromuscular block on Bispectral Index and Cerebral State Index in children under propofol anesthesia - a prospective randomized clinical trial. Paediatr Anaesth. 2010;20(8):697–703. doi:10.1111/j.1460-9592.2010.03327.x
14. Xiong W, Li M, Wang M, Zhang S, Yang Q. The Safety of Laparoscopic Cholecystectomy in the Day Surgery Unit Comparing with That in the Inpatient Unit: a Systematic Review and Meta-Analysis. Biomed Res Int. 2020;2020:1924134. doi:10.1155/2020/1924134
15. Martini CH, Boon M, Bevers RF, Aarts LP, Dahan A. Evaluation of surgical conditions during laparoscopic surgery in patients with moderate vs deep neuromuscular block. Br J Anaesth. 2014;112(3):498–505. doi:10.1093/bja/aet377
16. Vanlinthout LE, Mesfin SH, Hens N, et al. A systematic review and meta-regression analysis of mivacurium for tracheal intubation. Anaesthesia. 2014;69(12):1377–1387. doi:10.1111/anae.12786
17. Queiroz Rangel Micuci AJ, Vercosa N, Filho PAG, et al. Effect of pretreatment with magnesium sulphate on the duration of intense and deep neuromuscular blockade with rocuronium: a randomised controlled trial. Eur J Anaesthesiol. 2019;36(7):502–508. doi:10.1097/EJA.0000000000001003
18. Savarese JJ, Ali HH, Basta SJ, et al. The clinical neuromuscular pharmacology of mivacurium chloride (BW B1090U). A short-acting nondepolarizing ester neuromuscular blocking drug. Anesthesiology. 1988;68(5):723–732. doi:10.1097/00000542-198805000-00010
19. Naguib M, Brull SJ, Kopman AF, et al. Consensus Statement on Perioperative Use of Neuromuscular Monitoring. Anesth Analg. 2018;127(1):71–80. doi:10.1213/ANE.0000000000002670
20. Palsen S, Wu A, Beutler SS, et al. Investigation of intraoperative dosing patterns of neuromuscular blocking agents. J Clin Monit Comput. 2019;33(3):455–462. doi:10.1007/s10877-018-0186-4
21. Brueckmann B, Sasaki N, Grobara P, et al. Effects of sugammadex on incidence of postoperative residual neuromuscular blockade: a randomized, controlled study. Br J Anaesth. 2015;115(5):743–751. doi:10.1093/bja/aev104
22. Murphy GS, Brull SJ. Residual neuromuscular block: lessons unlearned. Part I: definitions, incidence, and adverse physiologic effects of residual neuromuscular block. Anesth Analg. 2010;111(1):120–128. doi:10.1213/ANE.0b013e3181da832d
23. Saager L, Maiese EM, Bash LD, et al. Incidence, risk factors, and consequences of residual neuromuscular block in the United States: the prospective, observational, multicenter RECITE-US study. J Clin Anesth. 2019;55:33–41. doi:10.1016/j.jclinane.2018.12.042
24. Fortier LP, McKeen D, Turner K, et al. The RECITE Study: a Canadian Prospective, Multicenter Study of the Incidence and Severity of Residual Neuromuscular Blockade. Anesth Analg. 2015;121(2):366–372. doi:10.1213/ANE.0000000000000757
25. Murphy GS, Szokol JW, Avram MJ, et al. Intraoperative acceleromyography monitoring reduces symptoms of muscle weakness and improves quality of recovery in the early postoperative period. Anesthesiology. 2011;115(5):946–954. doi:10.1097/ALN.0b013e3182342840
26. Yu B, Ouyang B, Ge S, et al. Incidence of postoperative residual neuromuscular blockade after general anesthesia: a prospective, multicenter, anesthetist-blind, observational study. Curr Med Res Opin. 2016;32(1):1–9. doi:10.1185/03007995.2015.1103213
27. Grabitz SD, Rajaratnam N, Chhagani K, et al. The Effects of Postoperative Residual Neuromuscular Blockade on Hospital Costs and Intensive Care Unit Admission: a Population-Based Cohort Study. Anesth Analg. 2019;128(6):1129–1136. doi:10.1213/ANE.0000000000004028
28. Wu L, Wei SW, Xiang Z, et al. Effect of neuromuscular block on surgical conditions during short-duration paediatric laparoscopic surgery involving a supraglottic airway. Br J Anaesth. 2021;127(2):281–288. doi:10.1016/j.bja.2021.04.031
29. Schumacher PM, Stadler KS, Wirz R, et al. Model-based control of neuromuscular block using mivacurium: design and clinical verification. Eur J Anaesthesiol. 2006;23(8):691–699. doi:10.1017/S0265021506000524
30. Chen Y, Wang B, Yao L, Feng Z. Maximum dose of continuous infusion of mivacurium for thyroid surgery under total intravenous anesthesia: a sequential trial of monitoring neurological function in 30 patients. Nan Fang Yi Ke Da Xue Xue Bao. 2021;41(1):64–68. doi:10.12122/j.issn.1673-4254.2021.01.08
31. Jan GS, Tong WN, Chan AM, Hui TW, Lo JW. Recovery from mivacurium block with or without anticholinesterase following continuous infusion in obstetric patients. Anaesth Intensive Care. 1996;24(5):585–589. doi:10.1177/0310057X9602400514
32. Ahn EK, Bai SJ, Cho BJ, Shin YS. The infusion rate of mivacurium and its spontaneous neuromuscular recovery in magnesium-treated parturients. Anesth Analg. 1998;86(3):523–526. doi:10.1097/00000539-199803000-00014
33. Woloszczuk-Gebicka B. Mivacurium infusion requirement and spontaneous recovery of neuromuscular transmission in children anaesthetized with nitrous oxide and fentanyl, halothane, isoflurane or sevoflurane. Paediatr Anaesth. 2002;12(6):511–518. doi:10.1046/j.1460-9592.2002.00901.x
34. Ezzine S, Donati F, Varin F. Mivacurium arteriovenous gradient during steady state infusion in anesthetized patients. Anesthesiology. 2002;97(3):622–629. doi:10.1097/00000542-200209000-00016
35. Savarese JJ, Lien CA, Belmont MR, Wastila WB. The clinical pharmacology of new benzylisoquinoline-diester compounds, with special consideration of cisatracurium and mivacurium. Anaesthesist. 1997;46(10):840–849. doi:10.1007/s001010050477
36. Chung DY, Hardman J. Prolonged paralysis following mivacurium administration. Anaesth Intensive Care. 2002;30(3):360–363. doi:10.1177/0310057X0203000316
37. Andersson ML, Moller AM, Wildgaard K. Butyrylcholinesterase deficiency and its clinical importance in anaesthesia: a systematic review. Anaesthesia. 2019;74(4):518–528. doi:10.1111/anae.14545
38. Brinch JHW, Soderstrom CM, Gatke MR, Madsen MV. Reversal of mivacurium-induced neuromuscular blockade with a cholinesterase inhibitor: a systematic review. Acta Anaesthesiol Scand. 2019;63(5):564–575. doi:10.1111/aas.13304
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

