Back to Journals » Infection and Drug Resistance » Volume 12
Comparative effectiveness of antifungal agents in patients with hematopoietic stem cell transplantation: a systematic review and network meta-analysis
Authors Su HC, Hua YM, Feng IJ, Wu HC
Received 30 January 2019
Accepted for publication 24 April 2019
Published 15 May 2019 Volume 2019:12 Pages 1311—1324
DOI https://doi.org/10.2147/IDR.S203579
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sahil Khanna
Hui-Chen Su,1,* Yi-Ming Hua,1,* I Jung Feng,2 Hung-Chang Wu3
1Department of Pharmacy, Chi Mei Medical Center, Tainan City, Taiwan; 2Department of Medical Research, Chi Mei Medical Center, Tainan City, Taiwan; 3Division of Hematology-Oncology, Department of Internal medicine, Chi Mei Medical Center, Tainan City, Taiwan
*These authors contributed equally to this work
Purpose: The aim of this study was to use a network meta-analysis to evaluate the relative efficacy of various agents at preventing invasive fungal infections (IFIs). In this way, suitable prophylactic regimens may be selected for patients with hematopoietic stem cell transplantation (HSCT).
Methods: We conducted a systematic review of randomized controlled trials comparing the prophylactic effects of two antifungal agents or an antifungal agent and a placebo administered to patients with HSCT. Relevant studies were found in the PubMed and Cochrane databases. Unpublished studies were collected from the ClinicalTrials.gov registry.
Results: Sixteen two-arm studies were identified. Compared with placebo, all six antifungal agents (amphotericin B, fluconazole, itraconazole, micafungin, posaconazole, and voriconazole) presented with greater efficacy at controlling proven IFIs. OR ranged from 0.08 to 0.29. Voriconazole (surface under the cumulative ranking curve [SUCRA]=71.6%), posaconazole (SUCRA=68.9%), and itraconazole (SUCRA=64.7%) were the three top-ranking drugs for preventing proven IFIs. Itraconazole ranked highest (SUCRA=83.1%) and had the greatest efficacy at preventing invasive candidiasis. Posaconazole and micafungin were the two top-ranking drugs (SUCRA=81.3% and 78.4%, respectively) at preventing invasive aspergillosis. Micafungin and voriconazole were the drugs of choice because they lowered mortality more than the other agents (SUCRA=74.6% and 61.1%, respectively).
Conclusion: This study is the first network meta-analysis to explore the prophylactic effects of antifungal agents in patients with HSCT. Voriconazole was the best choice for the prevention of proven IFIs in HSCT patients.
Keywords: antifungal agents, hematopoietic stem cell transplantation, network meta-analysis
Introduction
Invasive fungal infections (IFIs) are a major cause of morbidity and death in immunocompromised populations including patients with hematological malignancies or hematopoietic stem cell transplant (HSCT). HSCT patients are at high risk of prolonged severe neutropenia and graft-versus-host disease (GVHD).1,2 The aforementioned diseases and immunosuppressive agents for the treatment of GVHD are associated with high IFI rates. The all-cause mortality rate is 5% in autologous HSCT and 19% in allogeneic HSCT at 4 months after transplantation.3
Before HSCT, patients should receive conditioning therapy during the pre-engraftment phase. However, severe neutropenia is associated with conditioning therapy. Thus, the risk of IFI is particularly high during the pre-engraftment and early engraftment phases in the first month after transplantation. Candida and Aspergillus are the most common fungal genera causing IFIs.4 As IFI has a strong impact in these populations, prophylaxis must be initiated before engraftment either at the start or immediately after the end of conditioning therapy.4
Numerous antifungal agents such as polyenes, triazoles, and echinocandins are currently available. Nevertheless, they have certain drawbacks and limitations. The polyene antifungal amphotericin B is nephrotoxic and has largely been replaced by relatively safer agents. The first-generation azoles fluconazole and itraconazole reduce morbidity in HSCT recipients and were recommended for IFI prophylaxis. However, their efficacy is constrained by poor bioavailability and/or drug–drug interactions. In contrast, the newer triazoles such as voriconazole and posaconazole have proven to be comparatively safe and efficacious. Echinocandins such as micafungin are alternative prophylactic agents. Anti-mold agents such as voriconazole and posaconazole may be preferable for patients with GVHD.
Recently, randomized controlled trials (RCTs) and meta-analyses5-11 showed that triazoles and echinocandins significantly reduced IFIs in patients at risk of contracting them. The relative prophylactic efficacy of these agents in patients with hematological malignancies, chemotherapy, or HSCT was evaluated by three network meta-analyses.12–14 Posaconazole is apparently the most effective prophylactic.12,13 On the other hand, itraconazole and posaconazole proved to be more cost-effective in a Singapore health care setting study.14 These clinical assessments were based on patients being administered chemotherapy or HSCT. However, the clinical condition and disease severity in patients on chemotherapy were markedly different from those for patients with HSCT. To the best of our knowledge, no similar evaluations have been conducted on more severely affected patient populations such as those with HSCT who have highly specificity and are strongly immunocompromised. Therefore, the antifungal agents most appropriate for them must be identified. A network meta-analysis integrates both direct and indirect RCT evidence by head–head comparison and relative to the comparator drugs of interest, respectively. In this manner, the relative efficacies of all interventions may be judged using a single model.15
The aim of the present study was to compare the relative primary IFI prevention efficacies of multiple antifungal agents via a network meta-analysis. In this way, a prophylactic regimen for HSCT patients can be optimized.
Materials and methods
Inclusion criteria
RCTs were included to compare the efficacies of prophylactically administered antifungal agents against other drugs or placebo in patients who received HSCT. Studies were sought which clearly indicated patient inclusion and exclusion criteria, type of stem cell transplantation (allogeneic or autologous), the definition and evaluation of infection, and the drug administration strategy. Trials meeting at least one of the following criteria were excluded: 1) chemotherapy 2) empirical or therapeutic use 3) pediatric populations, or 4) economic studies.
Systematic search and study selection
Relevant studies published up until June 2018 were identified in the PubMed and Cochrane databases. The following Medical Subject Headings terms were used: “stem cell transplant”, “prophyla*”, “antifung*” OR “azole” OR “echinocandin” OR “polyene” and all antifungals on the market. Please see Table S1 for an example of the search strategy used in this study. The “related articles” option in PubMed broadened the search. All abstracts, studies, and citations retrieved were reviewed. Other studies were also identified using the reference sections of pertinent articles and through correspondence with field experts. Unpublished studies were collected from the ClinicalTrials.gov registry (
Data extraction
Baseline and outcome data were independently evaluated by two reviewers (HCS and YMH). Study designs, population characteristics, inclusion and exclusion criteria, drug administration strategies, intervention times, stem cell transplantation types, infection types, and follow-up times were extracted. The decisions recorded by the reviewers were compared and discrepancies were resolved by a third reviewer (IJF).
Methodological quality appraisal
HCS and YMH also independently assessed the methodological quality of each study using the bias risk method recommended by the Cochrane Collaboration.16 Bias was rated as a judgment (high, low, or unclear) for individual elements in seven domains including randomization adequacy, allocation concealment, patient and outcome assessor blinding, patient information concerning study withdrawal, intention-to-treat analysis, and freedom from other biases.
Outcomes
The primary outcome was the incidence of proven IFI. For most studies, IFIs were defined and graded according to international consensus.17 Proven IFIs met criteria which allowed them to be diagnosed with certainty. These included confirmation of the presence of fungi by tissue biopsy, needle aspiration, microbial cultures, or histological examination. Probable IFIs were defined by the presentation of host factor and microbiological and clinical criteria. Here, we focused exclusively on the proven IFI outcome to obtain precise definitions. Secondary outcomes included the incidences of invasive candidiasis (IC), invasive aspergillosis (IA), and all-cause mortality. The definitions of IC and IA used here resembled those for IFIs based on the international consensus.17
Statistical analyses
In the present study, a random-effects meta-analysis with a frequentist setting was performed in STATA v. 15.0 (StataCorp, College Station, TX, USA). Differences in study characteristics, including dosage form, regimen, and durations were combined to evaluate the summarized treatment efficacy. The heterogeneity variable τ2 was used to estimate between-study variance in treatment effects and was assumed to be the same for all comparisons. Transitivity, a required property, was assumed in this study. It represents the difference between treatments A and B which can be validly estimated by indirect comparison. Inconsistencies between direct and indirect evidence were assessed using a design-by-treatment interaction model for the entire network test and a side-splitting model for the local test. A network plot was generated for all direct comparisons. The summarized treatment-effect estimates of pairwise comparisons were presented as OR and 95% CI. The likelihood of each rank order was estimated and presented as the surface under the cumulative ranking curve (SUCRA). In the SUCRA plot, the horizontal and vertical axes represent, respectively, the possible rank from best to worst and the cumulative probability of being a corresponding rank order. SUCRA is the ratio of the area under the curve to the entire plot area. The value of SUCRA ranges from 0% to 100%. Treatments in the top rank order have SUCRA values approaching 100% while those in the bottom rank have SUCRA values near 0%.18 Small-study effects were evaluated with Egger’s test.
Results
Characteristics of included studies and patients
The process flow depicting the present systematic literature review is shown in Figure 1. The initial search strategy yielded 3,040 studies. After removing duplicates, 1,473 were deemed ineligible according to the title- and abstract-screening criteria. Therefore, the full texts of 39 studies were retrieved. However, most studies were excluded from our final review because 13 included patients with hematologic malignancies receiving both transplantation and chemotherapy, four evaluated the effects of empirical treatment, one only accounted for a pediatric population, and five involved the assessment of economic outcome. Sixteen two-arm studies published between 1994 and 2016 were identified19-34 of which six were open-label RCTs.19,20,23,26,27,29
 | Figure 1 PRISMA process flow of study selection. |
A total of 4,961 participants were enrolled in the review (Table 1). The trials were distributed across North America, South America, Europe, Asia, and Africa. Six studies included patients receiving only allogeneic HSCT.21,22,24,26–28 The times at which antifungal therapy was initiated were based on the courses of conditional therapy and occurred within 24 or 48 hrs before the onset of HSCT. Certain antifungal agents were initiated simultaneous to pre-HSCT conditioning therapy. Antifungal therapy was discontinued under any of the following conditions: <2–5 d after engraftment (defined as an absolute neutrophil count of >500 cells mm−3 after the nadir absolute count); treatment days 21–42 after HSCT; development of IFI; unacceptable drug toxicity.
 | Table 1 Study baselines |
Network meta-analysis
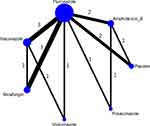
For the 16 identified studies, the seven reported arms were 1) intravenous amphotericin B, 2) fluconazole, 3) itraconazole, 4) voriconazole, 5) posaconazole, 6) micafungin, and 7) placebo. The integral network for the six antifungal agents and placebo is presented in Figure 2. The first-generation azoles fluconazole and itraconazole were the most extensively studied treatments (12 reports for fluconazole and five studies on itraconazole).
Study quality
Study quality was generally moderate (Table 2). Most trials presented with randomized procedures and allocation concealment. Nine studies had adequate randomization and sequence descriptions. In nine studies, patients were stratified by transplant, age, conditioning regimen, and donor relatedness. Ten studies were double-blind and most of them indicated the masking procedure used. On the other hand, six studies had an open-label design. Seven studies furnished no detailed descriptions on their assessor blinding method. Most of the studies used the intention-to-treat analytical method.
 | Table 2 Detailed quality assessment by Cochrane Collaboration |
Proven IFI in the overall population
Thirteen studies reported proven IFI outcomes. In nine studies, the IFIs were defined and graded according to the international consensus.19,21–27,29 In another seven studies, the IFIs were identified by positive culture, clinical evidence of blood or tissue infection, or biopsies.20,28,30–34 Figure 3 shows the integrated OR of the proven IFIs and a forest plot.19,20,22,23,26–34 Compared with the placebo, amphotericin B, fluconazole, itraconazole, micafungin, voriconazole, and posaconazole all had greater efficacy against proven IFIs and their OR ranged from 0.08 to 0.29. Except for posaconazole, all treatments show significantly greater efficacy at preventing proven IFIs. According to the summarized OR and SUCRA, voriconazole (SUCRA=71.6%), posaconazole (SUCRA=68.9%), and itraconazole (SUCRA=64.7%) are three top-ranking drugs for the prevention of proven IFIs, followed by micafungin (SUCRA=64%), fluconazole (SUCRA=44.9%), amphotericin B (SUCRA=34%), and placebo (SUCRA=2%) (Tables S2 and S6).
 | Figure 3 Summarized OR and corresponding 95% CI for multiple treatment comparisons of proven IFI. |
IC in the overall population
Combining the IC outcomes of 11 trails with seven arms,19–22,24–27,30,31,33 the network meta-analysis showed consistency among the antifungal agents investigated and they all had greater efficacy at preventing IC infection than the placebo. However, only fluconazole and itraconazole displayed significantly greater efficacy than the placebo (OR=0.25, 95% CI=0.07–0.91; OR=0.12, 95% CI=0.02–0.73, respectively; Figure 4). Itraconazole was ranked the highest (SUCRA=83.1%) and had the strongest efficacy at preventing IC compared to the other five drugs and the placebo. Amphotericin B and fluconazole were in second and third place, respectively (SUCRA=68.2% and 53%) (Tables S3 and S7).
 | Figure 4 Summarized OR and corresponding 95% CI for multiple treatment comparisons of IC infection. |
IA in the overall population
The IA outcomes of 10 identified studies are summarized.19–22,24–27,30,31 Compared with the placebo, amphotericin B was the only drug with relatively lower efficiency at preventing IA (OR=1.16, 95% CI=0.03–39.03). Posaconazole and micafungin were the most efficacious at preventing IA (SUCRA=81.3% and 78.4%, respectively). Posaconazole was significantly more efficacious at preventing IA than fluconazole (OR=0.32, 95% CI=0.11–0.91) but not the other agents (Figure 5; Tables S4 and S8).
 | Figure 5 Summarized OR and corresponding 95% CI for multiple treatment comparisons of IA infection. |
All-cause mortality in the overall population
We analyzed 12 studies (730 cases) reporting overall mortality rates.19,21,23–27,30–34 Compared with patients receiving placebo, patients being administered antifungal agents presented with lower risks of all-cause mortality (Figure 6). The network meta-analysis and SUCRA indicated that the lowest risks of death were associated with micafungin and voriconazole relative to the other comparators (SUCRA=74.6% and 61.1%, respectively) (Tables S5 and S9).
 | Figure 6 Summarized OR and corresponding 95% CI for multiple treatment comparisons of all-cause mortality. |
Figure 7 shows a scatterplot of the cumulative probabilities that each agent is the most efficacious prophylactic antifungal for both proven IF and mortality. An optimal cluster analysis indicated that micafungin, itraconazole, voriconazole, and posaconazole were clustered together in a group of agents associated with relatively lower risks of proven IF and death.
 | Figure 7 Scatterplot of cumulative probabilities of proven IF and mortality. |
Publication bias
A funnel plot and Egger’s test confirmed that there was no significant publication bias.
Inconsistency
The design-by-treatment interaction and side-splitting models identified no inconsistency factors (P>0.05 in all analyses). Thus, our network was robust.
Discussion
Several new antifungal agents were launched since the year 2000 including caspofungin in 2001, voriconazole in 2002, and posaconazole in 2006. We extensively searched databases and reports on them in the effort to determine the best therapeutic strategy for IFI. The evidence suggested that voriconazole is the most effective prophylaxis against proven IFI in patients with HSCT. In contrast, posaconazole and micafungin are relatively more efficacious at preventing IA. Itraconazole ranked highest at reducing the risks of IC and all-cause mortality. Overall, these newer antifungal agents were comparatively more efficacious at controlling proven IFI and reducing mortality.
The guidelines indicated that fluconazole is effective as a prophylactic antifungal in allogeneic HSCT patients when it is administered from the onset of conditioning therapy.35–39 However, fluconazole was relatively less efficacious against certain Candida species including C. krusei and C. glabrata. Therefore, it was not recommended as a first-line preventive therapy by the Centers for Disease Control and Prevention or the American Society for Blood and Marrow Transplantation.4,40 According to the European Leukemia Net and the Immunocompromised Host Society created by the European Conference on Infections in Leukemia (ECIL 3 group),39 fluconazole was highly recommended only when mold infection was definitively excluded. Otherwise, micafungin, itraconazole, voriconazole, or posaconazole were alternative agents of choice. The utility of itraconazole may be limited by its poor tolerance and oral toxicity.40 The Infectious Diseases Working Party (AGIHO) of the German Society for Hematology and Medical Oncology (DGHO) published that there is inadequate evidence to support the prophylactic use of itraconazole.38
The risk of infection by molds such as Aspergillus sp. increases with the duration of neutropenia. In this condition, anti-mold agents should be considered. Various dosage forms of amphotericin B have been administered to prevent aspergillosis, but there are limited data concerning their safety and efficacy in HSCT patients.38,40 Voriconazole therapy proved to be as effective as that of fluconazole and itraconazole for IC prophylaxis in allogeneic HSCT recipients.4,35 Two of the RCTs in our network meta-analysis showed that the incidence of IFI in the voriconazole group was lower than that in the itraconazole group (1.5% vs 2.0%) and the fluconazole group (1.6% vs 3.0%) during neutropenia and GVHD after HSCT.21,22 Therefore, the ECIL 3 group recommended the use of voriconazole in these phases of HSCT but they assigned a provisional AI grading to it.39 In the DGHO guideline,38 posaconazole was given priority and preference over fluconazole in the prevention of IFI during prolonged neutropenia. However, this decision was not based on data from hematopoietic stem cell recipients. The National Comprehensive Cancer Network in Taiwan recommended fluconazole or micafungin as a first choice prophylaxis for neutropenic allogeneic HSCT patients.36,37 Voriconazole, posaconazole, and amphotericin B were proposed as alternatives. According to a retrospective study, micafungin proved to be as efficacious as posaconazole in patients with hematological malignancies.4,38 For patients with GVHD, posaconazole or voriconazole was strongly recommended.4,36,37
Three network meta-analyses were recently conducted to evaluate the effects of antifungal agents for hematological patients receiving HSCT or chemotherapy.12–14 The network meta-analysis of Zhao et al focused exclusively on azoles and comprised 21 trials.14 The exclusion criteria were children and patients with GVHD. Only 33% of the studies consisted exclusively of HSCT patients. Posaconazole ranked highest in the prevention of IF, IA, and all-cause mortality. In 2017, two network meta-analyses explored the prophylactic effects of antifungal agents administered to hematological patients receiving HSCT or chemotherapy.12,13 Both studies enrolled patients with GVHD and at any age. Leonart et al focused only on RCTs while Lee et al included placebo-designed studies. Consequently, the former comprised 25 studies while the latter consisted of 54 investigations. The proportions of RCTs including patients exclusively receiving HSCT were 24% and 27%, respectively. The results for overall proven IFIs in Lee’s study12 demonstrated that posaconazole provided promising reduction (RR=0.14, 95% CI=0.05–0.36) and had the highest ranking. It was followed by micafungin, itraconazole, and fluconazole. Posaconazole was also the most efficacious at preventing IA (RR=0.13, 95% CI=0.03–0.65). Itraconazole was ranked first at preventing IC (RR=0.21, 95% CI=0.11–0.39). According to another network meta-analysis,13 posaconazole was the best option for IFI prophylaxis and IFI-related mortality reduction and voriconazole was second best. However, the relative preventative efficacies of IA and IC reported in that study differed from those presented by Lee. The overall ranking suggests that voriconazole is the preferred choice as a prophylactic against IA and IC.
The risk severity was high for our study population because they were in the queue for BMT and some of them had GVHD events. Our results were similar for those of prior network meta-analyses using IFI and IA as endpoints. Anti-mold such as posaconazole and voriconazole proved to be a priority for the prevention of IFI and IA in these patients. For IC prevention, Leonart et al advised voriconazole whereas Lee et al and the present study proposed itraconazole.
The probability ranking associated itraconazole with good prophylactic efficacy against IC. It also had positive tendencies for other endpoints. These findings corroborate those reported for other studies.12,14 In certain RCTs,20,26,27 itraconazole has potent activity against both IF and IC. However, itraconazole capsules are contraindicated for fungal prophylaxis in HSCT recipients due to poor bioavailability, long time to achieve steady-state serum level, interactions with other drugs, and gastrointestinal side effects.40 According to a meta-analysis,41 IFI risk was reduced with itraconazole cyclodextrin solution but not the capsule form. In our meta-analysis, only one study involved itraconazole capsules. Therefore, the apparently outstanding efficacy of itraconazole could be explained by its high bioavailability. One of the RCTs in our study26 strongly supported itraconazole administration. However, possible reasons for this conclusion are as follows: 1) baseline imbalance baseline; 2) most patients were maintained on highly bioavailable intravenous itraconazole; and 3) the fungal pathogens were more resistant to fluconazole than itraconazole. We removed this RCT to eliminate its influence. Nevertheless, we obtained the same results as before.
In the present study, micafungin and posaconazole were recommended for the prevention of IA whereas posaconazole alone was advised for this purpose by Lee et al12 and Zhao et al.14 Posaconazole has a similar activity level against Candida spp to that of itraconazole but both are ≥8× more efficacious than fluconazole for this application. Conversely, micafungin was only weakly effective against IA. This observation may be explained by inadequate follow-up. The incidence of IA in HSCT recipients occurred particularly at pre-engraftment and 3–4 months after engraftment.42 Nevertheless, the average follow-up time was <100 d during which time the onset of IA occurrence was probably overlooked. In the present study, there was no significant difference among antifungal agents in terms of all-cause mortality. In another two network meta-analyses, however, posaconazole was reported as the best option for mortality avoidance.13,14
The initiation time for antifungal agent therapy and the optimal treatment duration remain to be determined. The onset of treatment was aligned with the duration of conditioning therapy. Nine of the studies started treatment within 1–2 d before conditioning therapy began. In four investigations, patients received treatments from the beginning of conditioning therapy. The guideline recommends 90–100 d of antifungal primary prophylaxis for allogeneic HSCT.39 A post-hoc RCT analysis showed that when fluconazole was administered until day 75 post-transplant, it conferred prolonged protection against IC.4 These duration endpoints differed from those of the studies in our meta-analysis. Unfortunately, there were too few reports available to run a subgroup analysis.
To the best of our knowledge, this network meta-analysis is the first to explore the prophylactic efficacies of various antifungal agents in patients with HSCT. We endeavored to identify the best available treatment options. However, there were certain limitations. First, the best regimen was difficult to define because of discrepancies in dose and duration. Moreover, the dosage forms may have been changed according to clinical protocol or disease condition. Drug levels could have fluctuated because of relative differences in bioavailability. Second, the definition of IFI was inconsistent among the trials studied. To obtain more exact evidence, we extracted proven IFI as the primary outcome because it was defined by comparatively strict and narrow criteria. Third, HSCT type and intervention and duration timing might have influenced the prognosis. Nevertheless, we did not perform a subgroup analysis because of the small sample size.
Conclusions
The present network meta-analysis indicated that voriconazole and posaconazole are effective at preventing IFIs in HSCT recipients. The characteristics of these studies were highly variable, but the study number was too low to support the assessment of the associations between prognostic factors and outcomes. Further RCTs must be conducted to obtain more direct head-to-head evidence for these correlations.
Summary
Patients with certain types of cancers such as leukemias or lymphomas may require irradiation to destroy their diseased bone marrow so that it may be replaced either by their own or a donor’s healthy bone marrow. One function of the bone marrow is to produce the cells found in blood including white blood cells, which make up part of the immune system. Until the bone marrow graft takes, the patient may have very low white blood cell counts and be susceptible to bacterial or fungal infection. These patients are particularly sensitive to airborne molds. While these may cause only mild illness in healthy people, they could be fatal in bone marrow transplant patients. Therefore, certain drugs known to prevent fungal infections are given to these patients in order to protect them from these molds. Nevertheless, the older generations of drugs either damaged other organs such as the kidneys, reacted with other drugs, or were poorly absorbed in the body. Within the past 20 years, however, new safer and more effective medications were developed for this purpose. In this study, we searched numerous articles describing the safety and effectiveness of various antifungal drugs on patients who received bone marrow transplants. Indeed, we found that the latest generation of drugs designed to prevent fungal infection generally worked better and helped reduce the death rates more than the earlier, first-generation medications.
Abbreviation list
GVHD, graft-versus-host disease; HSCT, hematopoietic stem cell transplantation; IA, invasive aspergillosis; IC, invasive candidiasis; IFI, invasive fungal infections; SUCRA, surface under the cumulative ranking curve.
Author contributions
HSC designed the study, acquired the data, and prepared the manuscript. YMH conducted the article search, acquired the data, appraised the papers, and prepared the manuscript. IJF analyzed the data and prepared the manuscript. WHC interpreted the data and prepared the manuscript. All authors contributed to the data analysis, article drafting and revision, read and approved the final manuscript version for publication, and agreed to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Garcia-Vidal C, Upton A, Kirby KA, Marr KA. Epidemiology of invasive mold infections in allogeneic stem cell transplant recipients: biological risk factors for infection according to time after transplantation. Clin Infect Dis. 2008;47(8):1041–1050. doi:10.1086/59
2. Mikulska M, Raiola AM, Bruno B, et al. Risk factors for invasive aspergillosis and related mortality in recipients of allogeneic SCT from alternative donors: an analysis of 306 patients. Bone Marrow Transplant. 2009;44(6):361–370. doi:10.1038/bmt.2009.39
3. Kontoyiannis DP, Marr KA, Park BJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001-2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50(8):1091–1100. doi:10.1086/651263
4. Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow transplant. 2009;15(10):1143–1238. doi:10.1016/j.bbmt.2009.06.019
5. Bow EJ, Vanness DJ, Slavin M, et al. Systematic review and mixed treatment comparison meta-analysis of randomized clinical trials of primary oral antifungal prophylaxis in allogeneic hematopoietic cell transplant recipients. BMC Infect Dis. 2015;15:128. doi:10.1186/s12879-015-0855-6
6. Chen Q, Lin MH, Chen ML, Liu ZY, Chai D, Wang R. Efficacy and safety of micafungin for invasive candida infections: a meta-analysis of randomized controlled trials. Chin Med J. 2012;125(2):345–351.
7. Ping B, Zhu Y, Gao Y, Yue C, Wu B. Second- versus first-generation azoles for antifungal prophylaxis in hematology patients: a systematic review and meta-analysis. Ann Hematol. 2013;92(6):831–839. doi:10.1007/s00277-013-1693-5
8. Vardakas KZ, Michalopoulos A, Falagas ME. Fluconazole versus itraconazole for antifungal prophylaxis in neutropenic patients with haematological malignancies: a meta-analysis of randomised-controlled trials. Br J Haematol. 2005;131(1):22–28. doi:10.1111/j.1365-2141.2005.05727.x
9. Wang JF, Xue Y, Zhu XB, Fan H. Efficacy and safety of echinocandins versus triazoles for the prophylaxis and treatment of fungal infections: a meta-analysis of RCTs. Eur J Clin Microbiol Infect Dis. 2015;34(4):651–659. doi:10.1007/s10096-014-2287-4
10. Xu SX, Shen JL, Tang XF, Feng B. Newer antifungal agents for fungal infection prevention during hematopoietic cell transplantation: a meta-analysis. Transplant Proc. 2013;45(1):407–414. doi:10.1016/j.transproceed.2012.07.149
11. Ziakas PD, Kourbeti IS, Mylonakis E. Systemic antifungal prophylaxis after hematopoietic stem cell transplantation: a meta-analysis. Clin Ther. 2014;36(2):292–306.e291.
12. Lee CH, Lin C, Ho CL, Lin JC. Primary Fungal Prophylaxis in Hematological Malignancy: a Network Meta-Analysis of Randomized Controlled Trials. Antimicrob Agents Chemother. 2018;62(8). doi:10.1128/AAC.00355-18
13. Leonart LP, Tonin FS, Ferreira VL, et al. A network meta-analysis of primary prophylaxis for invasive fungal infection in haematological patients. J Clin Pharm Ther. 2017;42(5):530–538. doi:10.1111/jcpt.12579
14. Zhao YJ, Khoo AL, Tan G, et al. Network Meta-analysis and Pharmacoeconomic Evaluation of Fluconazole, Itraconazole, Posaconazole, and Voriconazole in Invasive Fungal Infection Prophylaxis. Antimicrob Agents Chemother. 2016;60(1):376–386. doi:10.1128/AAC.01985-15
15. Mills EJ, Ioannidis JP, Thorlund K, Schunemann HJ, Puhan MA, Guyatt GH. How to use an article reporting a multiple treatment comparison meta-analysis. Jama. 2012;308(12):1246–1253. doi:10.1001/2012.jama.11228
16. Green S, Higgins JP. Cochrane handbook for systematic reviews of interventions version 5.1. 0., on The Cochrane Collaboration. Available from:
17. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis. 2002;34(1):7–14. doi:10.1086/323335
18. Mbuagbaw L, Rochwerg B, Jaeschke R, et al. Approaches to interpreting and choosing the best treatments in network meta-analyses. Syst Rev. 2017;6(1):79. doi:10.1186/s13643-017-0473-z
19. Park S, Kim K, Jang JH, et al. Randomized trial of micafungin versus fluconazole as prophylaxis against invasive fungal infections in hematopoietic stem cell transplant recipients. J Infect. 2016;73:496–505. doi:10.1016/j.jinf.2016.06.011
20. Huang X, Chen H, Han M, et al. Multicenter, randomized, open-label study comparing the efficacy and safety of micafungin versus itraconazole for prophylaxis of invasive fungal infections in patients undergoing hematopoietic stem cell transplant. Biol Blood Marrow Transplant. 2012;18(10):1509–1516. doi:10.1016/j.bbmt.2012.02.002
21. Marks DI, Pagliuca A, Kibbler CC, et al. Voriconazole versus itraconazole for antifungal prophylaxis following allogeneic haematopoietic stem-cell transplantation. Br J Haematol. 2011;155(3):318–327. doi:10.1111/j.1365-2141.2010.08188.x
22. Wingard JR, Carter SL, Walsh TJ, et al. Randomized, double-blind trial of fluconazole versus voriconazole for prevention of invasive fungal infection after allogeneic hematopoietic cell transplantation. Blood. 2010;116(24):5111–5118. doi:10.1182/blood-2010-02-268151
23. Hiramatsu Y, Maeda Y, Fujii N, et al. Use of micafungin versus fluconazole for antifungal prophylaxis in neutropenic patients receiving hematopoietic stem cell transplantation. Int J Hematol. 2008;88(5):588–595. doi:10.1007/s12185-008-0196-y
24. Ullmann AJ, Lipton JH, Vesole DH, et al. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N Engl J Med. 2007;356(4):335–347. doi:10.1056/NEJMc063190
25. van Burik JA, Ratanatharathorn V, Stepan DE, et al. Micafungin versus fluconazole for prophylaxis against invasive fungal infections during neutropenia in patients undergoing hematopoietic stem cell transplantation. Clin Infect Dis. 2004;39(10):1407–1416. doi:10.1086/422312
26. Winston DJ, Maziarz RT, Chandrasekar PH, et al. Intravenous and oral itraconazole versus intravenous and oral fluconazole for long-term antifungal prophylaxis in allogeneic hematopoietic stem-cell transplant recipients. A multicenter, randomized trial. Ann Intern Med. 2003;138(9):705–713. doi:10.7326/0003-4819-138-9-200305060-00006
27. Marr KA, Crippa F, Leisenring W, et al. Itraconazole versus fluconazole for prevention of fungal infections in patients receiving allogeneic stem cell transplants. Blood. 2004;103(4):1527–1533. doi:10.1182/blood-2003-08-2644
28. Annaloro C, Oriana A, Tagliaferri E, et al. Efficacy of different prophylactic antifungal regimens in bone marrow transplantation. Haematologica. 1995;80(6):512–517.
29. Chaftari AM, Hachem RY, Ramos E, et al. Comparison of posaconazole versus weekly amphotericin B lipid complex for the prevention of invasive fungal infections in hematopoietic stem-cell transplantation. Transplantation. 2012;94(3):302–308. doi:10.1097/TP.0b013e318254dae1
30. Goodman JL, Winston DJ, Greenfield RA, et al. A controlled trial of fluconazole to prevent fungal infections in patients undergoing bone marrow transplantation. N Engl J Med. 1992;326(13):845–851. doi:10.1056/NEJM199203263261301
31. Koh LP, Kurup A, Goh YT, Fook-Chong SM, Tan PH. Randomized trial of fluconazole versus low-dose amphotericin B in prophylaxis against fungal infections in patients undergoing hematopoietic stem cell transplantation. Am J Hematol. 2002;71(4):260–267. doi:10.1002/(ISSN)1096-8652
32. Riley DK, Pavia AT, Beatty PG, et al. The prophylactic use of low-dose amphotericin B in bone marrow transplant patients. Am J Med. 1994;97(6):509–514. doi:10.1016/0002-9343(94)90345-X
33. Slavin MA, Osborne B, Adams R, et al. Efficacy and safety of fluconazole prophylaxis for fungal infections after marrow transplantation–a prospective, randomized, double-blind study. J Infect Dis. 1995;171(6):1545–1552.
34. Wolff SN, Fay J, Stevens D, et al. Fluconazole vs low-dose amphotericin B for the prevention of fungal infections in patients undergoing bone marrow transplantation: a study of the North American Marrow Transplant Group. Bone Marrow Transplant. 2000;25(8):853–859. doi:10.1038/sj.bmt.1702233
35. Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of america. Clin Infect Dis. 2011;52(4):e56–e93. doi:10.1093/cid/cir073
36. Ko BS, Chen WT, Kung HC, et al. 2016 guideline strategies for the use of antifungal agents in patients with hematological malignancies or hematopoietic stem cell transplantation recipients in Taiwan. J Microbiol Immunol Infect. 2018;51(3):287–301. doi:10.1016/j.jmii.2017.07.005
37. Baden LR, Swaminathan S, Angarone M, et al. Prevention and Treatment of Cancer-Related Infections, Version 2.2016, NCCN Clinical Practice Guidelines in Oncology. J Nat Compr Cancer Network. 2016;14(7):882–913. doi:10.6004/jnccn.2016.0093
38. Mellinghoff SC, Panse J, Alakel N, et al. Primary prophylaxis of invasive fungal infections in patients with haematological malignancies: 2017 update of the recommendations of the Infectious Diseases Working Party (AGIHO) of the German Society for Haematology and Medical Oncology (DGHO). Ann Hematol. 2018;97(2):197–207. doi:10.1007/s00277-017-3196-2
39. Maertens J, Marchetti O, Herbrecht R, et al. European guidelines for antifungal management in leukemia and hematopoietic stem cell transplant recipients: summary of the ECIL 3–2009 update. Bone Marrow Transplant. 2011;46(5):709–718. doi:10.1038/bmt.2010.175
40.
41. Glasmacher A, Prentice A, Gorschluter M, et al. Itraconazole prevents invasive fungal infections in neutropenic patients treated for hematologic malignancies: evidence from a meta-analysis of 3,597 patients. J Clin Oncol. 2003;21(24):4615–4626. doi:10.1200/JCO.2003.04.052
42. Hamza NS, Ghannoum MA, Lazarus HM. Choices aplenty: antifungal prophylaxis in hematopoietic stem cell transplant recipients. Bone Marrow Transplant. 2004;34(5):377–389. doi:10.1038/sj.bmt.1704603
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.