Back to Journals » Infection and Drug Resistance » Volume 14
Combined Pulmonary Tuberculosis with Pulmonary and Pleural Cryptococcosis in a Patient Receiving Ruxolitinib Therapy
Authors Kasemchaiyanun A , Suwatanapongched T, Incharoen P, Plumworasawat S, Bruminhent J
Received 4 July 2021
Accepted for publication 29 July 2021
Published 21 September 2021 Volume 2021:14 Pages 3901—3905
DOI https://doi.org/10.2147/IDR.S327821
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Héctor Mora-Montes
Akarawut Kasemchaiyanun,1 Thitiporn Suwatanapongched,2 Pimpin Incharoen,3 Sirithep Plumworasawat,3 Jackrapong Bruminhent4
1Division of Pulmonary and Critical Care Medicine, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 2Division of Diagnostic Radiology, Department of Diagnostic and Therapeutic Radiology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 3Department of Pathology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand; 4Division of Infectious Disease, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Correspondence: Jackrapong Bruminhent
Division of Infectious Disease, Department of Medicine, Faculty of Medicine Ramathibodi Hospital, Mahidol University, 270 Ramathibodi Hospital, Ratchathewi, Bangkok, Thailand
Tel/Fax +66 2 201 1581
Email [email protected]; [email protected]
Abstract: With an advance in therapy, there are increasing emerging and re-emerging opportunistic infections among patients with hematologic conditions and malignancy. Herein, we present a 56-year-old woman with primary myelofibrosis who developed combined tuberculosis (TB) and cryptococcosis with extensive pulmonary, pleural, and nodal involvement during ruxolitinib therapy. Marked clinical and radiologic improvements were undoubtedly evident after receiving anti-TB and antifungal therapies and pleural drainage. Hence, the presence of atypical clinical and radiologic manifestations and incomplete responses, despite receiving adequate antimicrobial treatment, should raise concerns regarding the combined emerging and re-emerging opportunistic infections and the possibility of unusual radiologic manifestations of cryptococcosis in a ruxolitinib-treated patient.
Keywords: ruxolitinib, Janus kinase inhibitor, cryptococcosis, tuberculosis, pleural effusion, treatment, primary myelofibrosis
Introduction
Ruxolitinib, an oral inhibitor of Janus kinase (JAK) 1 and JAK2, has been approved to treat intermediate- and high-risk myelofibrosis (MF) and polycythemia vera. Ruxolitinib selectively inhibits JAK2-driven Ba/F3 cell proliferation, which plays an essential role in the pathogenesis of bone marrow fibrosis in MF.1,2 Although ruxolitinib therapy helps reduce symptomatic splenomegaly and constitutional symptoms, it may exert substantial immunosuppression by impairing activation of T cells and downregulation of cytokines. As a result, it may reduce the control of silent infections and increase the risk of reactivation of opportunistic infections. These infections include herpes simplex, zoster, hepatitis B, tuberculosis (TB), and opportunistic fungal infections (Pneumocystis jirovecii, mucormycosis and cryptococcosis).3
At present, there are only seven case reports regarding ruxolitinib-associated cryptococcal infection.4 Nevertheless, the co-occurrence of cryptococcosis and other infections is extremely rare. Combined TB and potentially life-threatening cryptococcal infection in a ruxolitinib-treated patient has never been reported.
Herein, we report a patient with primary MF who developed pulmonary TB, followed by pulmonary cryptococcosis with associated pleural and nodal involvements, 10 and 12 months after receiving ruxolitinib therapy, respectively. The patient showed marked clinical and radiological improvements after receiving antituberculous and antifungal treatments and suspending of ruxolitinib.
Case Report
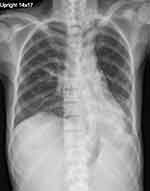
A 56-year-old woman presented with a 2-week history of shortness of breath, fever, and productive cough. She had a 3-year history of primary MF. Due to massive symptomatic splenomegaly, ruxolitinib was commenced in January 2020. She had never smoked. She worked as a government officer and had no known environmental exposure to pollutants or pets. She had received BCG vaccination in the past. There has no tuberculosis or cryptococcosis reported in household contacts of this patient. During ten months of ruxolitinib therapy, she presented to the outside hospital with a 3-week history of a cough and fever. The initial chest radiograph (Figure 1A) and computed tomography (CT) scan of the chest showed extensive airspace consolidation in the left lower lobe and numerous fine nodular opacities in both lungs. There were enlarged right supraclavicular and subcarinal lymph nodes without pleural effusion. She was diagnosed as having lobar pneumonia and received levofloxacin for seven days without any clinical improvement. Sputum staining for Gram- and acid-fast bacilli (AFB) were negative. The sputum polymerase chain reaction for Mycobacterium tuberculosis (PCR-MTB) was detected. Therefore, pulmonary TB was diagnosed, and isoniazid (H), rifampin (R), pyrazinamide (Z), and ethambutol (E) were initiated.
Despite negative sputum culture for TB, antituberculous therapy was continued, and the patient’s symptoms subsided. Two months later, she was admitted with a marked deterioration in the shortness of breath, especially on exertion, productive cough, and fever for two weeks. She was treated with oral amoxicillin/clavulanic acid for three days before admission. On evaluation, she was ill-appearing. Vital signs included a temperature of 36.5°C, blood pressure of 110/70 mmHg, heart rate of 92 /minute, and respiratory rate of 20 /minute. Her oxygen saturation was 96% at ambient air. Respiratory examination revealed bilateral crackles and decreased breath sound at the left lower lung. The patient’s abdominal examination revealed massive splenomegaly.
The complete blood count showed hemoglobin level of 9.3 g/dL, a white blood cell (WBC) count of 52,710 cells/uL (60% neutrophil, 7% lymphocyte, 10% eosinophil, 2% band form, 4% metamyelocyte, 5% myelocyte and 4% promyelocyte), and platelet count of 185,000 cells/uL. Anti-HIV testing was non-reactive. Other blood tests were unremarkable, except for increased serum lactate dehydrogenase (LDH, 1302 U/L) and alkaline phosphatase (181 U/L).
Follow-up chest radiography (Figure 1B) and chest CT scan (Figure 1C and D) showed marked progression of bilateral pulmonary abnormalities with newly developed and left pleural effusion and progressive nodal enlargement.
Ultrasound-guided thoracentesis yielded 400 mL of slightly turbid, yellow fluid. Pleural fluid analysis revealed WBC of 5266 cell/mm3 (mononuclear cell of 77%, neutrophil of 23%), protein of 4.62 g/dL (serum protein of 6.07 g/dL), and LDH of 780 U/L. Bronchoscopy performed later revealed minimal whitish secretion was seen along the lower trachea and mild narrowing of the left basal trunk. Bronchoscopy with bronchoalveolar lavage (BAL) and transbronchial biopsy were performed. PCR-MTB and AFB staining of BAL fluid were negative. Gomori methenamine silver (GMS) staining and cytology of BAL fluid showed budding yeasts (Figure 2A). Histopathological sections of the left lower lobe and right upper lobe biopsies showed non-necrotizing granulomatous inflammation (Figure 2B) and organizing pneumonia (Figure 2C), respectively, but showed no organisms on AFB and GMS stains.
However, BAL and pleural fluid cultures grew Cryptococcus neoformans, and serum cryptococcal antigen was positive with a titer of 1:1024. The left chest tube was introduced. The lumbar puncture and cerebrospinal fluid analysis were performed and showed no central nervous system involvement.
The final diagnosis of combined pulmonary TB and cryptococcosis with extensive pulmonary, pleural and nodal involvement was entertained.
She was started on amphotericin B deoxycholate (0.7 mg/kg per day) plus flucytosine (100 mg/kg per day orally) and administration of ruxolitinib was therefore discontinued. Over the following four weeks, the patient had significant clinical and radiographic improvements. Eleven months of oral fluconazole was chronologically prescribed after completing induction therapy for 28 days. She received completing 2 months of intensive phase anti-TB drugs (2HRZE) before cryptococcosis diagnosis then continued anti-tuberculous with 10 months of isoniazid and ethambutol due to possible drug interactions between rifampicin and fluconazole. Upon follow-up, further clinical and radiographic improvements (Figure 3) were observed without any adverse drug reactions.
Discussion
Ruxolitinib has been approved to treat intermediate and high-risk MF. It helps reduce the splenic size and constitutional symptoms and increase survival benefits.1,2 On the other hand, ruxolitinib has several unfavorable effects on innate and adaptive immunity. Ruxolitinib impaired natural killer cell maturation, attenuates the dendritic cell function and T cell response, causing suppression of multiple cytokines. Thus, it increases susceptibility to common intracellular pathogens, including Mycobacterium spp., Coccidioides spp. and Cryptococcus spp.3,4
At present, there are only two case reports having pulmonary cryptococcosis secondary to ruxolitinib treatment. Both the patients had isolated pulmonary involvement without cryptococcal antigenemia and showed clinical improvement after suspending ruxolitinib and receiving antifungal therapy.5,6
Moreover, combined cryptococcosis and other infections in ruxolitinib-treated patients are rare. Only two patients, one with disseminated histoplasmosis and cryptococcal meningitis7 and cryptococcal fungemia and another with cellulitis caused by Mycobacterium haemophilum,4 have been reported. Combined pulmonary TB and cryptococcosis with widespread pulmonary involvement, pleural empyema, lymphadenopathy and cryptococcal antigenemia, similar to the present case, has never been reported.
Due to nonspecific clinical and radiographic manifestations overlapping with pulmonary TB and other infections, the diagnosis of cryptococcosis is challenging. As shown in the present case, the presence of numerous fine nodular opacities, mediastinal lymphadenopathy, positive PCR-MTB, and clinical response to anti-TB therapy hindered pulmonary cryptococcosis diagnosis.8,9 Hence, an alternative diagnosis should always be considered when there is a slow resolution of clinical symptoms and radiographic abnormalities despite the appropriate treatment. Further workup, especially histopathological and microbiological studies, is crucial for the correct diagnosis and management.
We speculated that the immunomodulatory effects of both TB and cryptococcosis might increase susceptibility to each other.10,11 Thus far, it has remained inconclusive whether TB preceded cryptococcosis or the infections initially coincided in the present case. Continuous deterioration of T cell-mediated immunity induced by ruxolitinib and the delay in the diagnosis and treatment of cryptococcosis might reduce recruiting activated macrophages and clearing and quarantining organisms to form a granuloma and the progression to severe cryptococcosis in the present case. Our case clearly supported that ruxolitinib suppressed T-cell mediated immune response which cause increase susceptibility to many intracellular pathogens. Hence, suspension of ruxolitinib is recommended in order to restore cell-mediated immunity and had a favorable outcome as shown in the present case. Although a solid conclusion regarding a relationship between cryptococcosis and ruxolitinib therapy has not been established, a plausible association should be considered based on several case reports and series published in the literature.4 Although male sex seems to be predominated, a gender preference and this infection have not been confirmed. We believe an immune defect, especially cell-mediated immunity, would rather play a significant role in pathogenesis. Further studies to investigate for an explanation or pathogenesis are encouraged.
Organizing pneumonia can be idiopathic or secondary to various non-specific chronic inflammatory lung injuries, such as infection, drug toxicity, inhalation of toxic gas, and collagen vascular diseases.14 It represents a process of unusual lung repair and, therefore, can complicate infection or persist after the resolution of the infection. Interestingly, pulmonary cryptococcosis causing organizing pneumonia is rare, with only seven cases have been reported.12,13 The presence of combined granulomatous pneumonia and organizing pneumonia might be responsible for developing extensive consolidation and prolonged clinical course in the present case. Although the patient did not receive steroid treatment, clinical and radiographic improvements were undoubtedly evident after antifungal therapy and pleural drainage.
Conclusion
In summary, we presented the first patient with primary MF who developed combined TB and cryptococcosis during ruxolitinib therapy. Thus, clinicians should always be aware of combined emerging and re-emerging opportunistic infections in a ruxolitinib-treated patient, especially those with atypical clinical and radiologic manifestations and incomplete responses, despite receiving adequate antimicrobial treatment. Clinicians should be careful in the evaluation of the infective risk before administering ruxolitinib. TB screening, especially for those who live in endemic areas or with prior history of TB followed by chemoprophylaxis before ruxolitinib therapy is encouraged. While large-scale study is needed to define a solid evidence-based antifungal prophylactic strategy for cryptococcosis in these patients, meantime, patients needed to be closely clinical surveillance for their risk of the opportunistic infection.
Ethic and Consent Statement
This case report was approved by the Ethics Committee on Human Rights related to research involving human subjects at Faculty of Medicine Ramathibodi Hospital. The written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Disclosure
The authors declare that they have no conflicts of interest. The authors have indicated that they have no financial relationships relevant to this study to disclose.
References
1. Zhang S, Wang Y, Sanchez L, et al. Efficacy and safety of single-agent ruxolitinib therapy for myelofibrosis and polycythemia vera: a meta-analysis. Blood. 2015;126(23):1622. doi:10.1182/blood.V126.23.1622.1622
2. Zahr AA, Salama ME, Carreau N, et al. Bone marrow fibrosis in myelofibrosis: pathogenesis, prognosis and targeted strategies. Haematologica. 2016;101(6):660–671. doi:10.3324/haematol.2015.141283
3. Lussana F, Cattaneo M, Rambaldi A, et al. Ruxolitinib-associated infections: a systematic review and meta-analysis. Am J Hematol. 2018;93(3):339–347. doi:10.1002/ajh.24976
4. Sayabovorn N, Chongtrakool P, Chayakulkeeree M. Cryptococcal fungemia and Mycobacterium haemophilum cellulitis in a patient receiving ruxolitinib: a case report and literature review. BMC Infect Dis. 2021;21(1):27. doi:10.1186/s12879-020-05703-2
5. Wysham NG, Sullivan DR, Allada G. An opportunistic infection associated with ruxolitinib, a novel janus kinase 1,2 inhibitor. Chest. 2013;143(5):1478–1479. doi:10.1378/chest.12-1604
6. Hirano A, Yamasaki M, Saito N, et al. Pulmonary cryptococcosis in a ruxolitinib-treated patient with primary myelofibrosis. Respir Med Case Rep. 2017;22:87–90.
7. Prakash K, Richman D. A case report of disseminated histoplasmosis and concurrent cryptococcal meningitis in a patient treated with ruxolitinib. BMC Infect Dis. 2019;19(1):287. doi:10.1186/s12879-019-3922-6
8. Qu J, Zhang X, Lu Y, et al. Clinical analysis in immunocompetent and immunocompromised patients with pulmonary cryptococcosis in western China. Sci Rep. 2020;10:9387. doi:10.1038/s41598-020-66094-7
9. Liu K, Ding H, Xu B, et al. Clinical analysis of non-AIDS patients pathologically diagnosed with pulmonary cryptococcosis. J Thorac Dis. 2016;8(10):2813–2821. doi:10.21037/jtd.2016.10.36
10. Huang CT, Tsai YJ, Fan JY, et al. Cryptococcosis and tuberculosis co-infection at a university hospital in Taiwan, 1993–2006. Infection. 2010;38(5):373–379. doi:10.1007/s15010-010-0045-9
11. Fang W, Zhang L, Liu J, et al. Tuberculosis/cryptococcosis co-infection in China between 1965 and 2016. Emerg Microbes Infect. 2017;6(8):e73. doi:10.1038/emi.2017.61
12. Kessler AT, Al Kharrat T, Kourtis AP. Cryptococcus neoformans as a cause of bronchiolitis obliterans organizing pneumonia. J Infect Chemother. 2010;16(3):206–209. doi:10.1007/s10156-010-0039-7
13. Chikumoto A, Oishi K, Hamada K, et al. Cryptococcosis as a cause of organizing pneumonia. Respir Med Case Rep. 2019;27:100851.
14. Cordier JF. Cryptogenic organising pneumonia. Eur Respir J. 2006;28(2):422–446. doi:10.1183/09031936.06.00013505
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.