Back to Journals » OncoTargets and Therapy » Volume 13
Combination of CpG Oligodeoxynucleotide and Anti-4-1BB Antibody in the Treatment of Multiple Hepatocellular Carcinoma in Mice
Authors Ma S, Yang X, Zhou H, Zhang C, Kang J, Sun D
Received 6 May 2020
Accepted for publication 30 June 2020
Published 20 July 2020 Volume 2020:13 Pages 6997—7005
DOI https://doi.org/10.2147/OTT.S260353
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Leo Jen-Liang Su
Shizhao Ma,* Xinying Yang,* Huifang Zhou, Chaoqun Zhang, Jiwen Kang, Dianxing Sun
The Liver Disease Center of PLA, The 980th Hospital of PLA Joint Logistics Support Force, Shijiazhuang 050082, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Dianxing Sun
The Liver Disease Center of PLA,The 980th Hospital of PLA Joint Logistics Support Force, Shijiazhuang 050082, People’s Republic of China
Tel +86 31187978434
Email [email protected]
Background: To investigate the effect of topical application of CpG oligodeoxynucleotide (CpG-ODN) combined with anti-4-1BB antibodies on mouse HCC multiple tumor-bearing models and the degree of improvement of anti-tumor immune response in mice.
Materials and Methods: We inoculated each BALB/c male mouse subcutaneously with one tumor in the axillae of the four limbs and divided them into four groups. We only selected the tumor-bearing part of the left lower limb for drug treatment. We measured the tumor-bearing volume of mice in each group. Then, we tested the organ coefficients of mice, the concentrations of IL-12 and IFN-γ in peripheral blood, the ratio of spleen Tregs and CD8+T cells, the spleen CTL killing activity, and the survival time of mice.
Results: We found that the tumor-bearing volume decreased significantly after the combination of CpG-ODN and anti-4-1BB antibody (P< 0.001). The organ coefficients of treated mice were not significantly different from normal mice (P> 0.05). The concentration of IL-12 and IFN-in serum and the ratio of CD8+T cells in spleen were increased, while the ratio of spleen Tregs was decreased. CTL activity of spleen was increased. The survival time of mice was significantly prolonged (P< 0.001).
Conclusion: The treatment programme combining CpG-ODN with an anti-4-1BB antibody can significantly reduce tumor growth at the treatment site, slow the growth rate of metastases and improve host prognosis.
Keywords: hepatocellular carcinoma, 4-1BB, CpG-ODN, immunity therapy
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors in the clinic and is highly invasive. According to reports, nearly 750,000 new cases of HCC occur worldwide each year.1 The incidence and mortality rates of HCC in China are higher than the world averages,2 and HCC exhibits a progressive growth trend, with the affected population becoming younger.3 Since most tumor patients are diagnosed at an advanced stage, often with multiple metastatic lesions, frequently used treatments may not be able to achieve the desired therapeutic effects.4 Although early-stage liver cancer can feasibly be treated by surgical resection, there is still the possibility of postoperative recurrence.5
The poor treatment of tumors is mainly due to changes in the tumor microenvironment (TME).6 Inhibition of the immune system promotes tumor progression. Numerous studies have shown that we can improve the anti-tumor immune effect of the body by applying immunotherapy to improve TME.7,8
CpG-ODN is an oligodeoxynucleotide containing unmethylated CpG, which is recognized by TLR9 and triggers a defence mechanism of the body.9 CpG-ODN acts as an immunopotentiator to non-specifically activate cellular immunity. CpG-ODN not only promotes the production of pro-inflammatory Th1 cytokines and the maturation and activation of APCs but also enhances the immunogenicity of peptide tumor vaccines.10,11 It induces a cytokine microenvironment that promotes a helper T cell response.12 Studies have shown that CpG-ODN can be used as an adjuvant to improve the therapeutic effect of radiotherapy or radiofrequency ablation on HCC.13,14 However, the effect of CpG-ODN alone on liver tumor suppression is not satisfactory, and the progress of metastatic tumors cannot be controlled. The conventional treatment plan is still dominant, or CpG-ODN is further modified to improve the therapeutic effect.15
As a new type of immune checkpoint molecule, the costimulatory molecule 4-1BB has been studied in recent immune-related research. The 4-1BB molecule is a type I transmembrane protein belonging to the tumor necrosis factor receptor superfamily and is expressed on the surface of activated T lymphocytes.16,17 The expression of 4-1BB mRNA can be detected within a few hours after T cells are stimulated and activated.18 Studies have shown that the anti-tumor effect of anti-4-1BB antibodies mainly depends on CD8+T cells,19 and IFN and CD40 are also required for this reaction.20,21 In addition, anti-4-1BB antibodies have a synergistic enhancing effect with IL-12 against tumors.22 4-1BB molecules can be expressed on DCs, while CpG-ODN can promote DC activation, which in turn promotes the expression of 4-1BB molecules. Therefore, we envisage that the addition of an anti-4-1BB antibody to CpG-ODN therapy is likely to achieve a stronger immunotherapeutic effect than CpG-ODN or anti-4-1BB antibody alone. In this experiment, we had focussed on evaluating the difference in efficacy before and after antibody addition. And some indicators were detected to evaluate changes in anti-tumor immune-mediated effects.
Materials and Methods
Primary Reagents
The CpG-ODN1826 sequence was 5ʹ-TCCATGACGTTCCTGACGTT-3ʹ23 and CpG-ODN was subjected to non-methylation treatment, total thiol modification, and HPLC purification. This sequence was synthesized by Shanghai Shenggong Bioengineering Technology Service Co., Ltd. Anti-4-1BB antibody (BioXCell, BE0239), FITC-anti-CD3 antibody (eBioscience, 85-11-0032-80), APC-anti-CD8 antibody (eBioscience, 85-17-0081-81), PE-Cy5-anti-CD4 antibody (eBioscience, 85-18-0041-81), PE-anti-Foxp3 antibody (eBioscience, 85-12-4771-80), FITC-anti-CD25 antibody (Biogems, BG-07312-50-25), mouse serum IL-12p70 ELISA kits (NeoBioscience, EMC006), mouse serum IFN-γ ELISA kits (NeoBioscience, EMC101g), and CCK-8 kit (Hangzhou Lianke Biotechnology Co., Ltd., CCK801) were purchased.
Cell Line and Experimental Animals
Mouse H22 liver cancer cells were preserved by the Hepatology Laboratory of the 980th Hospital of the PLA Joint Logistics Support Force. BALB/c male mice (SPF grade, 6–8 weeks old, weighing 18–22 g) were purchased from the Experimental Animal Center of Hebei Medical University (certificate number: 1811007) and raised by the experimental animal centre of the hospital. The research protocol was approved by the Experimental Animal Ethics Committee of the 980th Hospital of the PLA Joint Logistics Support Force (batch number: dwsy-20180515) and complied with laboratory animal management and use guidelines.
Establishment of a Mouse Liver Cancer Model and Calculation of Tumor Volume Changes for Each Group After Treatment
H22 hepatoma cells were cultured and subcultured 3 times in the peritoneal cavity of mice. The 3rd-passage ascites were harvested under aseptic conditions and processed into a cell suspension with a concentration of 1 × 107/mL using serum-free medium. A 0.2-mL suspension of the cells was injected subcutaneously into the axillary fossae of each mouse. Forty mice with a detectable tumor were selected 7 days later and randomly divided into a model control group, a CpG group, a 4-1BB group and a CpG+4-1BB group, and drugs were injected into the tumor in the left lower extremity on the 7th, 9th, and 11th days after tumor inoculation. Ten normal mice were selected as the normal group, which was used as a blank control. The model group was given physiological saline (0.05 mL/injection). The CpG group was administered CpG-ODN (60 μg/0.05 mL/injection). The 4-1BB group was administered anti-4-1BB antibody (60 μg/0.05 mL/injection). The CpG+4-1BB group was given CpG-ODN (60 μg/0.05 mL/injection) and an anti-4-1BB antibody (60 μg/0.05 mL/injection). The long diameter and the width diameter of each tumor were measured 1 time before treatment on the 7th day and every 3 days beginning on the 9th day. Tumor volume was calculated according to the formula: tumor volume (mm3) = (long diameter × width diameter2)/2. The treated tumor volume and distant tumor volumes were calculated in each group of mice.
Weighing the Heart, Lungs, Liver and Kidneys and Calculating Organ Indexes
After mice were sacrificed, we dissected and isolated the heart, lungs, liver and kidneys of the mice and weighed these organs. Organ coefficients were calculated according to the following formula: organ coefficient (%) = organ weight (mg)/body weight (g) × 10. Differences among the organ control group, the CpG group and the CpG+4-1BB group were compared to determine whether the mice experienced immune-related side effects after treatment.
Measurement of the Concentrations of IL-12 and IFN-γ in the Serum by ELISA
On the 30th day after modelling, the mice in each group were sacrificed by cervical dislocation. Blood samples were collected by eyeball extraction. The blood samples from the mice in the four groups were collected in 2-mL EP tubes, allowed to stand at room temperature for 2 hours and centrifuged at 4000 rpm for 10 minutes to separate the serum. Regarding the preparation and operation methods of the indicator test, we strictly followed the instructions of the kits.
Proportion of CD8+ T Cells in the Spleen Lymphocyte Population Detected by Flow Cytometry
After the mouse spleen was removed, a lymphocyte suspension was prepared, and the cell concentration was adjusted to 1 × 107/mL with PBS. Then, 100 μL of FITC-conjugated anti-CD3 antibody and 0.125 μg of APC-conjugated anti-CD8 antibody were added, and the mixture was mixed and incubated at 4 °C for 30 minutes in the dark. After washing with PBS, 0.5 mL of 1% paraformaldehyde was added to resuspend the cells, and the cells were detected within 24 hours.
Flow Cytometry for the Detection of the Percentage of CD4+CD25+Foxp3+ Tregs in the Spleen Lymphocyte Population
In total, 0.06 μg of PE-Cy5-conjugated anti-CD4 antibody and 0.125 μg of FITC-conjugated anti-CD25 antibody were added to 100 μL of splenocyte suspension, mixed, and incubated at 4 °C for 30 minutes in the dark. After washing with PBS once, 100 μL of membrane-permeabilization solution was added to the specimen, and incubation was again performed in the dark for 30 minutes. After washing again with PBS and centrifuging, 0.25 μg of PE-conjugated anti-Foxp3 antibody was added to the specimen, and the mixture was incubated at 4 °C for 90 minutes in the dark. After washing with PBS, 0.5 mL of 1% paraformaldehyde was added to resuspend the cells, and the cells were detected within 24 hours.
Detection of the CTL Killing Activity of Mouse Splenocytes
The mice in the model group, the CpG group, the 4-1BB group and the CpG+4-1BB group were sacrificed by cervical dislocation 1 week before the end of the treatment regimen and the end of the last administration, and the spleen was aseptically processed to prepare a cell suspension that was used as the effector population. H22 cells in a logarithmic growth cycle were adjusted to a concentration of 1 x 107/mL. A total of 100 μL per well was inoculated into a 96-well culture plate, and 100 μL of splenocyte suspension was added at an effector: target ratio of 5:1, 25:1 or 50:1. Three replicate wells were set up for each group, and a target cell group, an effector cell group, and a blank control group were simultaneously established. The cells were cultured for 4 hours at 37 °C in a 5% CO2 incubator, and then 10 μL of CCK-8 reagent was added to each well and incubated for an additional 4 hours. The absorbance (A) of each well at a wavelength of 450 nm was measured with a microplate reader. CTL killing activity was calculated according to the formula: killing activity (%) = [1 - (target mixed cell average A value – effector cell average A value)/target cell average A value] × 100%.
Evaluation of Mouse Survival After Intervention
The survival of mice after treatment was evaluated to determine the potential of CpG-ODN combined with an anti-4-1BB monoclonal antibody. The viability and survival time of each group of mice were observed from the first administration (day 7 after modelling) to 90 days.
Statistical Analysis
All of the above experiments were repeated. Statistical analysis was performed using SPSS19.0 software. Measurement data are presented as the mean ± standard deviation. Repeated measures were used to design analysis of variance (ANOVA). If necessary, comparisons between groups were performed by the LSD-t test. The survival rate of each group of mice (except the normal control group) was analysed by the Log rank test. A P value < 0.05 was considered to be statistically significant. All statistical tests were two-sided.
Results
Tumor Volume Was Significantly Reduced in Mice Treated with CpG-ODN and an Anti-4-1BB Antibody
We divided model mice into four groups according to the treatment plan and implemented the treatment plan according to the above plan. On the 7th day after modelling, that is, before the first dose of treatment was administered, we measured and calculated tumor volumes. The average volume of each of the four tumors of the 10 mice in each group was between 117 and 120 mm3, and there were no significant differences, which met the screening requirements of the experimental model.
After 3 doses, we measured tumor volume on the 13th day after modelling and found a significant change in the tumor volumes of the CpG group, the 4-1BB group and the CpG+4-1BB group (F = 1084.64, P< 0.001, Figure 1A. The data results are shown in Table 1A in the Supplementary Materials.). The average tumor volume in the left lower limb in the model control group was 316.75±7.21 mm3. The average volume in the CpG group (252.73±6.95 mm3) and 4-1BB group (216.48±5.83 mm3) were significantly smaller than that in the model group, and the volume of 4-1BB group was smaller than that of CpG group, while that in the CpG+4-1BB group was only 189.20±3.49 mm3. Then, we found new discoveries. After the 13th day, the tumors in the left lower limb of the CpG+4-1BB group showed a trend of regression, and the volume measured on the 29th day was only 47.25±0.93 mm3. The differences between the CpG+4-1BB group and the model control group, the CpG group or the 4-1BB group were significant (F = 5470.29, P< 0.001).
In observing changes in tumor volume, we also focused on changes in distant untreated homologous metastases. We found that the treatment effect did not meet our expectations. The 3 metastases in the model control group and the CpG group showed progressive growth trends that were not significantly different (P> 0.05). In the 4-1BB group, only a few tumors in the three untreated tumors showed a slower growth rate, but this was not what we want. After the addition of the anti-4-1BB monoclonal antibody, the therapeutic effect was significantly improved. Compared with that in the model control group, the CpG group and the 4-1BB group, the growth rate of the three metastases in the CpG+4-1BB was slower [P< 0.001, Figure 1B–D. The data results are shown in Table 1B–D in the Supplementary Materials].
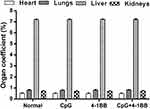
There Were No Obvious Autoimmune Lesions in the Organs of Immunotherapy-Treated Mice
Comparisons showed that there were no significant differences in the organ coefficients among the model control group, the CpG group, the 4-1BB group and the CpG-4-1BB group (P> 0.05, Figure 2. The data results are shown in Table 2 in the Supplementary Materials). Because the overall drug doses were small, there were no obvious immune-related side effects.
Increased Serum IL-12 and IFN-γ Levels After Anti-4-1BB Antibody Treatment
We first tested the serum IL-12 and IFN-γ concentrations in normal mice for comparison with the mice in the model control group. The results showed that the concentrations of these two cytokines in the model group were lower than those in the normal mice (P< 0.001). Next, we compared differences in cytokine concentrations among the model group, the CpG group, the 4-1BB group and the CpG+4-1BB group. The results showed that the concentrations of these two cytokines were significantly different among the three groups (F(IL-12) = 104.08, P< 0.001, Figure 3A; F(IFN-γ) = 171.41; P< 0.001, Figure 3B. The data results are shown in Table 3 in the Supplementary Materials). Compared with the CpG group and the 4-1BB group, the concentrations of both cytokines were significantly increased in the CpG+4-1BB group (P< 0.001).
Combination Therapy with CpG-ODN and the Anti-4-1BB Antibody Inhibited Treg Proliferation and Enhanced CD8+T Cell Proliferation
We first compared changes in the proportions of two immune cell types in the spleen lymphocytes of the mice in the normal and model groups. The results showed that the proportion of CD4+CD25+Foxp3+Tregs in the spleen lymphocyte population was higher in the model group than in the normal group (P < 0.001, Figure 4A), and the proportion of spleen CD8+T cells was lower in the model group than in the normal group (P< 0.001, Figure 4B). Next, we examined the proportions of these two immune cell types in the mouse spleen lymphocyte population in the CpG group, the 4-1BB group and the CpG+4-1BB group. Compared with that in the model control group, the proportions of CD4+CD25+Foxp3+Tregs in the CpG group, the 4-1BB group and the CpG+4-1BB group were decreased (F = 123.91, P< 0.001, Figure 4A), and the proportions of CD8+T cells were increased (F = 343.94, P< 0.001, Figure 4B). Comparing groups showed that the proportion of CD4+CD25+Foxp3+Tregs in the CpG+4-1BB group was lower than that in the CpG group and the 4-1BB group (P< 0.001, Figure 4A) and the proportion of CD8+T cells in the CpG+4-1BB group was higher than that in the CpG group and the 4-1BB group (P< 0.001, Figure 4B). The data results are shown in Table 4 in the Supplementary Materials.
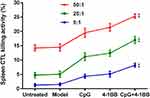
Enhancement of the Killing Ability of Spleen CTLs in Mice Treated with CpG-ODN and the Anti-4-1BB Antibody
To further demonstrate the ability of CpG-ODN in combination with anti-4-1BB antibody treatment to enhance antitumor immunity, we performed an in vitro cytotoxic T lymphocyte (CTL) killing activity assay. Among three different target ratios, the CTL killing effect of the CpG + 4-1BB group was significantly higher than that of the model group, CpG group and 4-1BB group (F = 311.88, P< 0.001, Figure 5. The data results are shown in Table 5 in the Supplementary Materials). And in the same group, with the increase of the effective target ratio, the killing activity of CTL is also stronger.
The Anti-4-1BB Antibody Significantly Prolonged the Survival Time of Tumor-Bearing Mice
With tumor progression, all 10 mice in the model group died, with an average survival time of 44.9 days. The mice in the CpG group were all died, with an overall mean survival time of 46.0 days. After statistical analysis, there was no significant difference between the two groups (P> 0.05). Only one mouse in the 4-1BB group survived, and the overall average survival time in this group was 63.6 days. Compared with the model group and CpG group, the survival time was prolonged (P< 0.05). When mice were given the CpG-ODN and anti-4-1BB antibody treatment, the general state of the mice was significantly improved, and the overall average survival time was 86.9 days (χ2 = 48.766, P< 0.001, Figure 6. The data results are shown in Table 6 in the Supplementary Materials). However, there were still 3 mice that died, which may be due to differences in individual tolerance of the treatment.
Discussion
The TME is the key component affecting tumor progression. Changes in the TME inhibit the body’s anti-tumor immune response so that a patient’s own immune system cannot resist tumor cell invasion, which in turn affects the patient’s therapeutic effect.24 Different types of tumors or homologous tumors occur in different patients, the TME undergoes subtle changes, and we cannot accurately discern its state. However, under certain conditions, the TME will behave in a specific manner. We can associate these typical manifestations with the mechanism of tumor progression and find breakthrough points to achieve tumor control and even tumor elimination.25,26 We evaluated some indicators in normal mice in the experiment and compared these indicators with those in tumor-bearing mice. The reason for this inclusion was to verify that tumor production had a negative impact on the body’s immune function. The comparison showed that CD8+T cell numbers decreased, secreted cytokine levels decreased, and Treg numbers increased significantly due to tumor progression. It can be seen that the combined effects of tumor cells and various negative changes in immune function-related factors make the body’s immunity against tumors decline and promote the occurrence of tumor immune escape, which directly affects the therapeutic effect.
In this experiment, we inoculated tumors in four different parts of the mouse body to establish a model of homologous tumor metastasis in liver cancer. The tumor growth at the injection site was significantly reduced after the addition of the antibody, and the tumor growth in the untreated distant sites was significantly inhibited. This result indicates that when a tumor in one site is selected for treatment, homologous tumors in other sites may also experience different degrees of therapeutic effects, showing the specificity of the therapeutic effect direct against tumor antigens in different sites.27 It also indicates that local immunomodulation induced by in situ injection into the tumor is sufficient to elicit a systemic anti-tumor immune response.28 Correlation analysis of the safety and tolerability of CpG-ODN and anti-4-1BB antibodies was performed in most previous studies, and the results were satisfactory. Long-term use may cause some flu-like symptoms such as fever, headache, and nausea, and some patients may experience blood system diseases and some strong immune reactions,15,29,30 but these side effects have not yet reached lethality. However, the dose we used was small, the number of injections was small, and no histological damage to organs was observed upon mouse necropsy. In view of the safety of drugs, we should also carry out the detection of liver function, alpha-fetoprotein and other blood indicators, which are commonly used and convincing indicators in clinical practice. However, due to poor laboratory practice, we do not have more serum samples for detection of related indicators.
Tregs, CD8+T cells, IL-12 and IFN-γ are important components of the TME and play crucial roles in the antitumor immune response.31 By comparing the test indexes of the normal group and the model group, we can see that the growth of a tumor disrupts the balance of the TME. The increase in the number of Tregs and the reductions in the number of CD8+T cells and levels of IL-12 and IFN-γ all promoted the occurrence of tumor immune escape. When we injected CpG-ODN or anti-4-1BB antibody into the tumor, the tumor growth rate at the injected drug site slowed, and the indicators were improved compared with those of the model group. However, CpG group had no therapeutic effect in three distant sites, while 4-1BB group had only a partial slow-down in distant sites. When we added the anti-4-1BB antibody, the growth of the tumor at the site of administration not only slowed within a few days of treatment but tumor volume also decreased afterwards. The three tumors that were not treated also showed obvious therapeutic effects, and the tumor growth rate slowed. The indicators for post-testing also improved significantly. Although tumor volume and various indicators were improved after CpG-ODN administration alone, the three untreated tumors in the CpG-ODN group were not different from those in the model group. This finding indicates that CpG-ODN has a relatively small enhancing effect on the anti-tumor immune effect and local application does not induce an enhancement of the systemic anti-tumor immune response. Although the anti-4-1BB antibody has a stronger therapeutic effect than CpG-ODN, it is also ineffective in treating distant tumors. However, when CpG-ODN and the anti-4-1BB antibody are combined, the local anti-tumor immune effect is extended to the whole body due to the synergy between the two therapeutics, reducing the growth rates of the three untreated tumors and significantly improving test indicators.
We want to achieve the goal of curing HCC via the combination of CpG-ODN and an anti-4-1BB antibody. However, this ideal goal was not achieved through this experiment. The reason for this may be that the doses of drugs we used were small or the route of administration was not suitable. If we increase the drug doses appropriately or administer the drugs intravenously, the treatment outcome may be more desirable. These changes will be the key issues we need to think about in subsequent experiments. This experiment was performed to observe only the preliminary effects of CpG-ODN combined with anti-4-1BB antibody for the treatment of liver cancer. In the future, we will design combinations of different doses of the two drugs to test to determine the best treatment. At the same time, differences in efficacy among different routes of administration will be compared.
This study is only about the therapeutic effect of HCC, but it is not clear about the specificity of the therapeutic regimen for HCC. We will also study the efficacy of CpG-ODN in combination with anti-4-1BB antibody in the treatment of various types of tumors in order to evaluate the specificity of the second treatment regimen in the treatment of HCC. The treatment of liver cancer is still the focus of current research. At present, in the international arena, people have established HCC immunotherapy as a top research priority, and the research results are endless. Immunotherapy will eventually replace the existing treatment programme, which will bring new hope for survival to HCC patients.
Acknowledgments
The authors thank Professor Dianxing Sun for his guidance on the experiment and writing of this paper, Kiwen Kang for his guidance on experimental techniques, Chaoqun Zhang and Huifang Zhou for their help with experimental data collection, and Xinying Yang for her help with data analysis. This study was supported by grants from the National Natural Science Foundation of China (No.30571667).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Greten TF, Duffy AG, Korangy F. Hepatocellular carcinoma from an immunologic perspective. Clin Cancer Res. 2013;19(24):6678–6685. doi:10.1158/1078-0432.CCR-13-1721
2. Grandhi MS, Kim A, Ronnekleivkelly SM, et al. Hepatocellular carcinoma: from diagnosis to treatment. Surg Oncol. 2016;25(2):74–85. doi:10.1016/j.suronc.2016.03.002
3. Gomes MA, Priolli DG, Tralhao JG, et al. Carcinoma hepatocelular: epidemiologia, biologia, diagnóstico e terapias. Rev Assoc Med Bras. 2013;59(5):514–524. doi:10.1016/j.ramb.2013.03.005
4. Sherman M. Hepatocellular carcinoma. Gastroenterologist. 2016;20(4):703–720.
5. Molinari M, Coutere SD, Krahn M, et al. Patients’ preferences and trade-offs for the treatment of early stage hepatocellular carcinoma. J Surg Res. 2014;189(1):57–67. doi:10.1016/j.jss.2014.02.015
6. Ye L, Zhang Q, Cheng Y, et al. Tumor-derived exosomal HMGB1 fosters hepatocellular carcinoma immune evasion by promoting TIM-1 + regulatory B cell expansion. J Immunother Cancer. 2018;6(1):1–15. doi:10.1186/s40425-018-0451-6
7. Lv Y, Zhao Y, Wang X, et al. Increased intratumoral mast cells foster immune suppression and gastric cancer progression through TNF-α-PD-L1 pathway. J Immunother Cancer. 2019;7(1):1–15. doi:10.1186/s40425-019-0530-3
8. Capasso A, Lang J, Pitts TM, et al. Characterization of immune responses to anti-PD-1 mono and combination immunotherapy in hematopoietic humanized mice implanted with tumor xenografts. J Immunother Cancer. 2019;7(1):37. doi:10.1186/s40425-019-0518-z
9. Sommariva M, Noci VL, Storti C, et al. Activation of NK cell cytotoxicity by aerosolized CpG-ODN/Poly(I:C) against lung melanoma metastases is mediated by alveolar macrophages. Cell Immunol. 2017;313:52–58. doi:10.1016/j.cellimm.2017.01.004
10. Lai C, Duan S, Ye F, et al. The enhanced antitumor-specific immune response with mannose- and CpG-ODN-coated liposomes delivering TRP2 peptide. Theranostics. 2018;8(6):1723–1739. doi:10.7150/thno.22056
11. Li J, Li J, Aipire A, et al. The combination of Pleurotus ferulae water extract and CpG-ODN enhances the immune responses and antitumor efficacy of HPV peptides pulsed dendritic cell-based vaccine. Vaccine. 2016;34(31):3568–3575. doi:10.1016/j.vaccine.2016.05.022
12. Ignacio RMC, Kim CS, Kim YD, et al. Therapeutic effect of Active Hexose-Correlated Compound (AHCC) combined with CpG-ODN (oligodeoxynucleotide) in B16 melanoma murine model. Cytokine. 2015;76(2):131–137. doi:10.1016/j.cyto.2015.06.002
13. Liang S, Hu J, Xie Y, et al. A polyethylenimine-modified carboxyl-poly(styrene/acrylamide) copolymer nanosphere for co-delivering of CpG and TGF-β receptor I inhibitor with remarkable additive tumor regression effect against liver cancer in mice. Int J Nanomedicine. 2016;11:6753–6762. doi:10.2147/IJN.S122047
14. Zhang Q, Yuan RF, Li XH, et al. Clinical effects of CpG-based treatment on the efficacy of hepatocellular carcinoma by skewing polarization toward M1 macrophage from M2. Cancer Biother Radiopharm. 2017;32(6):215–219. doi:10.1089/cbr.2017.2240
15. Yang X, Lai C, Liu A, et al. Anti-tumor activity of mannose-CpG-oligodeoxynucleotides-conjugated and hepatoma lysate-loaded nanoliposomes for targeting dendritic cells in vivo. J Biomed Nanotechnol. 2019;15(5):1018–1032. doi:10.1166/jbn.2019.2755
16. Compte M, Harwood SL, Munoz IG, et al. A tumor-targeted trimeric 4-1BB-agonistic antibody induces potent anti-tumor immunity without systemic toxicity. Nat Commun. 2018;9(1):4809. doi:10.1038/s41467-018-07195-w
17. Menk AV, Scharping NE, Rivadeneira DB, et al. 4-1BB costimulation induces T cell mitochondrial function and biogenesis enabling cancer immunotherapeutic responses. J Exp Med. 2018;215(4):1091–1100. doi:10.1084/jem.20171068
18. Chester C, Sanmamed MF, Wang J, et al. Immunotherapy targeting 4-1BB: mechanistic rationale, clinical results, and future strategies. Blood. 2017;131(1):49–57. doi:10.1182/blood-2017-06-741041
19. Buchan SL, Dou L, Remer M, et al. Antibodies to costimulatory receptor 4-1BB enhance anti-tumor immunity via T regulatory cell depletion and promotion of CD8 T cell effector function. Immunity. 2018;49(5):958–970. doi:10.1016/j.immuni.2018.09.014
20. Misumi T, Tanabe K, Fujikuni N, et al. Stimulation of natural killer cells with rhCD137 ligand enhances tumor-targeting antibody efficacy in gastric cancer. PLoS One. 2018;13(10):e0204880. doi:10.1371/journal.pone.0204880
21. Fromm G, De Silva S, Giffin L, et al. Gp96-Ig/Costimulator (OX40L, ICOSL, or 4-1BBL) combination vaccine improves T-cell priming and enhances immunity, memory, and tumor elimination. Cancer Immunol Res. 2016;4(9):766–778. doi:10.1158/2326-6066.CIR-15-0228
22. Pan P, Gu P, Li Q, et al. Regulation of dendritic cell function by NK cells: mechanisms underlying the synergism in the combination therapy of IL-12 and 4-1BB activation. J Immunol. 2004;172(8):4779–4789. doi:10.4049/jimmunol.172.8.4779
23. Sfondrini L, Sommariva M, Tortoreto M, et al. Anti-tumor activity of CpG-ODN aerosol in mouse lung metastases. Int J Cancer. 2013;133(2):383–393. doi:10.1002/ijc.28028
24. Horsman MR, Vaupel P. Pathophysiological basis for the formation of the tumor microenvironment. Front Oncol. 2016;6:66. doi:10.3389/fonc.2016.00066
25. Ventola CL. Cancer immunotherapy, Part 3: challenges and future trends. P T. 2017;42(8):514.
26. Li X, Shao C, Shi Y, et al. Lessons learned from the blockade of immune checkpoints in cancer immunotherapy. J Hematol Oncol. 2018;11(1):31. doi:10.1186/s13045-018-0578-4
27. Lussier DM, Woolf EC, Johnson JL, et al. Enhanced immunity in a mouse model of malignant glioma is mediated by a therapeutic ketogenic diet. BMC Cancer. 2016;16(1):310. doi:10.1186/s12885-016-2337-7
28. Nouri N, Garbe C. Intralesional immunotherapy as a strategy to treat melanoma. Expert Opin Biol Ther. 2016;16(5):619–626. doi:10.1517/14712598.2016.1157161
29. Ioannou XP, Gomis S, Hecker R, et al. Safety and efficacy of CpG-containing oligodeoxynucleotides as immunological adjuvants in rabbits. Vaccine. 2003;21(27):4368–4372. doi:10.1016/S0264-410X(03)00437-7
30. Ascierto PA, Simeone E, Sznol M, et al. Clinical experiences with anti-CD137 and anti-PD1 therapeutic antibodies. Semin Oncol. 2010;37(5):508–516. doi:10.1053/j.seminoncol.2010.09.008
31. Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299(5609):1057–1061. doi:10.1126/science.1079490
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.