Back to Journals » Neuropsychiatric Disease and Treatment » Volume 10
Cognitive impairment and memory dysfunction after a stroke diagnosis: a post-stroke memory assessment
Authors Al-Qazzaz N, Ali S, Ahmad SA, Islam S, Mohamad K
Received 3 May 2014
Accepted for publication 27 May 2014
Published 9 September 2014 Volume 2014:10 Pages 1677—1691
DOI https://doi.org/10.2147/NDT.S67184
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Noor Kamal Al-Qazzaz,1,5 Sawal Hamid Ali,1 Siti Anom Ahmad,2 Shabiul Islam,3 Khairiyah Mohamad4
1Department of Electrical, Electronic and Systems Engineering, Faculty of Engineering and Built Environment, Universiti Kebangsaan Malaysia, Bangi, Selangor, Malaysia; 2Department of Electrical and Electronic Engineering, Faculty of Engineering, Universiti Putra Malaysia, Serdang, Selangor, Malaysia; 3Institute of Microengineering and Nanoelectronics (IMEN), Universiti Kebangsaan Malaysia, Bangi, Selangor, Malaysia; 4Neurology Unit, Department of Medicine, Universiti Kebangsaan Malaysia Medical Center, Cheras, Kuala Lumpur, Malaysia; 5Department of Biomedical Engineering, Al-Khwarizmi College of Engineering, Baghdad University, Baghdad, Iraq
Abstract: Cognitive impairment and memory dysfunction following stroke diagnosis are common symptoms that significantly affect the survivors’ quality of life. Stroke patients have a high potential to develop dementia within the first year of stroke onset. Currently, efforts are being exerted to assess stroke effects on the brain, particularly in the early stages. Numerous neuropsychological assessments are being used to evaluate and differentiate cognitive impairment and dementia following stroke. This article focuses on the role of available neuropsychological assessments in detection of dementia and memory loss after stroke. This review starts with stroke types and risk factors associated with dementia development, followed by a brief description of stroke diagnosis criteria and the effects of stroke on the brain that lead to cognitive impairment and end with memory loss. This review aims to combine available neuropsychological assessments to develop a post-stroke memory assessment (PSMA) scheme based on the most recognized and available studies. The proposed PSMA is expected to assess different types of memory functionalities that are related to different parts of the brain according to stroke location. An optimal therapeutic program that would help stroke patients enjoy additional years with higher quality of life is presented.
Keywords: dementia, vascular dementia, memory, neuropsychological assessment
Introduction
Cognitive impairment and memory loss are common after a stroke. Approximately 30% of stroke patients develop dementia within 1 year of stroke onset.1 Stroke affects the cognitive domain, which includes attention, memory, language, and orientation. The most affected domains are attention and executive functions; at the time of stroke diagnosis, memory problems are often prominent. Post-stroke dementia (PSD), particularly vascular dementia (VaD), reflects the vascular risk factors that are mostly correlated with cerebral vascular disease (CVD). Post-stroke cognitive impairment is the evolution of CVD that predisposes individuals to the vascular cognitive impairment (VCI) spectrum. Thus, understanding the VCI spectrum stages is necessary to evaluate the mental state of post-stroke patients, particularly the cognitive dysfunction and memory decline during the period following a stroke diagnosis. Until recently, no specific neuropsychological assessment to evaluate PSD including memory loss existed. Current efforts are focused on combining more than one of the available neuropsychological assessments to obtain a significant diagnosis of cognitive decline severity following a stroke. The aim of this study was to develop a post-stroke memory assessment (PSMA) based on the most popular and available neuropsychological assessments. The proposed PSMA is expected to assess different types of memory functionalities that are related to different parts of the brain according to the affected memory. Results are then correlated and related to the stroke location and severity. PSMA may provide a promising tool for evaluating post-stroke VaD and assisting medical doctors and clinicians in the assessment as well as evaluation of post-stroke memory impairment severity.
Stroke types
Stroke is considered a major cause of long-term physical disabilities in adults; it is the second most common cause of cognitive impairment and dementia and the third leading cause of death after coronary artery diseases and cancer.2,3
A stroke is a “brain attack” that is caused either by reducing blood and oxygen flow to the brain or by bleeding. Stroke can be classified into two main types: ischemic and hemorrhagic. Transient ischemic attack (TIA) is sometimes considered as the third type of stroke and can be referred to as a “mini-stroke.”4 Stroke characteristics are listed in Table 1.
  | Table 1 Classification of stroke |
Vascular risk factors and stroke diagnosis criteria
Numerous risk factors band to cause a stroke: modifiable risk factors, including age, sex, ethnicity, genetics; and non-modifiable risk factors, including CVD, heart disease, diabetes mellitus, hyperlipidemia, cigarette smoking, and alcohol abuse, as shown in Figure 1.5,6 Stroke, which is considered a CVD, is an influential risk factor for cognitive impairment that eventually leads to the development of PSD.7 Thus, stroke survivors require immediate medical control of these risk factors, which are modifiable, to reduce stroke prevalence.
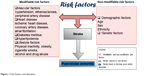  | Figure 1 Risk factors and dementia. |
Clinically, stroke can usually be diagnosed through typical symptoms and signs. Medical history is an early step of diagnosis and includes stroke onset, course, and patient information taken from patients’ careers or relatives, followed by physical and neurological examinations of the patients. The neurological examination can be performed using the formal stroke scale developed by the National Institution of Health Stroke Scale8 to classify early stroke severity. Laboratory testing is the next step; at this stage, blood tests are used to determine the blood sugar level and cholesterol level. This step is followed by an examination of the computer tomography/magnetic resonance imaging scan and electrocardiography recording to indicate stroke location and pulse irregularity, such as cardiovascular status, carotid bruits, fundus examination, peripheral vascular disease, and hypertension.9 Electroencephalography is used to help differentiate between seizure and TIA or between lacunar and cortical infarction in occasional patients, as illustrated in Figure 2.10
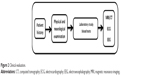  | Figure 2 Clinical evaluation. |
Stroke effects on brain cerebrovascular function
The brain requires a constant supply of blood to carry oxygen and nutrients to the cortical neurons in order for it to function in a normal manner. Numerous arteries cooperate to achieve this demand. In the case where an ischemic or hemorrhagic stroke occurs in one or more of these arteries and/or their branches, it causes damage to a specific neuroanatomic location (ie, right hemisphere cortex, left hemisphere cortex, or subcortex, which can then be localized further to the frontal lobe, temporal lobe, parietal lobe, thalamus, for example). Thus, the part of the brain that does not get the blood it needs starts to die. Brain cellular damage and death within minutes of stroke onset is called the core, whereas the zone in which the blood decreases or marginal perfusion occurs is called the ischemic penumbra, as shown in Figure 3.4,11
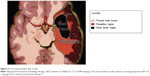  | Figure 3 Core and penumbra after stroke. |
Owing to the complexity of the neuronal networks concerned in cortical processes, the ischemic or hemorrhagic stroke that occurs in a specific vascular distribution and the damage to a neuroanatomic site typically impairs more than one cognitive function. Moreover, some stroke events may involve multiple neurologic systems that cause cognitive decline based on vascular distribution (ie, perceptual and sensory or motor and sensory), as tabularized in Table 2.12
  | Table 2 Stroke outcome due to vessel infarction |
Cognitive disorder following a stroke
Dementia is associated with neurodegenerative disorder diversity, neuronal dysfunction, and neuronal death. Dementia occurs when the brain is affected by a specific disease or condition that causes cognitive impairment.13 In the case of a stroke, one or more cognitive domains may be affected, including attention, memory, language, and orientation. The highest impact of stroke at the time of diagnosis is on the attention and executive functions rather than on memory, which may be impaired at various post-stroke intervals. Previous studies show that post-stroke memory prevalence varies from 23% to 55% 3 months after stroke, ending with a decline from 11% to 31% 1 year after stroke onset.3,14 Cognitive impairment after a stroke is common and leads to PSD. PSD includes all dementia types that occur after a stroke, including VaD; degenerative dementia, particularly Alzheimer’s disease (AD); or mixed dementia (VaD plus AD).2 VaD, the second leading cause of dementia in the world after AD, occurs as a result of stroke. Between 1% and 4% of elderly people aged 65 years and older suffer from VaD, and its prevalence will double every 5–10 years after this age.15,16 VaD is characterized by impairment in the cognitive function due to vascular lesion and infarction resulting from the stroke. The clinical manifestation of VaD varies based on the size, location, and type of cerebral damage.15 Figure 4 illustrates the cognitive impairment sequences which predispose individuals to the VCI spectrum.
The VCI spectrum can be viewed as a cognitive consequence in the cognitive domain, starting from mild cognitive impairment (MCI) and ending with severe dementia. The period beyond dementia in which the brain is at risk is called “cognitive impairment no dementia.”17
MCI causes a more considerable decline in cognitive function with respect to individual age and education level, but not notably with the activities of daily life.18,19 Clinically, MCI is the transitional stage between early normal cognition and late severe dementia, and it is considered heterogeneous because some MCI patients develop dementia while others stay and continue as MCI patients for many years. However, by default, patients diagnosed with MCI have a high potential to develop dementia within the third month from the time dementia symptoms begin to arise.2,20 The most observed symptoms of MCI are limited to memory, but the patient’s daily living activities are preserved.21 This article is focused on VaD as a common cause of cognitive impairment following a stroke and the effect of VaD on memory loss. It likewise discusses the available neuropsychological assessments that assess and predict the effect of dementia based on the dementia spectrum as well as aids in detecting signs of dementia, particularly memory disturbance. A number of diagnosis criteria and clinical neuropsychological assessments are combined. The most common diagnosis criteria are developed and characterized by the National Institute of Neurological Disorders and Stroke and Association Internationale pour la Recherché et l’Enseignement en Neurosciences for VaD22–26 and Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition criteria.27 The severity of cognitive symptoms could be assessed using the Clinical Dementia Rating Scale.28 The most usable test to evaluate the early dementia stages, even severity of dementia in clinical practice, is the Mini-Mental State Examination (MMSE).29
Brain memory and causes of memory loss
The brain memory system refers to the process of how our brain transmits and stores available information for future use, with or without conscious awareness. The human brain memory system is a complex structure, with different functionalities, as shown in Table 3. Based on stroke location and severity, memory disorder may occur for one or more memory types, eventually ending in memory decline and loss.30
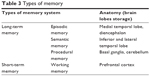  | Table 3 Types of memory |
Memory loss can be caused by several factors, such as lifestyle, brain injury, infection, thyroid dysfunction, aging, MCI, and dementia (Table 4).31
This article focuses on stroke as the major cause of cognitive impairment resulting in memory decline. The effect of stroke varies based on its type, location, and severity.2 After a stroke, the most prominent impairment can be recognized in the patient’s processing speed, attention, and executive function. Note that 20%–50% of stroke patients suffer from memory intricacy that manifests during the period following a stroke diagnosis. PSD, particularly VaD, causes slowing in cognitive flexibility, perceptual disorder, and impairment information retrieval at the time of stroke diagnosis. This period corresponds to MCI in the VCI spectrum, followed by a decline in episodic memory function in case of dementia, and ending in severe dementia and impairment of all cognitive properties.32–35
Cognitive domain and memory assessment after a stroke
Cognitive impairment, particularly memory problems following a stroke, can be evaluated and assessed through neuropsychological assessments. Clinically, different neuropsychological assessments are used to assess cognitive dysfunction in terms of cognitive domain.36 A set of standardized neuropsychological assessments have been selected due to their sensitivity for MCI and to cover different cognitive domains including memory; for example, MMSE,29 Montreal Cognitive Assessment (MoCA),37 and Addenbrooke’s Cognitive Examination Revised (ACE-R)38 are widely used to assess the cognitive dysfunction of patients. Several validated clinical neuropsychological assessments are used to assess cognitive domain, including (but not limited to) Trail Making Test (TMT)39 and Clock Drawing Test (CDT)40 for attention and executive function (both are short tests that evaluate executive function),18 Rey Osterrieth Figure Copy41 for construction praxis test, and Phonological and Semantic Fluency Token test for language test.42 Other tests (eg, Frontal Assessment Battery [FAB]43) can be used as a quick and easy battery test. The Cambridge Examination for Mental Disorders of the Elderly,44 is a standardized instrument that is used to investigate the cognitive domains required to diagnose dementia in multiple domains, including memory. The most common tests to assess memory evaluate memory in terms of retention, retrieval, and encoding (eg, the Wechsler Memory Scale (WMS)-Revised45 may be employed to distinguish amnesia from dementia in patients). For verbal memory, numerous assessments are used, including the WMS,46 Rey Auditory Verbal Learning Test,47 Rivermead Behavioral Memory Test (RBMT),48 and California Verbal Learning Test.48 Memory disorder in elderly dementia patients can be assessed using the Free and Cued Selective Reminding Test. This test aids in distinguishing dementia from normal aging with acceptable accuracy.36
Until recently, no specific assessment was developed specifically to assess short-term memory, working memory, and long-term memory impairment following stroke VaD. Thus, evaluating memory in terms of its types to predict stroke effect on memory retrieval is important.
PSMA
The decline in memory as a result of stroke VaD and the characterization of memory complaint based on VaD development can be assessed through a PSMA. This assessment is based on the most popular studies and is a combination of available neuropsychological assessment tests.49,50 Memory evaluation is proposed to be associated with memory types. Thus, short-term memory and working memory refer to the perceptual and learning areas of the cognitive domain, which are processed by the frontal lobe. Episodic and semantic long-term memory refers to memory, language, and visuospatial domains, which are processed by the parietal, medial temporal lobe, and hippocampus. Procedural memory refers to the procedural domain and is processed by the cerebellum and basil ganglia. Table 5 describes the proposed PSMA, which achieves this demand. The concept integrated the most usable neuropsychological assessments (MMSE, ACE-R, MoCA, WMS-IV, RBMT, TMT A and B, CDT, FAB, Wechsler Adult Intelligence Scale – Fourth Edition, and others) and reconstructed them to evaluate memory types.50
PSMA was designed with inspected administration time of 30 minutes, as illustrated in the Supplementary materials. The test examines the following:
- Orientation: in time and place
- Short-term memory: a seven-digit number, phone number, and postal code
- Working memory: attention and concentration, verbal working memory, and visuospatial working memory
- Explicit long-term memory: episodic memory and semantic memory
- Procedural memory.
Discussion
Neuropsychological assessments are used in evaluating and assessing cognitive impairment and dementia. Specific assessment is urgently needed to evaluate different types of memory functionalities after stroke. The present study focused on using available neuropsychological assessments to develop a PSMA scheme based on scientific knowledge, which is available through neuropsychological testing. PSMA may help provide impetus to detect the earliest stages of dementia before significant mental decline. Therefore, efforts are being exerted to use more than one assessment to evaluate cognitive impairment and memory dysfunction. For instance, the MMSE is a brief test with extensive international usage; however, several studies have mentioned that the MMSE alone can be used in a sensitive test to detect cognitive impairment, except if cutoff is increased or combined with other neuropsychological tests.51,52 Therefore, the MMSE was used with MoCA and ACE-R to detect MCI because the last two assessments are used to assess early stages of dementia and executive function, as well as identify frontal subcortical infarction.50,53,54 In addition, ACE-R has good sensitivity for dementia, whereas MoCA is specifically used in MCI screening. Moreover, TMT, Stroop, and CDT tests can be used with the MMSE to evaluate frontal lesion verbal fluency, and visuospatial skills can be evaluated through Rey Osterrieth figure recall. FAB has been reported to identify frontal temporal lobe dysfunction.55 MMSE, ACE-R, MoCA, and FAB characteristics are shown in Table 6. It can be noticed from the table that the administration time ranged from 35–45 minutes for four assessments. The PSMA administration time was reduced approximately to 30 minutes. PSMA has been designed to incorporate more than one neuropsychological assessment to evaluate short-term, working, and long-term memory with less time consumed compared with multiple test usage. Using more than one assessment to evaluate patient mentality takes a longer time, resulting in patient difficulty in concentrating on the assessment items. PSMA evaluates the cognitive domain and focuses on memory types that are affected by VaD.
Conclusion
Currently, no specific neuropsychological assessment to assess memory in terms of its types exists. This article provides an overview of the effects of stroke on the brain and on cognitive impairment, including memory evaluation with the most commonly used neuropsychological tests. The article proposes a PSMA to assess different types of memory based on the available assessments. It likewise uses the widely available neuropsychological assessments to study the association between memory as a part of cognitive domain and cognitive impairment, which lead to memory decline in the period following stroke onset.
Disclosure
The authors declare that there are no conflicts of interest in this work.
References
Cullen B, O’Neill B, Evans JJ, Coen RF, Lawlor BA. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry. 2007;78(8):790–799. | ||
Leys D, Hénon H, Mackowiak-Cordoliani M-A, Pasquier F. Poststroke dementia. Lancet Neurol. 2005;4(11):752–759. | ||
Cumming TB, Marshall RS, Lazar RM. Stroke, cognitive deficits, and rehabilitation: still an incomplete picture. Int J Stroke. 2013;8(1):38–45. | ||
Mohr JP. Stroke: Pathophysiology, Diagnosis, and Management. Elsevier Health Sciences; 2004. | ||
Sahathevan R, Brodtmann A, Donnan GA. Dementia, stroke, and vascular risk factors: a review. Int J Stroke. 2012;7(1):61–73. | ||
Iemolo F, Duro G, Rizzo C, Castiglia L, Hachinski V, Caruso C. Pathophysiology of vascular dementia. Immun Ageing. 2009;6(1):13. | ||
Sibolt G, Curtze S, Melkas S, et al. Poststroke dementia is associated with recurrent ischaemic stroke. J Neurol Neurosurg Psychiatry. 2013; 84(7):722–726. | ||
Brott T, Adams H, Olinger CP, et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke. 1989;20(7):864–870. | ||
Demarin V, Zavoreo I, Kes VB. Carotid artery disease and cognitive impairment. J Neurol Sci. 2012;322(1–2):107–111. | ||
Sacco RL, Adams R, Albers G, et al; American Heart Association/American Stroke Association Council on Stroke; Council on Cardiovascular Radiology and Intervention; American Academy of Neurology. Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co-sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline. Circulation. 2006;113(10):e409–e449. | ||
Foreman B, Claassen J. Quantitative EEG for the detection of brain ischemia. Crit Care. 2012;16(2):216. | ||
Donovan NJ, Kendall DL, Heaton SC, Kwon S, Velozo CA, Duncan PW. Conceptualizing functional cognition in stroke. Neurorehabil Neural Repair. 2008;22(2):122–135. | ||
Borson S, Frank L, Bayley PJ, et al. Improving dementia care: the role of screening and detection of cognitive impairment. Alzheimers Dement. 2013;9(2):151–159. | ||
Snaphaan L, de Leeuw F-E. Poststroke memory function in nondemented patients: a systematic review on frequency and neuroimaging correlates. Stroke. 2007;38(1):198–203. | ||
McVeigh C, Passmore P. Vascular dementia: prevention and treatment. Clin Interv Aging. 2006;1(3):229. | ||
Ruitenberg A, Ott A, van Swieten JC, Hofman A, Breteler MM. Incidence of dementia: does gender make a difference? Neurobiol Aging. 2001;22:575–580. | ||
Jacova C, Kertesz A, Blair M, Fisk JD, Feldman HH. Neuropsychological testing and assessment for dementia. Alzheimer Dement. 2007;3(4): 299–317. | ||
Korczyn AD, Vakhapova V, Grinberg LT. Vascular dementia. J Neurol Sci. 2012;322(1–2):2–10. | ||
Ankolekar S, Geeganage C, Anderton P, Hogg C, Bath PM. Clinical trials for preventing post stroke cognitive impairment. J Neurol Sci. 2010;299(1–2):168–174. | ||
Winblad B, Palmer K, Kivipelto M, et al. Mild cognitive impairment – beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med. 2004; 256(3):240–246. | ||
Andrade C, Radhakrishnan R. The prevention and treatment of cognitive decline and dementia: an overview of recent research on experimental treatments. Indian J Psychiatry. 2009;51(1):12–25. | ||
Sheng B, Cheng LF, Law CB, Li HL, Yeung KM, Lau KK. Coexisting cerebral infarction in Alzheimer’s disease is associated with fast dementia progression: applying the National Institute for Neurological Disorders and Stroke/Association Internationale pour la Recherche et l’Enseignement en Neurosciences Neuroimaging Criteria in Alzheimer’s Disease with Concomitant Cerebral Infarction. J Am Geriatr Soc. 2007;55(6):918–922. | ||
Jack CR Jr, Albert M, Knopman DS, et al. Introduction to revised criteria for the diagnosis of Alzheimer’s disease: National Institute on Aging and the Alzheimer Association workgroups. Alzheimers Dement. 2011; 7(3):257–262. | ||
Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):270–279. | ||
McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263–269. | ||
Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):280–292. | ||
Association AP. Diagnostic and statistical manual of mental disorders fourth edition. Washington, DC: American Psychiatric Association; 1994. | ||
Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL. A new clinical scale for the staging of dementia. Br J Psychiatry. 1982;140(6):566–572. | ||
Folstein MF, Folstein SE, McHugh PR. Mini-mental state. A prac-32. 1998. | ||
D’Esposito M. Chapter 11. Working memory. In: Goldenberg G, Miller BL, editors. Handbook of Clinical Neurology. Volume 88. Elsevier; 2008:237–247. | ||
Buckner RL. Memory and executive function in aging and AD: multiple factors that cause decline and reserve factors that compensate. Neuron. 2004;44(1):195–208. | ||
Lim C, Alexander MP. Stroke and episodic memory disorders. Neuropsychologia. 2009;47(14):3045–3058. | ||
Snaphaan L, Rijpkema M, van Uden I, Fernandez G, de Leeuw FE. Reduced medial temporal lobe functionality in stroke patients: a functional magnetic resonance imaging study. Brain. 2009;132(Pt 7):1882–1888. | ||
Planton M, Peiffer S, Albucher JF, et al. Neuropsychological outcome after a first symptomatic ischaemic stroke with “good recovery”. Eur J Neurol. 2012;19(2):212–219. | ||
Cooper S, Greene JD. The clinical assessment of the patient with early dementia. J Neurol Neurosurg Psychiatry. 2005;76(Suppl 5):v15–v24. | ||
Pasquier F. Early diagnosis of dementia: neuropsychology. J Neurol. 1999;246(1):6–15. | ||
Smith T, Gildeh N, Holmes C. The Montreal Cognitive Assessment: validity and utility in a memory clinic setting. Can J Psychiatry. 2007; 52(5):329–332. | ||
Mathuranath P, Nestor P, Berrios G, Rakowicz W, Hodges J. A brief cognitive test battery to differentiate Alzheimer’s disease and frontotemporal dementia. Neurology. 2000;55(11):1613–1620. | ||
Amodio P, Wenin H, Del Piccolo F, et al. Variability of trail making test, symbol digit test and line trait test in normal people. A normative study taking into account age-dependent decline and sociobiological variables. Aging Clin Exp Res. 2002;14(2):117–131. | ||
Shulman KI. Clock-drawing: is it the ideal cognitive screening test? Int J Geriatr Psychiatry. 2000;15(6):548–561. | ||
Caffarra P, Vezzadini G, Dieci F, Zonato F, Venneri A. Rey-Osterrieth complex figure: normative values in an Italian population sample. Neurol Sci. 2002;22(6):443–447. | ||
Carlesimo G, Caltagirone C, Gainotti G, et al. The Mental Deterioration Battery: normative data, diagnostic reliability and qualitative analyses of cognitive impairment. Eur Neurol. 1996;36(6):378–384. | ||
Dubois B, Slachevsky A, Litvan I, Pillon B. The FAB: a frontal assessment battery at bedside. Neurology. 2000;55(11):1621–1626. | ||
Roth M. CAMDEX-R: the Cambridge examination for mental disorders of the elderly. Cambridge University Press; 1998. | ||
Wechsler D. Wechsler Memory Scale – Revised. San Antonio, TX: Psychological Corporation; 1987. | ||
Wechsler D. Wechsler memory scale. New York: Psychological Corporation; 1945. | ||
Osterrieth PA. Le test de copie d’une figure complexe. Arch Psychol. 1944;30:206–356. | ||
Wilson BA, Cockburn J, Baddeley AD. The Rivermead Behavioural Memory Test. Suffolk, UK: Thames Valley Test Company; 1991. | ||
Pendlebury ST, Mariz J, Bull L, Mehta Z, Rothwell PM. MoCA, ACE-R, and MMSE Versus the National Institute of Neurological Disorders and Stroke – Canadian Stroke Network Vascular Cognitive Impairment Harmonization Standards Neuropsychological Battery After TIA and Stroke. Stroke. 2012;43(2):464–469. | ||
Bagnoli S, Failli Y, Piaceri I, et al. Suitability of neuropsychological tests in patients with vascular dementia (VaD). J Neurol Sci. 2012; 322(1–2):41–45. | ||
Moulaert VR, Verbunt JA, van Heugten CM, Wade DT. Cognitive impairments in survivors of out-of-hospital cardiac arrest: a systematic review. Resuscitation. 2009;80(3):297–305. | ||
Cao M, Ferrari M, Patella R, Marra C, Rasura M. Neuropsychological findings in young-adult stroke patients. Arch Clin Neuropsychol. 2007; 22(2):133–142. | ||
Sikaroodi H, Yadegari S, Miri SR. Cognitive impairments in patients with cerebrovascular risk factors: a comparison of Mini Mental Status Exam and Montreal Cognitive Assessment. Clin Neurol Neurosurg. 2013;115(8):1276–1280. | ||
Kandiah N, Wiryasaputra L, Narasimhalu K, et al. Frontal subcortical ischemia is crucial for post stroke cognitive impairment. J Neurol Sci. 2011;309(1–2):92–95. | ||
Bagnoli S, Failli Y, Piaceri I, et al. Suitability of neuropsychological tests in patients with vascular dementia (VaD). J Neurol Sci. 2012; 322(1):41–45. | ||
Summers D, Malloy R. CT and MR imaging in the acute ischemic stroke patient: a nursing perspective. J Radiol Nurs. 2011;30(3):104–115. |
Supplementary materials
    |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.