Back to Journals » Clinical Interventions in Aging » Volume 17
Clinical Value of Inflammatory Cytokines in Patients with Aneurysmal Subarachnoid Hemorrhage
Authors Luo C , Yao J , Bi H, Li Z, Li J, Xue G, Li K, Zhang S, Zan K, Meng W, Zhang Z, Chen H
Received 17 February 2022
Accepted for publication 20 April 2022
Published 26 April 2022 Volume 2022:17 Pages 615—626
DOI https://doi.org/10.2147/CIA.S362854
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nandu Goswami
Cong Luo,1 Jiaxin Yao,1 Haoran Bi,2 Zhen Li,1 Ju Li,3 Guosong Xue,3 Ke Li,3 Shenyang Zhang,3 Kun Zan,3 Wenqing Meng,3 Zunsheng Zhang,3 Hao Chen3
1Graduate School, Xuzhou Medical University, Xuzhou, Jiangsu, People’s Republic of China; 2Department of Biostatistics, Xuzhou Medical University, Xuzhou, Jiangsu, People’s Republic of China; 3Department of Neurology, The Affiliated Hospital of Xuzhou Medical University, Xuzhou, Jiangsu, People’s Republic of China
Correspondence: Zunsheng Zhang; Hao Chen, Department of Neurology, The Affiliated Hospital of Xuzhou Medical University, 99 Huaihai West Road, Quanshan District, Xuzhou, Jiangsu, People’s Republic of China, Tel +86-13913473179 ; +86-15252006510, Email [email protected]; [email protected]
Background: Inflammation is closely associated with prognosis in patients with aneurysmal subarachnoid hemorrhage (aSAH), which is orchestrated by inflammatory cytokines. Therefore, this study aimed to investigate the levels of inflammatory cytokines in the early stage of aSAH and their predictive value for prognosis.
Methods: In this retrospective study, 206 patients with aSAH were recruited and assigned to a severe group (WFNS grade ≥ 4) and a mild group (WFNS grade < 4) according to the severity of patients on admission. Flow cytometry was performed to detect the levels of 12 inflammatory cytokines in the serum of patients. Then, patients were grouped into a poor prognosis group (mRS score ≥ 4) and a good prognosis group (mRS score < 4) based on their prognosis after 3 months of discharge to compare the relationship between cytokines and prognosis. Propensity score matching (PSM) was utilized to control confounding factors. The correlation between inflammatory factors and prognosis was determined using Spearman correlation, and the predictive efficacy of inflammatory factors was tested by a receiver operating characteristic curve.
Results: Serum IL-1β, IL-5, IL-6, IL-8, IL-10, IFN-γ, and TNF-α levels were significantly higher in the mild group than in the severe group and in the poor prognosis group than in the good prognosis group. After PSM, the differences in IL-1β, IL-5, IFN-α, and IFN-γ levels disappeared between the two groups, whereas IL-2, IL-6, IL-8, IL-10, and TNF-α levels remained higher in the poor prognosis group than in the good prognosis group. Additionally, IL-2, IL-6, IL-8, and IL-10 levels were positively correlated with mRS scores. Moreover, the predictive value was found to be the highest for IL-6 and the lowest for TNF-α.
Conclusion: Inflammation degree was related to the severity of aSAH. Inflammatory markers, including IL-6, IL-10, IL-8, IL-2, and TNF-α, might predict the poor prognosis of aSAH.
Keywords: aneurysmal subarachnoid hemorrhage, inflammation, cytokine, prognosis, propensity score matching
Introduction
As a typical clinical emergency, subarachnoid hemorrhage (SAH) accounts for approximately 5% of all strokes with high mortality and disability, among which about 80% is aneurysmal SAH (aSAH).1 It is primarily characterized by the rupture of the aneurysm, resulting in a sudden flow of blood into the subarachnoid space filled with cerebrospinal fluid (CSF). Despite tremendous advances in the early treatment of aSAH with the help of aneurysm clipping and coil embolization,2 the overall mortality rate remains high at 32–67%.3 Furthermore, the majority of survivors suffer from varying degrees of disability.4 Therefore, it is critical to early identify biomarkers associated with poor prognosis and intervention. In addition, brain injury following aSAH leads to systemic inflammation, such as altered inflammatory cytokines, which is correlated with poor prognosis.5 It has been reported that elevated serum concentrations of inflammatory markers, like tumor necrosis factor (TNF)-α, interleukin (IL)-1, IL-6, and other cytokines are associated with the poor prognosis of SAH patients.6–8 However, the aforementioned cytokines are inconclusive for the outcome of aSAH, and sometimes the conclusions are even contradictory. Moreover, the relationship between the levels of some inflammatory cytokines (such as IL-2 and IL-5) and the prognosis of aSAH remains poorly identified. Thus, this study set out to determine whether the expression of 12 serum cytokines [IL-1, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, IL-17, interferon (IFN)-α, interferon (IFN)-γ, and TNF-α] was differential in patients with different severity of aSAH and healthy populations and to ascertain whether the concentrations of the aforesaid cytokine differed in aSAH patients with different outcomes and whether they can be adopted as early predictors to better guide clinical decisions.
Materials and Methods
Research Subjects
A total of 206 patients with aSAH admitted to the Affiliated Hospital of Xuzhou Medical University from January 15, 2020, to August 1, 2021, were enrolled in this study as study subjects. The inclusion criteria were as follows: (1) SAH confirmed by cranial computed tomography (CT) or lumbar puncture; (2) the presence of aneurysm confirmed by CT angiography or digital subtraction angiography after admission to the hospital; and (3) access to the hospital within 24 hours.
Exclusion criteria included: (1) the history of long-term immunomodulatory therapy, including cortisol and other drugs; (2) severe cardiovascular or pulmonary disease, AIDS, autoimmune system disease, or malignancy; (3) co-infection; (4) insufficient clinical data; (5) death while in the hospital; (6) SAH caused by trauma, arteriovenous malformation, or smog or SAH in which the source of the hemorrhage could not be determined by cerebral angiography; and (7) re-rupture hemorrhage after aneurysm surgery.
Additionally, 86 healthy people examined in our hospital during the same period were collected as controls. Our research was ratified by the Ethics Committee of Xuzhou Medical University (Ethical Batch Number: XYFY2021-KL232-01). Patients were not required to provide informed consent, as the study was retrospective and the data was anonymous.
Clinical Data Collection
The clinical information of the patients, including gender, age, mean arterial pressure on admission [(2 × diastolic + systolic)/3], surgical procedure, time from onset to admission, previous history of hypertension, diabetes, smoking, and alcohol consumption, was obtained through the electronic medical record system of the hospital. The location (anterior or posterior circulation), diameter, and the number of the aneurysm were clarified by the imaging examination results of patients and course records during hospitalization, followed by the recording of the occurrence of aSAH-related complications, such as acute hydrocephalus, delayed cerebral ischemia (DCI, which was evaluated as per previously published guidelines9) and pneumonia. The World Federation of Neurosurgery Surgeons (WFNS) grade10 was employed to assess the severity of the disease (WFNS grade ≥ 4 as the severe group, and WFNS grade < 4 as the mild group). The CT-based modified Fisher Scale (MFS)11 was utilized to assess the degree of hemorrhage. All of these were assessed by two attending neurologists. When the conclusions of the two assessors were inconsistent, the assessment was performed by a third associate neurologist.
The laboratory findings on the admission of patients were harvested by a neurology resident. The laboratory indices of patients were collected, which consisted of white blood cells, neutrophils, lymphocytes, platelets, red blood cell count, blood glucose, platelet count, and serum cytokines (IL-1, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12p70, IL-17, IFN-α, IFN-γ, and TNF-α).
In addition, the data of serum cytokines were attained from 165 patients who were examined at our physical examination center during the same period to facilitate the comparison with the healthy population. A total of 86 healthy individuals were matched for age and gender to the case group and enrolled as the control group of this study.
The mRS score12 was selected as a scale to reflect the prognosis of patients, reflecting the degree of disability, which was more straightforward and easier to use. This score was acquired through the telephone follow-up of patients by one resident 90 days after discharge: the mRS score of 0–3 was defined as a good prognosis and the score of 4–6 as a poor prognosis.
Statistical Analysis
SPSS 25.0 was utilized to analyze the comparative trial data. Continuous variables conforming to normal distribution were expressed as means ± standard deviation, whilst those with abnormal distribution were summarized as median and quartiles [median (P25, P75)]. Group comparisons conforming to normal distribution and variance were analyzed using two independent samples t-tests, and the corrected t-test was used when the used variance was not equal. For data with abnormal distribution and rank data, comparisons among three groups were analyzed using Kruskal–Wallis test with p value corrected by Bonferroni, and comparisons between two groups were analyzed using Mann–Whitney U-test. Besides, categorical variables were shown as rates (%). Data between groups were compared using Pearson χ2 test, Fisher exact test, and continuity correction χ2 test. Propensity score (PS) was conducted using the PS matching (PSM) 3.04 program package of R software 3.3.3, and PSM was calculated using logistic regression. The covariates entered into the PSM model, which comprised MFS, WFNS, age, sex, history of smoking, history of alcohol consumption, history of diabetes mellitus, history of hypertension, admission time, aneurysm location, aneurysm size, surgical procedure, white blood cell count, neutrophil count, blood glucose, lymphocyte count, and PSM using a 1:2 matching method with a caliper value of 0.4. Spearman correlation was utilized to analyze the association between grade information and information with skewed distribution. After PSM, the best cut-off values of statistically significant variables were analyzed using the receiver operating characteristic (ROC) curve. The area under the curve (AUC) was calculated to determine the predictive ability.
Results
The Baseline Data of aSAH Patients
A total of 206 patients (97 males and 109 females) with SAH were included in this study with a mean age of 59.69 ± 12.93 years. Among these patients, 18 patients (8.74%) smoked, 12 patients (5.83%) drank, 28 patients (13.59%) developed diabetes mellitus, and 121 patients (58.74%) suffered from hypertension. The WFNS grade was generally considered to accurately reflect the severity of SAH. Therefore, patients were classified in the light of the WFNS grade, with patients with WFNS ≥ 4 as the severe group and those with WFNS < 4 as the mild group. The mild group consisted of 117 patients, while the severe group consisted of 89 patients. The baseline characteristics of patients classified as per the disease severity are depicted in Table 1. The severe group exhibited higher MFS, white blood cell count, and neutrophil count, and a lower blood glucose level than the mild group (P < 0.05). Additionally, it was discovered that the probability of developing aSAH-related complications, such as acute hydrocephalus, delayed cerebral ischemia, and pulmonary infection was significantly higher in the severe group than in the mild group (P < 0.05). There was no statistically significant difference between these two groups regarding other indicators.
 |
Table 1 Baseline Characteristics and Clinical Data on the Admission in the Mild and Severe Groups |
Comparison of the Cytokines of the Mild, Severe, and Control Groups
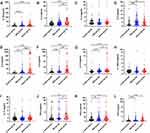
Serum levels of IL-1, IL-5, IL-6, IL-8, IL-10, IFN-γ, and TNF-α were obviously higher in patients with mild aSAH than in healthy controls (P < 0.05). The above cytokines were also elevated in the severe group compared to the control group (P < 0.05). Furthermore, in contrast to the mild group, the severe group had substantially higher serum IFN-α and IL-2 concentrations than the control group (P < 0.05). However, serum IFN-α and IL-2 concentrations did not statistically differ between the mild and control groups (P > 0.05). The severe group was also compared with the mild group in this study. It was found that the concentrations of serum IL-2, IL-6, IL-8, IL-10, and IFN-α were higher in the severe group than in the mild group (P < 0.05). However, IL-1β, IL-5, IFN-γ, and TNF-α levels were not conspicuously different between patients with mild and severe aSAH (P > 0.05). In addition, no statistically significant differences were found in serum IL-4, IL-12p70, and IL-17 concentrations in aSAH patients compared to healthy controls (P > 0.05; Table 2, Figure 1).
 |
Table 2 Comparison of Inflammatory Cytokines Between Groups |
The Effects of Cytokines on the Prognosis of aSAH
Furthermore, WFNS, MFS, aneurysm diameter, neutrophils, white blood cell count, and blood glucose level were higher in patients with poor prognosis than in patients with good prognosis. In contrast, lymphocyte count was lower in the poor prognosis group than in the good prognosis group (P < 0.05). DCI, acute hydrocephalus, and SAH-associated pneumonia in the poor prognosis group were evidently enhanced in the poor prognosis group versus the good prognosis group (P < 0.05). More importantly, it was noted higher IL-1β, IL-5, IL-2, IL-6, IL-8, IL-10, IFN-α, IFN-γ, and TNF-α levels in the poor prognosis group than in the good prognosis group (P < 0.05).
Meanwhile, the prognosis of aSAH patients is afflicted by several factors, especially WFNS, MFS, and DCI, which were considered factors related to poor prognosis in previous studies. Therefore, to control the confounding factors in the baseline that interfered with the prognosis, PSM was employed to correct the baseline. There was no significant difference between groups after matching (P > 0.05). Even after matching, serum IL-2, IL-6, IL-8, IL-10, and TNF-α levels were considerably higher in patients with poor prognosis than in those with good prognosis (P < 0.05). However, it was also observed that the variability of IL-1β, IFN-α, and IFN-γ levels disappeared between groups. Imbalance in baseline characteristics most likely to cause variability (P > 0.05) (Tables 3 and 4).
 |
Table 3 Comparison of Baseline Characteristics Before and After PSM in the Good and Poor Prognosis Groups |
 |
Table 4 Comparison of Inflammatory Cytokine Profiles Before and After PSM in the Good and Poor Prognosis Groups |
The Correlation Between Cytokines and Prognosis and Their Diagnostic Value
This research discovered that the serum concentrations of IL-2, IL-6, IL-8, IL-10, and TNF-α were augmented in patients with poor prognosis in comparison to patients with good prognosis after matching by propensity score. First, the correlation between inflammatory factors and mRS, a scale reflecting the prognosis of patients, was investigated. There exhibited the positive correlations of IL-2, IL-6, IL-8, and IL-10 levels and mRS (r = 0.296, P = 0.007; r = 0.513, P < 0.001; r = 0.345, P = 0.002; r = 0.432, P < 0.001). TNF-α shared a positive correlation with mRS, but it was not statistically significant (r = 0.165, P = 0.142). Similarly, the ROC analysis revealed that the above cytokines could be utilized as predictors for the favorable or poor prognosis of aSAH, with AUCs of 0.676, 0.794, 0.708, 0.746, and 0.643 for IL-2, IL-6, IL-8, IL-10, and TNF-α, respectively. Their best cut-off values for the diagnosis were 2.16 pg/mL, 34.32 pg/mL, 13.78 pg/mL, 2.16 pg/mL, and 5.91 pg/mL, respectively (Figure 2, Table 5).
 |
Table 5 The Diagnostic Efficacy of IL-2, IL-6, IL-8, IL-10, and TNF-α in Patients with aSAH |
Discussion
The association between serum levels of 12 cytokines and the severity and prognosis of patients with aSAH was assessed. We revealed that the cytokines IL-1β, IL-2, IL-5, IL-6, IL-8, IL-10, IFN-α, IFN-γ, and TNF-α were elevated following aSAH, among which IL-2, IL-6, IL-8, IL-10, and IFN-α were associated with the severity of aSAH. We further evaluated the influences of the aforementioned cytokines on prognosis and found that the levels IL-1β, IL-5, IL-2, IL-6, IL-8, IL-10, IFN-α, IFN-γ, and TNF-α were enhanced in patients with poor prognosis versus patients with good prognosis. Because the prognosis of aSAH was impacted by numerous factors, the baseline was corrected by PSM. Following correction, the results manifested that only five cytokines, IL-2, IL-6, IL-8, IL-10, and TNF-α, were still remarkably elevated in the poor prognosis group compared to the good prognosis group. Our research further evaluated the predictive value of these five cytokines for the prognosis of aSAH patients, which confirmed that IL-6 had the optimal predictive value. A positive correlation was noted between IL-6 level and the increased mRS score that reflected the prognosis of patients. Meanwhile, TNF-α had a relatively poor predictive value and did not correlate with the mRS score.
Upregulated inflammatory cytokines in the serum of aSAH patients reflect the significant inflammatory load of the organism during the early stages of diseases. When the aneurysm ruptures, a large amount of blood rush into the subarachnoid space, thus triggering a cascade of inflammatory responses. At this time, the immune cells (especially oligodendrocytes and astrocytes) inherent in the central nervous system are activated and release chemokines,13 which results in the recruitment of a large number of immune cells, especially neutrophils, in the peripheral blood into the subarachnoid,14 the secretion of pro-inflammatory cytokines, and the further exacerbation of inflammation. Although a mild inflammation is thought to aid in injury repair, excessive inflammation can aggravate brain edema, mitochondrial dysfunction, disruption of the blood-brain barrier (BBB), and neuronal apoptosis, all of which cause the further impairment of consciousness and are believed to contribute to the poor prognosis of patients with aSAH.15,16 In addition, inflammation activation also occurs in the peripheral immune system.17 Immune cell activation in the peripheral blood after aSAH is influenced by the sympathetic nervous system and the hypothalamic-pituitary-adrenal regulation.18 Changes in the peripheral inflammatory state and associated symptoms can manifest as systemic inflammatory response syndrome (SIRS), which triggers leukocytosis, lymphocytopenia, and cytokine release.19
Due to the disruption of BBB caused by local inflammation within the brain, plenty of inflammatory factors and neutrophils from peripheral blood enter the brain,20 further amplifying the intracranial inflammation. This vicious cycle may exacerbate the complications of brain injury. Among prior studies, Beseoglu et al21 found that the blood glucose level on admission was correlated with poor neurological status, which might be associated with the SIRS induced by aSAH and the release of inflammatory cytokines that aggravate stress response. Zeng et al22 discovered a positive correlation between neutrophil count and the severity of the condition. Also, Al-Mufti et al23 observed a correlation between the neutrophil to lymphocyte ratio at the time of admission and the condition of patients. Existing studies have validated SIRS as an independent risk factor for DCI, HCP, and other related complications.24 These findings are concordant with our results observed at baseline, indicating that the more intense the inflammation, the more severe the condition of aSAH, which is most prominently characterized by a persistent increase in the degree of impaired consciousness in patients. The early onset of impaired consciousness in aSAH (generally no longer than 72 h; commonly the patients with WFNS classification ≥ 4) can also be regarded as a result of early brain injury (EBI). EBI has been a hot topic of research in recent years, with complex pathophysiological mechanisms, among which inflammation is a vital one.25 Inflammation also was observed in the severe group of our research, where the aforementioned inflammatory indexes were appreciably higher in patients with severe aSAH than in patients with mild aSAH, corroborating the correlation of EBI with inflammation.
In addition to leukocytes, cytokines, as inflammatory markers, were previously reported to be augmented following aSAH.26 In the present study, nine cytokines were upregulated, while IL-4, IL-12p70, and IL-17 levels were not statistically different in patients with aSAH compared with controls. IL-4 is considered an anti-inflammatory cytokine with neuroprotective and cognitive improvement impacts in neurodegenerative diseases,27 which was not found to be increased in aSAH in our study. The cytokines, IL-5 and IL-10, are also documented to assume a pivotal anti-inflammatory role. Although IL-5 has been elaborated as a cerebroprotective agent that dampens the inflammation in ischemic stroke,28 its role in hemorrhagic stroke, particularly aSAH, remains enigmatic. There has hitherto been little research about the precise role of elevated IL-5 in aSAH though no difference in IL-5 expression was observed between mild and severe patients in our study and IL-5 was not utilized as a predictor of disease outcome. Future research about IL-5 should focus on the elucidation of the molecular mechanism underlying IL-5 induction in aSAH. Also, researches with larger sample sizes and a higher level of evidence in clinical studies are warranted.
Although IL-10 is regarded as an anti-inflammatory cytokine, its elevation in both hemorrhagic and ischemic stroke correlates to the severity of disease and poorer prognosis in patients,29,30 which is concurrent with the observation of this study. At present, it is believed that the increased levels of IL-10 reflect the balance between pro-inflammatory cytokines and anti-inflammatory cytokines. High levels of IL-10 are thought to be caused by an overreaction to an inflammatory stimulus. Studies have confirmed the significant correlation between IL-10 and the pro-inflammatory cytokine IL-6,31 and IL-10 plays an immunosuppressive role in balancing the inflammatory response and increases the risk of infection.29 In short, IL-10 can inhibit inflammation, however, its anti-inflammatory effects are masked by a large number of pro-inflammatory cytokines released. IL-17 is a pro-inflammatory cytokine that plays a part in the disruption of BBB and the progression of brain injury in ischemic stroke.32 However, its influence remains under-studied in hemorrhagic stroke. Chaudhry et al33 found the elevation of IL-17 level after aSAH, whereas it was not associated with clinical outcomes. Our research unveiled no marked enhancement of IL-17 level. The different conclusions may be related to the sample size, and the role IL-17 assumes in aSAH still needs to be studied prospectively in studies with a larger sample. IL-12p70 plays an essential role as the principal component of IL-12 that exerts biological effects on numerous autoimmune diseases or chronic diseases, like multiple sclerosis, diabetes, and psoriasis.34 In the present research, no noticeable augmentation of IL-12p70 was found in aSAH, which might be explained by the fact that IL-12 does not play an important role in diseases with acute onset and rapid progression. In addition to IL-10, the cytokines associated with disease severity were IL-2, IL-6, IL-8, and IFN-α. Previously, upregulated IL-6 and IL-8 were associated with disease severity,8,35 whereas there are no clinical studies on the relationship between IL-2 and IFN-α levels with aSAH. Our findings demonstrated no difference in IL-2 and IFN-α levels between patients with mild aSAH and healthy populations. Interestingly, IL-2 and IFN-α levels were observably higher in severe patients, and IL-2 was associated with the poor prognosis of aSAH in a follow-up study. Small doses of IL-2 may exhibit a therapeutic effect on rats with SAH and improve the prognosis of rats.36 In this regard, we speculate that low serum IL-2 levels may act as a protective factor against aSAH. Nonetheless, the toxic concentration of IL-2 may be more significant than a specific threshold. Some studies have unraveled that the in vivo injection of IL-2 at a high concentration can induce lymphocyte apoptosis37 and exert multi-organ toxic effects on the nervous system, heart, and kidneys,38 which may explain why high IL-2 level is associated with the poor prognosis of aSAH. IFN-α expression increases early in inflammation and further amplifies the inflammation, thus having a detrimental effect on brain injury.39 For the first time, our research presented a substantial increase in IFN-α expression in patients with severe aSAH, suggesting that IFN-α might be a helpful reference marker for the severity of aSAH. In conclusion, the comparative analysis of cytokines in our research revealed that during the early stages of aSAH, the body exhibited systemic inflammation due to stress and bleeding. Among the elevated cytokines, pro-inflammatory cytokines were predominant, whereas anti-inflammatory cytokines were not dramatically elevated. The sharp increase of these pro-inflammatory cytokines was particularly obvious in patients with severe aSAH. Inflammation tended to be amplified in individuals with severe aSAH.
We continued to dissect the relationship between the prognosis of patients and the levels of inflammatory cytokines. The data uncovered that several cytokines showed signally higher serum levels in patients with poor prognosis than in those with good prognosis. Intriguingly, these cytokines related to prognosis were consistent with the elevated inflammatory cytokines following aSAH, revealing that the prognosis of aSAH was relatively poor when patients developed robust inflammation. However, when baseline data were compared between patients with good and poor prognoses, it was discovered that patients with poor prognoses had higher WFNS grade and MFS, more extensive aneurysms, and the same variability in leukocytes, neutrophils, lymphocytes, and glucose. Corroborating findings were reported in a previous study,40 where factors, such as the severity of disease and bleeding volume could affect the prognosis of aSAH patients. Additionally, this previous study indicated the same differences in the levels of inflammatory cytokines between patients with severe and mild aSAH. Several confounding factors may afflict the elevated cytokines in the serum of patients with poor prognosis. Hence, we further assessed the level of which serum cytokine differed between patients with good and poor prognoses given the same baseline information. Two cohorts were screened using PSM, and the baseline information was deemed statistically indistinguishable. It was manifested in our research that in the matched cohort, serum IL-2, IL-6, IL-8, IL-10, and TNF-α levels remained higher in the poor prognosis group than in the good prognosis group. Simultaneously, the variability of IL-1β, IL-5, IFN-α, and IFN-γ disappeared between the two groups. These results illustrated that the upregulation of five cytokines, IL-2, IL-6, IL-8, IL-10, and TNF-α, in the early stage of the disease could predict the poor prognosis of patients under the exact condition of disease severity and bleeding. In this regard, the ROC curve was applied to assess the predictive value of these five cytokines for poor prognosis, and the AUC value was utilized to identify their predictive ability. The predictive values were IL-6, IL-10, IL-8, IL-2, and TNF-α in descending order. The same conclusion was obtained in the correlation analysis of these five cytokines with mRS. Specifically, IL-6 level was positively correlated with mRS score with the strongest correlation, while TNF-α level was statistically insignificant although it shared a positive correlation with mRS scores.
Controlling as many confounding factors as possible, these five cytokines were associated with a poor prognosis. They could serve as biomarkers to predict prognosis, thus providing a potential target for the future treatment of aSAH. Admittedly, inflammation is currently thought to play a key role in the pathophysiological mechanisms of aSAH. Still, the role of inflammation is complicated, and its impacts on the organism cannot be solely seen as causing damage. Therefore, it seems undesirable to completely repress inflammation.41 Instead, the treatment of diseases by immunomodulation should consider the more precise and selective blocking of the harmful effects of inflammation. Currently, it has been documented that the peripheral inflammation in aSAH can be diminished using the IL-1 receptor antagonist, IL-1Ra, but further researches are warranted to validate whether it can improve the prognosis.42 In the future, more in-depth studies on the pathways and interventions of other cytokines, such as IL-2, IL-6, and IL-10, may indicate new avenues for the treatment of aSAH.
Despite these encouraging observations, the current study has to be interpreted as hypothesis-generating due to several limitations. First, although PSM was utilized in this study to control confounding factors, we could not control for many potential confounding variables that could interfere with the results. Second, the dynamic changes in cytokines over time following aSAH are not clearly defined. The prognostic value of cytokines for aSAH at different time points needs further investigation. Moreover, due to related data insufficiency, the correlation between cytokine levels and other markers of acute inflammation has not been fully evaluated. Lastly, the sample size of this single-center study was small. The conclusions obtained are warranted to be further verified by multi-center and extensive sample clinical studies.
Conclusion
In summary, this study confirmed that systemic inflammation occurred early after aSAH and that the expression of inflammatory factors was related to the severity of the disease. The inflammatory markers, IL-6, IL-10, IL-8, IL-2, and TNF-α, could be adopted to predict the poor prognosis of aSAH, which may become potential therapeutic targets for clinical use in the future.
Abbreviations
aSAH, aneurysmal subarachnoid hemorrhage; SAH, subarachnoid hemorrhage; IFN, interferon; WFNS, World Federation of Neurosurgery Surgeons; MFS, modified Fisher Scale; PS, propensity score; PSM, PS matching; ROC, receiver operating characteristic; AUC, area under the curve; SIRS, systemic inflammatory response syndrome; EBI, early brain injury; CSF, cerebrospinal fluid.
Ethics Approval and Consent to Participate
This study was designed in accordance with the Declaration of Helsinki and approved by the ethics committee of the Affiliated Hospital of Xuzhou Medical University.
Consent for Publication
Patients were not required to provide informed consent, as the study was retrospective and the data was anonymous.
Acknowledgment
We would like to acknowledge the reviewers for their helpful comments on this paper.
Author Contributions
All authors made a significant contribution to the work reported, whether in conception, study design, execution, acquisition of data, analysis and interpretation, or all these areas, took part in drafting, revising, or critically reviewing the article, gave final approval to the version to be published, have agreed on the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
Funding
The work is funded by The opening Project of Provincial key Laboratory of Colleges and Universities in Jiangsu Province (XZSYSKF2020010); The Youth Medical Science and Technology Innovation Project of Xuzhou Municipal Health Commission (XWKYHT20200001); The Science and Technology Development Fund Project of the affiliated Hospital of Xuzhou Medical University in 2020 (XYFZ2020008).
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Macdonald RL, Schweizer TA. Spontaneous subarachnoid haemorrhage. Lancet. 2017;389(10069):655–666. doi:10.1016/s0140-6736(16)30668-7
2. Owen CM, Montemurro N, Lawton MT. Microsurgical management of residual and recurrent aneurysms after coiling and clipping: an experience with 97 patients. Neurosurgery. 2015;62(Suppl 1):92–102. doi:10.1227/neu.0000000000000791
3. Grasso G, Alafaci C, Macdonald RL. Management of aneurysmal subarachnoid hemorrhage: state of the art and future perspectives. Surg Neurol Int. 2017;8:11. doi:10.4103/2152-7806.198738
4. English SW. Long-term outcome and economic burden of aneurysmal subarachnoid hemorrhage: are we only seeing the tip of the iceberg?. Neurocrit Care. 2020;33(1):37–38. doi:10.1007/s12028-020-00943-1
5. Saand AR, Yu F, Chen J, Chou SH. Systemic inflammation in hemorrhagic strokes - A novel neurological sign and therapeutic target?. J Cereb Blood Flow Metab. 2019;39(6):959–988. doi:10.1177/0271678X19841443
6. Muroi C, Hugelshofer M, Seule M, et al. Correlation among systemic inflammatory parameter, occurrence of delayed neurological deficits, and outcome after aneurysmal subarachnoid hemorrhage. Neurosurgery. 2013;72(3):367–375. doi:10.1227/NEU.0b013e31828048ce
7. Fassbender K, Hodapp B, Rossol S, et al. Inflammatory cytokines in subarachnoid haemorrhage: association with abnormal blood flow velocities in basal cerebral arteries. J Neurol Neurosurg Psychiatry. 2001;70(4):534–537. doi:10.1136/jnnp.70.4.534
8. Zhong W, Zhang Z, Zhao P, et al. The impact of initial systemic inflammatory response after aneurysmal subarachnoid hemorrhage. Turk Neurosurg. 2017;27(3):346–352. doi:10.5137/1019-5149.Jtn.16193-15.1
9. Vergouwen MD, Vermeulen M, van Gijn J, et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke. 2010;41(10):2391–2395. doi:10.1161/strokeaha.110.589275
10. Drake CG. Report of world federation of neurological surgeons committee on a universal subarachnoid hemorrhage grading scale. J Neurosurg. 1988;68(6):985–986. doi:10.3171/jns.1988.68.6.0985
11. Claassen J, Bernardini GL, Kreiter K, et al. Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid hemorrhage: the fisher scale revisited. Stroke. 2001;32(9):2012–2020. doi:10.1161/hs0901.095677
12. Broderick JP, Adeoye O, Elm J. Evolution of the modified rankin scale and its use in future stroke trials. Stroke. 2017;48(7):2007–2012. doi:10.1161/strokeaha.117.017866
13. Zhou Y, Wang Y, Wang J, Anne Stetler R, Yang QW. Inflammation in intracerebral hemorrhage: from mechanisms to clinical translation. Prog Neurobiol. 2014;115:25–44. doi:10.1016/j.pneurobio.2013.11.003
14. Osuka K, Watanabe Y, Suzuki C, Iwami K, Miyachi S. Sequential expression of neutrophil chemoattractants in cerebrospinal fluid after subarachnoid hemorrhage. J Neuroimmunol. 2021;357:577610. doi:10.1016/j.jneuroim.2021.577610
15. Wu F, Liu Z, Li G, et al. Inflammation and oxidative stress: potential targets for improving prognosis after subarachnoid hemorrhage. Front Cell Neurosci. 2021;15:739506. doi:10.3389/fncel.2021.739506
16. Sehba FA, Hou J, Pluta RM, Zhang JH. The importance of early brain injury after subarachnoid hemorrhage. Prog Neurobiol. 2012;97(1):14–37. doi:10.1016/j.pneurobio.2012.02.003
17. Gris T, Laplante P, Thebault P, et al. Innate immunity activation in the early brain injury period following subarachnoid hemorrhage. J Neuroinflammation. 2019;16(1):253. doi:10.1186/s12974-019-1629-7
18. Schiller M, Ben-Shaanan TL, Rolls A. Neuronal regulation of immunity: why, how and where?. Nat Rev Immunol. 2021;21(1):20–36. doi:10.1038/s41577-020-0387-1
19. Giede-Jeppe A, Reichl J, Sprügel MI, et al. Neutrophil-to-lymphocyte ratio as an independent predictor for unfavorable functional outcome in aneurysmal subarachnoid hemorrhage. J Neurosurg. 2019;132(2):400–407. doi:10.3171/2018.9.Jns181975
20. Li R, Yuan Q, Su Y, Chopp M, Yan T, Chen J. Immune response mediates the cardiac damage after subarachnoid hemorrhage. Exp Neurol. 2020;323:113093. doi:10.1016/j.expneurol.2019.113093
21. Beseoglu K, Steiger HJ. Elevated glycated hemoglobin level and hyperglycemia after aneurysmal subarachnoid hemorrhage. Clin Neurol Neurosurg. 2017;163:128–132. doi:10.1016/j.clineuro.2017.10.037
22. Hanhai Z, Bin Q, Shengjun Z, et al. Neutrophil extracellular traps, released from neutrophil, promote microglia inflammation and contribute to poor outcome in subarachnoid hemorrhage. Aging. 2021;13(9):13108–13123. doi:10.18632/aging.202993
23. Al-Mufti F, Amuluru K, Damodara N, et al. Admission neutrophil-lymphocyte ratio predicts delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. J Neurointerv Surg. 2019;11(11):1135–1140. doi:10.1136/neurintsurg-2019-014759
24. Kuo LT, Huang AP. The pathogenesis of hydrocephalus following aneurysmal subarachnoid hemorrhage. Int J Mol Sci. 2021;22(9). doi:10.3390/ijms22095050
25. Rass V, Helbok R. Early brain injury after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep. 2019;19(10):78. doi:10.1007/s11910-019-0990-3
26. Macdonald RL. Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol. 2014;10(1):44–58. doi:10.1038/nrneurol.2013.246
27. Sholl-Franco A, da Silva AG, Adão-Novaes J. Interleukin-4 as a neuromodulatory cytokine: roles and signaling in the nervous system. Ann NY Acad Sci. 2009;1153:65–75. doi:10.1111/j.1749-6632.2008.03962.x
28. Sheikh AM, Nagai A, Wakabayashi K, et al. Mesenchymal stem cell transplantation modulates neuroinflammation in focal cerebral ischemia: contribution of fractalkine and IL-5. Neurobiol Dis. 2011;41(3):717–724. doi:10.1016/j.nbd.2010.12.009
29. Chaudhry SR, Kahlert UD, Kinfe TM, et al. Elevated systemic il-10 levels indicate immunodepression leading to nosocomial infections after Aneurysmal Subarachnoid Hemorrhage (SAH) in patients. Int J Mol Sci. 2020;21(5):1569. doi:10.3390/ijms21051569
30. Garcia JM, Stillings SA, Leclerc JL, et al. Role of interleukin-10 in acute brain injuries. Front Neurol. 2017;8:244. doi:10.3389/fneur.2017.00244
31. Savarraj J, Parsha K, Hergenroeder G, et al. Early brain injury associated with systemic inflammation after subarachnoid hemorrhage. Neurocrit Care. 2018;28(2):203–211. doi:10.1007/s12028-017-0471-y
32. Waisman A, Hauptmann J, Regen T. The role of IL-17 in CNS diseases. Acta Neuropathol. 2015;129(5):625–637. doi:10.1007/s00401-015-1402-7
33. Chaudhry SR, Güresir E, Vatter H, et al. Aneurysmal subarachnoid hemorrhage lead to systemic upregulation of IL-23/IL-17 inflammatory axis. Cytokine. 2017;97:96–103. doi:10.1016/j.cyto.2017.05.025
34. Ullrich KA, Schulze LL, Paap EM, Müller TM, Neurath MF, Zundler S. Immunology of IL-12: an update on functional activities and implications for disease. EXCLI J. 2020;19:1563–1589. doi:10.17179/excli2020-3104
35. Savarraj JPJ, Parsha K, Hergenroeder GW, et al. Systematic model of peripheral inflammation after subarachnoid hemorrhage. Neurology. 2017;88(16):1535–1545. doi:10.1212/wnl.0000000000003842
36. Dong G, Li C, Hu Q, et al. Low-dose il-2 treatment affords protection against subarachnoid hemorrhage injury by expanding peripheral regulatory t cells. ACS Chem Neurosci. 2021;12(3):430–440. doi:10.1021/acschemneuro.0c00611
37. Wu K, Ma J, Bai W, et al. Short-term intratracheal use of PEG-modified IL-2 and glucocorticoid persistently alleviates asthma in a mouse model. Sci Rep. 2016;6:31562. doi:10.1038/srep31562
38. Dutcher JP, Schwartzentruber DJ, Kaufman HL, et al. High dose interleukin-2 (Aldesleukin) - expert consensus on best management practices-2014. J Immunother Cancer. 2014;2(1):26. doi:10.1186/s40425-014-0026-0
39. Roselli F, Chandrasekar A, Morganti-Kossmann MC. Interferons in traumatic brain and spinal cord injury: current evidence for translational application. Front Neurol. 2018;3:458. doi:10.3389/fneur.2018.00458
40. Cai L, Zeng H, Tan X, Wu X, Qian C, Chen G. The role of the blood neutrophil-to-lymphocyte ratio in aneurysmal subarachnoid hemorrhage. Front Neurol. 2021;12:671098. doi:10.3389/fneur.2021.671098
41. Kipnis J. Multifaceted interactions between adaptive immunity and the central nervous system. Science. 2016;353(6301):766–771. doi:10.1126/science.aag2638
42. Galea J, Ogungbenro K, Hulme S, et al. Reduction of inflammation after administration of interleukin-1 receptor antagonist following aneurysmal subarachnoid hemorrhage: results of the subcutaneous interleukin-1Ra in SAH (SCIL-SAH) study. J Neurosurg. 2018;128(2):515–523. doi:10.3171/2016.9.Jns16615
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
The Role of Inflammasomes in Mediating Urological Disease: A Short Literature Review
Shidid S, Bluth MH, Smith-Norowitz TA
Journal of Inflammation Research 2022, 15:4359-4365
Published Date: 1 August 2022