Back to Journals » OncoTargets and Therapy » Volume 11
Clinical manifestations of pulmonary mucosa-associated lymphoid tissue lymphoma: single-center experience with 18 patients
Authors Zhao S, Zhang L, Gu Z , Zhu C, Fang S, Yang N, Wang F, Guan L, Luo L, Gao C
Received 26 July 2017
Accepted for publication 19 December 2017
Published 24 January 2018 Volume 2018:11 Pages 555—561
DOI https://doi.org/10.2147/OTT.S147275
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jianmin Xu
Shasha Zhao,1,2 Lin Zhang,3 Zhenyang Gu,1 Chengying Zhu,1,2 Shu Fang,1 Nan Yang,1 Feiyan Wang,1,2 Lixun Guan,1 Lan Luo,1 Chunji Gao1
1Department of Hematology, Chinese People’s Liberation Army (PLA) General Hospital, Beijing, 2School of Medicine, Nankai University, Tianjin, 3Department of Hematology, First Hospital of Qinhuangdao, Qinhuangdao, China
Purpose: Pulmonary mucosa-associated lymphoid tissue (MALT) lymphoma is a rare entity. To date, the optimal treatment for this disease is still under debate. The aim of this study was to analyze and summarize the clinical manifestations and therapeutic experience of 18 pulmonary MALT lymphoma patients to collect information about the optimal treatment modality.
Patients and methods: A retrospective analysis was performed in patients who were diagnosed with pulmonary MALT lymphoma at the Chinese People’s Liberation Army General Hospital from April 1995 to April 2016.
Results: Clinical data of 18 patients were available. The median age was 55 (range, 34–67) years. Also, 61.1% of the patients were male. Only 33.3% had a history of smoking and 27.8% of the patients had tuberculosis. Treatment modalities included surgery alone in 1 patient (5.6%), chemotherapy in 10 patients (55.5%), surgery in combination with chemotherapy in 6 patients (33.3%) and observation in 1 patient (5.6%). Over the median observation period of 93 months, 2 patients died, the median progression-free survival was 6 years, and the estimated 5- and 10-year overall survival rates were 94.1% and 83.7%, respectively. The survival data confirmed the indolent nature of the disease. There was no difference in progression-free survival between the chemotherapy group and the surgery in combination with chemotherapy group.
Conclusion: Pulmonary MALT lymphoma tended to be an indolent disease. In order to preserve the lung function and reduce the risks associated with surgery, chemotherapy might be an optimal choice for the treatment of pulmonary MALT lymphoma.
Keywords: mucosa-associated lymphoid tissue lymphoma, lung, clinical features, chemotherapy
Introduction
Pulmonary marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT) that arises from the bronchial-associated lymphoid tissue is known as bronchial-associated lymphoid tissue lymphoma.1,2 Pulmonary MALT lymphoma is a rare disease constituting <1% of all malignant lymphomas, but it is the most common histologic subtype of lung lymphoma.3
Pulmonary MALT tends to occur in old-aged patients. The majority of patients are asymptomatic; in symptomatic patients, nonspecific pulmonary symptoms (such as cough, sputum, chest pain, dyspnea) are the most common.4 Pulmonary lesions are commonly discovered via a routine chest radiographic study incidentally, and the majority of patients present with localized disease. The commonest presentation is that of a solitary, well-defined nodule found on routine X-ray examination. Single or multiple nodules or areas of consolidation constitute the most frequently observed patterns on computerized tomography (CT) scans.5,6 Less commonly, there may be multiple nodules and/or a pleural effusion.7 Treatment options range from simple surgical excision to surgery followed by radiotherapy or chemotherapy alone.4,7 Due to the lack of randomized controlled trials and favorable response to different therapeutic modalities incorporated during the course of the disease, there are no standard treatment protocols or guidelines for pulmonary MALT lymphoma. Although quite a few reports have been published regarding pulmonary MALT lymphoma, the optimal treatment for this condition is not thoroughly elucidated. In this retrospective analysis, we summarize the clinical features and our therapeutic experience of 18 primary pulmonary MALT lymphoma patients in order to collect information about the optimal treatment modality.
Patients and methods
Patients
We retrospectively evaluated clinical data of 18 patients who were diagnosed with pulmonary MALT lymphoma from April 1995 to April 2016 at the Chinese People’s Liberation Army General Hospital. Inclusion in this study required an initial diagnosis of pulmonary MALT lymphoma according to the World Health Organization criteria and primary site in the lungs.8
The following clinical data were obtained from the records: patients’ gender, age, performance status (PS), clinical stage, International Prognostic Index (IPI), respiratory symptoms, the presence of B symptoms, past history, blood count, erythrocyte sedimentation rate (ESR), lactic dehydrogenase level, β2-microglobulin (β2-MG) level, radiologic findings, bone marrow findings, date of diagnosis, diagnosis procedures, histology, type of treatment, treatment response, date of progression, date of last follow-up and cause of death. Response criteria were defined according to the World Health Organization criteria for response.9 The IPI score was recorded according to the published criteria.10 The staging was based upon the Ann Arbor staging system.11
The study was conducted in accordance with the Declaration of Helsinki. All the patients signed written informed consent, and this study was approved by the Ethical Committee of the People’s Liberation Army General Hospital.
Statistical methodology
SPSS version 17.0 (SPSS, Chicago, IL, USA) was used for data processing and analysis. Overall survival (OS) and progression-free survival (PFS) curves were estimated by the Kaplan–Meier method. OS was measured from the date of diagnosis to the date of death or the last follow-up visit. PFS was calculated from the end of induction therapy to the date of disease progression/relapse or death in patients who received any treatment. Duration of follow-up was measured from the date of diagnosis to the last follow-up or date of death. All patients with no progression or who were alive at the last follow-up date were considered right censored. Survival rates were compared for statistical differences by log-rank analyses. All statistical tests were two-sided, and P<0.05 was considered statistically significant.
Results
Patient characteristics
From our database, we could identify a total of 18 consecutive patients with pulmonary MALT lymphoma diagnosed and treated at our institution from April 1995 to April 2016. The clinical characteristics of the 18 patients are presented in Table 1. The male:female ratio of the 18 patients was 11:7. The median age at diagnosis was 55 (range 34–67) years. Seven patients (38.9%) were older than 60 years. Ten patients (55.6%) presented good PS (0–1), while 14 patients (77.8%) presented at diagnosis with late stage disease (stages III–IV). IPI was low risk in 1 patient, low-intermediate risk in 11 patients, high–intermediate risk in 2 patients and high risk in 4 patients. B symptoms were observed in 3 patients. Six patients (33.3%) had a history of smoking and 5 patients (27.8%) had tuberculosis. With the exception of 4 patients (22.2%) who exhibited no symptoms, patients initially presented with cough (38.9%), expectoration (33.3%), dyspnea (27.8%) and chest pain (33.3%). Hemoglobin level and serum lactate dehydrogenase (LDH) activity were normal in most patients (94.4%). Ten patients (10/14, 71.4%) had elevated β2-MG, and 6 patients (6/15, 40%) showed elevated ESR. Four patients (22.2%) were infected with Helicobacter pylori. None of the evaluated patients were infected by human immunodeficiency virus, while 5 patients (27.8%) were infected with hepatitis B virus.
Diagnostic procedures
In symptomatic patients with pulmonary MALT lymphoma, the median duration of symptoms before definitive diagnosis was 3 (range 1–60) months. All patients underwent thoracic CT scan or chest X-ray. A common radiologic pattern, such as single or multiple pulmonary nodules, masses or mass-like areas of consolidation, localized in the center or in the periphery of parenchyma was not definable.
Diagnostic procedures of patients with pulmonary MALT lymphoma are presented in Table 2. Surgical lung biopsy was performed in 6 patients (video-assisted thoracoscopy, n=2; open lung biopsy, n=4), transbronchial lung biopsy in 7 patients and CT-guided transthoracic lung biopsy in 5 patients.
Histology
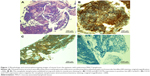
The pulmonary structure was effaced by abnormal lymphocyte infiltration, predominantly localized along bronchovascular bundles, interlobular septa and visceral pleura. Morphologically, the lymphoma cells exhibited a spectrum of cytologic features (small round cells, centrocyte-like cells, monocytoid cells). The presence of lymphoepithelial lesion was frequent and involved bronchiolar and bronchial epithelial structures. Immunohistochemical staining was performed in all 18 cases, with all neoplastic lymphocytes staining positive for CD20 and CD79a expression. There was also scattered positive staining for BCL2 in 10 cases. CD10, CD5 and CyclinD1 expression were negative for most neoplastic lymphocytes. Examples of representative pathologic images of tissue from patients in this study are shown in Figure 1.
Treatment and response
Treatment and response are reported in Table 3. Of the 18 patients, 1 patient did not receive any treatment, while 17 patients received various combinations of treatment. One patient underwent surgery alone, 6 patients were treated with surgery plus chemotherapy and 10 patients received chemotherapy alone. R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine and prednisolone) and CHOP (cyclophosphamide, doxorubicin, vincristine and prednisolone) were administered to 9 and 2 patients, respectively. R-COP (rituximab, cyclophosphamide, vincristine and prednisolone) was administered to 1 patient. Two patients were treated with R-FND (rituximab, fludarabine, mitoxantrone and dexamethasone). R-FC (rituximab, fludarabine and cyclophosphamide) was administered to 1 patient. Rituximab was used in combination with chemotherapy in 14 patients, including surgery followed by rituximab-containing chemotherapy in 4 patients and rituximab-containing chemotherapy alone in 10 patients. No patient was treated with radiotherapy. The responses to treatment were a complete response (CR) in 8 patients, including 1 who received surgery alone, partial response (PR) in 6 patients and stable disease in 3 patients.
Outcome
The median time between diagnosis and start of treatment was 12 (range 0–68) days. Five patients experienced progression/relapse after first-line treatment (median time to progression was 72 months, range 1–204). With a median follow-up of 93 (range 12–204) months, 2 patients died from unknown causes. Figure 2 shows the Kaplan–Meier curves of OS and PFS of the entire population. The median PFS was 6 years and the estimated 5- and 10-year OS rates were 94.1% and 83.7%, respectively. Figure 3 shows the differences in PFS according to treatment modality; we found no difference between the chemotherapy group and the surgery in combination with chemotherapy group (P=0.24).
  | Figure 3 Progression-free survival curve according to surgery and chemotherapy in pulmonary MALT lymphoma patients. |
Discussion
Our data confirm the previously described indolent behavior of pulmonary MALT lymphoma.12–15 The median age of the 18 patients in our study was 55 years, similar to those of the other series.12,16 Although previous studies reported female predominance16–18 or an equal number of male and female patients,19,20 we found male predominance, which was consistent with some previous studies.12,15,21 In this study, about 77.8% of patients were symptomatic and they usually reported nonspecific pulmonary symptoms such as cough, expectoration, dyspnea and chest pain, while a minority of patients (16.7%) experienced constitutional symptoms such as fever, night sweats and weight loss. These characteristics are summarized in Table 4. As mentioned in the preceding text, 77.8% of the patients were in stage III/IV, and therefore, we cannot exclude that lung was not the primary site of the disease. Most patients presented good PS; although 71.4% patients had elevated β2-MG levels, most patients had no adverse biologic prognostic factors such as anemia, elevated LDH or elevated ESR. Our series confirmed the characteristic indolent clinical presentation of pulmonary MALT lymphoma.
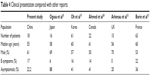  | Table 4 Clinical presentation compared with other reports |
Many investigators believed that pulmonary MALT lymphoma is not a normal constituent of human lungs and rather its development is driven by long-term exposure to various antigenic stimuli.22–24 Smoking, chronic inflammatory disease and various autoimmune diseases, such as Sjoren’s syndrome and rheumatoid arthritis, have been believed to be the etiological agents for pulmonary MALT lymphoma, but the pathogenesis of this disease remains unclear.7,9,25–27 In this study, 33.3% of patients had a smoking history, which was lower than that of other studies.15,20,28 So, we cannot definitively exclude its possible role in pathogenesis of the disease. Further studies are needed to address this issue. Another notable finding in our study was that 27.8% of the patients had a history of tuberculosis.
In our series, the radiographic appearance of pulmonary MALT lymphoma was represented as pulmonary nodules and as lung masses or mass-like areas of consolidation. Pleural effusion and mediastinal or hilar lymphadenopathy were rare in our series, consistent with previous observations.5,7,29 This suggested that the radiographic presentation of pulmonary MALT lymphomas is not specific, but variable in different patients. Pulmonary MALT lymphoma could be diagnosed only by pathologic methods. CT-guided transthoracic lung biopsy and open thoracotomy or video-assisted thoracoscopy lung biopsy are the common methods to obtain samples. A previous study had reported the positive predictive rate of puncture biopsy under CT guidance for diagnosing pulmonary non-Hodgkin’s lymphoma was only 25%.16 However, in our study, using ancillary immunohistochemical staining, 12 (66.7%) patients were diagnosed by minimally invasive procedures, including CT-guided transthoracic lung biopsy and transbronchial lung biopsy without surgery. This finding may be attributed to the wide use of examination of immunohistochemical staining and less-invasive diagnostic procedures before performing surgery.
In agreement with the previous observations, our data showed the impressively favorable clinical course of pulmonary MALT lymphoma regardless of the treatment. The optimal management of lung extranodal marginal zone lymphomas with regard to surgery, chemotherapy and radiation therapy alone or in combination, as well as abstention from therapy, has to be clearly determined. Some experts have suggested that surgery should not be the first choice of treatment, the aim of which should be to preserve lung function.30–32 Oh et al15 found no differences in OS between patients given chemotherapy and those who underwent surgery. But Lee et al found complete surgical resection may lead to favorable treatment outcomes for low-stage pulmonary MALT lymphoma.33 However, in our study, 14 patients presented at diagnosis with late stage disease (stages III–IV) and only 1 patient accepted surgery alone. Most of our patients were managed by chemotherapy. Rituximab was used in combination with chemotherapy in 14 patients. We observed 6 cases of CR, 5 PR cases and 3 cases of stable disease. Ahmed et al20 reported the results of 10 patients treated with rituximab alone or in combination with chemotherapy, with 2 achieving CR and 8 achieving PR. It is, therefore, possible that rituximab has a potential curative power in patients with pulmonary MALT lymphoma. During the follow-up period, 2 cases died, disease progression was noted in 5 patients overall (27.8%) and 1 of them developed disease progression during the initial observation period. The estimated 5- and 10-year OS rates were 94.1% and 83.7%, respectively. Our results were in agreement with most of the previous observations that showed a slow rate of progression and a favorable course in pulmonary MALT lymphoma.13,15,20,34 In our series, we noted no difference in PFS between chemotherapy and surgery combined with chemotherapy. So, operation might not be the first choice to preserve the lung function and avoid the risks of surgery.
Limitations of this study should be acknowledged. First, this was a retrospective study with a relatively small number of patients. Second, patients with pulmonary MALT lymphoma were included in this study based on a single tertiary care medical center. Therefore, it should be further confirmed through multicenter, large-scale clinical data.
Conclusion
Our study revealed that pulmonary MALT lymphoma tended to be an indolent disease. We found a male preponderance. The elevation of LDH was rare in our patients. Lung nodules and masses or mass-like areas of consolidation were the common radiographic findings. With CT-guided transthoracic lung biopsy and transbronchial lung biopsy, invasive procedures might be a suitable option for diagnosis of pulmonary MALT lymphoma. In order to preserve the lung function and reduce the risks associated with surgery, chemotherapy might be an optimal choice for the treatment of pulmonary MALT lymphoma.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No 81270642 and 81070451) and Beijing Natural Science Foundation (No 7162175).
Disclosure
The authors report no conflicts of interest in this work.
References
Harris NL, Jaffe ES, Stein H, et al. A revised European-American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood. 1994;84(5):1361–1392. | ||
Isaacson P, Wright DH. Malignant lymphoma of mucosa-associated lymphoid tissue. A distinctive type of B-cell lymphoma. Cancer. 1983;52(8):1410–1416. | ||
Ahmed S, Siddiqui AK, Rai KR. Low-grade B-cell bronchial associated lymphoid tissue (BALT) lymphoma. Cancer Invest. 2002;20(7–8):1059–1068. | ||
Wannesson L, Cavalli F, Zucca E. Primary pulmonary lymphoma: current status. Clin Lymphoma Myeloma. 2005;6(3):220–227. | ||
Bae YA, Lee KS, Han J, et al. Marginal zone B-cell lymphoma of bronchus-associated lymphoid tissue: imaging findings in 21 patients. Chest. 2008;133(2):433–440. | ||
King LJ, Padley SP, Wotherspoon AC, Nicholson AG. Pulmonary MALT lymphoma: imaging findings in 24 cases. Eur Radiol. 2000;10(12):1932–1938. | ||
Zinzani PL, Poletti V, Zompatori M, et al. Bronchus-associated lymphoid tissue lymphomas: an update of a rare extranodal maltoma. Clin Lymphoma Myeloma. 2007;7(9):566–572. | ||
Swerdlow SH. Lymphoma classification and the tools of our trade: an introduction to the 2012 USCAP Long Course. Mod Pathol. 2013;26(Suppl 1):S1–S14. | ||
Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer. 1981;47(1):207–214. | ||
International Non-Hodgkin’s Lymphoma Prognostic Factors P. A predictive model for aggressive non-Hodgkin’s lymphoma. N Engl J Med. 1993;329(14):987–994. | ||
Carbone PP, Kaplan HS, Musshoff K, Smithers DW, Tubiana M. Report of the Committee on Hodgkin’s Disease Staging Classification. Cancer Res. 1971;31(11):1860–1861. | ||
Arkenau HT, Gordon C, Cunningham D, Norman A, Wotherspoon A, Chau I. Mucosa associated lymphoid tissue lymphoma of the lung: the Royal Marsden Hospital experience. Leuk Lymphoma. 2007;48(3):547–550. | ||
Borie R, Wislez M, Thabut G, et al. Clinical characteristics and prognostic factors of pulmonary MALT lymphoma. Eur Respir J. 2009;34(6):1408–1416. | ||
Imai H, Sunaga N, Kaira K, et al. Clinicopathological features of patients with bronchial-associated lymphoid tissue lymphoma. Intern Med. 2009;48(5):301–306. | ||
Oh SY, Kim WS, Kim JS, et al. Pulmonary marginal zone B-cell lymphoma of MALT type – what is a prognostic factor and which is the optimal treatment, operation, or chemotherapy?: Consortium for Improving Survival of Lymphoma (CISL) study. Ann Hematol. 2010;89(6):563–568. | ||
Graham BB, Mathisen DJ, Mark EJ, Takvorian RW. Primary pulmonary lymphoma. Ann Thorac Surg. 2005;80(4):1248–1253. | ||
Huang H, Lu ZW, Jiang CG, Li J, Xu K, Xu ZJ. Clinical and prognostic characteristics of pulmonary mucosa-associated lymphoid tissue lymphoma: a retrospective analysis of 23 cases in a Chinese population. Chin Med J (Engl). 2011;124(7):1026–1030. | ||
Kurtin PJ, Myers JL, Adlakha H, et al. Pathologic and clinical features of primary pulmonary extranodal marginal zone B-cell lymphoma of MALT type. Am J Surg Pathol. 2001;25(8):997–1008. | ||
Zinzani PL, Tani M, Gabriele A, et al. Extranodal marginal zone B-cell lymphoma of MALT-type of the lung: single-center experience with 12 patients. Leuk Lymphoma. 2003;44(5):821–824. | ||
Ahmed S, Kussick SJ, Siddiqui AK, et al. Bronchial-associated lymphoid tissue lymphoma: a clinical study of a rare disease. Eur J Cancer. 2004;40(9):1320–1326. | ||
Ogusa E, Tomita N, Ishii Y, et al. Clinical manifestations of primary pulmonary extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue in Japanese population. Hematol Oncol. 2013;31(1):18–21. | ||
Gould SJ, Isaacson PG. Bronchus-associated lymphoid tissue (BALT) in human fetal and infant lung. J Pathol. 1993;169(2):229–234. | ||
Pabst R. Is BALT a major component of the human lung immune system? Immunol Today. 1992;13(4):119–122. | ||
Pabst R, Gehrke I. Is the bronchus-associated lymphoid tissue (BALT) an integral structure of the lung in normal mammals, including humans? Am J Respir Cell Mol Biol. 1990;3(2):131–135. | ||
Meuwissen HJ, Hussain M. Bronchus-associated lymphoid tissue in human lung: correlation of hyperplasia with chronic pulmonary disease. Clin Immunol Immunopathol. 1982;23(2):548–561. | ||
Nakamura N, Yamada G, Itoh T, et al. Pulmonary MALT lymphoma with amyloid production in a patient with primary Sjogren’s syndrome. Intern Med. 2002;41(4):309–311. | ||
Sato A, Hayakawa H, Uchiyama H, Chida K. Cellular distribution of bronchus-associated lymphoid tissue in rheumatoid arthritis. Am J Respir Crit Care Med. 1996;154(6 Pt 1):1903–1907. | ||
Sammassimo S, Pruneri G, Andreola G, et al. A retrospective international study on primary extranodal marginal zone lymphoma of the lung (BALT lymphoma) on behalf of International Extranodal Lymphoma Study Group (IELSG). Hematol Oncol. 2016;34(4):177–183. | ||
Cordier JF, Chailleux E, Lauque D, et al. Primary pulmonary lymphomas. A clinical study of 70 cases in nonimmunocompromised patients. Chest. 1993;103(1):201–208. | ||
Girinsky T, Paumier A, Ferme C, et al. Low-dose radiation treatment in pulmonary mucosa-associated lymphoid tissue lymphoma: a plausible approach? A single-institution experience in 10 patients. Int J Radiat Oncol Biol Phys. 2012;83(3):e385–e389. | ||
Troch M, Streubel B, Petkov V, Turetschek K, Chott A, Raderer M. Does MALT lymphoma of the lung require immediate treatment? An analysis of 11 untreated cases with long-term follow-up. Anticancer Res. 2007;27(5B):3633–3637. | ||
Wang L, Xia ZJ, Zhang YJ, Huang HQ, Lin TY, Lu Y. Radical surgery may be not an optimal treatment approach for pulmonary MALT lymphoma. Tumor Biol. 2015;36(8):6409–6416. | ||
Lee H, Yang B, Nam B, et al. Treatment outcomes in patients with extranodal marginal zone B-cell lymphoma of the lung. J Thorac Cardiovasc Surg. 2017;154(1):342–349. | ||
Zinzani PL, Magagnoli M, Galieni P, et al. Nongastrointestinal low-grade mucosa-associated lymphoid tissue lymphoma: analysis of 75 patients. J Clin Oncol. 1999;17(4):1254. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.