Back to Journals » Journal of Pain Research » Volume 13
Clinical Implications of Vertebral Endplate Disruptions After Lumbar Discectomy: 3-Year Results from a Randomized Trial of a Bone-Anchored Annular Closure Device
Authors Kuršumović A, Bouma G , Miller LE , Assaker R, Van de Kelft E, Hes R, Kienzler JC
Received 8 August 2019
Accepted for publication 7 March 2020
Published 31 March 2020 Volume 2020:13 Pages 669—675
DOI https://doi.org/10.2147/JPR.S226480
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor E Alfonso Romero-Sandoval
Adisa Kuršumović,1 Gerrit Joan Bouma,2 Larry E Miller,3 Richard Assaker,4 Erik Van de Kelft,5 Robert Hes,6 Jenny C Kienzler7
1Department of Neurosurgery, Donauisar Klinikum Deggendorf, Deggendorf, Germany; 2Department of Neurosurgery, OLVG-West and Academic Medical Center, Amsterdam, the Netherlands; 3Miller Scientific, Johnson City, TN, USA; 4Department of Neurosurgery, Centre Hospitalier Régional Universitaire de Lille, Lille, France; 5Department of Neurosurgery, AZ Nikolaas, Sint-Niklaas and University of Antwerp, Antwerpen, Belgium; 6Department of Neurosurgery, AZ Klina, Brasschaat, Belgium; 7Department of Neurosurgery, Kantonsspital Aarau, Aarau, Switzerland
Correspondence: Adisa Kuršumović
Department of Neurosurgery, Donauisar Klinikum Deggendorf, Deggendorf, Germany
Email [email protected]
Objective: Vertebral endplate disruptions (VEPD) are common findings on imaging after lumbar surgery. The objective of this study was to explore the clinical implications of VEPD development following lumbar discectomy with or without implant with a bone-anchored annular closure device (ACD).
Methods: This was a multicenter randomized controlled trial of patients with large postsurgical annular defects after limited lumbar discectomy who were randomized to additionally receive an ACD or no additional treatment. VEPD were identified on computed tomography and confirmed by an imaging core laboratory. Clinical outcomes included recurrent herniation, reoperation, Oswestry Disability Index, leg pain, and back pain. Patient follow-up in this study was 3 years.
Results: In the ACD group (n=272), the risk of reoperation was lower in patients with vs without VEPD (8% vs 24%, p< 0.01), but no other clinical outcomes differed when stratified by VEPD prevalence or size. In the Control group (n=278), the risk of symptomatic reherniation was higher in patients with VEPD (41% vs 23%, p< 0.01) and patients with the largest VEPD had the highest reoperation rates. Patient-reported outcomes were not associated with VEPD prevalence or size in the Control group.
Conclusion: VEPD had no significant influence on patient-reported outcomes at 3 years after lumbar discectomy. VEPD increased the risk of recurrence in patients treated with lumbar discectomy only, but had no negative influence in patients treated with the ACD.
Keywords: annular closure, disc herniation, lumbar discectomy, randomized controlled trial, sciatica, vertebral endplate
Introduction
Vertebral endplate disruptions (VEPD) are common findings on magnetic resonance imaging or computed tomography (CT) in asymptomatic adults and in patients with low back pain.1–3 New VEPD may also develop after lumbar surgery.4–6 However, the natural history and long-term clinical implications of VEPD are unclear. Results from a randomized controlled trial demonstrated that patients with lumbar disc herniation treated with limited lumbar discectomy and a bone-anchored annular closure device (ACD) had a lower risk of symptomatic recurrence and reoperation versus those treated with limited lumbar discectomy only, even though the ACD group had a higher frequency of VEPD in follow-up.7 Since the typical patient undergoing lumbar discectomy is 40 to 50 years of age, it is imperative to understand the natural history and clinical consequences of VEPD over the long term. This study presents long-term data on the association between VEPD and clinical outcomes from a randomized controlled trial of patients with large annular defects undergoing limited lumbar discectomy with or without bone-anchored ACD.
Methods
Details of the study design, patients, procedure, and VEPD assessment methods have been previously reported.8 Briefly, this randomized controlled trial was conducted to determine whether implantation of a bone-anchored ACD following lumbar discectomy reduced the risk of recurrent herniation compared to lumbar discectomy alone in high-risk patients with large annular defects. The clinical trial was conducted according to the principles of the Helsinki Declaration, was approved by local ethics review boards (Supplement Table 1), and all participants provided written informed consent. This study was prospectively registered at ClinicalTrials.gov (NCT01283438). Important study eligibility criteria were single-level lumbar disc herniation, disc height ≥ 5 mm, attempted nonsurgical treatment for at least 6 weeks, back-related disability at least 40 points (0–100 scale) on the Oswestry Disability Index (ODI),9 and leg pain severity at least 40 points (0–100 scale) on a visual analogue scale.10
Eligible patients underwent limited lumbar discectomy where only extradiscal tissue was removed. After completion of the discectomy, patients with a large annular defect (4–6 mm tall and 6–10 mm wide) were intraoperatively randomized to limited discectomy only or additional implantation with a bone-anchored ACD (Figure 1). The annular defect size criterion was implemented since the risk of symptomatic reherniation and reoperation in patients with large annular defects is more than double that compared to those with small annular defects.11 Patients with small annular defects or defects larger than 10 mm were excluded from further study participation. Patients returned for follow-up visits at 6 weeks, 3 months, 6 months, and annually thereafter.
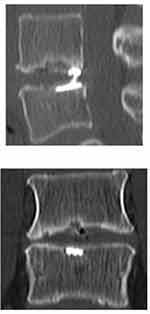
VEPD were evaluated at baseline and at annual follow-up visits by multiplanar, low-dose CT with two-dimensional reconstruction at the index level. CT images were read by board-certified radiologists at an independent imaging core laboratory who were blinded to patient outcomes. VEPD was defined as any osseous disruption of the superior or inferior vertebral endplate observed on CT that could not be explained by the overall shape of the endplate, recognizing the variation in morphology among vertebral endplates.12 Main clinical outcomes included ODI, leg pain severity, back pain severity, symptomatic reherniation confirmed by clinical and imaging findings, and reoperations.
Statistical analyses were performed on a modified intention-to-treat population, which included all randomized patients in whom the intended procedure was attempted. Baseline patient characteristics are presented as means and standard deviations for continuous variables and numbers and percentages for categorical variables. Group comparisons were made using Student’s t-test for continuous data or Fisher’s exact test for categorical data. Time-to-event data were analyzed using Kaplan-Meier methods with Log-rank tests for group comparisons. Statistical significance was set at P<0.05 and hypothesis testing was two-sided. Statistical analyses were performed using SAS v9.4 (SAS Institute) and R v3.3.2 (R Foundation for Statistical Computing).
Results
The modified intention-to-treat population consisted of 272 patients in the ACD group and 278 in the Control group. VEPD were present at baseline in 18% of the ACD group and 15% of Controls. In follow-up, there were 436 VEPD in the ACD group and 140 VEPD in Controls (P<0.001). Based on an a priori analysis plan to evaluate the association of VEPD with clinical outcomes at the time in which over 90% of VEPD achieved growth stability based on change in annual EPC size, presence of a sclerotic margin, and presence of reactive edema, we present these associations using 3-year follow-up data. Among patients with at least 1 VEPD at the 3-year follow-up visit, the relative frequency of VEPD at any vertebral endplate location was higher in the ACD group. Independent of VEPD frequency, the location of VEPD tended to be comparable among groups, with the highest frequencies observed in the central or central-posterior portion of the endplate (Figure 2).
Overall, outcomes at 3 years favored the ACD group for symptomatic reherniation (14.8% vs 29.5%; P<0.001), reoperation (11.0% vs 19.3%; P=0.007), leg pain (21 vs 30; P<0.01), back pain (23 vs 30; P=0.01), and ODI (18 vs 23; P=0.02). The frequency of all-cause serious adverse events was comparable between groups (33.7% vs 36.7%; P=0.47).
In the ACD group, the risk of reoperation was lower in patients with vs without VEPD (8% vs 24%, p<0.01), but no other clinical outcomes differed by VEPD prevalence (Table 1). In the Control group, the risk of symptomatic reherniation was higher in patients with VEPD (41% vs 23%, p<0.01); no other outcomes in this group differed by VEPD prevalence. In the ACD group, clinical outcomes were comparable across VEPD size tertiles (Table 2; Figure 3). In the Control group, patients with the largest VEPD had the highest reoperation rates (Figure 4).
 |
Table 1 Patient-Reported Outcomes at 3 Years by Treatment Group and VEPD Prevalence |
 |
Table 2 Patient-Reported Outcomes at 3 Years by Treatment Group and VEPD Area Tertiles |
Discussion
VEPD are distinct radiographic findings that are frequently observed in asymptomatic and symptomatic patients alike. Some types of VEPD, such as Schmorl’s nodes, have been implicated in the development of low back pain.13 While implantation with an ACD following limited lumbar discectomy in high-risk patients with large annular defects increases the risk for VEPD development, the clinical implications of VEPD have remained unclear. In this study, we performed an analysis investigating the association of clinical outcomes with VEPD prevalence and size. In each treatment group, VEPD characteristics had no association with patient-reported outcomes at 3 years follow-up. However, VEPD may influence recurrence risk, which differs by treatment group. In the ACD group, patients with VEPD had a lower risk of reoperation in follow-up. In Control patients, VEPD was associated with higher risk for symptomatic reherniation and reoperation.
A nonrandomized study of 85 patients undergoing limited lumbar discectomy with or without additional ACD implantation reported similar results where VEPD prevalence was higher in those treated with ACD (52% vs 10%), yet the reherniation rate was lower with ACD (5% vs 50%).14 Two-year results from this trial failed to identify clinical correlations associated with VEPD.8 Here, we present 3-year results that corroborate the findings from prior studies. In this respect, the current study represents the most comprehensive known analysis with the longest follow-up of the impact of VEPD following lumbar discectomy, with or without ACD implant. While speculative, it is plausible that the positive clinical results among patients with VEPD might be attributed to more secure seating of the occlusion component against the opposing endplate, resulting in a subsequent lower risk of reherniation.
Several aspects related to the design of this study may influence the generalizability of findings to routine clinical practice. VEPD were assessed with CT scan (Figure 5) and images were read by an imaging core laboratory. Therefore, it is unclear how these findings may relate to those observed in routine clinical practice where x-rays and MRI but not CT scan are imaging modalities of choice, especially in the postoperative patient. This was a randomized controlled trial with a large sample size in which clinical outcomes and CT findings over 3 years follow-up were reviewed by independent assessors. In this respect, the conclusions derived from this trial are robust and important potential risks of bias are minimized. Nonetheless, several important limitations of this study deserve additional clarification. First, while use of CT follow-up with imaging core laboratory assessments is a study strength, there is a chance of over-detection since no minimum VEPD size was specified. That is, VEPD observed on CT were included in this analysis, regardless of size or whether they were also detected on MRI. Second, results of this study are only generalizable to patients undergoing limited lumbar discectomy with a large post-surgical annular defect, which comprises about 30% of discectomy cases.11 Third, VEPD is a general term that may include erosions, Schmorl’s nodes, Modic changes types 1 and 2, fractures, or calcification,1 but we did not categorize VEPD type in this study. Fourth, we were unable to differentiate between VEPD occurring due to surgery versus those occurring postoperatively since the first postoperative CT scan was performed at the 1-year follow-up visit. Finally, given the relatively young age of this patient population, it will be important to continue with routine patient follow-up and to report these same associations of imaging and clinical findings once final 5-year data from this trial become available.
Conclusion
VEPD had no association with patient-reported outcomes (leg pain, back pain, ODI) at 3 years after lumbar discectomy. VEPD increased the risk of recurrence in patients treated with lumbar discectomy only, but had no negative influence in patients treated with the ACD.
Abbreviations
ACD, annular closure device; CT, computed tomography; MCS, Mental Component Summary; MRI, magnetic resonance imaging; ODI, Oswestry Disability Index, PCS, Physical Component Summary; VEPD, vertebral endplate disruption.
Data Sharing Statement
All authors have agreed to the submission and publication of this manuscript. The authors confirm that requests for data underlying the findings described in this manuscript may be made to the corresponding author starting 1 year following publication of this article.
Acknowledgments
We thank Greg Maislin, PhD of Biomedical Statistical Consulting for assistance with data analysis.
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
- Dr. Kuršumović reports personal fees from Intrinsic Therapeutics Inc. during and outside the submitted work.
- Dr. Bouma reports grants from Intrinsic Therapeutics Inc. during the conduct of the study.
- Dr. Miller reports personal fees from Intrinsic Therapeutics Inc. during the conduct of the study.
- Dr. Assaker reports personal fees from Medtronic and DePuy Synthes outside the submitted work.
- Dr. Hes reports personal fees from consultant for Intrinsic Therapeutics Inc. outside the submitted work.
- Drs. Van de Kleft and Kienzler have nothing to disclose and received no compensation, monetary or otherwise, from the study sponsor.
- The authors report no other conflicts of interest in this work.
References
1. Wang Y, Videman T, Battie MC. Lumbar vertebral endplate lesions: prevalence, classification, and association with age. Spine (Phila Pa 1976). 2012;37(17):1432–1439. doi:10.1097/BRS.0b013e31824dd20a
2. Sahoo MM, Mahapatra SK, Kaur S, et al. Significance of vertebral endplate failure in symptomatic lumbar disc herniation. Global Spine J. 2017;7:230–238. doi:10.1177/2192568217694142
3. Rajasekaran S, Bajaj N, Tubaki V, et al. ISSLS prize winner: the anatomy of failure in lumbar disc herniation: an in vivo, multimodal, prospective study of 181 subjects. Spine (Phila Pa 1976). 2013;38:1491–1500. doi:10.1097/BRS.0b013e31829a6fa6
4. Weiner BK, Vilendecic M, Ledic D, et al. Endplate changes following discectomy: natural history and associations between imaging and clinical data. Eur Spine J. 2015;24(11):2449–2457. doi:10.1007/s00586-014-3734-8
5. Rahme R, Moussa R, Bou-Nassif R, et al. What happens to Modic changes following lumbar discectomy? Analysis of a cohort of 41 patients with a 3- to 5-year follow-up period. J Neurosurg Spine. 2010;13(5):562–567. doi:10.3171/2010.5.SPINE09818
6. Barth M, Diepers M, Weiss C, et al. Two-year outcome after lumbar microdiscectomy versus microscopic sequestrectomy: part 2: radiographic evaluation and correlation with clinical outcome. Spine (Phila Pa 1976). 2008;33(3):273–279. doi:10.1097/BRS.0b013e31816201a6
7. Kienzler JC, Klassen PD, Miller LE, et al. Three-year results from a randomized trial of lumbar discectomy with annulus fibrosus occlusion in patients at high risk for reherniation. Acta Neurochir (Wien). 2019;161(7):1389–1396. doi:10.1007/s00701-019-03948-8
8. Kursumovic A, Kienzler JC, Bouma GJ, et al. Morphology and clinical relevance of vertebral endplate changes following limited lumbar discectomy with or without bone-anchored annular closure. Spine (Phila Pa 1976). 2018;43:1386–1394.
9. Stromqvist B, Fritzell P, Hagg O, et al. The Swedish spine register: development, design and utility. Eur Spine J. 2009;18(Suppl 3):294–304. doi:10.1007/s00586-009-1043-4
10. Collins SL, Moore AR, McQuay HJ. The visual analogue pain intensity scale: what is moderate pain in millimetres? Pain. 1997;72(1):95–97. doi:10.1016/S0304-3959(97)00005-5
11. Miller LE, McGirt MJ, Garfin SR, et al. Association of annular defect width after lumbar discectomy with risk of symptom recurrence and reoperation: systematic review and meta-analysis of comparative studies. Spine (Phila Pa 1976). 2018;43(5):E308–E315. doi:10.1097/BRS.0000000000002501
12. Oetgen ME, Yue JJ, la Torre JJ, et al. Does vertebral endplate morphology influence outcomes in lumbar total disc arthroplasty? Part II: clinical and radiographic results as evaluated utilizing the vertebral endplate Yue-Bertagnoli (VEYBR) classification. SAS J. 2008;2:101–106. doi:10.1016/S1935-9810(08)70025-2
13. Kyere KA, Than KD, Wang AC, et al. Schmorl’s nodes. Eur Spine J. 2012;21(11):2115–2121. doi:10.1007/s00586-012-2325-9
14. Barth M, Fontana J, Thome C, et al. Occurrence of discal and non-discal changes after sequestrectomy alone versus sequestrectomy and implantation of an annulus closure device. J Clin Neurosci. 2016;34:288–293. doi:10.1016/j.jocn.2016.09.013
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.