Back to Journals » Journal of Inflammation Research » Volume 14
Clinical Implications of Intestinal Barrier Damage in Psoriasis
Authors Sikora M ![]() , Stec A
, Stec A ![]() , Chrabaszcz M
, Chrabaszcz M ![]() , Giebultowicz J, Samborowska E, Jazwiec R
, Giebultowicz J, Samborowska E, Jazwiec R ![]() , Dadlez M, Olszewska M, Rudnicka L
, Dadlez M, Olszewska M, Rudnicka L
Received 18 November 2020
Accepted for publication 24 December 2020
Published 27 January 2021 Volume 2021:14 Pages 237—243
DOI https://doi.org/10.2147/JIR.S292544
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ning Quan
Mariusz Sikora,1 Albert Stec,1 Magdalena Chrabaszcz,1 Joanna Giebultowicz,2 Emilia Samborowska,3 Radoslaw Jazwiec,3 Michal Dadlez,3,4 Malgorzata Olszewska,1 Lidia Rudnicka1
1Department of Dermatology, Medical University of Warsaw, Warsaw, Poland; 2Department of Bioanalysis and Drug Analysis, Faculty of Pharmacy with the Laboratory Medicine Division, Medical University of Warsaw, Warsaw, Poland; 3Mass Spectrometry Laboratory, Institute of Biochemistry and Biophysics, Polish Academy of Sciences, Warsaw, Poland; 4Institute of Genetics and Biotechnology, Biology Department, Warsaw University, Warsaw, Poland
Correspondence: Mariusz Sikora
Department of Dermatology, Medical University of Warsaw, Koszykowa 82A, Warsaw 02-008, Poland
Tel +48 22 502 13 24
Fax +48 22 502 21 06
Email [email protected]
Background: An increasing amount of evidence suggests an association between increased intestinal permeability and the pathogenesis of chronic inflammatory diseases. However, the clinical significance of gut barrier dysfunction in psoriasis remains to be established.
Objective: To evaluate whether there are differences in disease activity, the severity of gastrointestinal symptoms and the blood concentration of bacterial metabolites in psoriatic patients with a normal and altered intestinal barrier.
Patients and Methods: Gut barrier integrity was assessed with the serum concentrations of claudin-3, a modulator of intestinal tight junctions and an intestinal fatty acid-binding protein, a marker of enterocyte damage. Gastrointestinal symptoms were evaluated with a validated questionnaire. The concentration of trimethylamine N-oxide (TMAO), a gut microbiota-associated metabolite, was measured with high-performance liquid chromatography.
Results: One hundred and fourteen patients with psoriasis were finally enrolled in the study – 68 with an altered gut barrier and 46 with a properly functioning intestinal barrier. Patients with an altered gut barrier showed a significantly higher score in the Gastrointestinal Symptom Rating Scale (3.20 vs 1.46, p< 0.001). Moreover, patients with psoriasis and a disrupted intestinal barrier demonstrated a higher disease activity (PASI: 19.7 vs 10.3, p< 0.001) and systemic inflammatory parameters (neutrophil-to-lymphocyte ratio: 2.86 vs 1.71, p< 0.001; C-reactive protein 3.76 vs 1.92; p< 0.05). The marker of bacterial translocation was significantly higher in psoriatic patients with damaged gut integrity (TMAO: 375.7± 51.9 vs 119.4± 27.5 ng/mL; p< 0.05).
Conclusion: The altered gut barrier in psoriasis is associated with gastrointestinal symptoms, systemic inflammatory profile and the increased blood concentration of gut microbiota-derived metabolite – TMAO. Intestinal barrier modulation represents a new promising therapeutic approach.
Keywords: gut barrier, microbiome, psoriasis, systemic sclerosis, TMAO
Introduction
Psoriasis is a chronic, immune-mediated disease that affects the skin and exerts multiple systemic effects.1 Increasing evidence suggests that disturbances within the gut microbial composition, their metabolic products and intestinal permeability may exacerbate pathophysiologic pathways in a number of inflammatory disorders.2,3 An altered gut barrier allows the translocation of luminal contents into the underlying tissues and then into the circulation.4 An increased passage of bacterial components (lipopolysaccharide, DNA, toxins), which are potent pro-inflammatory triggers, results in local and (or) systemic immune response with potential clinical implications.5 Emerging data indicated that such a chronic low-grade inflammation was the hallmark of psoriasis and its related comorbidities, such as obesity, insulin resistance, atherosclerosis and nonalcoholic fatty liver disease.6,7
Gut microbiota profiling in psoriasis confirmed significant alterations in its biodiversity and composition.8 Several studies assessed the intestinal barrier in psoriasis by measuring the plasma concentrations of intestinal fatty acid-binding protein (a marker of enterocyte damage),9,10 zonulin (a protein that specifically and reversibly regulates intestinal permeability),11,12 claudin-3 (a component of tight junctions),13,14 lipopolysaccharide (a bacterial endotoxin)12,15 and bacterial DNA.16,17 However, the results were heterogeneous, with some patients presenting significantly affected intestinal integrity, and others showing a properly functioning gut barrier.
Therefore, the aim of our study was to compare those two groups of patients in order to establish the clinical significance of altered intestinal barrier in psoriasis. The specific objectives were as follows: (a) to determine whether there are differences in disease activity between psoriatic patients with a normal and damaged intestinal barrier, (b) to investigate the presence and severity of gastrointestinal symptoms among psoriatic patients with altered gut integrity, (c) to evaluate the blood concentration of trimethylamine N-oxide (TMAO), a gut microbiota-associated metabolite, depending on the function of the intestinal barrier.
Patients and Methods
Study Participants
A prospective cohort of 120 patients between 18 and 60 years of age, with chronic plaque psoriasis qualified for systemic treatment or phototherapy, was enrolled in the study between January 2018 and December 2018 in a tertiary referral dermatological center (Department of Dermatology, Medical University of Warsaw, Warsaw, Poland). The exclusion criteria were as follows:
- Acute infection during the last 3 months prior to the study.
- The intake of the potential modifiers of gut microbiota or intestinal integrity (prebiotics, probiotics, antibiotics, antifungal agents, proton pump inhibitors) within the previous 3 months.
- Medications altering gastrointestinal passage (laxative or antidiarrheal drugs) within 3 months before the enrollment.
- Dietary restrictions during the last 3 months.
- A history of chronic gastrointestinal diseases (inflammatory bowel diseases, irritable bowel syndrome, celiac disease, diverticular disease, food allergies).
- A history of gastrointestinal surgery during the last 6 months.
- Phototherapy, biologics or conventional systemic therapies for psoriasis during the last 3 months.
- Acute or chronic liver, renal and cardiovascular disorders.
- A history of malignancy.
- Drug or alcohol abuse.
- Pregnancy and the period of breastfeeding.
Clinical and Laboratory Procedures
The patients underwent a thorough physical examination, whole blood count, C-reactive protein, lipid profile, liver and kidney function tests. Disease severity was determined with the Psoriasis Area Severity Index score (PASI).
Endoscopy, abdominal ultrasonography, computed tomography, serum or fecal analyses were performed to exclude an organic disease of the gastrointestinal tract, if indicated.
The blood was collected once after an overnight fast. Blood samples were centrifuged at 4000 rpm (1500 g) for 10 min. within 15 min. of sample collection. The plasma was subsequently collected and frozen at −80°C to be analyzed later.
Intestinal Barrier Assessment
Gastrointestinal barrier integrity was assessed via the measurement of serum claudin-3 (CLDN-3) and intestinal fatty acid-binding protein (I-FABP) with the use of the commercially available enzyme-linked immunosorbent assay (ELISA) kits (EIAab, Wuhan, China).9,13,14
Based on biomarker concentrations, the participants were allocated into two groups: with disrupted gut barrier and with properly functioning gut barrier. Patients were considered as having a disrupted gut barrier with CLDN-3 over 49.4 ng/mL and I-FABP over 412.3 pg/mL. Those cut-off values were defined as the mean concentration with two standard deviations and based on the results of healthy subjects published in our previous studies.9,13,14
Evaluation of TMAO Concentration
The blood plasma concentration of TMAO was determined using liquid chromatography coupled with triple-quadrupole mass spectrometry as previously described.36,37 The limit of quantification for TMAO was 20.3 ng/mL.
Gastrointestinal Symptoms
The occurrence of gastrointestinal symptoms was evaluated with the Gastrointestinal Symptoms Rating Scale (GSRS). The GSRS is a reliable and validated questionnaire that utilizes a seven-level Likert scale (1–7), depending on the intensity and frequency of symptoms experienced during the previous week. A higher score mainly indicates inconvenient symptoms. Sixteen questions are clustered into five domains: reflux, abdominal pain, indigestion, diarrhea and constipation.38,39 Patients completed the questionnaire before blood collection. By the time of the final analysis, the results of the questionnaire were blinded to the investigators.
Ethical Statement
All participants gave their written informed consent before entering the study. The study was conducted in accordance with the Helsinki declaration and the protocol approved by the institutional Ethics Committee (Medical University of Warsaw, Warsaw, Poland).
Statistical Analysis
The Shapiro–Wilk test was used to assess the normality of distribution. The categorical variables were summarized as frequencies and percentages and were compared with a chi-square test. The continuous variables were not normally distributed. Therefore, they were presented as medians with interquartile ranges (IQR). The non-parametric Mann–Whitney U-test was applied to compare differences between the groups.
All statistical analyses were performed with STATISTICA 13 software (StatSoft, Inc., USA). The p value of <0.05 was considered statistically significant.
Results
One hundred and fifty patients with psoriasis were initially included in the study. According to the exclusion criteria, 24 of them could not participate. The subjects were 20- to 60-year-old (mean age 43.7±14.1) men (n=87) and women (n=39) with the disease duration ranging from 6 months to 35 years. Based on the adopted cut-off values for CLDN-3 and I-FABP, 68 patients were qualified to the group with an altered intestinal barrier and 46 to the group with a properly functioning intestinal barrier. Twelve patients were excluded due to the inconsistent result of gut integrity biomarkers.
Table 1 describes the anthropometric characteristics, clinical data and laboratory findings of the psoriatic patients with an altered and normal gut barrier. No statistically significant differences were observed for age, sex and BMI between the groups. The patients with psoriasis and an altered intestinal barrier demonstrated a higher disease activity assessed with the PASI score (19.7 [16.7–21.1] vs 10.3 [6.3–12.7]; p<0.001). Compared to the patients with a normal barrier, those with altered intestinal integrity also had the higher values of systemic inflammation biomarkers, i.e. neutrophil-to-lymphocyte ratio; NLR (2.86 [2.20–4.42] vs 1.71 [1.47–2.04]; p<0.001) and C-reactive protein (CRP) concentration (3.76 [2.35–5.67] vs 1.92 [0.70–3.60]; p<0.05).
 |
Table 1 Anthropometric, Clinical and Laboratory Data of Patients with Psoriasis According to Normal and Altered Intestinal Barrier |
As for the GSRS scores, both groups showed significant differences (Table 2). The patients with an altered gut barrier had a higher total score in the GSRS (3.20 [2.53–3.67] vs 1.46 [1.07–1.67]; p<0.001), as well as in individual values for particular sections.
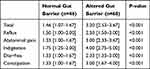 |
Table 2 Gastrointestinal Symptom Rating Scale (GSRS) in Patients with Psoriasis According to Normal and Altered Intestinal Barrier |
Figure 1 presents TMAO concentration in the serum of psoriatic patients with a normal and altered gut barrier. The latter group had a significantly higher circulating level of this bacterial metabolite (375.7±51.9 vs 119.4±27.5 ng/mL; p<0.05).
 |
Figure 1 Plasma concentration of trimethylamine N-Oxide (TMAO) in psoriatic patients with normal and altered gut barrier (*p<0.05). |
Discussion
Several studies previously revealed increased intestinal permeability in patients with psoriasis.12,14 However, the clinical significance of this phenomenon still remains unclear. To our knowledge, it is the first study that evaluates disease activity, self-reported gastrointestinal symptoms and TMAO concentration in patients with psoriasis according to the presence of a normal or altered gut barrier.
We found that psoriatic patients with an altered gut barrier experienced gastrointestinal symptoms much more frequently and intensely. Their results obtained in the GSRS were significantly higher in the total score as well as for individual symptom domains. Gastrointestinal symptoms are quite common among patients with psoriasis. Feldman et al conducted an online-based survey and found that several gastrointestinal signs and symptoms (pain, abdominal bloating, diarrhea, mucus in stool, blood in stool, and unintentional weight loss) were more prevalent in the respondents with moderate-to-severe psoriasis.18 However, literature sources regarding the relationship between gastrointestinal symptoms and increased gut permeability are scarce. So far, gastrointestinal symptoms have been found to be associated with the biomarkers of a disrupted gut barrier in the elderly,19 after intensive exercise20 and in patients with irritable bowel syndrome.21 Impaired gut integrity may facilitate the translocation of luminal content to the inner layers of the intestinal wall and lead to a local immune response. The low-grade inflammation in the intestinal mucosa alters gastrointestinal reflexes and activates the visceral sensory system, subsequently promoting the occurrence of gastrointestinal symptoms.22 Patients with psoriasis and psoriatic arthritis presented the signs of subclinical gut inflammation reflected by an increased fecal calprotectin concentration, without the clinical and endoscopic features of inflammatory bowel diseases.23
A higher disease activity and systemic inflammation are other characteristic features of an altered intestinal barrier in patients with psoriasis. It is unknown whether increased gut permeability is an early event in the pathogenesis of psoriasis or the consequence of the disease. Previous findings indicated a correlation between intestinal integrity biomarkers and the PASI score or an inflammatory state, reflected by an increased CRP concentration and neutrophil-to-lymphocyte ratio.9,13 The loss of barrier function allows the translocation of bacterial antigens into the circulation, where they may contribute to immune activation and act as a trigger of psoriasis exacerbation. The increased blood concentration of lipopolysaccharide, an endotoxin derived from Gram-negative bacterial cell walls and a strong proinflammatory molecule, was confirmed in psoriatic patients.12,15 Cell-free bacterial DNA was also identified in the blood of patients with psoriasis.16,17 Nucleotide sequencing indicates that the detected DNA fragments correspond to the microbiota commonly found in the gastrointestinal tract.
Not only antigens but also bacterial metabolites translocating to the systemic circulation might be crucial in the interactions between gut microbiota, a compromised intestinal barrier, gastrointestinal symptoms and psoriasis activity. Trimethylamine N-oxide is one of the most intensively studied gut-microbiome-derived metabolite in the recent years.24 Dietary L-carnitine, choline and lecithin are metabolized by the intestinal microbiome to trimethylamine (TMA), which is subsequently absorbed and converted into TMAO in the liver by flavin-containing monooxygenase 3.25 An increased circulating TMAO concentration was demonstrated to activate pro-inflammatory signaling pathways and positively correlate with cardiovascular risks.26,27 A high TMAO concentration was linked to several psoriasis comorbidities, i.e. obesity,28 hypertension,29 metabolic syndrome,30 nonalcoholic fatty liver disease31 and insulin resistance.32 Before our study was conducted, TMAO levels had not been assessed in psoriasis, although its concentration correlates significantly with skin and joint involvement in patients with psoriatic arthritis.33 According to some authors, it was not TMAO itself, but rather its precursor – TMA which was responsible for the observed disorders and TMAO as the product of oxygenation was a biomarker of the increased intestinal translocation of TMA.34,35
We hypothesize that TMAO or TMA translocation in the presence of an altered intestinal barrier is a potential mechanism linking gut dysbiosis to the immune activation, influence on disease activity and/or the development of psoriasis comorbidities. The modulation of the gut microbiota, the reinforcement of the intestinal barrier or direct TMAO inactivation may thus become an important target in the prevention and treatment of psoriasis.
Conclusion
In the present paper, we demonstrated that an altered gut barrier in psoriasis is associated with gastrointestinal symptoms, systemic inflammatory profile and the increased blood concentration of gut microbiota-derived metabolite – TMAO. The findings provide an important basis for future studies to investigate how to optimally modify the gut microbiome and intestinal barrier in order to beneficially influence psoriasis with its comorbidities.
Abbreviations
TMAO, trimethylamine N-oxide; PASI, Psoriasis Area Severity Index score; CLDN-3, claudin-3; I-FABP, intestinal fatty acid-binding protein; ELISA, enzyme-linked immunosorbent assay; GSRS, Gastrointestinal Symptoms Rating Scale; IQR, interquartile ranges; BMI, Body Mass Index; NLR, neutrophil-to-lymphocyte ratio; CRP, C-reactive protein; TMA, trimethylamine.
Data Sharing Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
The authors thank Prof. Robert Gniadecki for his critical comments on the manuscript. The authors would also like to thank AstraZeneca for permission to use Polish adaptation of the Gastrointestinal Symptom Rating Scale.
Funding
This research was funded by the Polish Ministry of Science and Higher Education, grant number MNiSW/2020/220/DIR/NN4.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182(4):840–848. doi:10.1111/bjd.18245
2. Tong Y, Marion T, Schett G, Luo Y, Liu Y. Microbiota and metabolites in rheumatic diseases. Autoimmun Rev. 2020;19(8):102530. doi:10.1016/j.autrev.2020.102530
3. Tajik N, Frech M, Schulz O, et al. Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis. Nat Commun. 2020;11(1):1995. doi:10.1038/s41467-020-15831-7
4. Camilleri M. Leaky gut: mechanisms, measurement and clinical implications in humans. Gut. 2019;68(8):1516–1526. doi:10.1136/gutjnl-2019-318427
5. Gomez-Hurtado I, Gallego-Duran R, Zapater P, et al. Bacterial antigen translocation and age as BMI-independent contributing factors on systemic inflammation in NAFLD patients. Liver Int. 2020. doi:10.1111/liv.14571
6. Tilg H, Zmora N, Adolph TE, Elinav E. The intestinal microbiota fuelling metabolic inflammation. Nat Rev Immunol. 2020;20(1):40–54. doi:10.1038/s41577-019-0198-4
7. Prussick RB, Miele L. Nonalcoholic fatty liver disease in patients with psoriasis: a consequence of systemic inflammatory burden? Br J Dermatol. 2018;179(1):16–29. doi:10.1111/bjd.16239
8. Sikora M, Stec A, Chrabaszcz M, et al. Gut microbiome in psoriasis: an updated review. Pathogens. 2020;9(6):463. doi:10.3390/pathogens9060463
9. Sikora M, Stec A, Chrabaszcz M, et al. Intestinal fatty acid binding protein, a biomarker of intestinal barrier, is associated with severity of psoriasis. J Clin Med. 2019;8(7). doi:10.3390/jcm8071021
10. Stehlikova Z, Kostovcik M, Kostovcikova K, et al. Dysbiosis of skin microbiota in psoriatic patients: co-occurrence of fungal and bacterial communities. Front Microbiol. 2019;10:438. doi:10.3389/fmicb.2019.00438
11. Gerdes S, Osadtschy S, Buhles N, Mrowietz U. Zonulin may not be a marker of autoimmunity in patients with psoriasis. Acta Derm Venereol. 2012;92(2):171–172. doi:10.2340/00015555-1208
12. Richetta AG, Grassi S, Moliterni E, et al. Increased intestinal barrier permeability in patients with moderate to severe plaque-type psoriasis. J Dermatol. 2020;47(10). doi:10.1111/1346-8138.15361
13. Sikora M, Chrabaszcz M, Waskiel-Burnat A, Rakowska A, Olszewska M, Rudnicka L. Claudin-3 - a new intestinal integrity marker in patients with psoriasis: association with disease severity. J Eur Acad Dermatol Venereol. 2019;33(10):1907–1912. doi:10.1111/jdv.15700
14. Sikora M, Chrabaszcz M, Maciejewski C, et al. Intestinal barrier integrity in patients with plaque psoriasis. J Dermatol. 2018;45(12):1468–1470. doi:10.1111/1346-8138.14647
15. Romani J, Caixas A, Escote X, et al. Lipopolysaccharide-binding protein is increased in patients with psoriasis with metabolic syndrome, and correlates with C-reactive protein. Clin Exp Dermatol. 2013;38(1):81–84. doi:10.1111/ced.12007
16. Codoner FM, Ramirez-Bosca A, Climent E, et al. Gut microbial composition in patients with psoriasis. Sci Rep. 2018;8(1):3812. doi:10.1038/s41598-018-22125-y
17. Ramirez-Bosca A, Navarro-Lopez V, Martinez-Andres A, et al. Identification of bacterial DNA in the peripheral blood of patients with active psoriasis. JAMA Dermatol. 2015;151(6):670–671. doi:10.1001/jamadermatol.2014.5585
18. Feldman SR, Srivastava B, Abell J, et al. Gastrointestinal signs and symptoms related to inflammatory bowel disease in patients with moderate-to-severe psoriasis. J Drugs Dermatol. 2018;17(12):1298–1308.
19. Ganda Mall JP, Ostlund-Lagerstrom L, Lindqvist CM, et al. Are self-reported gastrointestinal symptoms among older adults associated with increased intestinal permeability and psychological distress? BMC Geriatr. 2018;18(1):75. doi:10.1186/s12877-018-0767-6
20. Karhu E, Forsgard RA, Alanko L, et al. Exercise and gastrointestinal symptoms: running-induced changes in intestinal permeability and markers of gastrointestinal function in asymptomatic and symptomatic runners. Eur J Appl Physiol. 2017;117(12):2519–2526. doi:10.1007/s00421-017-3739-1
21. Linsalata M, Riezzo G, D’Attoma B, Clemente C, Orlando A, Russo F. Noninvasive biomarkers of gut barrier function identify two subtypes of patients suffering from diarrhoea predominant-IBS: a case-control study. BMC Gastroenterol. 2018;18(1):167. doi:10.1186/s12876-018-0888-6
22. Akiho H, Ihara E, Nakamura K. Low-grade inflammation plays a pivotal role in gastrointestinal dysfunction in irritable bowel syndrome. World J Gastrointest Pathophysiol. 2010;1(3):97–105. doi:10.4291/wjgp.v1.i3.97
23. Adarsh MB, Dogra S, Vaiphei K, Vaishnavi C, Sinha SK, Sharma A. Evaluation of subclinical gut inflammation using faecal calprotectin levels and colonic mucosal biopsy in patients with psoriasis and psoriatic arthritis. Br J Dermatol. 2019;181(2):401–402. doi:10.1111/bjd.17745
24. Nowinski A, Ufnal M. Trimethylamine N-oxide: a harmful, protective or diagnostic marker in lifestyle diseases? Nutrition. 2018;46:7–12. doi:10.1016/j.nut.2017.08.001
25. Ufnal M, Zadlo A, Ostaszewski R. TMAO: a small molecule of great expectations. Nutrition. 2015;31(11–12):1317–1323. doi:10.1016/j.nut.2015.05.006
26. Farhangi MA, Vajdi M. Novel findings of the association between gut microbiota-derived metabolite trimethylamine N-oxide and inflammation: results from a systematic review and dose-response meta-analysis. Crit Rev Food Sci Nutr. 2020;60(16):2801–2823. doi:10.1080/10408398.2020.1770199
27. Yang S, Li X, Yang F, et al. Gut microbiota-dependent marker TMAO in promoting cardiovascular disease: inflammation mechanism, clinical prognostic, and potential as a therapeutic target. Front Pharmacol. 2019;10:1360. doi:10.3389/fphar.2019.01360
28. Dehghan P, Farhangi MA, Nikniaz L, Nikniaz Z, Asghari-Jafarabadi M. Gut microbiota-derived metabolite trimethylamine N-oxide (TMAO) potentially increases the risk of obesity in adults: an exploratory systematic review and dose-response meta- analysis. Obes Rev. 2020;21(5):e12993. doi:10.1111/obr.12993
29. Ge X, Zheng L, Zhuang R, et al. The gut microbial metabolite trimethylamine N-oxide and hypertension risk: a systematic review and dose-response meta-analysis. Adv Nutr. 2020;11(1):66–76. doi:10.1093/advances/nmz064
30. Papandreou C, More M, Bellamine A. Trimethylamine N-oxide in relation to cardiometabolic health—cause or effect? Nutrients. 2020;12(5):1330. doi:10.3390/nu12051330
31. Leon-Mimila P, Villamil-Ramirez H, Li XS, et al. Trimethylamine N-oxide levels are associated with NASH in obese subjects with type 2 diabetes. Diabetes Metab. 2020. doi:10.1016/j.diabet.2020.07.010
32. Roy S, Yuzefpolskaya M, Nandakumar R, Colombo PC, Demmer RT, Hu C. Plasma trimethylamine-N-oxide and impaired glucose regulation: results from the Oral Infections, Glucose Intolerance and Insulin Resistance Study (ORIGINS). PLoS One. 2020;15(1):e0227482. doi:10.1371/journal.pone.0227482
33. Coras R, Kavanaugh A, Boyd T, et al. Choline metabolite, trimethylamine N-oxide (TMAO), is associated with inflammation in psoriatic arthritis. Clin Exp Rheumatol. 2019;37(3):481–484.
34. Jaworska K, Bielinska K, Gawrys-Kopczynska M, Ufnal M. TMA (trimethylamine), but not its oxide TMAO (trimethylamine-oxide), exerts haemodynamic effects: implications for interpretation of cardiovascular actions of gut microbiome. Cardiovasc Res. 2019;115(14):1948–1949. doi:10.1093/cvr/cvz231
35. Jaworska K, Hering D, Mosieniak G, et al. TMA, A forgotten uremic toxin, but not TMAO, is involved in cardiovascular pathology. Toxins (Basel). 2019;11(9):490. doi:10.3390/toxins11090490
36. Ufnal M, Jazwiec R, Dadlez M, Drapala A, Sikora M, Skrzypecki J. Trimethylamine-N-oxide: a carnitine-derived metabolite that prolongs the hypertensive effect of angiotensin II in rats. Can J Cardiol. 2014;30(12):1700–1705. doi:10.1016/j.cjca.2014.09.010
37. Wolyniec W, Kasprowicz K, Giebultowicz G, et al. Changes in water soluble uremic toxins and urinary acute kidney injury biomarkers after 10- and 100-km runs. Int J Environ Res Public Health. 2019;16(21):4153. doi:10.3390/ijerph16214153
38. Kulich KR, Regula J, Stasiewicz J, et al. Psychometric validation of the polish translation of the Gastrointestinal Symptom Rating Scale (GSRS) and Quality of Life in Reflux and Dyspepsia (QOLRAD) questionnaire in patients with reflux disease. Pol Arch Med Wewn. 2005;113(3):241–249.
39. Kulich KR, Madisch A, Pacini F, et al. Reliability and validity of the Gastrointestinal Symptom Rating Scale (GSRS) and Quality of Life in Reflux and Dyspepsia (QOLRAD) questionnaire in dyspepsia: a six-country study. Health Qual Life Outcomes. 2008;6(1):12. doi:10.1186/1477-7525-6-12
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
