Back to Journals » OncoTargets and Therapy » Volume 7
Clinical efficacy and safety of imatinib in the management of Ph+ chronic myeloid or acute lymphoblastic leukemia in Chinese patients
Received 3 December 2013
Accepted for publication 16 January 2014
Published 6 March 2014 Volume 2014:7 Pages 395—404
DOI https://doi.org/10.2147/OTT.S38846
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Yu Zhu, Si-Xuan Qian
Department of Hematology, The First Affiliated Hospital of Nanjing Medical University, Jiangsu Province Hospital, Nanjing, Jiangsu Province, People's Republic of China
Abstract: Imatinib mesylate is considered the standard first-line systemic treatment for patients with chronic myeloid leukemia (CML) and functions by targeting BCR-ABL tyrosine kinases. Imatinib has substantially changed the clinical management and improved the prognosis of CML and Philadelphia chromosome-positive acute lymphocytic leukemia (Ph+ ALL). Here, we review the pharmacology, mode of action, and pharmacokinetics of imatinib; Chinese efficacy studies in CML and Ph+ ALL; safety and tolerability; patient-focused perspectives, such as quality of life, patient satisfaction, acceptability, and adherence; and uptake of imatinib.
Keywords: imatinib, Philadelphia chromosome-positive acute lymphocytic leukemia, chronic myeloid leukemia, safety, clinical efficacy
Introduction
The development of imatinib represents one of the most important milestones in the history of medicine. In the 1990s, allogeneic stem cell transplantation (allo-SCT) was the frontline therapy in eligible patients with chronic myeloid leukemia (CML); however, this therapy was accompanied by high mortality, even when using human leukocyte antigen (HLA)-matched donors. Imatinib mesylate, a BCR-ABL tyrosine kinase activity inhibitor (TKI), was the first targeted therapy available for patients with CML, providing major clinical advantages and better quality of life outcomes compared with previous interferon (IFN)-based treatments.1,2 Today, the majority of CML patients can expect to have a generally normal quality of life and much better survival/prognosis, even with the possibility of discontinuing long-term therapy safely.
Imatinib is a selective inhibitor of the BCR-ABL tyrosine kinase and is generally well tolerated by the majority of patients. Other tyrosine kinases inhibited by imatinib include c-KIT and platelet-derived growth factor receptors (PDGFRs).3 The well-known International Randomised Study (IRIS) of Interferon versus Imatinib, which randomly assigned 1,106 patients with newly diagnosed CML to receive 400 mg/day of imatinib or IFN (5,000,000 U/m2/day) plus 10-day cycles of cytarabine (20 mg/m2/day) every month, resulted in a 69% 12-month complete cytogenetic response (CCyR) rate and a 76.2% 18-month CCyR rate for imatinib, versus 14.5% for IFN plus cytarabine (P<0.001)4 in 2003. In 2006,5 after a median follow-up of 60 months in IRIS, the cumulative CCyR rose to 87%. Hochhaus et al6 published 6-year follow-up data and reported no further cases of disease progression. Cumulative CCyR rates were 82%, and 57% of the patients who achieved CCyR remained adherent to imatinib therapy after randomization to the imatinib therapy group, and these numbers were stable at the 7-year follow-up.7 At the 8-year follow-up,8 55% of patients initially randomized to the imatinib were still on study treatment, and an estimated overall survival (OS) rate of 85% was reported.
Apart from CML, imatinib is also a powerful agent for the treatment of Philadelphia chromosome-positive acute lymphocytic leukemia (Ph+ ALL). Single-agent imatinib therapy for relapsed or refractory Ph+ ALL and lymphoid CML cells in Blast Crisis (CML-BC) was associated with response rates of 40%–50%; complete response (CR) rates were 5%–7%, with a median survival of 2–5 months.9–11 These data led to a number of studies of imatinib as a first-line treatment in elderly patients, especially patients who could not tolerate allogeneic stem cell transplantation (allo-SCT). The current consensus is that imatinib, administered along with chemotherapy, improves outcomes compared with chemotherapy alone.12–16 The number of patients who are able to receive allo-SCT, and the outcomes of allo-SCT, have improved as well.13,14,17,18 When imatinib was combined during induction, consolidation, and maintenance phases of therapy, 96% of patients with Ph+ ALL achieved CR. A complete molecular response (CMR) was achieved in 60% of patients. Half of the patients were able to proceed with allo-SCT. Compared with previous chemotherapy regimens without imatinib, the combination did not result in unexpected toxicities, and published follow-up studies reported a 2-year disease-free survival (DFS) of 87% with hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (hyper-CVAD) and imatinib, far superior to historical outcomes with hyper-CVAD alone, with 2-year DFS rates of 28% (P<0.001).19
Pharmacokinetics of imatinib
Studies have evaluated the absolute bioavailability of a single oral dose of imatinib mesylate (Glivec®; Novartis, Basel, Switzerland) 400 mg (capsules versus [vs] oral solution), compared with imatinib 100 mg (intravenous [iv] infusion) in 12 healthy subjects. Both the rate and extent of imatinib absorption, as measured by maximum concentration (Cmax), partial area under the curve (AUC), and total AUC, were similar for the oral solution and the imatinib capsule intended for the market. Absolute bioavailability values for the imatinib capsule and oral solution were 98.3% and 97.2%, respectively;20 this outcome showed that the 400 mg oral dose of imatinib, as a capsule or a solution, was completely absorbed and was almost completely bioavailable (>97%).20 Peak plasma concentrations (Cmax) of imatinib were detected after a mean time of 3.84 and 4.07 hours following oral intake of 400 or 600 mg imatinib, respectively.21
The main elimination pathway is via the biliary–fecal route.22,23 Potential inactivation of imatinib through excessive binding to the plasma protein α1-acid glycoprotein may result in decreased levels of active drug, which consequently may result in the induction of resistance.22
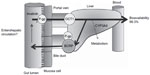
The main circulating metabolite of imatinib after it is metabolized by cytochrome P450 (CYP) 3A4 is N-desmethyl-imatinib (CGP74588), which has in vitro activity similar to that of imatinib, and the systemic exposure represents approximately 10%–15% of that for imatinib.21–23 Imatinib is absorbed and interacts slightly with P-glycoprotein (P-gp) or adenosine triphosphate (ATP)-binding cassette (ABC) transporters at the membranes of intestinal epithelial cells and is then transported to the intestinal lumen.20,21,24 In the liver, imatinib is transported into hepatocytes by organic cation transporter 1 (OCT1), where it may undergo metabolism to CGP74588 by hepatic CYP3A4 and a number of other enzymes, such as CYP1A2, CYP2D6, CYP2C9, and CYP2C19 (Figure 1).25–27 Imatinib is known to be a substrate for CYP3A4, and there is great potential for drug interactions with coadministered drugs, food, and nutritional supplements, potentially leading to subtherapeutic exposure or concentrations associated with greater-than-acceptable toxicity for imatinib.25
The data from Phase I and pharmacokinetic studies28 demonstrate that urinary excretion of imatinib and CGP74588 accounts for less than 10% of the imatinib dose and is not significantly different among the various groups, indicating that imatinib pharmacokinetics will not be significantly affected in patients with compensated renal failure.22
Degradation of imatinib predominantly occurs in a CYP3A4-dependent manner, leading to the formation of CGP74588.25 Both imatinib and CGP74588 are mainly glucuronidated to inactive O- and N-glucuronides23 by UDP-glucuronosyltransferases. These glucuronides are excreted into the bile, where they may be converted back to the parent drug and CGP74588 by bacterial glucuronidases in the gut lumen and reabsorbed through the process of enterohepatic recirculation, which is evidenced by a secondary plasma peak in the concentration–time profile of imatinib. However, the extent of biliary excretion of imatinib glucuronic acid conjugates, which are present in the plasma and urine, but are not detected in the feces, as well as their metabolites, has not yet been reported.23
Nevertheless, imatinib has a restricted ability to penetrate the blood–brain barrier.18,22,29–31 le Coutre et al22 detected imatinib and CGP74588 levels in cerebrospinal fluid (CSF) probes from 17 patients treated with 400 or 600 mg per day imatinib. The mean imatinib concentration in the CSF was 38 ng/mL (39 and 34 ng/mL in the CSF from patients without and with meningeosis, respectively), indicating that only a small fraction of plasmatic imatinib crosses the blood–brain barrier, regardless of the presence of meningeosis leukemia. Moreover, peak levels of CGP74588 were below 10 ng/mL. Imatinib is therefore ineffective at preventing or treating central nervous system leukemia.
Mode of action of imatinib
The BCR-ABL tyrosine kinase is a constitutively active kinase that functions by binding to ATP and transferring a phosphate group from ATP to tyrosine residues on various substrates. This activity causes the excess proliferation of myeloid cells that is characteristic of CML. Imatinib functions by blocking the binding of ATP to the BCR-ABL tyrosine kinase, thus inhibiting the activity of the kinase. In the absence of tyrosine kinase activity, substrates required for BCR-ABL function cannot be phosphorylated.32
Pharmacology of imatinib
Significant correlations between imatinib trough plasma levels (Cmin) and cytogenetic and molecular responses of Caucasian patients with CML have been found.32–34 In a pharmacokinetics/pharmacodynamics (PK/PD) analysis from the IRIS study, imatinib plasma Cmin levels higher than 1,000 ng/mL correlated significantly with improved cytogenetic and molecular response rates.33 In the Tyrosine Kinase Dose Optimization Study (TOPS), evaluation of the effects of imatinib 400 mg twice daily compared with imatinib 400 mg daily in previously untreated patients with newly diagnosed CML cells in Chronic Phase (CML-CP) showed that imatinib trough plasma levels were proportional to dose and stable over time.35 Patients with imatinib Cmin in the lowest quartile showed a lower major molecular response (MMR) at 12 months, whereas patients in the highest Cmin quartile showed a higher frequency of all grades of some adverse events.35
Another data analysis from patients with newly diagnosed untreated CML-CP from both the IRIS and TOPS trials confirmed that the higher steady-state imatinib levels correlated with better complete CCyR and MMR, but also resulted in more grade 3 and 4 treatment-related toxicities.36 In a study by Singh et al, mean plasma levels in nonresponders were shown to be significantly lower than those in responders (413 vs 1,380 ng/mL; P=0.002) in 40 patients with CML (20 responders and 20 nonresponders to imatinib).34
However, the imatinib trough level in Chinese patients has a higher imatinib Cmin than that of Caucasian patients; Li et al37 reported that the Cmin values for imatinib 400 mg and 600 mg daily were 1,325.61 and 1,550.90 ng/mL, respectively, in 46 CML patients. Zhong et al38 confirmed that the plasma imatinib trough concentration in 59 CML patients treated with a dose of 400 mg/day was 1,252 ng/mL. Different from the results above and some other studies,39 molecular responses were not correlated with imatinib Cmin values in the study by Li et al.37 This outcome was consistent with research by Zhou et al,40 who found that the median imatinib Cmin was 1,271 ng/mL in 416 Chinese CML patients, and that no significant difference was observed in terms of imatinib Cmin in patients reaching CCyR. However, Chen et al41 found that the mean Cmin of imatinib was significantly higher in the CCyR group (1,472 ng/mL) than in the non-CCyR group (1,067 ng/mL) after the administration of imatinib for 1 year. These inconsistent results did not confirm the imatinib concentration–response relationship in Chinese patients and demonstrated a major metabolic distinction between Chinese and Caucasian individuals.
For the 400 and 600 mg/day dose regimens, the reported Cmin values were 1,058 and 1,444 ng/mL, respectively, in a study conducted in France.39 In Canada, these same values were 1,065 and 999 ng/mL, respectively.42 These values are clearly lower than the Cmin values achieved in Chinese patients. However, similar to the Chinese patients, Korean patients exhibited a Cmin value of 1,378 ng/mL in the group treated with 400 mg/day imatinib. Based on these results, we speculate that the different imatinib trough plasma levels from Chinese and Caucasian patients could be attributed to body weight. The relatively high Cmin may account for the fact that some Chinese studies have failed to find a correlation between the Cmin and CCyR or MMR, if only because the majority of the population reached the effective imatinib concentration.
Chinese efficacy studies and comparative studies of imatinib
Imatinib (Glivec®) launched in mainland China in 2001, and a large number of CML and Ph+ ALL patients achieved good efficacy with imatinib treatment. The Glivec International Patient Assistance Program (GIPAP) made it possible for thousands of Chinese patients to receive imatinib in this developing country. Hence, we will summarize the relatively large amount of clinical research in imatinib in the past decade in the People’s Republic of China (Table 1 and Table 2).
  | Table 2 The clinical efficacy of imatinib in Chinese Ph+ ALL patients |
In a retrospective study from GIPAP in Ruijin Hospital in Shanghai,43 the relative efficacy and toxicity of imatinib therapy in 151 CML patients were described. In CML-CP, with a median follow-up duration of 21.5 months, complete hematological remission (CHR), major cytogenetic remission (MCyR), CCyR, and CMR were 96.9%, 82.6%, 76.1%, and 29.4%, respectively. According to the Sokal scoring system, the rates of MCyR and CCyR in low-risk patients were significantly higher than those in intermediate-risk and high-risk patients (P=0.0013 and P=0.0024, respectively). The adverse events experienced after imatinib treatment were acceptable. A univariate analysis from 241 CML patients44 suggested that factors such as the course of disease before treatment, hemoglobin count, white blood cell count, CCyR achievement, and MMR achievement influenced imatinib resistance.
With a 6-year follow-up of molecular responses in late CML-CP patients after CCyR with imatinib, Qin et al45 also found that imatinib could induce most late CML-CP patients to achieve long-term durable responses after achieving CCyR. Both the time when CCyR was first achieved and the depth of BCR-ABL reduction after CCyR are relevant to long-term event-free survival (EFS). A recent analysis of 171 patients with CML-CP who received imatinib monotherapy at the People’s Hospital of Peking University in Beijing compared the 7-year response to imatinib monotherapy as initial treatment (n=73) and retreatment (n=98).46 Similar to the results of the IRIS study,47 the expected 7-year progression-free survival (PFS) in patients with CCyR plus MMR in both groups was 100% at 12 months. There was a significant benefit to the use of imatinib as an initial treatment, compared with its use as a second-line retreatment. The 7-year EFS was significantly higher in the initial treatment group than in the retreatment group (92% vs 70%, respectively; P=0.049).
Two studies evaluated the efficacy and safety of imatinib in the treatment of adult patients with CML cells in Accelerated Phase (CML-AP) and CML-BC.48,49 Both studies also showed high hematological response (HR: 93.3%) and CHR (46.7%–85.3%). The estimated 4-year PFS and OS rates were 48.2% and 52.2%, respectively, in patients with HR.49
Imatinib can rescue patients with CML-AP, but almost half of these patients relapse within 4–5 years. In order to clarify the role of allo-hematopoietic (H)SCT in the treatment of CML-AP, a recent study50 was designed to compare the outcomes of imatinib (n=87) versus allo-HSCT treatment (n=45) in CML-AP patients. Both treatments resulted in similar survival rates in low-risk (no factor) patients, with 6-year EFS, OS, and PFS rates of more than 80.0% each. In intermediate-risk patients, EFS and OS did not differ in terms of therapy, with 6-year EFS and OS rates for imatinib (n=43) and allo-HSCT (n=16) of 47.1% versus 61.9% (P=0.788) and 61.3% versus 81.3% (P=0.773), respectively. However, in high-risk patients, imatinib was by far inferior to allo-HSCT, with 5-year EFS, OS, and PFS rates for imatinib (n=27) and allo-HSCT (n=6) of 9.3% versus 66.7% (P=0.030), 17.7% versus 100% (P=0.008), and 18.8% versus 100% (P=0.006), respectively. Therefore, allo-HSCT is a viable option for all patients with CML-AP; it is superior to imatinib, conferring significant survival advantages to high- and intermediate-risk patients.
More recently, a randomized study was conducted to compare dasatinib (100 mg/day) with imatinib (400 mg/day) in 37 CML-CP patients.51 The cumulative CCyR and MMR at 36 months were higher in the dasatinib group than in the imatinib group; however, no significant differences were observed.
Imatinib in combination with chemotherapy can significantly improve the CR rate, molecular CR rate, and CR duration for Ph+ ALL patients.52,53 Therefore, application of imatinib pretransplant might have benefits for patients with Ph+ ALL.54 In a retrospective analysis of 69 patients with Ph+ ALL,54 44 patients received imatinib therapy, including 24 pretransplant, 9 post-transplant, and 11 both pre- and post-transplant. The 3-year estimated OS and DFS were 62.3% and 53.6%, respectively, in patients who received imatinib therapy pretransplant, and 25.9% and 23.6%, respectively, in patients who did not receive imatinib therapy pretransplant. Moreover, additional chromosome abnormalities and blasts with myeloid lineage conferred a survival disadvantage in terms of OS and DFS. Huang et al53 reported that patients who received imatinib combined with intensive chemotherapy pretransplant significantly increased the CR rate and improved OS. The estimated OS and DFS at 3 years were 85.9% and 83.9%, respectively, and the cumulative relapse incidences at 3 years were 16.1% and 14.1%, respectively.53 Chen et al55 retrospectively compared the outcomes of allo-HSCT with and without imatinib in 35 patients. The data showed that the first complete remission (CR1) in the imatinib group was higher than that in the control group (20/23 vs 6/12, P=0.038). The estimated 5-year OS was significantly better with allo-HSCT plus imatinib than for allo-HSCT without imatinib. Importantly, there is no evidence that imatinib has an adverse effect on transplant-related morbidity or mortality.
Safety and tolerability of imatinib
Generally, imatinib is well tolerated overall, though mild-to-moderate toxicities are common in both CML and Ph+ ALL patients, with almost all patients experiencing at least one mild or moderate treatment-related adverse event (grade 1–2).2,8 The toxicity of imatinib is closely associated with the disease phase and drug dose. Obviously, patients with advanced-phase disease,56,57 receiving higher doses,58,59 suffered from more frequent and severe toxicities. Patients with advanced disease had more usual dose interruptions and reductions because of the higher dose of imatinib administration.58,59 Successful treatment in these patients relies on prompt and effective management of adverse events.
In the IRIS trial, a large percentage of CML-CP patients experienced grade 3 or 4 hematological toxicity from the administration of imatinib 400 mg.2 Despite high rates of neutropenia (14.3%) and thrombocytopenia (7.8%), grade 3–4 anemia was observed in only 3% of patients.2 Respective infectious and bleeding complications remained rare. Zhou et al43 reported that grade 3–4 neutropenia, anemia, and thrombocytopenia occurred in 17.8%, 5.9%, and 21.8% of Chinese patients with CML-CP, respectively. Grade 3–4 hematological toxicity presented in 40%–60% of patients with advanced disease.
As expected, higher doses of imatinib (600–800 mg/day) in CML-CP patients resulted in higher rates of grades 3–4 hematologic adverse events in several single-armed studies60 and in the Phase III study (TOPS) that compared imatinib 400 mg versus 800 mg in untreated CML-CP.59 Moreover, myelotoxicity is generally an early event, more commonly observed during the first 12 months of treatment with imatinib.61 In a Phase II single-arm trial of imatinib in CML-CP after failure of IFN after 12 months of treatment, neutropenia decreased from 33% to 13%, thrombocytopenia decreased from 18% to 1.9%, and anemia decreased from 6% to 1.4%.61 More frequent grade 3–4 hematological toxicity has also been observed in CML-AP patients compared with CML-CP patients; grade 3–4 neutropenia was observed in 60% of CML-CP patients receiving 600 mg once daily, while severe anemia was observed in 47% and thrombocytopenia in 43% of CML-AP patients.57 Similarly, in CML-BC, grade 3–4 neutropenia, thrombocytopenia, and anemia were observed in 64%, 62%, and 52% of patients, respectively. CML-BC patients undergoing imatinib treatment experienced more frequent fever than CML-CP and CML-AP patients.56 However, it is suggested not to withdraw treatment or reduce the dose of imatinib, and continuation of imatinib despite myelotoxicity may be appropriate in some patients.62 No hematological toxicity has been reported in Chinese patients receiving higher doses of imatinib.
Gastrointestinal toxicity (GI), presenting either as nausea and vomiting or diarrhea, is among the most common side effects of imatinib in CML patients. In CML, nausea is the most common adverse event, occurring in 43%–65% of patients taking 400–600 mg/day imatinib.63 However, grade 3–4 nausea occurs in less than 1% of patients.2,59 Diarrhea is also common, occurring in 45% of imatinib-treated patients, while severe diarrhea occurs in between 1.8% and 2.5% of patients.63 Fluid retention is the most typical nonhematological adverse reaction to imatinib in patients with CML and is the main reason for dose reductions in patients taking high-dose imatinib.64 The majority of patients have nonspecific edema,5,65 which is usually grade 1–2, dose dependent, and present in the periorbital regions and ankles. The imatinib-induced fluid retention syndrome is a characteristic side effect. Skin toxicity, mostly as a rash or dermatitis, is frequently reported32 and typically mild to moderate, usually being observed shortly after beginning treatment with imatinib. All grades of rash have been reported; with an incidence of 40% in CML patients.2,59 High-grade reactions are rare, with an incidence of 2%–3% with 400 mg imatinib and 5%–7% with 800 mg imatinib.2,59,66 Severe dermatologic reactions, including erythema multiform and Stevens–Johnson syndrome, have been reported in both diseases in individual case studies.67 Other commonly observed nonhematologic toxicities in CML patients treated with imatinib include fatigue, musculoskeletal toxicity cramps, myalgias and arthralgias, headache, dizziness, cough, and anorexia. Severe toxicity (grade 3–4) has been observed more frequently in patients receiving imatinib 800 mg/day.59 Nonhematological side effects have not been carefully researched in Chinese patients; however, because Chinese patients have higher plasma concentrations of imatinib, nonhematological toxicities will be more frequent.
Patient-focused perspectives
In the targeted therapy (TT) era, dramatic increases in clinical response rates and expected survival durations have rarely been shown in solid tumors. However, patients who are treated with TKIs need to continue treatment on a daily basis and for their entire life to control the disease. The long-term effects of such treatments on patients’ health-related quality of life (HRQOL) have been studied.68
In an IRIS study,1 HRQOL was evaluated in 1,049 CML-CP patients. Patients receiving IFN plus low-dose cytarabine experienced a large decline in the trial outcome index (TOI), whereas those receiving imatinib maintained their baseline level. Treatment differences at each visit were significant (P<0.001) and clinically relevant in favor of imatinib. Mean social and family well-being (SFWB), emotional well-being (EWB), and utility scores were also significantly better for patients taking imatinib. Patients who crossed over to imatinib experienced a large increase in TOI; significant differences (P<0.001) were observed between patients who did and did not cross over in favor of imatinib; this was noted to be one of the largest changes ever seen in HRQOL trial-based reports.
Recent data from the ADAGIO (Adherence Assessment with Glivec: Indicators and Outcomes) study,69 which recruited a total of 202 patients suggested that nonadherence is associated with poorer response to imatinib. One-third of all patients were considered to be nonadherent, and only 14.2% of patients were perfectly adherent, with 100% of prescribed imatinib taken. Patients with suboptimal responses had significantly higher mean percentages of nonadherence to imatinib treatment (23.2%) than did those with optimal responses (7.3%, P=0.005).
A study by Hirji et al69 aimed to assess the impact of patient-reported treatment restrictions and negative medication experiences (NMEs) on satisfaction among patients with CML treated with oral TKIs. Among 303 patients with CML, approximately 30% of patients reported treatment difficulties; treatment difficulties were higher among patients taking nilotinib (63.3%) than among those taking dasatinib (2.6%) or imatinib (19.2%; P<0.0001). Nonadherence was generally low; however, patients on nilotinib versus imatinib reported missing doses more often (P<0.05). Treatment satisfaction was associated with significantly increased HRQOL (P<0.05) and lower activity impairment (P<0.01). NMEs were associated with decreased treatment satisfaction (P<0.01) and HRQOL (P<0.05) and greater activity impairment (P<0.01). Higher overall treatment restrictions were associated with greater treatment difficulty (P<0.001), which correlated with nonadherence (P<0.01).
To investigate patient-reported personal factors associated with adherence behavior, Efficace et al70 analyzed adherence behavior in 413 CML patients receiving long-term therapy with imatinib by the Morisky Medication Adherence Scale and investigated the following personal factors: quality of life, perceived social support, fatigue, symptom burden, psychological well-being, desire for additional information, key sociodemographic factors, and treatment-related factors. Univariate and multivariate logistic regression analyses showed that 53% of patients present optimal adherence behaviors. The final multivariate model retained the following variables as independent predictors of optimal adherence to therapy: desire for more information (odds ratio [OR]: 0.43; P<0.001); social support (higher score representing greater support; OR: 1.29; P<0.001); and concomitant drug burden (OR: 1.82; P<0.006), which suggests that a higher level of social support, satisfaction with information received, and concomitant drug burden are the main factors associated with greater adherence to long-term imatinib therapy.
Place in therapy
The introduction of TKIs has changed the landscape of therapy for CML. Imatinib, as the first TKI for BCR-ABL-positive disease, has greatly changed the treatment and management of CML and Ph+ ALL. Second-generation TKIs, such as nilotinib and dasatinib, have now been developed, and this has sparked heated discussions as to whether these therapies could take the place of imatinib as the first-line therapy for untreated CML patients or act as more appropriate combination therapies in Ph+ ALL patients.
Second-generation TKIs show slight, but significant, increases in the proportion of patients achieving early CCyR on nilotinib (Evaluating Nilotinib Efficacy and Safety in clinical Trials-newly diagnosed patients [ENESTnd])71 and dasatinib (DASatinib versus Imatinib Study In treatment-Naive CML patients [DASISION])72 compared with patients receiving imatinib. Furthermore, both studies showed that nilotinib and dasatinib induced deep MMR, such as MMR3 and MMR4, in a larger proportion of patients than did imatinib. However, the most appropriate treatment choice is not only the most efficacious, and it is also important to consider other factors, such as side effects and pharmacoeconomic characteristics. Notably, the proportion of patients who are still taking the study drug allocated at randomization 2 years prior is practically identical to the proportion still taking imatinib in both the DASISION and the ENESTnd trials, indicating that the superior efficacies of nilotinib and dasatinib are offset by other factors, most likely the drugs’ side effects. Therefore, with a longer duration of follow-up, the place of imatinib for CML and Ph+ ALL treatment will be more accurately assessed.
Disclosure
This work was supported by the National Natural Science Foundation of China (No 81070437, 81270614, 81300379). The authors have no conflicts of interest in this work.
References
Hahn EA, Glendenning GA, Sorensen MV, et al; IRIS Investigators. Quality of life in patients with newly diagnosed chronic phase chronic myeloid leukemia on imatinib versus interferon alfa plus low-dose cytarabine: results from the IRIS Study. J Clin Oncol. 2003;21(11):2138–2146. | |
O’Brien SG, Guilhot F, Larson RA, et al; IRIS Investigators. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348(11):994–1004. | |
Rix U, Hantschel O, Durnberger G, et al. Chemical proteomic profiles of the BCR-ABL inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood. 2007;110(12):4055–4063. | |
Hughes TP, Kaeda J, Branford S, et al; International Randomised Study of Interferon versus STI571 (IRIS) Study Group. Frequency of major molecular responses to imatinib or interferon alfa plus cytarabine in newly diagnosed chronic myeloid leukemia. N Engl J Med. 2003;349(15):1423–1432. | |
Druker BJ, Guilhot F, O’Brien SG, et al; IRIS Investigators. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355(23):2408–2417. | |
Hochhaus A, O’Brien SG, Guilhot F, et al; IRIS Investigators. Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia. 2009;23(6):1054–1061. | |
O’Brien SG, Guilhot F, Goldman JM, et al. International randomized study of interferon versus ST1571 (IRIS) 7-year follow-up: sustained survival, low rate of transformation and increased rate of major molecular response (MMR) in patients (pts) with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP) treated with imatinib (IM): Proceedings of Blood: 50th Annual Meeting of the American Society of Hematology, San Diego, CA, USA, December 6–9, 2008. Washington, DC: American Society of Hematology; 2008: 76. | |
Deininger M, O’Brien SG, Guilhot F, et al. International Randomized Study of Interferon vs STI571 (IRIS) 8-year follow up: sustained survival and low risk for progression or events in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP) treated with imatinib: Proceedings of Blood: 51st Annual Meeting of the American Society of Hematology, New Orleans, LA, USA, December 5–8, 2009. Washington, DC: American Society of Hematology; 2009: 462. | |
Druker BJ, Sawyers CL, Kantarjian H, et al. Activity of a specific inhibitor of the BCR-ABL tyrosine kinase in the blast crisis of chronic myeloid leukemia and acute lymphoblastic leukemia with the Philadelphia chromosome. N Engl J Med. 2001;344(14):1038–1042. | |
Wassmann B, Pfeifer H, Scheuring U, et al. Therapy with imatinib mesylate (Glivec) preceding allogeneic stem cell transplantation (SCT) in relapsed or refractory Philadelphia-positive acute lymphoblastic leukemia (Ph+ALL). Leukemia. 2002;16(12):2358–2365. | |
Ottmann OG, Druker BJ, Sawyers CL, et al. A phase 2 study of imatinib in patients with relapsed or refractory Philadelphia chromosome-positive acute lymphoid leukemias. Blood. 2002;100(6):1965–1971. | |
de Labarthe A, Rousselot P, Huguet-Rigal F, et al. Imatinib combined with induction or consolidation chemotherapy in patients with de novo Philadelphia chromosome-positive acute lymphoblastic leukemia: results of the GRAAPH-2003 study. Blood. 2007;109(4):1408–1413. | |
Yanada M, Takeuchi J, Sugiura I, et al. High complete remission rate and promising outcome by combination of imatinib and chemotherapy for newly diagnosed BCR-ABL-positive acute lymphoblastic leukemia: a phase II study by the Japan Adult Leukemia Study Group. J Clin Oncol. 2006;24(3):460–466. | |
Wassmann B, Pfeifer H, Goekbuget N, et al. Alternating versus concurrent schedules of imatinib and chemotherapy as front-line therapy for Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL). Blood. 2006;108(5):1469–1477. | |
Delannoy A, Delabesse E, Lheritier V, et al. Imatinib and methylprednisolone alternated with chemotherapy improve the outcome of elderly patients with Philadelphia-positive acute lymphoblastic leukemia: results of the GRAALL AFR09 study. Leukemia. 2006;20(9):1526–1532. | |
Lee KH, Lee JH, Choi SJ, et al. Clinical effect of imatinib added to intensive combination chemotherapy for newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Leukemia. 2005;19(9):1509–1516. | |
Lee S, Kim YJ, Min CK, et al. The effect of first-line imatinib interim therapy on the outcome of allogeneic stem cell transplantation in adults with newly diagnosed Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood. 2005;105(9):3449–3457. | |
Burke MJ, Trotz B, Luo X, et al. Allo-hematopoietic cell transplantation for Ph chromosome-positive ALL: impact of imatinib on relapse and survival. Bone Marrow Transplant. 2009;43(2):107–113. | |
Thomas DA, Faderl S, Cortes J, et al. Treatment of Philadelphia chromosome-positive acute lymphocytic leukemia with hyper-CVAD and imatinib mesylate. Blood. 2004;103(12):4396–4407. | |
Peng B, Dutreix C, Mehring G, et al. Absolute bioavailability of imatinib (Glivec) orally versus intravenous infusion. J Clin Pharmacol. 2004;44(2):158–162. | |
Wang Y, Chia YL, Nedelman J, et al. A therapeutic drug monitoring algorithm for refining the imatinib trough level obtained at different sampling times. Ther Drug Monit. 2009;31(5):579–584. | |
le Coutre P, Kreuzer KA, Pursche S, et al. Pharmacokinetics and cellular uptake of imatinib and its main metabolite CGP74588. Cancer Chemother Pharmacol. 2004;53(4):313–323. | |
Green H, Skoglund K, Rommel F, et al. CYP3A activity influences imatinib response in patients with chronic myeloid leukemia: a pilot study on in vivo CYP3A activity. Eur J Clin Pharmacol. 2010;66(4):383–386. | |
Gschwind HP, Pfaar U, Waldmeier F, et al. Metabolism and disposition of imatinib mesylate in healthy volunteers. Drug Metab Dispos. 2005;33(10):1503–1512. | |
van Erp NP, Gelderblom H, Karlsson MO, et al. Influence of CYP3A4 inhibition on the steady-state pharmacokinetics of imatinib. Clin Cancer Res. 2007;13(24):7394–7400. | |
O’Brien SG, Meinhardt P, Bond E, et al. Effects of imatinib mesylate (STI571, Glivec) on the pharmacokinetics of simvastatin, a cytochrome p450 3A4 substrate, in patients with chronic myeloid leukaemia. Br J Cancer. 2003;89(10):1855–1859. | |
Hu S, Franke RM, Filipski KK, et al. Interaction of imatinib with human organic ion carriers. Clin Cancer Res. 2008;14(10):3141–3148. | |
Gibbons J, Egorin MJ, Ramanathan RK, et al; National Cancer Institute Organ Dysfunction Working Group. Phase I and pharmacokinetic study of imatinib mesylate in patients with advanced malignancies and varying degrees of renal dysfunction: a study by the National Cancer Institute Organ Dysfunction Working Group. J Clin Oncol. 2008;26(4):570–576. | |
Dai H, Marbach P, Lemaire M, et al. Distribution of STI-571 to the brain is limited by P-glycoprotein-mediated efflux. J Pharmacol Exp Ther. 2003;304(3):1085–1092. | |
Pfeifer H, Wassmann B, Hofmann WK, et al. Risk and prognosis of central nervous system leukemia in patients with Philadelphia chromosome-positive acute leukemias treated with imatinib mesylate. Clin Cancer Res. 2003;9(13):4674–4681. | |
Takayama N, Sato N, O’Brien SG, et al. Imatinib mesylate has limited activity against the central nervous system involvement of Philadelphia chromosome-positive acute lymphoblastic leukaemia due to poor penetration into cerebrospinal fluid. Br J Haematol. 2002;119(1):106–108. | |
Mauro MJ, Druker BJ. STI571: a gene product-targeted therapy for leukemia. Curr Oncol Rep. 2001;3(3):223–227. | |
Larson RA, Druker BJ, Guilhot F, et al. Imatinib pharmacokinetics and its correlation with response and safety in chronic-phase chronic myeloid leukemia: a subanalysis of the IRIS study. Blood. 2008;111(8):4022–4028. | |
Singh N, Kumar L, Meena R, et al. Drug monitoring of imatinib levels in patients undergoing therapy for chronic myeloid leukaemia: comparing plasma levels of responders and non-responders. Eur J Clin Pharmacol. 2009;65(6):545–549. | |
Guilhot F, Hughes TP, Cortes J, et al. Plasma exposure of imatinib and its correlation with clinical response in the Tyrosine Kinase Inhibitor Optimization and Selectivity Trial. Haematologica. 2012;97(5):731–738. | |
Larson RA, Chia YL, Granvil C, et al. Steady-state imatinib trough levels as well as dose interruptions are associated with clinical response (CCyR and MMR) and adverse events (AEs) in patients with chronic myeloid leukemia (CML) receiving IM as frontline therapy: Proceedings of Blood: 51st Annual Meeting of the American Society of Hematology, New Orleans, LA, USA, December 5–8, 2009. Washington, DC: American Society of Hematology; 2009: 872. | |
Li QB, Chen C, Chen ZC, et al. Imatinib plasma trough concentration and its correlation with characteristics and response in Chinese CML patients. Acta Pharmacol Sin. 2010;31(8):999–1004. | |
Zhong JS, Meng FY, Xu D, et al. [Study on imatinib trough concentration, efficacy and their relation in chronic myelocytic leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2012;33(3):177–182. Chinese. | |
Picard S, Titier K, Etienne G, et al. Trough imatinib plasma levels are associated with both cytogenetic and molecular responses to standard-dose imatinib in chronic myeloid leukemia. Blood. 2007;109(8):3496–3499. | |
Zhou L, Meng FY, Jin J, et al. [Analysis of plasma trough level of imatinib in Chinese CML patients]. Zhonghua Xue Ye Xue Za Zhi. 2012;33(3):183–186. Chinese. | |
Chen C, Wang W, Xu CG, et al. [Monitoring of plasma concentration of imatinib mesylate in patients with chronic myeloid leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2011;32(7):450–453. Chinese. | |
Forrest DL, Trainor S, Brinkman RR, et al. Cytogenetic and molecular responses to standard-dose imatinib in chronic myeloid leukemia are correlated with Sokal risk scores and duration of therapy but not trough imatinib plasma levels. Leuk Res. 2009;33(2):271–275. | |
Zhou L, Wang AH, Wang L, et al. [Efficacy and safety of imatinib in treatment of 151 chronic myeloid leukemia patients]. Zhonghua Xue Ye Xue Za Zhi. 2008;29(1):13–17. Chinese. | |
Zhou M, Qiu HY, He GS, et al. [Multivariate analysis of imatinib resistance-related factors during the treatment of chronic myeloid leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2013;34(5):395–398. Chinese. | |
Qin Y, Jiang B, Jiang Q, et al. Molecular responses of late chronic phase chronic myeloid leukemia patients after achieving complete cytogenetic responses with imatinib treatment: a 6-year follow-up. Ann Hematol. 2009;88(1):37–41. | |
Jiang H, Chen SS, Jiang B, et al. Seven-year response to imatinib as initial treatment versus re-treatment in Chinese patients with chronic myelogenous leukemia in the chronic phase. Ann Hematol. 2011;90(1):41–46. | |
Shah NP. Loss of response to imatinib: mechanisms and management. Hematology Am Soc Hematol Educ Program. 2005:183–187. | |
Jiang Q, Chen SS, Jiang B, et al. [Efficacy and safety evaluation of imatinib in the treatment of patients with chronic granulocytic leukemia in accelerated phase]. Zhonghua Xue Ye Xue Za Zhi. 2004;25(6):333–336. Chinese. | |
Jiang Q, Chen SS, Jiang B, et al. [The efficacy of imatinib mesylate for 124 patients with chronic myeloid leukemia in accelerated and blastic phase]. Zhonghua Xue Ye Xue Za Zhi. 2007;28(11):721–726. Chinese. | |
Jiang Q, Xu LP, Liu DH, et al. Imatinib mesylate versus allogeneic hematopoietic stem cell transplantation for patients with chronic myelogenous leukemia in the accelerated phase. Blood. 2011;117(11):3032–3040. | |
Zhou L, Wang JX, Huang XJ, et al. [Preliminary comparison of efficacy and safety of dasatinib and imatinib in newly diagnosed chronic myeloid leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2013;34(2):93–97. Chinese. | |
Li CX, Wu DP, Liu H, et al. [Clinical study of imatinib for treatment of Philadelphia chromosome positive acute lymphoblastic leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2010;31(3):181–185. Chinese. | |
Huang J, Zou DH, Fu MW, et al. [Imatinib combined with modified hyper-CVAD/MA followed by allogeneic hematopoietic stem cell transplantation in CR1 as the front-line therapy for adult Ph(+) acute lymphoblastic leukemia]. Zhonghua Xue Ye Xue Za Zhi. 2011;32(10):673–678. Chinese. | |
Zhang FH, Ling YW, Zhai X, et al. The effect of imatinib therapy on the outcome of allogeneic stem cell transplantation in adults with Philadelphia chromosome-positive acute lymphoblastic leukemia. Hematology. 2013;18(3):151–157. | |
Chen J, Wu DP, Chen F, et al. [Efficacies of hematopoietic stem cell transplantation plus imatinib in the treatment of Philadelphia chromosome-positive acute lymphoblastic leukemia: a comparative study]. Zhonghua Yi Xue Za Zhi. 2013;93(8):583–587. Chinese. | |
Sawyers CL, Hochhaus A, Feldman E, et al. Imatinib induces hematologic and cytogenetic responses in patients with chronic myelogenous leukemia in myeloid blast crisis: results of a phase II study. Blood. 2002;99(10):3530–3539. | |
Talpaz M, Silver RT, Druker BJ, et al. Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia: results of a phase 2 study. Blood. 2002;99(6):1928–1937. | |
Verweij J, van Oosterom A, Blay JY, et al. Imatinib mesylate (STI-571 Glivec, Gleevec) is an active agent for gastrointestinal stromal tumours, but does not yield responses in other soft-tissue sarcomas that are unselected for a molecular target. Results from an EORTC Soft Tissue and Bone Sarcoma Group phase II study. Eur J Cancer. 2003;39(14):2006–2011. | |
Cortes JE, Baccarani M, Guilhot F, et al. Phase III, randomized, open-label study of daily imatinib mesylate 400 mg versus 800 mg in patients with newly diagnosed, previously untreated chronic myeloid leukemia in chronic phase using molecular end points: tyrosine kinase inhibitor optimization and selectivity study. J Clin Oncol. 2010;28(3):424–430. | |
Hughes TP, Branford S, White DL, et al. Impact of early dose intensity on cytogenetic and molecular responses in chronic-phase CML patients receiving 600 mg/day of imatinib as initial therapy. Blood. 2008;112(10):3965–3973. | |
Hochhaus A, Druker B, Sawyers C, et al. Favorable long-term follow-up results over 6 years for response, survival, and safety with imatinib mesylate therapy in chronic-phase chronic myeloid leukemia after failure of interferon-alpha treatment. Blood. 2008;111(3):1039–1043. | |
Van Glabbeke M, Verweij J, Casali PG, et al. Predicting toxicities for patients with advanced gastrointestinal stromal tumours treated with imatinib: a study of the European Organisation for Research and Treatment of Cancer, the Italian Sarcoma Group, and the Australasian Gastro-Intestinal Trials Group (EORTC-ISG-AGITG). Eur J Cancer. 2006;42(14):2277–2285. | |
Mauro MJ, Deininger MW. Management of drug toxicities in chronic myeloid leukaemia. Best Pract Res Clin Haematol. 2009;22(3):409–429. | |
Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet. 2004;364(9440):1127–1134. | |
Zalcberg JR, Verweij J, Casali PG, et al. Outcome of patients with advanced gastro-intestinal stromal tumours crossing over to a daily imatinib dose of 800 mg after progression on 400 mg. Eur J Cancer. 2005;41(12):1751–1757. | |
Scheinfeld N. Imatinib mesylate and dermatology part 2: a review of the cutaneous side effects of imatinib mesylate. J Drugs Dermatol. 2006;5(3):228–231. | |
Efficace F, Cocks K, Breccia M, et al. Time for a new era in the evaluation of targeted therapies for patients with chronic myeloid leukemia: inclusion of quality of life and other patient-reported outcomes. Crit Rev Oncol Hematol. 2012;81(2):123–135. | |
Noens L, van Lierde MA, De Bock R, et al. Prevalence, determinants, and outcomes of nonadherence to imatinib therapy in patients with chronic myeloid leukemia: the ADAGIO study. Blood. 2009;113(22):5401–5411. | |
Hirji I, Gupta S, Goren A, et al. Chronic myeloid leukemia (CML): association of treatment satisfaction, negative medication experience and treatment restrictions with health outcomes, from the patient’s perspective. Health Qual Life Outcomes. 2013;11(1):167. | |
Efficace F, Baccarani M, Rosti G, et al. Investigating factors associated with adherence behaviour in patients with chronic myeloid leukemia: an observational patient-centered outcome study. Br J Cancer. 2012;107(6):904–909. | |
Saglio G, Kim DW, Issaragrisil S, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362(24):2251–2259. | |
Kantarjian H, Shah NP, Hochhaus A, et al. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2010;362(24):2260–2270. | |
Wang GR, Zhao YZ, Qian LS, et al. [Analysis of long-term treatment outcome and related factors in 95 chronic myeloid leukemia patients treated with imatinib]. ZhonghuaXue Ye XueZaZhi. 2008;29(1):18–22. Chinese. | |
Zou WY, Xu DR, Su C, Li J, Luo SK. [Therapeutic effects of imatinib on chronic myeloid leukemia in different phases and the factors affecting the effects]. Nan Fang Yi Ke Da XueXueBao. 2008;28(9):1660–1662. Chinese. | |
Zhao Y, Liu L, Wang Y, et al. Efficacy and prognosis of chronic myeloid leukemia treated with imatinib mesylate in a Chinese population.Int J Hematol. 2009;89(4):445–451. | |
Zhou KS, Wang YY, Zhao YZ, et al. [Analysis of efficacy and related factors in 85 chronic myeloid leukemia patients treated with imatinib mesylate]. Zhongguo Shi Yan Xue Ye XueZaZhi. 2010;18(1):208–212. Chinese. | |
Wu SH, Zheng CP, Xu J. [A retrospective study of chronic myelocytic leukemia treatment with imatinib and interferon-α]. ZhonghuaXue Ye XueZaZhi. 2012;33(4):311–315. | |
He H, Shen Y, Zhu Y,Chen S. Prognostic analysis of chronic myeloid leukemia in Chinese population in an imatinib era. Front Med. 2012;6(2):204–211. | |
Li Y, Qiu L, Zou D, Zhao Y, Mi Y, Wang J. Additional chromosomal abnormalities and their prognostic significance in adult Philadelphia-positive acute lymphoblastic leukemia: with or without imatinib in chemotherapy. Ann Hematol. 2009;88(11):1069–1077. | |
Guo Z, Chen HR, Liu XD,et al. [Application of imatinib plus Hyper-CVAD chemotherapy regimen in patients with Ph chromosome positive acute lymphocytic leukemia]. Zhonghua Yi XueZaZhi. 2011;91(20):1384–1387. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.