Back to Journals » ClinicoEconomics and Outcomes Research » Volume 9
Clinical and economic analysis of outcomes of dose tapering or withdrawal of tumor necrosis factor-α inhibitors upon achieving stable disease activity in rheumatoid arthritis patients
Authors Aletaha D , Snedecor SJ, Ektare V, Xue M, Bao Y, Garg V
Received 7 March 2017
Accepted for publication 16 May 2017
Published 28 July 2017 Volume 2017:9 Pages 451—458
DOI https://doi.org/10.2147/CEOR.S136327
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Giorgio Colombo
Daniel Aletaha,1 Sonya J Snedecor,2 Varun Ektare,2 Mei Xue,2 Yanjun Bao,3 Vishvas Garg3
1Division of Rheumatology, Department of Internal Medicine, Medical University of Vienna, Vienna, Austria; 2Pharmerit International, Bethesda, MD, 3AbbVie Inc., North Chicago, IL, USA
Objective: To compare the real-world, 5-year clinical and cost impact of maintaining treatment with the tumor necrosis factor-α inhibitors (anti-TNFs) adalimumab, etanercept, or infliximab vs dose tapering or withdrawal in rheumatoid arthritis (RA) patients who have achieved remission (defined as a 28-joint count Disease Activity Score [DAS28] < 2.6) or low disease activity (LDA; DAS28 < 3.2).
Methods: Using a 5-year Markov model with 1-month cycle length, we examined the clinical and cost impact of three treatment strategies: withdrawal, tapering, or maintenance of anti-TNFs among RA patients in remission or who have achieved LDA. This model assessed the time to loss of disease control, time to regaining control after treatment reinitiation, and associated medical and anti-TNF costs. To determine the risk of losing disease control, 14 studies (2309 patients) were meta-analyzed, adjusted for treatment strategy, anti-TNF, RA patient type (early or established RA), and model entry criterion (remission or LDA).
Results: Anti-TNF withdrawal and tapering incurred comparable 5-year total costs (€37,900–€59,700 vs €47,500–€59,200), which were lower than those incurred by anti-TNF maintenance (€67,100–€72,100). Established RA patients had higher total costs than early RA patients (€45,900–€72,100 vs €37,900–€71,700). Maintenance was associated with the longest time to loss of disease control (range, 27.3–47.1 months), while withdrawal had the shortest (range, 6.9–30.5 months).
Conclusion: Dose tapering or withdrawal of anti-TNFs results in similar reduction of health care costs but less time in sustained disease control compared to maintaining therapy. Future research is needed to understand the long-term clinical consequences of these strategies and patient preferences for treatment withdrawal.
Keywords: economic analysis, dose reduction, flare, biologics withdrawal
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease characterized by persistent destructive synovial inflammation contributing to progressive tissue damage1,2 and irreparable joint damage.3 RA has a global prevalence of 0.24% and contributes substantially to worldwide disability.4 The economic costs of RA are also substantial: for example, the estimated annual cost per patient in Spain ranged from US $7914 to $12,922 (in 2001), with national annual costs estimated at more than $2 billion.5
The introduction of tumor necrosis factor-α inhibitors (anti-TNFs) improved outcomes for many RA patients, particularly those who did not respond to conventional synthetic disease-modifying antirheumatic drugs such as methotrexate. Anti-TNFs improve RA signs and symptoms, inhibit radiographic progression, and have been found to be cost-effective for patients with moderate-to-severe RA.6
Treatment strategies investigating the possibility of anti-TNF dose tapering or withdrawal among patients achieving disease control are gaining popularity in order to optimize the benefits compared to the risks with respect to factors such as the high costs of biologics and patient preferences.7 In one example, the National Health Insurance Administration of Taiwan mandated RA patients taper anti-TNFs after 2 years of disease control followed by withdrawal after 1 additional year if control is maintained.8 In a more recent case, the Spanish Rheumatology Society and Hospital Pharmacy Society published a consensus statement that careful dose tapering could be undertaken in most patients with RA.9
Although these guidelines suggest tapering or withdrawing anti-TNF therapy after patients achieve disease control, patients do usually relapse after withdrawal9 and few data are available describing the clinical or economic implications of these strategies. The objective of this study is to assess, from the Spanish health care payer’s perspective, the health economic and clinical impact of withdrawing, tapering, or maintaining anti-TNF treatment in RA patients who have achieved stable disease control.
Methods
Model structure
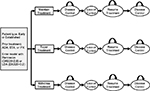
We constructed a 5-year Markov model with a 1-month cycle length to estimate the health care costs and time to loss of disease control associated with anti-TNF maintenance, dose tapering, or withdrawal. These 3 treatment strategies were compared in 4 different scenarios that vary by the type of RA patient modeled (early [within 3 years of diagnosis] or established) and the model entry criterion (having achieved disease remission [28-joint count Disease Activity Score, DAS28 < 2.6] or low disease activity [LDA; DAS28 < 3.2] at baseline). The model does not consider patients’ specific duration of disease control below these thresholds due to lack of robust scientific data on this topic. Given the availability of published evidence, this study focuses on the anti-TNFs adalimumab (ADA), etanercept (ETA), and infliximab (IFX).
Patients enter the model with controlled disease (Figure 1). At the end of each monthly Markov cycle, patients undergoing any of the 3 treatment strategies either retain or lose disease control (defined as loss of DAS28 < 2.6 or DAS28 < 3.2, depending on patients’ model entry criteria). Upon loss of disease control, patients undergoing tapering or withdrawal are modeled to reinitiate the full dose of their original anti-TNF, after which disease control may be achieved again. For all treatment strategies, it was assumed that those who regained disease control remain in control until the end of the model. Patients who did not regain disease control within the model horizon were not modeled to subsequently augment or change therapy.
Model transition probabilities
Within the 3 treatment strategies, separate monthly probabilities of loss of disease control were estimated for each anti-TNF (ADA, ETA, and IFX), RA patient type (early or established), and model entry criterion (remission or LDA). To supplement recent systematic reviews of anti-TNF withdrawal studies,7,10 we conducted a literature search in PubMed to identify clinical studies published from January 2010 to October 2014 that examined the time to loss of disease control after withdrawal, dose tapering, or maintenance with ADA, ETA, or IFX. The search terms used for the review were (spac* OR stop* OR cessation OR reduc* OR tapering OR “dose reduction” OR discontinuation OR “dose de-escalation” OR “de-escalation” OR withdrawal OR drug administration schedule [MeSH]) AND (“anti-tnf” OR tumor necrosis factor-alpha [MeSH] OR anti-inflammatory agents [MeSH] OR immunosuppressive agents [MeSH]) AND (arthritis, rheumatoid [MeSH] OR (arthritis [All Fields] AND rheumatoid [All Fields]) OR “rheumatoid arthritis” [All Fields]). Only English language articles were considered. European League Against Rheumatism (EULAR) and American College of Rheumatology (ACR) conference abstracts from the years 2011–2014 were also searched using the keywords taper, spaci, reduc, discont, contin, withdr, low dis, remissio, stop, dos, cessation, and escalat.
Data on time to loss of disease control were synthesized within a meta-analytic regression model to determine the monthly probabilities upon withdrawal, tapering, or maintenance for each anti-TNF, RA patient type, and model entry criterion. Most studies provided aggregated cohort-level data of the proportion of patients who lost disease control at various follow-up times; 1 study provided the length of time prior to loss of disease control for individual study subjects. To meta-analyze the available data, we assumed the time to loss of disease control followed a Weibull or exponential survival distribution. We analyzed fixed-effect and random-effects models for both distributions; the fixed-effect exponential distribution was found to be most appropriate for the available data set. This analysis was conducted within a Bayesian framework executed in WinBUGS software (The Medical Research Council, Cambridge, UK, and Imperial College of Science, Technology and Medicine, London, UK),11 which allowed the underlying exponential parameter to be estimated from cohort and individual patient data. Another advantage to this methodology is that results can be adjusted for treatment strategy, anti-TNF, RA patient type, and model entry criterion, and can generate outcomes for combinations of patient and treatment characteristics even when not observed in the clinical studies.
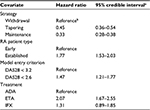
Meta-regression results were the monthly hazard of losing disease control for the reference case (ADA, treatment withdrawal, early RA, and LDA model entry criterion) and the hazard ratios associated with other covariates. Estimated monthly hazards were converted into monthly probabilities using the following formula: probability = 1 − exp (−hazard).
Fewer data were available to estimate the probability of regaining remission/LDA after reinitiation of treatment following initial loss of disease control. These reinduction data were analyzed similarly, but were unadjusted and assumed to be constant for all treatment strategies, RA patient types, and anti-TNFs.
Cost data
Total costs for the months spent in each health state were calculated for 4 combinations of scenarios: early or established RA and patients entering the model with remission or LDA. A PubMed literature search was performed to obtain disease and treatment costs in Spain, using the search terms (rheumatoid arthritis) AND DAS* AND (cost or econ*) and the search period January 2004 to October 2014. Abstracts from ACR and EULAR conferences were also searched from the year 2011 onward using the same keywords. Because this model is from the Spanish payer’s perspective, we considered only direct medical costs of disease and treatment and did not include indirect costs such as lost work or productivity.
Spanish costs of disease for patients in remission (DAS28 < 2.6), LDA (DAS28 ≤ 3.2), and flare (DAS28 > 3.2) were obtained from a study by Beresniak et al12 that estimated RA treatment costs in Spain by the level of disease activity (remission: €48.69, LDA: €57.77, and flare: €1012.68). Costs considered included rheumatologist and other specialist visits, general practitioner visits, laboratory tests, hospitalization, imaging, physiotherapy, surgery, medical transportation, and nursing. Monthly anti-TNF drug costs were based on labeled dosages in Spain. All costs are expressed in 2014 Euro.
Results
Published literature
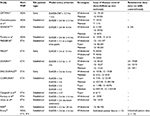
Fourteen published trials including 2309 patients using 1 of the 3 anti-TNF agents of interest were identified (Table 1).2,13–27 Eight studies evaluated ETA, 4 ADA, 1 IFX, and 1 all 3 anti-TNFs. Most of the studies included established RA patients. Five studies required achievement of LDA (DAS28 < 3.2) for treatment withdrawal or tapering, whereas 9 studies required achievement of remission (DAS28 < 2.6). One study used both definitions in respective subgroups and evaluated them separately. One study that reported data using the DAS44 withdrawal criterion was excluded.28 Follow-up time ranged from 6 to 24 months. Only 3 studies reported outcomes after treatment reinduction that was followed by loss of disease control. Other studies evaluating dose tapering and/or withdrawal were identified but were excluded as the data were not reported with sufficient detail to be used in our analysis.
Time to loss of disease control
Analysis of the published data revealed the adjusted risk of losing disease control upon dose tapering or maintenance of anti-TNFs was less than half compared to treatment withdrawal (Table 2). Patients with established RA had significantly higher risk of losing disease control than patients with early RA. Further, risk of losing disease control with ETA was found to be significantly higher than ADA, irrespective of the treatment strategy. Thus, the withdrawal strategy was associated with the fewest number of months until loss of disease control, followed by taper and maintenance. Established RA patients had fewer months until loss of disease control than early RA patients, and patients on ADA had longer time to loss of disease control than ETA or IFX (Figure 2). Upon treatment reinitiation after loss of disease control, the time to regain remission or LDA was estimated to have a monthly probability of 22% and was assumed equal for all patients.
Health care costs
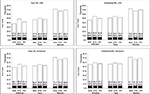
Patients whose anti-TNF treatment was withdrawn incurred the lowest overall 5-year health care costs, followed by patients whose doses were tapered or maintained; however, withdrawal was only marginally lower compared to tapering (Figure 3). Established RA patients had higher total costs than early RA patients across all treatment strategies. ADA was associated with the lowest costs in the withdrawal and taper strategies, but the highest cost in the maintenance strategy. The proportion of total costs due to anti-TNFs was similar across the 3 treatments, but was lowest for withdrawal and highest for maintenance.
Discussion
Through a meta-regression of the published evidence, our model found that patients whose anti-TNF (ADA, ETA, or IFX) treatments were withdrawn or tapered incurred lower 5-year health care costs but had worse clinical outcomes than patients who maintained. Further, the marginally lower costs incurred by withdrawal compared to dose tapering were offset by substantially less time to loss of disease control.
We found that irrespective of entering the model in remission or the LDA state, early RA patients incurred lower health care costs and had a longer time to losing disease control upon withdrawal or tapering of anti-TNFs compared to established RA patients. This finding indicates a higher feasibility of withdrawing or tapering anti-TNFs in early RA patients compared to established RA patients. Nonetheless, the risk–benefit of anti-TNF dose tapering or withdrawal over maintenance should be carefully examined in both early and established RA patients, as treat-to-target recommendations suggest remission and remission or LDA as clear targets for RA treatment in patients with early or long-standing disease, respectively.29
Both early and established RA patients who entered the model in remission lost disease control upon withdrawal or tapering in less time than patients who entered the model in LDA. This may seem counterintuitive, as one may expect patients who have achieved a deeper level of response to maintain that response longer, but the definition of losing disease control for each of these model entry criteria must also be considered. Specifically, for patients entering model at DAS28 > 2.6, the definition of losing disease control is DAS28 > 2.6 and patients entering at DAS28 < 3.2 do not lose disease control until they reach the DAS28 > 3.2 threshold. Thus, the time to loss of remission (DAS28 > 2.6) is expected to be shorter, resulting in a higher calculated risk of losing disease control. Although not within the scope of this analysis, we also assessed the model results when the model entry criterion was DAS28 < 2.6, but loss of disease control was defined as loss of LDA (DAS28 > 3.2). As expected, the overall time to loss of disease control was longer, and overall costs were lower compared to the scenario where both model entry and loss of disease control were DAS28 < 2.6. However, the relative differences among the treatments and the treatment strategies remained the same.
Data in our model included a compilation of all available published evidence describing the proportion of patients who attained remission or LDA but subsequently lost disease control following treatment withdrawal, dose tapering, or maintenance. One advantage of our model’s meta-analytic regression analysis is that we were able to estimate outcomes for patient combinations not directly studied in clinical trials. One limitation of such analysis is that it assumed an exponential distribution for the time to loss of disease control. Individual patient data are needed to test the validity of this assumption. However, this was provided by only 1 study of 13 patients, too few to make reasonable conclusions. In addition, other factors such as duration of remission and patient age could be important predictors of relapse after attaining disease control. To properly account for these factors, patient-level data – which were unavailable for most trials – would have been required, since these data vary on the patient level and not the study level.
Two important reasons for dose tapering or withdrawing anti-TNFs fall outside the scope of our model. First, patient preference is a key factor to consider when evaluating the benefits of these 2 strategies. Patients who experience burdensome side effects or those who wish to reduce their medication burden due to other comorbidities may highly value the ability to reduce or withdraw anti-TNF treatment. Although the primary focus of our study was based on cost estimations, it is important to mention that patient preference will be an additional and key determinant of the decision to go with 1 of the 3 strategies. For example, patients who are responding well may be less willing to continue chronic medication, given the potential risks and side effects of therapy. Second, patients may have worse quality of life upon losing disease control, and the value of maintaining disease control should include the willingness to pay to avoid this adverse health state. The magnitude of these patient preferences is currently unknown, and studies should be conducted to understand how tapering and withdrawing biologics affects patients’ quality of life.
In any case, frequent switches between disease control and flare-ups may be associated with higher risk of structural progression of disease.30 The data presented here represent the point estimate of the costs and number of months in disease control over a 5-year period. We did not estimate patients’ health utility on or off treatment, so no quality-adjusted life years (QALYs) were generated to estimate if treatment withdrawal or tapering were cost-effective strategies. Nor did we calculate the cost per month in RA remission, as there is no generally accepted willingness-to-pay threshold for a remission month, as there is for an additional QALY (eg, €30,000 in Spain).31 Patient age and occupational status will likely influence the overall costs of each strategy, and possibly also to a different extent. Taken together, these results should be interpreted as guidelines to the potential effects of anti-TNF withdrawal, tapering, or maintenance strategies, and not as absolute declarations of value.
When evaluating the relative costs of anti-TNFs vs the costs of the increased likelihood of losing disease control over a 5-year time horizon, the model could not include any potential long-term differences in outcomes associated with dose tapering or withdrawal. Even after disease control is regained upon treatment reinitiation, long-term damage may have occurred during the loss of control period, which could not be accounted for in this model. Our model is from the health care payer perspective and, as such, includes only direct costs and no productivity costs. Indirect productivity costs may increase during periods of lost disease control due to increased pain-related presenteeism or absenteeism. Thus, the true cost of loss of disease control may be more than modeled here.
In conclusion, compared to maintenance of anti-TNFs, dose tapering or withdrawal in patients with early or established RA results in reduction of health care costs but less time in sustained disease control.
Acknowledgments
The authors thank Beth Lesher of Pharmerit International for editorial support for this manuscript. AbbVie Inc. provided financial support and participated in the interpretation of data, review, and approval of this manuscript.
Author contributions
SJS, VE, and MX identified study data and conducted the analysis. All authors were involved in study design, drafting of the manuscript, and approval of the final version for submission.
Disclosure
Daniel Aletaha has received grant/research support, consulting fees, or speaker fees from AbbVie, AstraZeneca, BMS, Eli Lilly, Grünenthal, Jansen, Medac, Merck, Mitsubishi Tanabe, Novo Nordisk, Pfizer, and Sanofi/Regeneron. Yanjun Bao and Vishvas Garg are employees and shareholders of AbbVie. Sonya J Snedecor, Varun Ektare, and Mei Xue are employees of Pharmerit International and were paid consultants to AbbVie in connection with the development of this study. The authors report no other conflicts of interest in this work.
References
Nawata M, Saito K, Nakayamada S, Tanaka Y. Discontinuation of infliximab in rheumatoid arthritis patients in clinical remission. Mod Rheumatol. 2008;18(5):460–464. | ||
Tanaka Y, Hirata S, Fukuyo S, et al. Discontinuation of adalimumab without functional and structural progress after attaining remission in patients with rheumatoid arthritis. Arthritis Rheum. 2011;63:2468. | ||
Fuchs HA, Kaye JJ, Callahan LF, Nance EP, Pincus T. Evidence of significant radiographic damage in rheumatoid arthritis within the first 2 years of disease. J Rheumatol. 1989;16(5):585–591. | ||
Cross M, Smith E, Hoy D, et al. The global burden of rheumatoid arthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73(7):1316–1322. | ||
Lajas C, Abasolo L, Bellajdel B, et al. Costs and predictors of costs in rheumatoid arthritis: a prevalence-based study. Arthritis Rheum. 2003;49(1):64–70. | ||
Schoels M, Wong J, Scott DL, et al. Economic aspects of treatment options in rheumatoid arthritis: a systematic literature review informing the EULAR recommendations for the management of rheumatoid arthritis. Ann Rheum Dis. 2010;69(6):995–1003. | ||
Kavanaugh A, Smolen JS. The when and how of biologic agent withdrawal in rheumatoid arthritis: learning from large randomised controlled trials. Clin Exp Rheumatol. 2013;31(4 Suppl 78): S19–S21. | ||
Ministry of Health and Welfare, National Health Insurance Administration, Secondary National Health Insurance Administration. Drug payment regulations. Available from: http://www.nhi.gov.tw/webdata/webdata.aspx?menu=18&menu_id=683&webdata_id=2919. Accessed June 15, 2017. | ||
González-Álvaro I, Martínez-Fernández C, Dorantes-Calderón B, et al; Spanish Rheumatology Society; Spanish Rheumatology Society. Spanish Rheumatology Society and Hospital Pharmacy Society consensus on recommendations for biologics optimization in patients with rheumatoid arthritis, ankylosing spondylitis and psoriatic arthritis. Rheumatology (Oxford). 2015;54(7):1200–1209. | ||
Yoshida K, Sung YK, Kavanaugh A, et al. Biologic discontinuation studies: a systematic review of methods. Ann Rheum Dis. 2014;73(3):595–599. | ||
Lunn DJ, Thomas A, Best N, Spiegelhalter D. WinBUGS - a Bayesian modelling framework: concepts, structure, and extensibility. Stat Comput. 2000;10(4):325–337. | ||
Beresniak A, Ariza-Ariza R, Garcia-Llorente JF, Ramirez-Arellano A, Dupont D. Modelling cost-effectiveness of biologic treatments based on disease activity scores for the management of rheumatoid arthritis in Spain. Int J Inflam. 2011;2011:727634. | ||
Smolen JS, Emery P, Fleischmann R, et al. Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet. 2014;383(9914):321–332. | ||
Chatzidionysiou K, Askling J, Eriksson J, Kristensen LE, van Vollenhoven R; ARTIS group. Effectiveness of TNF inhibitor switch in RA: results from the national Swedish register. Ann Rheum Dis. 2015;74(5):890–896. | ||
Hirata S, Saito K, Kubo S, et al. Discontinuation of adalimumab after attaining disease activity score 28-erythrocyte sedimentation rate remission in patients with rheumatoid arthritis (HONOR study): an observational study. Arthritis Res Ther. 2013;15(5):R135. | ||
Tanaka Y, Hirata S, Kubo S, et al. Discontinuation of adalimumab after achieving remission in patients with established rheumatoid arthritis: 1-year outcome of the HONOR study. Ann Rheum Dis. 2015;74(2):389–395. | ||
Smolen JS, Nash P, Durez P, et al. Maintenance, reduction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis (PRESERVE): a randomised controlled trial. Lancet. 2013;381(9870):918–929. | ||
Emery P, Hammoudeh M, Fitzgerald O, et al. Assessing maintenance of remission with reduced dose etanercept plus methotrexate, methotrexate alone, or placebo in patients with early rheumatoid arthritis who achieved remission with etanercept and methotrextate: the PRIZE study. Ann Rheum Dis. 2013;72(Suppl 3):A399. | ||
van Vollenhoven RF, Franck-Larsson K, Leirisalo-Repo M, et al. In rheumatoid arthritis patients with stable low disease activity on methotrexate plus etanercept, continuation of etanercept at 50 mg or 25 mg weekly are both clinically superior to discontinuation: results from a randomized, 3-arm, double-blind study. Ann Rheum Dis. 2013; 72(Suppl 3):A434–A435. | ||
Yamanaka H, Seto Y, Nagaoka S, et al. Discontinuation of etanercept in early rheumatoid arthritis patients who have achieved sustained remission: results of the randomized controlled trial in period 2 of the ENCOURAGE study. Ann Rheum Dis. 2014;73(Suppl 2):239. | ||
Greenberg JD, Shan Y, Reed GW, Bitman B, Collier D. Comparison of switching to reduced dose vs continuation of standard dose etanercept for rheumatoid arthritis patients in the CORRONA registry. Ann Rheum Dis. 2014;73(Suppl 2):241. | ||
Campbell RC, Sanders E, Leone F, Gilgeours E, Sibley M, Kiely PD. The cost savings associated with a departmental etanercept dose reduction pathway for patients with rheumatic diseases and maintained low disease activity. Arthritis Rheum. 2013;65(Suppl 10):487. | ||
Kamiya M, Sohen S, Kikuchi H. Assessment of etanercept withdrawal in rheumatoid arthritis patients in remission. Ann Rheum Dis. 2011;70(Suppl 3):414. | ||
Urata Y, Nakamura Y, Motomura S. Discontinuation of etanercept in rheumatoid arthritis patients in clinical remission: two-year outcome. Ann Rheum Dis. 2012;71(Suppl 3):525. | ||
Tanaka Y, Takeuchi T, Mimori T, Miyasaka N, Koike T; RRR study investigators. The possibility and predictive factors of maintaining low disease activity and joint structure after discontinuation of infliximab in RA patients: results from 3-year experience of RRR study. Ann Rheum Dis. 2013;72(Suppl 3):A443. | ||
Brocq O, Millasseau E, Albert C, et al. Effect of discontinuing TNFalpha antagonist therapy in patients with remission of rheumatoid arthritis. Joint Bone Spine. 2009;76(4):350–355. | ||
Pham T, Morel J, Alfaiate T, et al. Predictive factors of relapse or persistent stable remission for rheumatoid arthritis (RA) patients in remission in a TNF blocker-spacing strategy trial (STRASS trial). Arthritis Rheum. 2013;65(Suppl 10):S1021. | ||
van der Broek M, Klarenbeek NB, Dirven L, et al. Discontinuation of infliximab and potential predictors of persistent low disease activity in patients with early rheumatoid arthritis and disease activity score-steered therapy: subanalysis of the BeSt study. Ann Rheum Dis. 2011;70(8):1389–1394. | ||
Smolen JS, Breedveld FC, Burmester GR, et al. Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis. 2016;75(1):3–15. | ||
Welsing PM, Landewé RB, van Riel PL, et al. The relationship between disease activity and radiologic progression in patients with rheumatoid arthritis: a longitudinal analysis. Arthritis Rheum. 2004;50(7):2082–2093. | ||
Sacristán JA, Oliva J, Del Llano J, Prieto L, Pinto JL. [What is an efficient health technology in Spain?]. Gac Sanit. 2002;16(4):334–343. Spanish [with English abstract]. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.