Back to Journals » Cancer Management and Research » Volume 12
Clinical Analysis of 15 Cases of Gallbladder Neuroendocrine Carcinoma and Comparison with Gallbladder Adenocarcinoma Using a Propensity Score Matching
Authors Yan S , Wang Y , Chen X, Zhang Y, Huang Z, Zhao J, Zhou J, Li Z, Bi X, Luo Z , Cai J, Zhao H
Received 18 August 2019
Accepted for publication 24 December 2019
Published 26 February 2020 Volume 2020:12 Pages 1437—1446
DOI https://doi.org/10.2147/CMAR.S227501
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Xueqiong Zhu
Shida Yan,1,* Yingyi Wang,2,* Xiao Chen,1,* Yefan Zhang,1 Zhen Huang,1 Jianjun Zhao,1 Jianguo Zhou,1 Zhiyu Li,1 Xinyu Bi,1 Zhiwen Luo,1 Jianqiang Cai,1 Hong Zhao1
1Department of Hepatobiliary Surgery, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China; 2Department of Medical Oncology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jianqiang Cai; Hong Zhao
Department of Hepatobiliary Surgery, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, No. 17 Panjiayuan Nanli, Chaoyang District, Beijing 100021, People’s Republic of China
Email [email protected]; [email protected]
Purpose: This study aimed to investigate the clinicopathological features and prognosis of gallbladder neuroendocrine carcinoma (GB-NEC).
Patients and Methods: Fifteen patients with GB-NEC and 171 patients with gallbladder adenocarcinoma (GB-ADC) treated in two tertiary medical centers between 2009 and 2015 were included. The clinicopathological features and prognostic risk factors of GB-NEC were analyzed retrospectively. A propensity score matching in a 1:2 ratio was used to compare the prognosis of GB-NEC and GB-ADC.
Results: For patients with GB-NEC, the median age of patients was 58.4 years (range 26– 75), with a M:F ratio of 7:8. Based on 2010 WHO classification, ten cases were pathologically confirmed as NECs and five cases as MANECs. For TNM staging, eleven patients were stage III or above; while for Nevin staging, seven patients were stage IV or above. The 1-, 2-, and 3-year overall survival (OS) of GB-NEC were 60.0%, 38.8% and 31.1%, respectively, and the median survival time was 20.4 months. Patients with lymph node metastasis had significantly shorter survival than those without (OS: 10.4 vs 26.0 months, p< 0.05). Accordingly, patients of Nevin stage III had better OS than those of Nevin stage IV (p< 0.05), but other potential risk factors including gender, age, clinical symptoms, TNM stage, histopathologic subtype and treatment showed no significance. After the propensity score matching, the baseline variables had no significant difference between 15 patients with GB-NEC and 30 patients with GB-ADC, survival analysis showed GB-NEC had worse prognosis (3-year overall survival rate: 31.1% vs 63.8%, p< 0.01).
Conclusion: Nevin staging helps classify patients of GB-NEC with different prognosis and the lymph node metastasis is a strong negative prognostic factor for OS. The propensity score analysis revealed even with the similar stage and treatment, GB-NEC still had worse OS than GB-ADC.
Keywords: gallbladder, neuroendocrine carcinoma, adenocarcinoma, survival, propensity score matching
Introduction
Neuroendocrine neoplasms (NEN) are recognized as true neoplasms with characteristic neuroendocrine differentiation that arise from neuroendocrine cells throughout the whole body.1 Since it was first recognized as karzinoide (carcinoid) in 1907,2 NEN was considered as a fairly infrequent and indolent tumor. In the past several decades, with further researches to elucidate the mechanism and characteristics of NEN,3–5 it has been seen as a heterogenous disease and many sophisticated diagnosis and treatment guidelines including the World Health Organization (WHO) classification and the European Neuroendocrine Tumour Society (ENETS) guidelines were established.6–8 As the application of advanced medical imaging and endoscopic techniques, the incidence rate of NENs of many anatomic sites was increasing in the past half century.4,9 Based on the SEER database, the incidence of NENs is 5.25/100,000 in 2004, while the most common primary tumor sites are gastrointestinal and respiratory tracts.9 Primary gallbladder neuroendocrine tumor, which comprise only 0.5% of all NENs, is an extremely rare disease.10 Despite the rapid development of the recognition of the pathogenesis and treatment of NENs, little is known about gallbladder NENs due to the rarity. Unlike NENs from other common sites, primary gallbladder NENs are mostly asymptomatic initially and often diagnosed at aggressive stage. Therefore, once diagnosed, extensive surgery and careful follow-up are mandatory and radical surgical resection is thought to be the relatively optimal treatment while medical therapy will be favorable in high grade metastatic carcinomas.10–13 However, current treatment strategies cannot prevent patients from poor prognosis and overall survival time varied from 4 to 13.7 months,11,14,15 which was significantly worse than gallbladder adenocarcinoma (GB-ADC), the most common carcinoma of gallbladder. In this report, we introduced and analyzed 15 patients with gallbladder neuroendocrine carcinoma (GB-NEC) treated in two tertiary medical centers in The People's Republic of China between 2009 and 2015, then we compared them with patients of GB-ADC using a propensity score analysis and discussed their clinicopathological features and prognosis.
Materials and Methods
Patients
Searching for patients with GB-NEC was performed in two tertiary medical centers in The People's Republic of China. Patients with GB-NEC were chosen if the following inclusion criteria were satisfied: 1) the anatomic site of tumor was in the gallbladder which was confirmed by imaging and surgery; 2) the diagnosis of GB-NEC was made by experienced pathologists with cells of variable mitotic activity and positive expression of more than one type of immunohistochemical stain, including CgA, Syn and CD56, based on the 2010 WHO classification of tumors of the digestive system;6 and 3) patients had detailed and intact medical records and follow-up data. Patients’ medical records including basic demographic information, clinical symptoms, TNM staging, pathological classification, distant metastasis, treatment, recurrence, and survival time, were reviewed retrospectively (Table 1). To perform propensity score matching with GB-NEC, patients with GB-ADC were also selected. As with the patients with GB-NEC, their demographic information and clinicopathological features were also collected. Between 2009 and 2015, 15 cases of GB-NEC and 171 cases of GB-ADC which fulfilled the inclusion criteria were taken into analysis. The study was approved by the institutional review board at both medical centers.
 |
Table 1 Clinicopathological Features of 15 Cases of Gallbladder Neuroendocrine Carcinoma |
Pathological Classification and Staging
In this study, the diagnosis and staging of the GB-NEC and GB-ADC are based on the 2010 WHO classification of tumors of the digestive system, the AJCC cancer staging definition of gallbladder (eighth edition)16 and the Nevin staging system.17
The 2010 WHO classification divides neuroendocrine tumors into three histopathologic subtypes: neuroendocrine tumors (NETs), neuroendocrine carcinomas (NECs), and mixed adenoneuroendocrine carcinomas (MANECs).6 NETs are composed of well-differentiated cells with similar features of normal gastrointestinal endocrine cells and express neuroendocrine markers and hormones. According to ENETS consensus of grading proposal for foregut neuroendocrine tumors,7 NETs have two tier, G1 and G2, based on mitotic count and Ki-67 index: G1, mitotic count <2 per 10 HPF and/or ≤2% Ki 67 index; and G2, mitotic count 2–20 per 10 HPF and/or 3–20% Ki 67 index. In the meantime, NECs belong to G3, defined as mitotic count >20 per 10 HPF and/or >20% Ki 67 index. Moreover, NECs encompass small cell and large cell types, which both are poorly differentiated and highly malignant neoplasm, with highly cellular atypia, extensive necrosis and higher mitotic figures. MANECs are complexes of adenocarcinoma and NEC, and by arbitrary definition, each part should comprise at least 30% of the whole neoplasm.
The TNM stage is determined according to the pathological report, which clearly stated the invasion degree, lymph node and distant metastasis, based on the 8th AJCC cancer staging system of gallbladder.16 The Nevin staging system was first described by James E. Nevin in 1976.17 Lesions of Nevin stage I involve intramucosal only; stage II, mucosa and muscularis; stage III, all three layers involved. For Nevin stage IV, cystic lymph node metastasis is detected; while patients with stage V have distant metastasis including hepatic metastasis.
Statistical Analysis
The overall survival of GB-NEC was described using the Kaplan-Meier method. Univariate survival analysis was performed by the Log rank test. In addition, we collected a list of cases of GB-ADC to compare the clinical features and prognosis between two pathological types of biliary carcinomas. To minimize the impact of other confounding factors on prognosis, we used a propensity score analysis to match these two types of patients in a 1:2 ratio using the nearest neighbor matching. The confounders used for matching included gender, age, Nevin stage, and radical surgery. For both GB-NEC and GB-ADC, the extent of radical resection should include cholecystectomy, partial hepatectomy and hepatoduodenal lymphadenectomy. We used paired t test to compare two groups of matched patients. The overall survival difference between the two groups was compared by the Log rank test and estimated according to the Kaplan-Meier method. Two-sided P values were computed and P<0.050 was considered statistically significant. All aforementioned statistical analyses were implemented using SPSS version 22.0 for Windows (IBM Corporation, Armonk, NY, USA).
Results
General Characteristics and Clinical Presentations
Table 1 shows detailed characteristics of all 15 patients with gallbladder NEC. In the 15 cases, there were seven men and eight women, with a median age of 58.4 years (range 26–75). Lesions in all cases were located in the gallbladder. The clinical presentations had no specificity, while the most common symptoms were abdominal pain (8, 53.3%) and weight loss (6, 40%) and one patient was diagnosed with jaundice. None of NECs in these patients showed any neuroendocrine function.
Staging
For TNM staging, four cases were stage II, four cases stage IIIA, three cases stage IIIB and the rest four cases stage IVB. Of the patients with stage IVB, two of them had distant metastases in the liver, one in the liver and bone, and the other one in the right neck lymph nodes. Nevin staging system were also used to classify these patients: more than half of the patients (8, 54.3%) belonged to Nevin stage III, three patients were stage IV and the remaining four cases stage V.
Histopathological Features
According to the 2010 WHO classification, ten patients were diagnosed with NECs and five were MANECs. As for differentiated degree, most cases (10, 66.7%) were poorly differentiated and only one case was well-differentiated. The Ki-67 index was available in 12 patients, which were all >20% and mostly over 60%. The NEC biomarkers were evaluated by immunohistochemistry and the positive rate was high. CgA was positive in 12 of 13 patients (92.3%) and Syn was positive in all 13 tested patients, while the positive rate of CD56 and CK were 91.7% and 100% (11/12 and 4/4).
Treatments
Of the 15 cases, radical resections were performed in ten patients (66.7%) and among them, four received postoperative chemotherapy (40%) and three underwent chemotherapy when the recurrence of the disease appeared (30%). One patient only had cholecystectomy, whose postoperative pathology turned out to be T3 stage. Of the rest, one patient had palliative therapy, two patients (13.3%) who had distant metastases only received chemotherapy and one patient who had both liver and bone metastases had complex treatments including chemotherapy, interventional therapy and radiotherapy. Among the total 10 patients who had chemotherapy, eight of them received EP regimen (Etoposide+ Cisplatin) and one had GP regimen (Gemcitabine+ Cisplatin).
Clinical Outcomes
The 1, 2, and 3-year overall survival rates of patients with GB-NEC were 60.0%, 38.8%, and 31.1%, respectively and the median survival time was 20.4 months (range 0.5–40.0 months; Figure 1).
 |
Figure 1 The overall survival rate of all 15 cases with GB-NEC. |
Univariate survival analysis demonstrated factors including gender, age, clinical symptoms, TNM stage, histopathologic subtype (NEC or MANEC) and treatment choice (radical operation or not, chemotherapy or not) had no prognostic significance, however, the median overall survival time in patients without lymph node metastasis was significantly longer than in those with lymph node metastasis (26.0 vs 10.4 months, p<0.05; Figure 2).
 |
Figure 2 Lymph node metastasis is a strong prognostic factor for GB-NEC (p=0.015). |
Since TNM staging cannot stratify patients with diverse prognosis, Nevin staging was used in our patients. As a result, Nevin stage III showed a significant protective effect on overall survival compared to Nevin stage IV (p<0.05). Nevertheless, there were no significant differences between patients with Nevin stage III and V or patients with Nevin stage IV and V (stage III vs V, p=0.088; stage IV vs V, p=0.886; Figure 3).
 |
Figure 3 Nevin stage III had significantly better clinical outcomes than Nevin stage IV (stage III vs IV, p=0.006; stage III vs V, p=0.088; stage IV vs V, p=0.886). |
Propensity Score Analysis
A group of 30 patients with GB-ADC was formed using propensity score matching in a 1:2 ratio (Table 2). Among matched patients with GB-ADC, they had 13 male patients (43%) and the average age was 62.1 (range 50–78). The number of cases with TNM stage II, III, and IV were 10, 13, and 7, respectively. Accordingly, 16 patients were Nevin stage III, five cases were Nevin stage IV and nine cases Nevin stage V. Overall, 20 patients received radical cholecystectomy, and four of them had postoperative chemotherapy. A total of 6 patients underwent cholecystectomy and among them, postoperative chemotherapies were performed in two patients. In the rest, three patients had sole chemotherapy and one patient had comprehensive treatments including cholecystectomy, chemotherapy, and radiotherapy.
 |
Table 2 Clinicopathological Characteristics of Patients with Gallbladder Adenocarcinoma |
In regard to the 30 cases of GB-ADC, the 1-, 2-, and 3-year survival rates were 86.7%, 82.1%, and 63.8%, respectively, while the median survival time was not reached. Paired t test demonstrated that baseline variables had no statistically significant difference between matched samples (Table 3). The 3-year overall survival rates of GB-ADC were significantly better than GB-NEC (63.8% vs 31.1%, p<0.01) (Figure 4), which demonstrated that even with the similar age, gender, Nevin stage, and treatment, GB-NEC still had worse overall survival than GB-ADC.
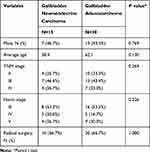 |
Table 3 Baseline Clinical Characteristics of Matched Samples by the Propensity Score |
 |
Figure 4 Patients with GB-NEC survived worse than patients with GB-ADC. Abbreviations: OS, overall survival; GB-ADC, gallbladder adenocarcinoma; GB-NEC, gallbladder neuroendocrine carcinoma. |
Discussion
Gallbladder cancer is a relatively uncommon neoplasm, of which adenocarcinoma (ADC) is the most common histological type.18 While NEC of gallbladder (GB-NEC) is extremely rare, comprising less than 1% of all NET and less than 3% of all gallbladder tumors.4,9,19 Due to its rarity, studies of GB-NEC are mostly case reports and only a few studies were single-center researches with small samples.11,13-15,20 Therefore, the data to evaluate the clinicopathological characteristics are limited. In our study, 15 patients with GB-NEC from two well-known tertiary medical centers were included, which makes it one of the largest series of GB-NEC in China.
The pathogenesis of GB-NEC is not elucidated clearly yet, previous literatures suggested that intestinal or gastric metaplasia of biliary epithelium, secondary to cholelithiasis and chronic inflammation, or multipotent cells of gallbladder could contribute to the disease.10 The neuroendocrine cells are thought to be derived from local multipotent stem cells instead of neural crest cells by migration.21 In addition, Li et al22 performed whole-genome sequencing in a case of metastatic small-cell GB-NEC and identified that some gene mutation associated with ERBB signaling, such as ERBB4, HRAS and NRG1, could play a role in gallbladder carcinogenesis.
The average age in our study was 58.4 (range 26–75), which shows no significant difference with previous studies.12,14,20 Some studies found more female patients in their cohorts,10,12,20 however, other studies including ours showed no clear female tendency.11,13,14 Most of the cases in our study had no specific clinical presentation, in accordance with most previous literatures,12–14 however, functional neuroendocrine tumors of gallbladder were also reported, including causing hyperinsulinemia, paraneoplastic sensory neuropathy and Cushing’s syndrome.23–25 Since no specific characteristics can be used to identify GB-NEC, all 15 cases were treated as patients with gallbladder adenocarcinoma in the beginning. However, Kimet al20 believed that GB-NECs had well-defined margins (intact overlying mucosa), larger hepatic and lymph node metastases than GB-ADCs, which can be used as CT discriminators.
The diagnosis of GB-NEC was mostly based on pathological and immunohistochemical (IHC) tests; meanwhile, the neoplasm must originate from the gallbladder instead of invasion of NEC from liver or other organs. The differential degree and Ki-67 index are necessary for the grading of GB-NEC. As the term “neuroendocrine” indicated, immunoexpression of neuroendocrine markers such as chromogranin A (CgA) and synaptophysin (Syn) is required to affirm a GB-NEC.26 Moreover, the plasma level of IHC biomarkers like CgA is deemed to correlate with tumor burden in untreated patients and poor-differentiated NECs tend to secrete less CgA than highly differentiated NECs, therefore they may be used to monitor efficacy and detect recurrence.27 In our cases, most of them were neuroendocrine carcinomas instead of mixed adenoneuroendocrine carcinomas, while small cell NECs predominated. Positive staining of CgA and Syn, as well as other biomarkers is present in our cases. In addition, the majority were poorly differentiated and had high Ki-67 values. Like other types of NECs, poorly differentiated cells, elevated Ki-67 and high mitotic index are believed to be predictive of a poor outcome in patients with GB-NEC.10 However, in our analysis, they did not show that tendency.
TNM staging is believed to be an important prognostic factor for neuroendocrine tumors. Analysis of pancreatic NETs demonstrated that patients with stage III and IV had significantly shorter 5-year survival.28 In the present study, eleven patients (73.3%) had stage III and above, nine of which (81.2%) survived less than 2 years. On the contrary, three of four patients (75.0%) with early stage lived more than 3 years. However, univariate analysis found no significant difference for overall survival between diverse TNM stages, which might be due to the relatively small case numbers. Instead, we used Nevin staging system to categorize patients and it turned out that patients of Nevin stage III had significantly better overall survival than stage IV (p<0.05). The main difference between Nevin stage III and IV is lymph node metastasis,17 which is in accordance with our finding that lymph node metastasis is a strong negative prognostic factor (median OS: 26.0 vs 10.4 months, p<0.05). Meanwhile, the limitation of case load might be the reason why patients of Nevin stage V did not show a similar trend.
Studies discussing the prognostic difference between GB-ADC and GB-NEC and their results are inconsistent. Chen et al12 and Kim et al20 found patients with GB-NEC had significantly shorter survival time, which they believed was due to the higher percentage of patients with advanced stage and lymphatic metastases; however, Yun’s study did not support this hypothesis.29 We suspected confounding factors between two types of patients might affect the conclusion, therefore our study used propensity score matching to minimize the influence of other confounding factors including age, gender, TNM and Nevin stage, and treatment choice. However, GB-NEC still had worse overall survival than GB-ADC (3-year survival rate: 31.1% vs 63.8%, p<0.01), which demonstrated that further investigation such as genomic analysis is needed to explore underlying reasons.
Therapeutic options are limited in GB-NECs due to its malignancy and delayed diagnosis. Radical resection is the first treatment choice if it is available. More than 70% of our cases received surgery. But the deficient number of cases might be the reason why our study did not show this significance. For patients with unresectable GB-NECs, the primary therapy would be chemotherapy. Although it is believed that chemotherapy could not improve survival and can only be used as a palliative choice in previous studies,10,30 it seems that in our data patients who received chemotherapies showed no worse survival than resectable patients. Besides, a patient of TNM stage 4B had comprehensive treatments including chemotherapy, interventional therapy and radiotherapy and he survived 36 months, which is distinctly longer than median survival time. This case emphasized the importance of the multi-disciplinary team (MDT) and personalized treatment for advanced patients.
This study had some limitations. First, due to the scarcity of patients with GB-NECs, the case numbers were relatively insufficient, which led to the difficulty of multivariate analysis. Second, it was a retrospective research and the data collection was limited. Therefore, it is necessary to conduct a prospective study with larger sample size to analyze the clinical features of GB-NECs in an attempt to find better treatments.
Conclusion
We have presented the clinical characteristics of 15 cases of gallbladder neuroendocrine carcinoma (GB-NECs), which is one of the biggest studies of this rare disease in The People's Republic of China so far. The median overall survival was 20.4 months and the 1-, 2-, and 3-year survival rates were 60.0%, 38.8%, and 31.1%, respectively. The univariate analysis showed that Nevin stage helped classify patients with different prognoses: patients of Nevin stage III showed better overall survival than ones of Nevin stage IV, in accordance with the finding that lymph node metastasis is a strong negative prognostic factor for overall survival. Moreover, propensity score analysis demonstrated that after eliminating the effect of confounding factors like age, gender, Nevin stage and treatment choice, GB-NEC still had worse overall survival than gallbladder adenocarcinoma. In addition, early diagnosis and radical surgery are recommended, while as for unresectable patients, multi-disciplinary and personalized treatments may improve the prognosis. Case recruitment is needed in the future to further discuss other prognostic factors and find better treatments.
Statement of Ethics
The study protocol has been approved by the ethics committee of Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College Hospital on human research. Written informed consent was obtained from all patients.
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
National Natural Science Foundation of China (81672461). CAMS Innovation Fund for Medical Sciences (CIFMS) (Grant no.2017-12M-4-002).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kunz PL. Carcinoid and neuroendocrine tumors: building on success. J Clin Oncol. 2015;33(16):1855–1863. doi:10.1200/JCO.2014.60.2532
2. Oberndorfer S. Karzinoide tumoren des dunndarms. Frankf Z Pathol. 1907;1:426–432.
3. Hemminki K, Li X. Incidence trends and risk factors of carcinoid tumors: a nationwide epidemiologic study from Sweden. Cancer. 2001;92(8):2204–2210. doi:10.1002/(ISSN)1097-0142
4. Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97(4):934–959. doi:10.1002/(ISSN)1097-0142
5. Panzuto F, Nasoni S, Falconi M, et al. Prognostic factors and survival in endocrine tumor patients: comparison between gastrointestinal and pancreatic localization. Endocr Relat Cancer. 2005;12(4):1083–1092. doi:10.1677/erc.1.01017
6. Bosman FT, Hruban RH, Theise ND. WHO Classification of Tumours of the Digestive System. World Health Organization; 2010.
7. Rindi G, Kloppel G, Alhman H, et al. TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including a grading system. Virchows Arch. 2006;449(4):395–401. doi:10.1007/s00428-006-0250-1
8. Oberg K, Castellano D. Current knowledge on diagnosis and staging of neuroendocrine tumors. Cancer Metastasis Rev. 2011;30(Suppl 1):3–7. doi:10.1007/s10555-011-9292-1
9. Yao JC, Hassan M, Phan A, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26(18):3063–3072. doi:10.1200/JCO.2007.15.4377
10. Eltawil KM, Gustafsson BI, Kidd M, Modlin IM. Neuroendocrine tumors of the gallbladder: an evaluation and reassessment of management strategy. J Clin Gastroenterol. 2010;44(10):687–695. doi:10.1097/MCG.0b013e3181d7a6d4
11. Lee KJ, Cho JH, Lee SH, et al. Clinicopathological characteristics of biliary neuroendocrine neoplasms: a multicenter study. Scand J Gastroenterol. 2017;52(4):437–441. doi:10.1080/00365521.2016.1261938
12. Chen C, Wang L, Liu X, Zhang G, Zhao Y, Geng Z. Gallbladder neuroendocrine carcinoma: report of 10 cases and comparision of clinicopathologic features with gallbladder adenocarcinoma. Int J Clin Exp Pathol. 2015;8(7):8218–8226.
13. Kim J, Lee WJ, Lee SH, et al. Clinical features of 20 patients with curatively resected biliary neuroendocrine tumours. Dig Liver Dis. 2011;43(12):965–970. doi:10.1016/j.dld.2011.07.010
14. Kamboj M, Gandhi JS, Gupta G, et al. Neuroendocrine carcinoma of gall bladder: a series of 19 cases with review of literature. J Gastrointest Cancer. 2015;46(4):356–364. doi:10.1007/s12029-015-9745-9
15. Kanetkar AV, Patkar S, Khobragade KH, Ostwal V, Ramaswamy A, Goel M. Neuroendocrine carcinoma of gallbladder: a step beyond palliative therapy, experience of 25 cases. J Gastrointest Cancer. 2018;50(2):298–303.
16. Byrd DR, Carducci MA, Compton CC, Fritz AG, Greene FL. AJCC Cancer Staging Manual.
17. Nevin Je Fau - Moran TJ, Moran Tj Fau - Kay S, Kay S Fau - King R, King R. Carcinoma of the gallbladder: staging, treatment, and prognosis. Cancer. 1976;37(0008–543X(Print)):141–148. doi:10.1002/1097-0142(197601)37:1<141::AID-CNCR2820370121>3.0.CO;2-Y
18. Randi G, Franceschi S, La Vecchia C. Gallbladder cancer worldwide: geographical distribution and risk factors. Int J Cancer. 2006;118(7):1591–1602. doi:10.1002/(ISSN)1097-0215
19. Duffy A, Capanu M, Abou-Alfa GK, et al. Gallbladder cancer (GBC): 10-year experience at Memorial Sloan-Kettering Cancer Centre (MSKCC). J Surg Oncol. 2008;98(7):485–489. doi:10.1002/jso.v98:7
20. Kim T-H, Kim SH, Lee KB, Han JK. Outcome and CT differentiation of gallbladder neuroendocrine tumours from adenocarcinomas. Eur Radiol. 2016;27(2):507–517. doi:10.1007/s00330-016-4394-3
21. Pearse AGE. The cytochemistry and ultrastructure of polypeptide hormone-producing cells of the apud series and the embryologic, physiologic and pathologic implications of the concept. J Histochem Cytochem. 1969;17(5):303–313. doi:10.1177/17.5.303
22. Li M, Liu F, Zhang Y, et al. Whole-genome sequencing reveals the mutational landscape of metastatic small-cell gallbladder neuroendocrine carcinoma (GB-SCNEC). Cancer Lett. 2017;391:20–27. doi:10.1016/j.canlet.2016.12.027
23. Ahn JE, Byun JH, Ko MS, Park SH, Lee MG. Case report: neuroendocrine carcinoma of the gallbladder causing hyperinsulinaemic hypoglycaemia. Clin Radiol. 2007;62(4):391–394. doi:10.1016/j.crad.2006.11.016
24. Uribe-Uribe NO, Jimenez-Garduno AM, Henson DE, Albores-Saavedra J. Paraneoplastic sensory neuropathy associated with small cell carcinoma of the gallbladder. Ann Diagn Pathol. 2009;13(2):124–126. doi:10.1016/j.anndiagpath.2007.08.003
25. Lin D, Suwantarat N, Kwee S, Miyashiro M. Cushing’s syndrome caused by an ACTH-producing large cell neuroendocrine carcinoma of the gallbladder. World J Gastrointest Oncol. 2010;2(1):56–58. doi:10.4251/wjgo.v2.i1.56
26. Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas. 2010;39(6):707–712. doi:10.1097/MPA.0b013e3181ec124e
27. Janson ET, Sorbye H, Welin S, et al. Nordic guidelines 2010 for diagnosis and treatment of gastroenteropancreatic neuroendocrine tumours. Acta Oncol. 2010;49(6):740–756. doi:10.3109/0284186X.2010.492791
28. Fischer L, Kleeff J, Esposito I, et al. Clinical outcome and long-term survival in 118 consecutive patients with neuroendocrine tumours of the pancreas. Br J Surg. 2008;95(5):627–635. doi:10.1002/(ISSN)1365-2168
29. Yun SP. Clinical outcomes of small cell neuroendocrine carcinoma and adenocarcinoma of the gallbladder. World J Gastroenterol. 2015;21(1):269. doi:10.3748/wjg.v21.i1.269
30. Deehan DJ, Heys SD, Kernohan N, Eremin O. Carcinoid tumour of the gall bladder: two case reports and a review of published works. Gut. 1993;34(9):1274. doi:10.1136/gut.34.9.1274
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
