Back to Journals » OncoTargets and Therapy » Volume 13
Circular RNAs and Bladder Cancer
Received 22 June 2020
Accepted for publication 3 September 2020
Published 29 September 2020 Volume 2020:13 Pages 9573—9586
DOI https://doi.org/10.2147/OTT.S268859
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr XuYu Yang
Zhonglin Cai, Hongjun Li
Department of Urology, Peking Union Medical College Hospital, Peking Union Medical College, Chinese Academy of Medical Sciences, Beijing, People’s Republic of China
Correspondence: Hongjun Li Tel +86 10 69156034
Email [email protected]
Abstract: Bladder cancer (BC) is the most common urinary system malignancy and is a serious threat to human health. Circular RNAs (circRNAs) are members of a newly defined class of noncoding RNAs (ncRNAs) that can regulate gene expression at the transcriptional or posttranscriptional level. Studies have shown that circRNAs are related to the clinicopathological characteristics, prognosis, and chemosensitivity of BC, and basic research has further confirmed that changes in the expression of circRNAs in BC are closely related to various tumor biological functions. CircRNAs promote tumor development by interacting with miRNAs to regulate transcription factors and both classical and nonclassical tumor signaling pathways. The nonclassical signaling pathways are related to cell cycle progression, epithelial–mesenchymal transition (EMT), extracellular matrix maintenance, and tumor stem cell maintenance. In this article, the relationships between circRNAs and the clinical characteristics of BC are reviewed, and the molecular mechanisms by which circRNAs promote tumor development are explored.
Keywords: circRNA, miRNA, bladder cancer, biomarker
Introduction
Bladder cancer (BC) is the most common malignant tumor in the urinary system and ranks 9th in global cancer incidence and 13th in global cancer-related mortality.1,2 In 2018, an estimated 549,393 new cases were diagnosed,3 75% of which were non-muscle-invasive BC, and nearly 200,000 deaths from BC occurred.4 In China, the mortality rate has increased significantly in the past few decades.5 A high recurrence rate is a representative feature of BC.6 Given the high incidence, recurrence rate and progression rate of BC, its five-year survival rate is still very low. Currently, surgical resection and chemotherapy are the main treatment methods for BC, but postoperative recurrence and the emergence of chemoresistance greatly limit the therapeutic effect of these treatments. Therefore, identifying a safe and effective molecular marker and treatment strategy is a pressing problem requiring resolution.
Noncoding RNAs (ncRNAs) are functional transcripts that are not translated into proteins and can be divided into two categories based on size: small ncRNAs (18–200 nucleotides) and long ncRNAs (lncRNAs, more than 200 nucleotides). Small ncRNAs, also known as microRNAs (miRNAs, usually 22–25 nucleotides), bind to miRNA response elements (MREs) in the 3ʹ-untranslated region (UTR) of the target messenger RNA (mRNA) to either promote mRNA degradation or inhibit protein translation. Circular RNAs (circRNAs) are members of a newly defined class of ncRNAs with a covalently closed loop structure that has neither 5ʹ-3ʹ polarity nor a polyadenylated tail.7 These characteristics prevent them from being degraded by RNA exonucleases.8 Therefore, circRNAs are more stable than linear RNAs. In addition, circRNAs can regulate gene expression at the transcriptional or posttranscriptional level. This article will discuss the role of several important circRNAs in BC, aiming to highlight new research directions for the prevention and treatment of BC.
CircRNA Biogenesis
CircRNAs can be divided into three types: 1) exonic circRNA (EcircRNA), 2) intronic circRNA (ciRNA) and 3) exon-intron circRNA (EIciRNA),9–11 which are produced from exons, introns or both (Figure 1). EcircRNAs are located mainly in the cytoplasm, and ciRNAs and EIciRNAs are more abundant in the nucleus.12 Among these three types of circRNAs, more than 80% are EcircRNAs.12,13 CircRNAs are widely believed to be spliced at canonical splice sites in a manner dependent on backsplicing.14 After pre-mRNA processing events are slowed, nascent mRNAs are processed through alternative pathways to promote backsplicing.15 Looping of the intron sequence flanking the upstream splice acceptor site and the downstream splice donor site is the key step in circRNA formation.12 CircRNA looping occurs via three mechanisms: 1) base pairing of inverted complementary sequences in downstream and upstream introns,16,17 2) binding of RNA-binding proteins to flanking introns via specific motifs,18 and 3) lariat formation during exon skipping and internal backsplicing of the lariat.16,19 These mechanisms can bring the upstream splice acceptor site and the downstream splice donor site into close proximity, resulting in looping of the circRNA.12,14
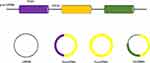 |
Figure 1 Three types of circRNAs produced from exons, introns or both. |
Relationships Among circRNAs, miRNAs and Target Genes
As a circular ncRNA, circRNAs are more stable than linear RNAs. An increasing number of scholars are referring to circRNAs as miRNA “sponges” because they competitively adsorb miRNAs owing to the presence of MREs, resulting in a reduction in the level of the target miRNA.20 miRNAs are endogenous single-stranded small ncRNAs of approximately 19–25 nucleotides in length.21 Recent literature has provided accumulating evidence that many miRNAs play roles similar to those of oncogenes or tumor suppressor genes and participate in the regulation of tumor cell proliferation, apoptosis, invasion, migration, angiogenesis, stem cell differentiation and other pathophysiological processes.22–24 miRNAs inhibit posttranscriptional translation or degrade target sequences by incompletely or completely pairing with the 3ʹ-UTR of the target mRNA, respectively, and these events regulate the expression of various cancer-related genes and thereby affect tumor biological characteristics.22–24 Therefore, the circRNA/miRNA/target gene regulatory axis plays an important role in the biological characteristics of tumors. By targeting any of these molecules, the tumor characteristics can be changed, suggesting new research avenues for tumor prevention and treatment.
Bladder Cancer-Associated circRNAs in Clinical Practice
CircRNAs were first discovered in RNA viruses, and were long considered byproducts of mRNA splicing.25 Advances in next-generation sequencing technology and bioinformatic approaches have led to the identification of thousands of circRNAs in different species.26 Researchers have conducted many studies aimed at elucidating the function of circRNAs in different tumors. Additionally, numerous circRNAs have been found to be differentially expressed in BC tissues and healthy adjacent tissues. These discovered circRNAs have a tremendous potential role in future BC research. The following section introduces the roles and mechanisms of circRNAs in BC.
Relationships Between circRNAs and Clinicopathological Characteristics
Research has shown that the BC-related functions of circRNAs can be divided into two natures: tumor promotion and tumor suppression. Many circRNAs, including circ-BPTF, circTFRC, circPDSS1, circZFR, circPRMT5, circPTK2, circASXL1, and circCASC15, have been found to have increased expression levels in BC tissues and cell lines compared to healthy adjacent tissues, normal tissues and normal cell lines and to play a role in promoting tumor development. Further analysis showed that high levels of these circRNAs are closely related to the clinicopathological stage of BC, which describes the tumor status, including the tumor size, presence of local and distant metastasis, and tumor pathological grade, and affect the survival of BC patients.27–34 Regarding tumor tissue classifications, the levels of circRNAs, including circ-BPTF, circTFRC, circZFR, circPTK2, and circASXL1, were found to be significantly higher in low-grade BC tissues than in high-grade BC tissues.27,28,30,32,33 Regarding the pathological TNM stage, circRNAs, including circTFRC, circZFR, circPRMT5, circPTK2, circASXL1 and circCASC15, were positively correlated with the T stage when patients were stratified into Ta–1 and T2–4 subgroups.28,30–34 Additionally, circTFRC, circZFR, circPRMT5, circPTK2 and circASXL1 were found to be closely related to lymph node status: BC patients with lymph node metastasis tended to have higher levels of these circRNAs.28,30–33 In addition, circASXL1 is associated with distant metastasis, and patients with BC with distant metastasis have higher levels of circASXL1.33 CircRNAs are related not only to pathological stage but also to clinical stage. For example, circPDSS1 levels differ across clinical stages of BC29—the higher the stage, the higher is the expression level. Clinically, BC tumors are divided into muscle-invasive and non-muscle-invasive tumors, and the former has worse outcomes. Compared with non-muscle-invasive BC tissue, muscle-invasive BC tissue has high levels of circ-BPTF.27 Tumor recurrence is an important factor affecting outcomes, and circ-BPTF and circZFR have been found to be closely related to the recurrence of BC; recurrent tumor tissue exhibits higher levels of circRNA than primary tumor tissue.27,30 In addition to the abovementioned findings regarding clinicopathological features, studies have shown that these circRNAs are not related to tumor size or vascular invasion. Because clinicopathological characteristics affect the prognosis of patients and circRNAs are closely related to the clinicopathological characteristics of BC, circRNAs may also be related to the prognosis of patients. Cumulative studies have confirmed that circ-BPTF, circTFRC, circZFR and circCASC15 are associated with overall survival in patients with BC;27,28,30,34 circZFR, circPRMT5 and circCASC15 are associated with disease-free survival;30,31,34 and hsa_circ_0137439 is associated with recurrence-free survival and overall survival.35 In general, higher levels of these circRNAs are associated with less favorable patient survival.
Cumulative studies have shown that many of the circRNAs discussed below act as tumor suppressors to inhibit BC. The expression levels of these circRNAs in primary BC tissues and BC cell lines are significantly reduced compared to those in adjacent tissues or normal cell lines. Moreover, the expression levels of these circRNAs are significantly negatively correlated with clinicopathological characteristics. CircFNDC3B, circACVR2A, circRIP2, circSLC8A1, circHIPK3, hsa_circ_0018069, circPICALM and circ-ZKSCAN1 are associated with pathological T stage.36–43 As the T stage advances, the expression levels of these circRNAs decrease significantly.36–43 Additionally, the expression levels of circFNDC3B, hsa_circ_0071662, circACVR2A, circRIP2, circHIPK3, circPICALM, circ-ZKSCAN1 and circMTO1 in patients with lymph node metastasis were found to be lower than those in patients with negative lymph nodes.36–38,40,42–45 Similarly, the expression levels of hsa_circ_0071662 and CircRIP2 in patients with distant metastasis and of CircHIPK3 in patients with vascular invasion were found to be lower than those in patients without distant metastasis and vascular invasion, respectively.38,40,44 Compared with low-grade bladder tumors, high-grade bladder tumors were found to exhibit lower levels of circFNDC3B, circACVR2A, circSLC8A1, circHIPK3, hsa_circ_0018069, circPICALM, circ-ZKSCAN1, hsa_circ_0077837 and hsa_circ_0004826.36,37,39–43,46 Additionally, the hsa_circ_0018069 expression level in muscle-invasive BC was found to be higher than that in non-muscle-invasive BC.41 Regarding survival, low levels of circFNDC3B, hsa_circ_0071662, circACVR2A, circRIP2, circPICALM, circ-ZKSCAN1 and circMTO1 are related to poor overall survival;36–38,42–45 similarly, circ-ZKSCAN1 and circMTO1 are positively correlated to disease-free survival;43,45 and hsa_circ_0077837 and hsa_circ_0004826 are also positively correlated with recurrence-free survival.46 In summary, regardless of whether the levels of circRNAs in BC tissue are increased or decreased, these circRNAs are closely related to clinicopathological characteristics (Table 1).
 |
Table 1 CircRNAs and Clinical Characteristics |
The close relationship between the expression levels of circRNAs and the clinicopathological characteristics of BC suggests that circRNAs will become diagnostic markers and prognostic predictors. However, molecular testing of tissues still has limitations for clinical application. Interestingly, circRNA expression in serum or urine has been found to be useful in evaluating clinicopathological features. In addition to identifying bladder tumors, hsa_circ_0137439 levels in urine can distinguish muscle-invasive from non-muscle-invasive BC.35 Serum has_circ_0003221 and hsa_circ_0000285 levels can also be used to identify bladder tumors,32,47 and the elevated serum levels of circFARSA, circSHKBP1 and circBANP in recurrent BC suggest that circRNAs can be used to verify tumor recurrence.48 In conclusion, the ability to detect the expression levels of circRNAs in serum and urine further increases the possibility of utilizing circRNAs as diagnostic and prognostic markers in BC.
circRNAs and Chemosensitivity
Chemotherapy is an important component of cancer treatment. Methyl jasmonate is a cyclopentanoic acid compound originally isolated from jasmine plants and is the most active anticancer derivative of natural jasmonate.49 Recently, an increasing number of basic studies have confirmed that methyl jasmonate exerts notable antitumor effects in many cancers, including colorectal cancer, non-small-cell lung cancer, gastric cancer, and prostate cancer, by inducing cancer cell apoptosis and blocking invasion, migration, and angiogenesis. Methyl jasmonate may thus be a promising new anticancer drug.50–53 One study showed that methyl jasmonate induces the expression of circRNA BCRC-3, which is derived from the PSMD1 gene. Overexpressed circRNA BCRC-3 in BC cells sponges miR-182-5p, thereby increasing the expression level of P27 (also called cell cycle-dependent kinase inhibitor 1B); this ultimately results in cell cycle inhibition in tumor cells and exerts an antitumor effect.54 Similarly, gambogic acid (GA), a natural product from Gambia, has been shown to perform an antitumor function in various tumors. In BC, GA induces the expression of circRNA BCRC4, which is expressed at low levels in BC. Increased expression of circRNA BCRC4 enhances the expression of miR-101 and subsequently downregulates EZH2 (which conventionally promotes tumor progression).55 Eventually, the sensitivity of BC cells to apoptosis is enhanced.55 In summary, antitumor drugs induce changes in the expression of circRNAs and thus inhibit tumor progression.
The sensitivity of tumors to chemotherapy can affect the survival and prognosis of patients. Dysregulation of circRNAs in BC induces chemoresistance. For example, the expression level of circELP3 is increased in bladder tumors, which consequently increases cisplatin resistance.56 Overexpression of circFNTA can regulate FNTA and KRA by interacting with miR-370-3p and increasing the chemotherapeutic resistance of BC to cisplatin.57 CircRNA Cdr1as mitigates APAF1 activity by binding to miR-1270, thereby increasing the sensitivity of cells to cisplatin.58 The ABC transport pathway is an important mechanism mediating tumor chemoresistance. Activity of this pathway reduces drug sensitivity by reducing the accumulation of drugs in tumor cells. Several studies have reported that the expression of ABC transport proteins in BC is upregulated, which can mediate multidrug resistance in tumors.59–64 Studies have shown that hsa_circ_102336, which is highly expressed in BC, binds to and inhibits miR-515-5p, thereby regulating the ABC transport pathway and subsequently the resistance of BC to cisplatin.65 In addition to affecting cisplatin resistance, circHIPK3 expression was found to be negatively correlated with gemcitabine resistance in bladder tumors, and overexpression of circHIPK3 was found to result in increased resistance of bladder tumors to gemcitabine.66 Additionally, hsa_circ_0000285 can promote chemosensitivity to cisplatin in BC. Compared with patients with chemosensitive bladder tumors, those with chemoresistant bladder tumors have lower expression of hsa_circ_0000285, suggesting that hsa_circ_0000285 increases chemosensitivity in BC.47 The above studies indicate that circRNAs are involved in the drug resistance of bladder tumors (Table 2) and will become important targets for reversing tumor drug resistance.
 |
Table 2 CircRNAs and Chemotherapy Drugs |
CircRNAs in Regulating the Biological Activity of Bladder Cancer
Cumulative studies have found that circRNAs interact with miRNAs to regulate the expression of target molecules and play an important role in the development of various biological characteristics of BC, including angiogenesis, invasion, metastasis, and chemoresistance.37,67–72 Understanding the mechanisms by which circRNAs affect the abovementioned biological functions of BC will help to identify important therapeutic targets.
Classical Tumor Signaling Pathways
Classical tumor signaling pathways are major pathways regulating the biological activity of tumors to promote tumor development and progression. Accumulating studies have shown that circRNAs are involved in classical tumor signaling pathways by interacting with miRNAs (Figure 2).
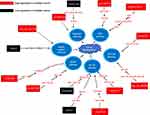 |
Figure 2 The detailed mechanisms of circRNAs on tumor classic pathways. |
The PI3K/AKT/mTOR pathway plays a key role in cell survival, cell physiology, and pathological changes, and activation of this signaling pathway can promote tumor progression.73 PTEN is a negative regulator of the PI3K/AKT/mTOR pathway, and it can regulate cell growth, proliferation, energy metabolism, and the immune response.74,75 CircZNF139 is highly expressed in BC cells and promotes the development of BC through the PI3K/AKT signaling pathway.76 The decreased expression of circSLC8A1 in BC reduces its interaction with miR-130b/miR-494, resulting in decreased PTEN expression and, consequently, increased activity of the downstream PI3K-AKT/PKB signaling pathway.39 In BC, PTEN is also regulated by circ-ITCH, and low expression of circ-ITCH reduces its interaction with miR-17 or miR-224, thereby increasing the PTEN level.77
Extracellular signal-regulated kinase (ERK) is generally considered a key regulator of biological activity in cells.78 It plays a key role in regulating various cellular functions, for example, promoting cell proliferation, differentiation and survival in response to extracellular signals.78 In addition, cumulative studies have shown that activation of ERK-related signaling leads to tumor formation and progression and is considered a promising target for tumor treatment.79–81 Elevated expression of circRNA MYLK in BC enhances sponging of miR-29a to activate VEGFA/VEGFR2 and the downstream Ras/ERK signaling pathway, which promotes tumor development.82 In addition, circRNA001418 is overexpressed in BC, thus reducing the miR-1297 level; this reduction results in an increase in EphA2 expression to activate the downstream ERK1/ERK2 signaling pathway, which promotes tumor progression.83
The NOTCH pathway has been shown to play both tumor-promoting and tumor-suppressing roles. In tumors, NOTCH1 functions as a tumor suppressor, while NOTCH2 acts as an oncogene that promotes tumor cell proliferation and metastasis through various mechanisms, including epithelial–mesenchymal transition (EMT) and cell cycle regulation.84 Overexpression of circKIF4A in BC can promote tumor development by sponging miR-375/1231, which consequently upregulates NOTCH2 expression.85 Similarly, hsa_circ_0008532 is overexpressed in BC and directly interacts with miR-155-5p and miR-330-5p to upregulate the MTGR1 expression and the downstream NOTCH signaling pathway.86
NF-κB can induce regulatory signals in response to different stimuli and has been shown to play a key regulatory role in the physiological and pathological processes of various diseases, including cancer.87 Compared with normal urethral epithelium, superficial BC tissue exhibits increased levels of NF-κB, while invasive BC tissue exhibits decreased levels; thus, the NF-κB-related pathway may function as a “double-edged sword” in tumor development.88 Circ-BPTF, which is highly expressed in BC, inhibits the oncogene RAB27A by directly interacting with miR-31-5p and weakening its inhibition of RAB27A, an action related to the regulation of NF-κB.28 CircUBXN7 inhibits miR-1247-3p and upregulates the B4GALT3 expression, which can inhibit NF-κB expression.89 Low expression of circUBXN7 in BC could thus result in increased NF-κB signaling to promote tumor progression.89
In addition to the abovementioned pathways, other classical pathways have been found to be regulated through circRNAs in BC. For example, circCEP128, which is highly expressed in BC, inhibits the expression of miR-145-5p, thereby upregulating MYD88 and other downstream proteins in the MAPK signaling pathway.90 In addition, overexpression of hsa_circ_0017247 in BC increases Wnt/β-catenin signaling activity and promotes tumor progression.91 These two pathways are involved in the development of numerous types of tumors. Additionally, some circRNAs are involved in more than one tumor signaling pathway in BC. The low expression of circRNA CiRs-6 in BC reduces March 1 expression by interfering with the interaction between miR-653 and March1, which can activate the NF-κB and Wnt/β-catenin signaling pathways.92 CircINTS4, which is highly expressed in BC, promotes the expression of CARMA4 by sponging miR-146b, thereby activating the NF-κB signaling pathway and inhibiting p38-MAPK signaling.93 In summary, the classical tumor signaling pathway is important and involved in the regulation of tumorigenesis and tumor progression. In BC, circRNAs are closely associated with classical tumor signaling pathways and contribute to the regulation of BC progression.
Nonclassical Tumor Pathways
In addition to classical tumor pathways, many circRNA-related pathways in BC have been found to be involved in the regulation of the cell cycle, EMT, extracellular matrix, and tumor stem cell-related functions in BC, resulting in promotion of tumor cell malignancy (Figure 3).
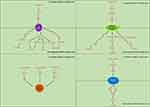 |
Figure 3 The detailed mechanisms of circRNAs on cell cycle, epithelial–mesenchymal transition, tumor stem cells and extracellular matrix. |
Regulation of the Cell Cycle
Tumor cells actively proliferate, and the cell cycle is closely related to cell proliferation. The cell cycle is regulated mainly by cyclins, cyclin-dependent protein kinases (CDKs), and CDK inhibitors.94 Many circRNAs have been found to be involved in the cell cycle in BC cells. Overexpression of circNR3C1 in BC was found to reduce the expression of cyclinD1 via sponging of miR-27a-3p; a similar relationship was found between hsa_circ_0058063, CDK6, and miR-145-5p.95,96 Therefore, low expression of circNR3C1 and hsa_circ_0058063 in BC accelerate the G1-to-S phase transition of tumor cells and enhance cell proliferation owing to decreased sponging of their target miRNAs and subsequently increased expression of cyclinD1 and CDK6, respectively.95,96 High expression levels of p21, a cyclin-dependent kinase inhibitor, can induce cell cycle arrest. The expression of p21 in BC tissues was significantly reduced compared with that in adjacent normal tissues.97 Studies have shown that circRNAs are involved in regulating p21 expression. In BC, reductions in the Cdr1as, circ-ZKSCAN1, and circ-ITCH expression levels weakened p21 activity via reducing binding to their respective target miRNAs (miR-135a, miR-1178-3p, miR-17, and miR-224), which ultimately led to downregulated expression of p21.77,98 The KIF2C protein is involved in the inhibition of cell cycle progression and mitosis.99 It is localized mainly in the cytoplasm throughout the cell cycle and participates in the DNA repair process.99 Studies have shown that KIF2C is associated with tumor progression.100–102 In BC, elevated expression of circRGNEF increases sponging of miR-548, which prevents the expression of its targetKIF2C and consequently promotes tumor progression.103 In summary, in BC, different circRNAs participate in the regulation of different cell cycle-related proteins, ultimately leading to accelerated tumor proliferation.
Regulation of EMT
The invasion and migration of tumor cells are closely related to EMT.104 CircRNAs also participate in tumor cell invasion and migration by regulating EMT.104 CircRNA-MYLK, which is highly expressed in BC, was found to sponge miR-29a to activate VEGFA/VEGFR signaling and downstream Ras/ERK pathways to promote EMT.82 In addition, VEGFA expression is increased via sponging of miR-205-05p by upregulated circ0001429 in BC.105 In addition to increased expression of VEGFA, increased expression of VEGF also promotes EMT in BC.40 CircHIPK3 was found to be expressed at low levels in BC, and circHIPK3 inhibits HPSE expression by sponging miR-558; therefore, the expression of the target mRNA VEGF is suppressed, resulting in inhibition of EMT.40 Low expression of circHIPK3 in BC increases the expression of VEGF and promotes EMT.40 FAK is a nonreceptor tyrosine kinase that contains multiple tyrosine phosphorylation sites, and phosphorylated FAK contributes to downstream signal transduction.106 Robust activation of the FAK signaling pathway is generally believed to promote EMT progression in tumors.107 One study showed that circPICALM sponges miR-1265 to promote the expression of its target STEAP4, and overexpressed STEAP4 can bind FAK to prevent autophosphorylation of the Y397 phosphorylation site on FAK and consequently inhibit EMT in bladder tumors.42 In BC, circPICALM is expressed at low levels; thus, EMT is promoted through the above mechanism.42 In addition, FAK is regulated by circFNDC3B via its interaction with miR-1178-3p, resulting in inhibition of G3BP2 and its downstream SRC/FAK signaling pathway.36 Low expression of circFNDC3B in BC abrogates its inhibitory effect on SRC/FAK.36 However, data regarding the effect of circFNDC3B on EMT are lacking, but it has been speculated that circFNDC3B affects EMT via FAK signaling. Low expression of circMTO1 in BC results in reduced binding of circMTO1 to miR-221, which alleviates the inhibition of the E-cadherin/N-cadherin pathways and subsequently promotes both EMT and the invasion and migration of bladder tumors.45 In summary, in bladder tumors, different circRNAs contribute to metastasis by inducing increases in the expression of EMT-related molecules, thereby promoting EMT.
Regulation of Tumor Stem Cells
Cancer stem cells retain the ability to drive tumor formation, metastasis, and drug resistance. Because of these characteristics, tumors are difficult to eliminate. CircRNA_103809 (hsa_circ_0072088) is essential in the maintenance of BC stem cell functions, including self-renewal and invasion and migration ability, by directly interacting with miR-511.108 CircGprc5a is upregulated in BC and BC stem cells.109 In BC stem cells, circGprc5a interacts with peptides and binds to Gprc5a, a key factor in bladder CSCs, in a peptide-dependent manner.109 Gprc5a is highly expressed in BC stem cells and promotes their self-renewal and metastasis.109 Expression of hsa_circ_0068307 is upregulated in tumor cell lines, and inhibiting the expression of this circRNA can slow cell invasion and proliferation.110 In BC stem cells, hsa_circ_0068307 knockdown upregulates miR-147 expression, and resulting in inhibition of c-Myc expression to hinder the differentiation of BC stem cells.110 In summary, circRNAs play an important role in BC stem cell maintenance and are important candidate molecules for eliminating BC stem cells and treating BC.
Regulation of Extracellular Matrix Maintenance
The extracellular matrix is an important component of the tumor microenvironment. Many studies have confirmed that the extracellular matrix has an important effect on tumor growth, invasion, migration, angiogenesis, and other behaviors.111–113 One report indicated that circHIPK3 inhibits HPSE expression by interacting with miR-558.40 However, in BC, a decrease in circHIPK3 expression leads to an increase in HPSE expression, which promotes the expression of MMP9 and VEGF to remodel the extracellular matrix and thereby advance tumor development.40 In addition, in BC, elevated expression of hsa_circ_0001361 upregulates MMP9 expression, which may affect the extracellular matrix by interacting with miR-491-5p.114 In summary, in BC, circRNAs have important roles in maintaining the extracellular matrix and promote tumor development by changing its composition.
Regulation of Transcription Factors
The literature discussed below indicates that in BC cells, circRNAs are involved in regulating the activity of transcription factors that control the expression of molecules related to tumor biology (Table 3). Myc, as a transcription factor, was found to be upregulated by hsa_circ_0067934 in BC via its sponging of miR-1304, and increased Myc expression activates growth factor-related molecules, such as VEGFR.115 As a member of the myc gene family, c-Myc is regulated by circCDYL in BC via direct interactions of circCDYL with miR-105-5p and activates cell cycle-related molecules.116 The current literature describes many circRNAs that have been found to be involved in regulating the activity of transcription factors, including transcriptional activators and repressors, in BC, but detailed information about the molecular transcripts that are regulated by these transcription factors is lacking. For example, in BC, circCEP128 and circDOCK1 are involved in mediating the expression of the Sox family transcription factors Sox11 and Sox5 by sponging miR-145-5p and hsa-miR-132-3p, respectively.117,118 The Sox family of proteins comprises transcriptional regulators involved in the development of various tissues.119–121 In addition, the KLF family comprises a group of evolutionarily conserved transcription factors that regulate a series of cellular processes, such as cell proliferation, apoptosis, differentiation, and metabolism.122–125 CircFUT8 and circPTPRA are involved in regulating the KLF family members KLF10 and KLF9 by directly binding to miR-570-3p and miR-636, respectively.126,127 CircRNA_0058063 (hsa_circ_0058063) directly interacts with miR-486-3p to regulate the levels of the FOXP4 transcription factor, and circCASC15 promotes the activation of CREB1 by sponging miR-1224-5p.34,128 Low expression of circFAM114A2 in BC weakens miR-762-mediated inhibition of ΔNP63, a major isomer of the transcription factor TP63.129 High expression of hsa_circ_0000144 in BC increases its direct binding to miR-217, thereby abrogating its inhibitory effect on the transcription factor RUNX2 and subsequently increasing RNX2 expression.130 In summary, circRNAs participate in the regulation of transcription factors related to BC tumors, but most research has not focused on which transcript is affected by these transcription factors; therefore, more in-depth studies are required.
 |
Table 3 CircRNAs and Transcript Factors |
Conclusions
An increasing number of studies have shown that some circRNAs are differentially expressed in BC tissues and that their dysregulation is related to the clinicopathological characteristics of BC patients. By acting as transcriptional regulators, these circRNAs are associated with the expression of target miRNAs and proteins related to BC cell proliferation, invasion, and metastasis and contribute to the development of BC. Given their unique advantages, such as superior stability, higher abundance, and presence in different body fluids, circRNAs are promising diagnostic and prognostic biomarkers as well as potential therapeutic targets for BC. In addition, circRNAs are involved in the development of tumor chemoresistance, although the detailed mechanisms of circRNAs in BC chemoresistance are unknown. Clarifying the detailed mechanism of chemoresistance is a highly important task to improve BC prevention and treatment. We believe that the discovery of additional circRNAs will lead to a new era of gene-targeted therapy.
Data Sharing Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This work is supported by the grant from National Natural Science Foundation of China (Grant No. 81871152).
Disclosure
The authors declare that they have no competing interests for this work. Disclosure of potential conflicts of interest: None.
References
1. Ploeg M, Aben KK, Kiemeney LA. The present and future burden of urinary bladder cancer in the world. World J Urol. 2009;27(3):289–293. doi:10.1007/s00345-009-0383-3
2. Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12):2893–2917. doi:10.1002/ijc.25516
3. Zhu G, Pei L, Li Y, Gou X. EP300 mutation is associated with tumor mutation burden and promotes antitumor immunity in bladder cancer patients. Aging. 2020;12(3):2132–2141. doi:10.18632/aging.102728
4. Witjes JA, Bruins HM, Cathomas R, et al. European association of urology guidelines on muscle-invasive and metastatic bladder cancer: summary of the 2020 guidelines. Eur Urol. 2020. doi:10.1016/j.eururo.2020.03.055
5. Pang C, Guan Y, Li H, Chen W, Zhu G. Urologic cancer in China. Jpn J Clin Oncol. 2016;46(6):497–501. doi:10.1093/jjco/hyw034
6. Zhao F, Lin T, He W, et al. Knockdown of a novel lincRNA AATBC suppresses proliferation and induces apoptosis in bladder cancer. Oncotarget. 2015;6(2):1064–1078. doi:10.18632/oncotarget.2833
7. Chen LL, Yang L. Regulation of circRNA biogenesis. RNA Biol. 2015;12(4):381–388. doi:10.1080/15476286.2015.1020271
8. Suzuki H, Tsukahara T. A view of pre-mRNA splicing from RNase R resistant RNAs. Int J Mol Sci. 2014;15(6):9331–9342. doi:10.3390/ijms15069331
9. Jeck WR, Sorrentino JA, Wang K, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA. 2013;19(2):141–157. doi:10.1261/rna.035667.112
10. Salzman J, Chen RE, Olsen MN, Wang PL, Brown PO. Cell-type specific features of circular RNA expression. PLoS Genet. 2013;9(9):e1003777. doi:10.1371/journal.pgen.1003777
11. Zhang Y, Zhang XO, Chen T, et al. Circular intronic long noncoding RNAs. Mol Cell. 2013;51(6):792–806. doi:10.1016/j.molcel.2013.08.017
12. Huang A, Zheng H, Wu Z, Chen M, Huang Y. Circular RNA-protein interactions: functions, mechanisms, and identification. Theranostics. 2020;10(8):3503–3517. doi:10.7150/thno.42174
13. Zaiou M. Circular RNAs as potential biomarkers and therapeutic targets for metabolic diseases. Adv Exp Med Biol. 2019;1134:177–191.
14. Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J. The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet. 2019;20(11):675–691. doi:10.1038/s41576-019-0158-7
15. Liang D, Tatomer DC, Luo Z, et al. The output of protein-coding genes shifts to circular RNAs when the pre-mRNA processing machinery is limiting. Mol Cell. 2017;68(5):940–954 e3. doi:10.1016/j.molcel.2017.10.034
16. Kelly S, Greenman C, Cook PR, Papantonis A. Exon skipping is correlated with exon circularization. J Mol Biol. 2015;427(15):2414–2417. doi:10.1016/j.jmb.2015.02.018
17. Zhang XO, Wang HB, Zhang Y, Lu X, Chen LL, Yang L. Complementary sequence-mediated exon circularization. Cell. 2014;159(1):134–147. doi:10.1016/j.cell.2014.09.001
18. Conn SJ, Pillman KA, Toubia J, et al. The RNA binding protein quaking regulates formation of circRNAs. Cell. 2015;160(6):1125–1134. doi:10.1016/j.cell.2015.02.014
19. Eger N, Schoppe L, Schuster S, Laufs U, Boeckel JN. Circular RNA splicing. Adv Exp Med Biol. 2018;1087:41–52.
20. Sen R, Ghosal S, Das S, Balti S, Chakrabarti J. Competing endogenous RNA: the key to posttranscriptional regulation. ScientificWorldJournal. 2014;2014:896206. doi:10.1155/2014/896206
21. Li X, Xu M, Ding L, Tang J. MiR-27a: a novel biomarker and potential therapeutic target in tumors. J Cancer. 2019;10(12):2836–2848. doi:10.7150/jca.31361
22. Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017;16(3):203–222. doi:10.1038/nrd.2016.246
23. Acunzo M, Romano G, Wernicke D, Croce CM. MicroRNA and cancer–a brief overview. Adv Biol Regul. 2015;57:1–9. doi:10.1016/j.jbior.2014.09.013
24. Sun Z, Shi K, Yang S, et al. Effect of exosomal miRNA on cancer biology and clinical applications. Mol Cancer. 2018;17(1):147. doi:10.1186/s12943-018-0897-7
25. Sanger HL, Klotz G, Riesner D, Gross HJ, Kleinschmidt AK. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc Natl Acad Sci U S A. 1976;73(11):3852–3856. doi:10.1073/pnas.73.11.3852
26. Lopez-Jimenez E, Rojas AM, Andres-Leon E. RNA sequencing and prediction tools for circular RNAs analysis. Adv Exp Med Biol. 2018;1087:17–33.
27. Wang Y, Mo Y, Gong Z, et al. Circular RNAs in human cancer. Mol Cancer. 2017;16(1):25. doi:10.1186/s12943-017-0598-7
28. Bi J, Liu H, Cai Z, et al. Circ-BPTF promotes bladder cancer progression and recurrence through the miR-31-5p/RAB27A axis. Aging. 2018;10(8):1964–1976. doi:10.18632/aging.101520
29. Yu Q, Liu P, Han G, Xue X, Ma D. CircRNA circPDSS1 promotes bladder cancer by down-regulating miR-16. Biosci Rep. 2020;40(1). doi:10.1042/BSR20191961
30. Zhang WY, Liu QH, Wang TJ, Zhao J, Cheng XH, Wang JS. CircZFR serves as a prognostic marker to promote bladder cancer progression by regulating miR-377/ZEB2 signaling. Biosci Rep. 2019;39(12). doi:10.1042/BSR20192779
31. Chen X, Chen RX, Wei WS, et al. PRMT5 circular RNA promotes metastasis of urothelial carcinoma of the bladder through sponging miR-30c to induce epithelial-mesenchymal transition. Clin Cancer Res. 2018;24(24):6319–6330. doi:10.1158/1078-0432.CCR-18-1270
32. Xu ZQ, Yang MG, Liu HJ, Su CQ. Circular RNA hsa_circ_0003221 (circPTK2) promotes the proliferation and migration of bladder cancer cells. J Cell Biochem. 2018;119(4):3317–3325. doi:10.1002/jcb.26492
33. Tang G, Xie W, Qin C, et al. Expression of circular RNA circASXL1 correlates with TNM classification and predicts overall survival in bladder cancer. Int J Clin Exp Pathol. 2017;10(8):8495–8502.
34. Zhuang C, Huang X, Yu J, Gui Y. Circular RNA hsa_circ_0075828 promotes bladder cancer cell proliferation through activation of CREB1. BMB Rep. 2020;53(2):82–87. doi:10.5483/BMBRep.2020.53.2.059
35. Song Z, Zhang Q, Zhu J, Yin G, Lin L, Liang C. Identification of urinary hsa_circ _0137439 as potential biomarker and tumor regulator of bladder cancer. Neoplasma. 2020;67(1):137–146. doi:10.4149/neo_2018_181214N970
36. Liu H, Bi J, Dong W, et al. Invasion-related circular RNA circFNDC3B inhibits bladder cancer progression through the miR-1178-3p/G3BP2/SRC/FAK axis. Mol Cancer. 2018;17(1):161. doi:10.1186/s12943-018-0908-8
37. Dong W, Bi J, Liu H, et al. Circular RNA ACVR2A suppresses bladder cancer cells proliferation and metastasis through miR-626/EYA4 axis. Mol Cancer. 2019;18(1):95. doi:10.1186/s12943-019-1025-z
38. Su Y, Feng W, Shi J, Chen L, Huang J, Lin T. circRIP2 accelerates bladder cancer progression via miR-1305/Tgf-beta2/smad3 pathway. Mol Cancer. 2020;19(1):23. doi:10.1186/s12943-019-1129-5
39. Lu Q, Liu T, Feng H, et al. Circular RNA circSLC8A1 acts as a sponge of miR-130b/miR-494 in suppressing bladder cancer progression via regulating PTEN. Mol Cancer. 2019;18(1):111. doi:10.1186/s12943-019-1040-0
40. Li Y, Zheng F, Xiao X, et al. CircHIPK3 sponges miR-558 to suppress heparanase expression in bladder cancer cells. EMBO Rep. 1646-1659;2017(18):9.
41. Li M, Wang Y, Liu Y, Zhang X, Liu J, Wang P. Low expression of hsa_circ_0018069 in human bladder cancer and its clinical significance. Biomed Res Int. 2019;2019:9681863.
42. Yan D, Dong W, He Q, et al. Circular RNA circPICALM sponges miR-1265 to inhibit bladder cancer metastasis and influence FAK phosphorylation. EBioMedicine. 2019;48:316–331. doi:10.1016/j.ebiom.2019.08.074
43. Bi J, Liu H, Dong W, et al. Circular RNA circ-ZKSCAN1 inhibits bladder cancer progression through miR-1178-3p/p21 axis and acts as a prognostic factor of recurrence. Mol Cancer. 2019;18(1):133. doi:10.1186/s12943-019-1060-9
44. Abulizi R, Li B, Zhang CG. Circ_0071662, a novel tumor biomarker, suppresses bladder cancer cell proliferation and invasion by sponging miR-146b-3p. Oncol Res. 2019. doi:10.3727/096504019X15740729375088
45. Li Y, Wan B, Liu L, Zhou L, Zeng Q. Circular RNA circMTO1 suppresses bladder cancer metastasis by sponging miR-221 and inhibiting epithelial-to-mesenchymal transition. Biochem Biophys Res Commun. 2019;508(4):991–996. doi:10.1016/j.bbrc.2018.12.046
46. Shen C, Wu Z, Wang Y, et al. Downregulated hsa_circ_0077837 and hsa_circ_0004826, facilitate bladder cancer progression and predict poor prognosis for bladder cancer patients. Cancer Med. 2020;9(11):3885–3903. doi:10.1002/cam4.3006
47. Chi BJ, Zhao DM, Liu L, et al. Downregulation of hsa_circ_0000285 serves as a prognostic biomarker for bladder cancer and is involved in cisplatin resistance. Neoplasma. 2019;66(2):197–202. doi:10.4149/neo_2018_180318N185
48. Pan J, Xie X, Li H, Li Z, Ren C, Ming L. Detection of serum long non-coding RNA UCA1 and circular RNAs for the diagnosis of bladder cancer and prediction of recurrence. Int J Clin Exp Pathol. 2019;12(8):2951–2958.
49. Sucu BO, Ipek OS, Kurtulus SO, Yazici BE, Karakas N, Guzel M. Synthesis of novel methyl jasmonate derivatives and evaluation of their biological activity in various cancer cell lines. Bioorg Chem. 2019;91:103146. doi:10.1016/j.bioorg.2019.103146
50. Raviv Z, Zilberberg A, Cohen S, et al. Methyl jasmonate down-regulates survivin expression and sensitizes colon carcinoma cells towards TRAIL-induced cytotoxicity. Br J Pharmacol. 2011;164(5):1433–1444. doi:10.1111/j.1476-5381.2011.01419.x
51. Zhang M, Su L, Xiao Z, Liu X, Liu X. Methyl jasmonate induces apoptosis and pro-apoptotic autophagy via the ROS pathway in human non-small cell lung cancer. Am J Cancer Res. 2016;6(2):187–199.
52. Ribera-Fonseca A, Jimenez D, Leal P, et al. The anti-proliferative and anti-invasive effect of leaf extracts of blueberry plants treated with methyl jasmonate on human gastric cancer in vitro is related to their antioxidant properties. Antioxidants. 2020;9(1):45.
53. Farooqi AA, Butt G, Razzaq Z. Algae extracts and methyl jasmonate anti-cancer activities in prostate cancer: choreographers of ‘the dance macabre’. Cancer Cell Int. 2012;12(1):50. doi:10.1186/1475-2867-12-50
54. Xie F, Li Y, Wang M, et al. Circular RNA BCRC-3 suppresses bladder cancer proliferation through miR-182-5p/p27 axis. Mol Cancer. 2018;17(1):144. doi:10.1186/s12943-018-0892-z
55. Li B, Xie F, Zheng FX, Jiang GS, Zeng FQ, Xiao XY. Overexpression of CircRNA BCRC4 regulates cell apoptosis and MicroRNA-101/EZH2 signaling in bladder cancer. J Huazhong Univ Sci Technolog Med Sci. 2017;37(6):886–890.
56. Su Y, Yang W, Jiang N, et al. Hypoxia-elevated circELP3 contributes to bladder cancer progression and cisplatin resistance. Int J Biol Sci. 2019;15(2):441–452. doi:10.7150/ijbs.26826
57. Chen J, Sun Y, Ou Z, et al. Androgen receptor-regulated circFNTA activates KRAS signaling to promote bladder cancer invasion. EMBO Rep. 2020;21(4):e48467. doi:10.15252/embr.201948467
58. Yuan W, Zhou R, Wang J, et al. Circular RNA Cdr1as sensitizes bladder cancer to cisplatin by upregulating APAF1 expression through miR-1270 inhibition. Mol Oncol. 2019;13(7):1559–1576. doi:10.1002/1878-0261.12523
59. Li H, Krstin S, Wang S, Wink M. Capsaicin and piperine can overcome multidrug resistance in cancer cells to doxorubicin. Molecules. 2018;23(3):557.
60. Stefan K, Schmitt SM, Wiese M. 9-deazapurines as broad-spectrum inhibitors of the ABC transport proteins P-glycoprotein, multidrug resistance-associated protein 1, and breast cancer resistance protein. J Med Chem. 2017;60(21):8758–8780. doi:10.1021/acs.jmedchem.7b00788
61. Nemcova-Furstova V, Kopperova D, Balusikova K, et al. Characterization of acquired paclitaxel resistance of breast cancer cells and involvement of ABC transporters. Toxicol Appl Pharmacol. 2016;310:215–228. doi:10.1016/j.taap.2016.09.020
62. Koshkin V, Krylov SN. Correlation between multi-drug resistance-associated membrane transport in clonal cancer cells and the cell cycle phase. PLoS One. 2012;7(7):e41368. doi:10.1371/journal.pone.0041368
63. Chen Y, Scully M, Petralia G, Kakkar A. Binding and inhibition of drug transport proteins by heparin: a potential drug transporter modulator capable of reducing multidrug resistance in human cancer cells. Cancer Biol Ther. 2014;15(1):135–145. doi:10.4161/cbt.27148
64. Choi YH, Yu AM. ABC transporters in multidrug resistance and pharmacokinetics, and strategies for drug development. Curr Pharm Des. 2014;20(5):793–807.
65. Gong P, Xu R, Zhuang Q, He X. A novel circular RNA (hsa_circRNA_102336), a plausible biomarker, promotes the tumorigenesis by sponging miR-515-5p in human bladder cancer. Biomed Pharmacother. 2020;126:110059. doi:10.1016/j.biopha.2020.110059
66. Xie F, Zhao N, Zhang H, Xie D. Circular RNA circHIPK3 promotes gemcitabine sensitivity in bladder cancer. J Cancer. 2020;11(7):1907–1912. doi:10.7150/jca.39722
67. Zeng Z, Zhou W, Duan L, et al. Circular RNA circ-VANGL1 as a competing endogenous RNA contributes to bladder cancer progression by regulating miR-605-3p/VANGL1 pathway. J Cell Physiol. 2019;234(4):3887–3896. doi:10.1002/jcp.27162
68. Yang C, Wu S, Wu X, Zhou X, Jin S, Jiang H. Silencing circular RNA UVRAG inhibits bladder cancer growth and metastasis by targeting the microRNA-223/fibroblast growth factor receptor 2 axis. Cancer Sci. 2019;110(1):99–106. doi:10.1111/cas.13857
69. Lin G, Sheng H, Xie H, et al. circLPAR1 is a novel biomarker of prognosis for muscle-invasive bladder cancer with invasion and metastasis by miR-762. Oncol Lett. 2019;17(3):3537–3547.
70. Zhang L, Xia HB, Zhao CY, Shi L, Ren XL. Cyclic RNA hsa_circ_0091017 inhibits proliferation, migration and invasiveness of bladder cancer cells by binding to microRNA-589-5p. Eur Rev Med Pharmacol Sci. 2020;24(1):86–96.
71. Mao W, Huang X, Wang L, et al. Circular RNA hsa_circ_0068871 regulates FGFR3 expression and activates STAT3 by targeting miR-181a-5p to promote bladder cancer progression. J Exp Clin Cancer Res. 2019;38(1):169. doi:10.1186/s13046-019-1136-9
72. Jiang Y, Wei T, Li W, Zhang R, Chen M. Circular RNA hsa_circ_0002024 suppresses cell proliferation, migration, and invasion in bladder cancer by sponging miR-197-3p. Am J Transl Res. 2019;11(3):1644–1652.
73. Aoki M, Fujishita T. Oncogenic roles of the PI3K/AKT/mTOR axis. Curr Top Microbiol Immunol. 2017;407:153–189.
74. Keppler-Noreuil KM, Parker VE, Darling TN, Martinez-Agosto JA. Somatic overgrowth disorders of the PI3K/AKT/mTOR pathway & therapeutic strategies. Am J Med Genet C Semin Med Genet. 2016;172(4):402–421. doi:10.1002/ajmg.c.31531
75. Papa A, Pandolfi PP. The PTEN(-)PI3K axis in cancer. Biomolecules. 2019;9(4):153. doi:10.3390/biom9040153
76. Yao J, Qian K, Chen C, et al. ZNF139/circZNF139 promotes cell proliferation, migration and invasion via activation of PI3K/AKT pathway in bladder cancer. Aging. 2020;12(10):9915–9934. doi:10.18632/aging.103256
77. Yang C, Yuan W, Yang X, et al. Circular RNA circ-ITCH inhibits bladder cancer progression by sponging miR-17/miR-224 and regulating p21, PTEN expression. Mol Cancer. 2018;17(1):19. doi:10.1186/s12943-018-0771-7
78. Maik-Rachline G, Hacohen-Lev-Ran A, Seger R. Nuclear ERK: mechanism of translocation, substrates, and role in cancer. Int J Mol Sci. 2019;20(5):1194. doi:10.3390/ijms20051194
79. Yanchun M, Yi W, Lu W, et al. Triptolide prevents proliferation and migration of esophageal squamous cell cancer via MAPK/ERK signaling pathway. Eur J Pharmacol. 2019;851:43–51. doi:10.1016/j.ejphar.2019.02.030
80. Qiu S, Hu W, Ma Q, Zhao Y, Li L, Ding Y. TIPE1 suppresses the invasion and migration of breast cancer cells and inhibits epithelial-to-mesenchymal transition primarily via the ERK signaling pathway. Acta Biochim Biophys Sin (Shanghai). 2019;51(10):1008–1015. doi:10.1093/abbs/gmz099
81. Sun QY, Ding LW, Johnson K, et al. SOX7 regulates MAPK/ERK-BIM mediated apoptosis in cancer cells. Oncogene. 2019;38(34):6196–6210. doi:10.1038/s41388-019-0865-8
82. Zhong Z, Huang M, Lv M, et al. Circular RNA MYLK as a competing endogenous RNA promotes bladder cancer progression through modulating VEGFA/VEGFR2 signaling pathway. Cancer Lett. 2017;403:305–317. doi:10.1016/j.canlet.2017.06.027
83. Peng G, Meng H, Pan H, Wang W. CircRNA 001418 promoted cell growth and metastasis of bladder carcinoma via EphA2 by miR-1297. Curr Mol Pharmacol. 2020;13. doi:10.2174/1874467213666200505093815
84. Teoh SL, Das S. Notch signalling pathways and their importance in the treatment of cancers. Curr Drug Targets. 2018;19(2):128–143. doi:10.2174/1389450118666170309143419
85. Shi YR, Wu Z, Xiong K, et al. Circular RNA circKIF4A sponges miR-375/1231 to promote bladder cancer progression by upregulating NOTCH2 expression. Front Pharmacol. 2020;11:605. doi:10.3389/fphar.2020.00605
86. Chen L, Yang X, Zhao J, et al. Circ_0008532 promotes bladder cancer progression by regulation of the miR-155-5p/miR-330-5p/MTGR1 axis. J Exp Clin Cancer Res. 2020;39(1):94. doi:10.1186/s13046-020-01592-0
87. Meyer PW, Anderson R, Ker JA, Ally MT. Rheumatoid arthritis and risk of cardiovascular disease. Cardiovasc J Afr. 2018;29(5):317–321. doi:10.5830/CVJA-2018-018
88. Levidou G, Saetta AA, Korkolopoulou P, et al. Clinical significance of nuclear factor (NF)-κB levels in urothelial carcinoma of the urinary bladder. Virchows Arch. 2008;452(3):295–304. doi:10.1007/s00428-007-0560-y
89. Liu H, Chen D, Bi J, et al. Circular RNA circUBXN7 represses cell growth and invasion by sponging miR-1247-3p to enhance B4GALT3 expression in bladder cancer. Aging. 2018;10(10):2606–2623. doi:10.18632/aging.101573
90. Sun M, Zhao W, Chen Z, et al. Circular RNA CEP128 promotes bladder cancer progression by regulating Mir-145-5p/Myd88 via MAPK signaling pathway. Int J Cancer. 2019;145(8):2170–2181. doi:10.1002/ijc.32311
91. Han CT, Bao QY, Cheng SJ, Liu M, Qian HN, Li D. Circular RNA hsa_circ_0017247 acts as an oncogene in bladder cancer by inducing Wnt/beta-catenin signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(3):1081–1087.
92. Su Y, Feng W, Zhong G, et al. ciRs-6 upregulates March1 to suppress bladder cancer growth by sponging miR-653. Aging. 2019;11(23):11202–11223. doi:10.18632/aging.102525
93. Zhang X, Liu X, Jing Z, et al. The circINTS4/miR-146b/CARMA3 axis promotes tumorigenesis in bladder cancer. Cancer Gene Ther. 2020;27(3–4):189–202. doi:10.1038/s41417-019-0085-y
94. Lim S, Kaldis P. Cdks, cyclins and CKIs: roles beyond cell cycle regulation. Development. 2013;140(15):3079–3093. doi:10.1242/dev.091744
95. Zheng F, Wang M, Li Y, et al. CircNR3C1 inhibits proliferation of bladder cancer cells by sponging miR-27a-3p and downregulating cyclin D1 expression. Cancer Lett. 2019;460:139–151. doi:10.1016/j.canlet.2019.06.018
96. Sun M, Zhao W, Chen Z, et al. Circ_0058063 regulates CDK6 to promote bladder cancer progression by sponging miR-145-5p. J Cell Physiol. 2019;234(4):4812–4824. doi:10.1002/jcp.27280
97. Aljabery F, Shabo I, Gimm O, Jahnson S, Olsson H. The expression profile of p14, p53 and p21 in tumour cells is associated with disease-specific survival and the outcome of postoperative chemotherapy treatment in muscle-invasive bladder cancer. Urol Oncol. 2018;36(12):530 e7–530 e18. doi:10.1016/j.urolonc.2018.05.025
98. Li P, Yang X, Yuan W, et al. CircRNA-Cdr1as exerts anti-oncogenic functions in bladder cancer by sponging microRNA-135a. Cell Physiol Biochem. 2018;46(4):1606–1616. doi:10.1159/000489208
99. Manning AL, Ganem NJ, Bakhoum SF, Wagenbach M, Wordeman L, Compton DA. The kinesin-13 proteins Kif2a, Kif2b, and Kif2c/MCAK have distinct roles during mitosis in human cells. Mol Biol Cell. 2007;18(8):2970–2979. doi:10.1091/mbc.e07-02-0110
100. Gan H, Lin L, Hu N, et al. KIF2C exerts an oncogenic role in nonsmall cell lung cancer and is negatively regulated by miR-325-3p. Cell Biochem Funct. 2019;37(6):424–431. doi:10.1002/cbf.3420
101. Song X, Zhang T, Wang X, et al. Distinct diagnostic and prognostic values of kinesin family member genes expression in patients with breast cancer. Med Sci Monit. 2018;24:9442–9464. doi:10.12659/MSM.913401
102. Chen J, Li S, Zhou S, et al. Kinesin superfamily protein expression and its association with progression and prognosis in hepatocellular carcinoma. J Cancer Res Ther. 2017;13(4):651–659. doi:10.4103/jcrt.JCRT_491_17
103. Yang C, Li Q, Chen X, et al. Circular RNA circRGNEF promotes bladder cancer progression via miR-548/KIF2C axis regulation. Aging. 2020;12(8):6865–6879. doi:10.18632/aging.103047
104. Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20(2):69–84.
105. Cao W, Zhao Y, Wang L, Huang X. Circ0001429 regulates progression of bladder cancer through binding miR-205-5p and promoting VEGFA expression. Cancer Biomark. 2019;25(1):101–113. doi:10.3233/CBM-182380
106. Zhao X, Guan JL. Focal adhesion kinase and its signaling pathways in cell migration and angiogenesis. Adv Drug Deliv Rev. 2011;63(8):610–615. doi:10.1016/j.addr.2010.11.001
107. Jin H, He Y, Zhao P, et al. Targeting lipid metabolism to overcome EMT-associated drug resistance via integrin beta3/FAK pathway and tumor-associated macrophage repolarization using legumain-activatable delivery. Theranostics. 2019;9(1):265–278. doi:10.7150/thno.27246
108. Tao T, Yuan S, Liu J, et al. Cancer stem cell-specific expression profiles reveal emerging bladder cancer biomarkers and identify circRNA_103809 as an important regulator in bladder cancer. Aging. 2020;12(4):3354–3370. doi:10.18632/aging.102816
109. Gu C, Zhou N, Wang Z, et al. circGprc5a promoted bladder oncogenesis and metastasis through Gprc5a-targeting peptide. Mol Ther Nucleic Acids. 2018;13:633–641. doi:10.1016/j.omtn.2018.10.008
110. Chen Q, Yin Q, Mao Y, et al. Hsa_circ_0068307 mediates bladder cancer stem cell-like properties via miR-147/c-Myc axis regulation. Cancer Cell Int. 2020;20:151. doi:10.1186/s12935-020-01235-6
111. Erdogan B, Webb DJ. Cancer-associated fibroblasts modulate growth factor signaling and extracellular matrix remodeling to regulate tumor metastasis. Biochem Soc Trans. 2017;45(1):229–236. doi:10.1042/BST20160387
112. Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15(12):1243–1253. doi:10.15252/embr.201439246
113. Gilkes DM, Semenza GL, Wirtz D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer. 2014;14(6):430–439.
114. Liu F, Zhang H, Xie F, et al. Hsa_circ_0001361 promotes bladder cancer invasion and metastasis through miR-491-5p/MMP9 axis. Oncogene. 2020;39(8):1696–1709. doi:10.1038/s41388-019-1092-z
115. Liu Q, Zhou Q, Zhong P. circ_0067934 increases bladder cancer cell proliferation, migration and invasion through suppressing miR-1304 expression and increasing Myc expression levels. Exp Ther Med. 2020;19(6):3751–3759.
116. Sun J, Zhang H, Tao D, et al. CircCDYL inhibits the expression of C-MYC to suppress cell growth and migration in bladder cancer. Artif Cells Nanomed Biotechnol. 2019;47(1):1349–1356. doi:10.1080/21691401.2019.1596941
117. Wu Z, Huang W, Wang X, et al. Circular RNA CEP128 acts as a sponge of miR-145-5p in promoting the bladder cancer progression via regulating SOX11. Mol Med. 2018;24(1):40. doi:10.1186/s10020-018-0039-0
118. Liu P, Li X, Guo X, et al. Circular RNA DOCK1 promotes bladder carcinoma progression via modulating circDOCK1/hsa-miR-132-3p/Sox5 signalling pathway. Cell Prolif. 2019;52(4):e12614. doi:10.1111/cpr.12614
119. Lefebvre V. Roles and regulation of SOX transcription factors in skeletogenesis. Curr Top Dev Biol. 2019;133:171–193.
120. Sarkar A, Hochedlinger K. The sox family of transcription factors: versatile regulators of stem and progenitor cell fate. Cell Stem Cell. 2013;12(1):15–30. doi:10.1016/j.stem.2012.12.007
121. Kamachi Y, Kondoh H. Sox proteins: regulators of cell fate specification and differentiation. Development. 2013;140(20):4129–4144. doi:10.1242/dev.091793
122. Nemer M, Horb ME. The KLF family of transcriptional regulators in cardiomyocyte proliferation and differentiation. Cell Cycle. 2007;6(2):117–121. doi:10.4161/cc.6.2.3718
123. Bureau C, Hanoun N, Torrisani J, Vinel JP, Buscail L, Cordelier P. Expression and function of kruppel like-factors (KLF) in carcinogenesis. Curr Genomics. 2009;10(5):353–360. doi:10.2174/138920209788921010
124. Park CS, Lewis A, Chen T, Lacorazza D. Concise review: regulation of self-renewal in normal and malignant hematopoietic stem cells by kruppel-like factor 4. Stem Cells Transl Med. 2019;8(6):568–574. doi:10.1002/sctm.18-0249
125. Kim CK, He P, Bialkowska AB, Yang VW. SP and KLF transcription factors in digestive physiology and diseases. Gastroenterology. 2017;152(8):1845–1875. doi:10.1053/j.gastro.2017.03.035
126. He Q, Huang L, Yan D, et al. CircPTPRA acts as a tumor suppressor in bladder cancer by sponging miR-636 and upregulating KLF9. Aging. 2019;11(23):11314–11328. doi:10.18632/aging.102530
127. He Q, Yan D, Dong W, et al. circRNA circFUT8 upregulates krupple-like factor 10 to inhibit the metastasis of bladder cancer via sponging miR-570-3p. Mol Ther Oncolytics. 2020;16:172–187. doi:10.1016/j.omto.2019.12.014
128. Liang H, Huang H, Li Y, Lu Y, Ye T. CircRNA_0058063 functions as a ceRNA in bladder cancer progression via targeting miR-486-3p/FOXP4 axis. Biosci Rep. 2020;40(3). doi:10.1042/BSR20193484
129. Liu T, Lu Q, Liu J, et al. Circular RNA FAM114A2 suppresses progression of bladder cancer via regulating NP63 by sponging miR-762. Cell Death Dis. 2020;11(1):47. doi:10.1038/s41419-020-2226-5
130. Huang W, Lu Y, Wang F, Huang X, Yu Z. Downregulation of circular RNA hsa_circ_0000144 inhibits bladder cancer progression via stimulating miR-217 and suppressing RUNX2 expression. Gene. 2018;678:337–342. doi:10.1016/j.gene.2018.08.036
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
