Back to Journals » OncoTargets and Therapy » Volume 11
circSMAD2 inhibits the epithelial–mesenchymal transition by targeting miR-629 in hepatocellular carcinoma
Authors Zhang XW, Luo P, Jing W, Zhou H, Liang CZ, Tu JC
Received 24 November 2017
Accepted for publication 27 January 2018
Published 16 May 2018 Volume 2018:11 Pages 2853—2863
DOI https://doi.org/10.2147/OTT.S158008
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yao Dai
Xianwei Zhang,1 Ping Luo,1 Wei Jing,1 Hu Zhou,2 Chunzi Liang,1 Jiancheng Tu1
1Department of Clinical Laboratory Medicine, Center for Gene Diagnosis, Zhongnan Hospital of Wuhan University, Wuhan, People’s Republic of China; 2Department of Blood Transfusion, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, People’s Republic of China
Background: Circular RNAs (circRNAs) are a class of widely distributed non-coding RNAs, which drew little attention for decades. Recent studies show that circRNAs are involved in cancer progression.
Methods: The circSMAD2 expression in HCC and adjacent non-tumor tissues was measured by quantitative real-time polymerase chain reaction, and the biological function of circSMAD2 was explored by proliferation, apoptosis, migration, invasion, and Western blot assays. Next, the dual-luciferase reporter assay was performed to identify the target miRNA of circSMAD2. Finally, circSMAD2 and its target miRNA were co-transfected in HCC cells to investigate their relationship to HCC progression.
Results: In this study, we found that circRNA SMAD2 (circSMAD2) expression was downregulated in hepatocellular carcinoma (HCC) tissues (P = 0.014) compared to the adjacent non-tumor tissues and markedly associated with the differentiation degree of the HCC tissues (P < 0.001). The in vitro experiments showed that overexpressed circSMAD2 inhibited the migration, invasion, and epithelial–mesenchymal transition (EMT) in HCC cells. Bioinformatics predicted that miR-629 is a potential target of circSMAD2, and the dual-luciferase reporter assay verified that miR-629 directly bound circSMAD2. In addition, we found that overexpression of circSMAD2 suppressed the expression of miR-629 in HCC cells, whereas knockdown of circSMAD2 upregulated the expression of miR-629. Furthermore, co-transfection of miR-629 mimics with circSMAD2 reversed the circSMAD2 effects of inhibiting the migration, invasion, and EMT of HCC cells.
Conclusion: Altogether, our data support that circSMAD2 inhibits the migration, invasion, and EMT of HCC cells by targeting miR-629.
Keywords: circular RNA, circSMAD2, hepatocellular carcinoma, epithelial–mesenchymal transition, microRNA-629
Introduction
Hepatocellular carcinoma (HCC) is the third leading cause of cancer-related deaths worldwide.1,2 Surgical resection during early stages is a curative treatment that improves the 5-year HCC survival rate.3 However, HCC patients are frequently diagnosed at an advanced stage, which indicates a poor prognosis.4 Therefore, a better understanding of the HCC progression mechanism and the identification of novel diagnostic markers and therapeutic targets for HCC are urgent.
Circular RNAs (circRNAs) are a large class of endogenous non-coding RNAs (ncRNAs) that covalently link the 5′ and 3′ ends together and form circular loops.5 CircRNAs are abundant, conserved, and stable and have tissue-specific expression in mammalian cells.5–8 CircRNAs play important roles in cellular activities, such as modulating the expression of parental genes9 and functioning as microRNA (miRNA) sponges.10 In addition, circRNAs are involved in different types of cancers, including HCC.11–15 For example, circMTO1 is significantly downregulated in HCC tissues, and the deceased amount of circMTO1 is a prognosis predictor for poor survival of patients.16
The circRNA SMAD2 (circSMAD2; circBase ID: Hsa_circ_0000847) is encoded by the SMAD2 gene and located at chr18:45391429-45423180. CircSMAD2 is an exon–intron circRNA (EIcircRNA) that is circularized with both exons and introns simultaneously. Several EIcircRNAs can upregulate the expression of their parental genes.9,17 SMAD2, the parental gene of circSMAD2, plays a key role in the epithelial–mesenchymal transition (EMT), which promotes the stationary epithelium-derived tumor cells to become migratory and invasive and plays a vital role in the progression of HCC.18 In addition, the expression of circSMAD2 is upregulated during TGF-β-induced EMT.19 Based on the abovementioned information, we investigated the relationship between circSMAD2 and HCC in this study.
Materials and methods
Patient data and specimen collection
A total of 86 pairs of HCC and adjacent non-tumor tissues were obtained from HCC patients (82 males and four females, mean age 54 ± 10 years) who underwent surgery without preoperative chemotherapy or radiotherapy at the Zhongnan Hospital of Wuhan University from 2011 to 2015. All patients were selected based on their pathology reports. Tumor staging (stages I, II, III, and IV) was defined according to the seventh edition of the AJCC Cancer Staging Manual. Tumor specimens and paired adjacent non-tumor tissues were stored at −80°C in RNAlater® RNA Stabilization Solution (Thermo Fisher Scientific, Waltham, MA, USA).
Ethical approval
The study conforms with the Code of Ethics of the World Medical Association (Declaration of Helsinki) printed in the British Medical Journal (18 July 1964). In addition, the study was approved by the ethics committee of Zhongnan Hospital of Wuhan University, and all patients signed the written informed consent.
RNA extraction and reverse transcription
Total RNA content of tissues and plasma was extracted using the TRIzol reagent (Thermo Fisher Scientific) according to the manufacturer’s instructions. The concentration and purity of the obtained RNA samples were quantified using the NanoDrop ND2000 (Thermo Fisher Scientific). The RNA samples were reverse transcribed to cDNA using the PrimeScript™ RT Reagent Kit with gDNA Eraser (Takara, Dalian, China) according to the manufacturer’s instructions.
Quantitative real-time polymerase chain reaction (qRT-PCR)
qRT-PCR was performed with SYBR® Premix Ex Taq™ II (Takara) on the Bio-Rad CFX96 (Bio-Rad Laboratories Inc., Hercules, CA, USA) according to the manufacturer’s instructions. The related primer sequences are listed in Table S1. The reactions began with an initial denaturation at 95°C for 5 min, followed by denaturation at 95°C for 30 s, annealing at 63.3°C for 30 s, and extension at 72°C for 30 s. The denaturation, annealing, and extension steps were repeated for 40 cycles. The glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and U6 genes were used as internal controls for circRNAs and miRNAs, respectively.
Plasmid construction
We used the pcDNA3.1(+) circRNA mini vector (Addgene plasmid 60648), which is a circRNA-forming plasmid obtained from Liang and Wilusz20 to subclone the circSMAD2 sequence for the transfection studies. In addition, psiCHECK2 (Promega Corporation, Fitchburg, WI, USA) was used in the dual-luciferase reporter assay. The circSMAD2cDNA was amplified using PCR with specific primers. The obtained PCR fragments were subcloned into the EcoRV and SacII sites of the pcDNA3.1(+) circRNA mini vectors. The wild-type circSMAD2 (circSMAD2-wt) and the mutant-type circSMAD2 (circSMAD2-mut) were subcloned into the XhoI and NotI sites of the psiCHECK2 vectors, respectively. Afterward, the reconstructed plasmids were validated using DNA sequencing. Finally, more copies of the reconstructed plasmids were produced and purified using the Mini Plasmid Preparation Kit (Axygen, Hangzhou, China) according to the manufacturer’s instructions. In addition, small interfering RNA of circSMAD2 (si-circSMAD2; GenePharma, Suzhou, China) was synthesized to knock down the expression of circSMAD2, and miR-629 mimics (GenePharma) was synthesized to overexpress miR-629 in vitro (Table S2).
Cell culture and plasmid transfection
The HCC cell lines HepG2, HCCLM9, HCCLM3, and MHCC97L as well as the immortalized human hepatic cell line L02 were obtained from the Cell Bank of Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). Cells were cultured in DMEM (Thermo Fisher Scientific) with 10% fetal bovine serum (FBS; Thermo Fisher Scientific) in a humidified incubator at 37°C with 5% CO2. A six-well plate was seeded with 5 × 105 cells and incubated for 24 h. The cells were then transfected with the related genes using Lipofectamine™ 2000 (Thermo Fisher Scientific) according to the manufacturer’s instructions.
Cell proliferation assay
Cell proliferation assays were conducted using the Cell Counting Kit-8 (CCK-8) (Dojindo, Kumamoto, Japan) according to the manufacturer’s instructions. In brief, transfected cells were seeded into 96-well plates (2,000 cells/well) and cultured for 0, 24, 48, and 96 h. Then, 10 μL of CCK-8 solution was added to each well, and the plates were incubated at 37°C for an additional 2 h. Finally, the solution was measured at 450 nm on the spectrophotometer (EnSpire; PerkinElmer Inc., Waltham, MA, USA).
Flow cytometric analysis
Transfected HepG2 cells were harvested after transfection for 24 h and stained using an Annexin V-FITC/PI Apoptosis Detection Kit (Beyotime, Shanghai, China) according to the manufacturer’s instructions. The cells were then analyzed using a Cytomics™ FC 500 flow cytometer (Beckman Coulter, Brea, CA, USA).
Cell migration and invasion assays
Transfected HepG2 cells were harvested after transfection for 24 h and seeded into the upper chambers (20,000 cells/chamber) of Transwell assay plates (Corning Incorporated, Corning, NY, USA) with 200 μL of serum-free DMEM to quantify cell migration. Similarly, transfected HepG2 cells were seeded into the upper chambers (40,000 cells/chamber) of Transwell plates with Matrigel-coated membranes (BD, Franklin Lakes, NJ, USA) in 200 μL serum-free DMEM to quantify cell invasion. The lower chambers were filled with DMEM containing 10% FBS. After an incubation period of 24 h, the medium was removed and cells were fixed with methanol for 20 min. The cells were stained with crystal violet for 20 min, air-dried, and photographed using a digital microscope. The number of cells was calculated from five random fields in each chamber.
Western blot
Cells were lysed for extraction of total protein. After measurement of protein concentration, the protein was separated using 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes (EMD Millipore, Billerica, MA, USA). After blocking with 5% non-fat milk, the membranes were incubated with primary antibodies: anti-SMAD2 (Abcam, Cambridge, MA, USA), anti-GAPDH (Abcam), anti-E-cadherin (Cell Signaling Technology, Danvers, MA, USA), anti-N-cadherin (Cell Signaling Technology), anti-Snail (Cell Signaling Technology), and anti-Vimentin (Cell Signaling Technology). Then the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies. Immunoblot signals were visualized using ECL Plus Kit (GE Healthcare, Fairfield, CT, USA) according to the manufacturer’s instructions.
Dual-luciferase reporter assay
HepG2 cells were co-transfected with 4 μg psiCHECK-2-circSMAD2-wt or orpsiCHECK-2-circSMAD2-mut plasmids and 20 nmol/L miR-629 mimics or miRNA negative control (miR-NC) using Lipofectamine™ 2000 (Thermo Fisher Scientific) according to the manufacturer’s instructions. After 48 h of incubation, the Firefly and Renilla luciferase activities were measured using a dual-luciferase reporter assay kit (Promega Corporation) according to the manufacturer’s instructions.
Statistical analyses
All statistical analyses were carried out using SPSS version 21.0 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism 5.0 (GraphPad Software, Inc., La Jolla, CA, USA). The numerical data are presented as mean (M) ± standard error of the mean (SEM) of at least three determinations. Results were considered as statistically significant at P < 0.05. The normality of distribution for each data set was tested using the Shapiro–Wilk test. Normally distributed data sets were analyzed using the Student’s t-tests, while non-normally distributed data sets were analyzed using Kruskal–Wallis variance analyses.
Results
CircSMAD2 was downregulated in HCC tissues and cell lines and markedly associated with the differentiation degree of HCC tissues
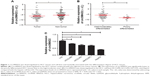
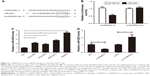
To elucidate the circSMAD2 profile in HCC, we performed qRT-PCR in 86 pairs of HCC and adjacent non-tumor tissues. CircSMAD2 was downregulated in HCC tissues compared with non-tumor tissues (P = 0.014; Figure 1A). Comparison of different clinical features (Table 1) indicated that the expression of circSMAD2 was significantly lower in the poorly differentiated HCC tissues than in the high/moderate ones (P < 0.001; Figure 1B). We further detected circSMAD2 expression in the HCC and hepatic cell lines. The qRT-PCR results showed that circSMAD2 was downregulated in the HCC cell lines HCCLM9, HCCLM3, HCC97L, and HepG2 compared with the hepatic cell line L02 (Figure 1C).
Overexpression of circSMAD2 inhibited the migration, invasion, and EMT of HCC cells
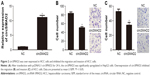
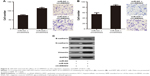
To investigate the biological function of circSMAD2, we overexpressed circSMAD2 in HepG2 cells (Figure 2A), which had the lowest circSMAD2 expression of the four HCC cell lines. The Transwell assay showed that overexpression of circSMAD2 inhibited the migration and invasion of HepG2 cells (Figure 2B and C), whereas the CCK-8 and flow cytometric assays showed no significant changes in the proliferation and apoptosis of these cells (Figure S1A and B). The EMT plays a key role in the tumor metastasis. Thus, we determined if circSMAD2 could regulate the EMT in HCC cells. Western blot assays showed that the epithelial cell marker E-cadherin was significantly upregulated, whereas the mesenchymal cell markers N-cadherin, Snail, and Vimentin were significantly reduced in circSMAD2-overexpressed HepG2 cells (Figure 3), suggesting that circSMAD2 inhibited the EMT process in HCC cells.
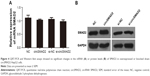
Several EIcircRNAs can regulate the expression of parental genes, and circSMAD2 is an EIcircRNA. In addition, SMAD2 protein plays a key role in the EMT process. Thus, we explored whether circSMAD2 regulated the EMT in HCC by regulating SMAD2 expression. However, the results indicated no significant change in SMAD2 expression with overexpressed or knocked down circSMAD2 (Figure S1C) HepG2 cells (Figure 4).
CircSMAD2 acts as an miRNA sponge for miR-629 in HCC cells
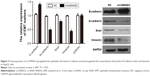
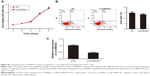
To explore whether circSMAD2 is serving as an miRNA sponge in HCC, we searched for potential target miRNAs using a bioinformatics database (circRNA interactome). Among the potential candidates, we focused on miRNAs involved in the EMT. miR-629 attracted our attention (Figure 5A) because it induces tumor initiation and growth of HCC and promotes the EMT in several cancer cell lines.21,22 To confirm whether miR-629 was the direct target of circSMAD2, we performed a dual-luciferase reporter assay. As shown in Figure 5B, the relative luciferase activity was significantly lower in HepG2 cells co-transfected with the psiCHECK2-circSMAD2-wt and miR-629 compared with controls, but no significant difference was detected in the HepG2 cells co-transfected with psiCHECK2-circSMAD2-mut and miR-629 compared with controls, suggesting that miR-629 directly bound circSMAD2. In contrast to the expression levels of circSMAD2, miR-629 was upregulated in the HCC cell lines HCCLM9, HCCLM3, HCC97L, and HepG2 compared with the hepatic cell line L02 (Figure 5C). In addition, overexpression of circSMAD2 reduced the expression of miR-629 in HepG2 cells, whereas knockdown of circSMAD2 upregulated the expression of miR-629 (Figure 5D). Collectively, circSMAD2 might act as an miRNA sponge for miR-629 in HCC cells.
miR-629 could reverse the circSMAD2 effects of inhibiting the migration, invasion, and EMT of HCC cells
To investigate whether circSMAD2 regulates the EMT in HCC cells by targeting miR-629, the miR-629 mimics and pcDNA 3.1-circSMAD2 were co-transfected into HepG2 cells (Figure S2). The Transwell and Western blot assays showed that co-transfection with miR-629 mimics and pcDNA 3.1-circSMAD2 reversed the circSMAD2 effect of inhibiting the migration, invasion, and EMT of HCC cells compared with co-transfection with miR-NC and pcDNA 3.1-circSMAD2 (Figure 6A–C). Collectively, these data suggest that circSMAD2 might inhibit the migration, invasion, and EMT by targeting miR-629 in HCC cells.
Discussion
Because of the development of RNA sequencing technologies and bioinformatics, the functions of circRNAs were revealed, such as modulating the expression of parental genes and serving as miRNA sponges. Furthermore, recent studies show that circRNAs participate in the initiation and progression of tumors, including HCC.12,16,23
In this study, we found that circSMAD2 expression was downregulated in HCC tissues compared with the adjacent non-tumor tissues, and the expression levels of circSMAD2 were significantly lower in the poorly differentiated tissues than in the well differentiated/moderately differentiated ones. The in vitro experiments showed that circSMAD2 overexpression inhibited the migration and invasion of HCC cells. The EMT is the mechanism that drives a transient and reversible de-differentiation of epithelial cells to a mesenchymal-like phenotype and contributes to tumor metastasis of various cancers, including HCC.24 Thus, we explored whether circSMAD2 was involved in the EMT in HCC cells. The results showed that overexpression of circSMAD2 inhibited the EMT in HCC cells. CircSMAD2 is an EIcircRNA, and several EIcircRNAs can regulate the expression of parental genes. In addition, SMAD2, the parental gene of circSMAD2, plays a key role in the EMT. Thus, we hypothesized that circSMAD2 might regulate the EMT by modulating SMAD2 expression. However, no significant change in the SMAD2 expression was found in HCC cells with overexpressed or knocked down circSMAD2, suggesting that circSMAD2 might have no influence on the SMAD2 expression.
A previous study showed that circRNAs are able to serve as miRNA sponges.10 To explore the underlying mechanism of circSMAD2 in HCC, the potential miRNA targets of circSMAD2 were predicted using bioinformatics. Then, we focused our attention on miR-629, because not only it is one of the high-ranked targets but also it plays a vital role in the HCC oncogenesis.21 The subsequent dual-luciferase reporter assay indicated that miR-629 directly bound circSMAD2. In contrast to the expression levels of circSMAD2, we found that miR-629 expression was upregulated in HCC cells. In addition, circSMAD2 overexpression induced miR-629 downregulation in HCC cells, whereas knockdown of circSMAD2 upregulated the expression of miR-629. Furthermore, co-transfection of circSMAD2 and miR-629 reversed the circSMAD2 effects of inhibiting the migration, invasion, and EMT of HCC cells. Hatziapostolou et al21 revealed that miR-629 is upregulated in both HCC cell lines and tumors and plays a key role in HCC initiation and growth. In addition, miR-629 promotes the EMT, motility, and invasion in clear renal cell carcinoma by downregulating the expression of tripartite motif-containing 33 (TRIM33), which regulates the EMT via inhibiting the TGF-β/Smad2 signaling pathway.22,25–27 TRIM33 expression is reduced in HCC tissues, and the reduced expression of TRIM33 promotes the HCC metastasis.28 Therefore, regulating miR-629 expression and its downstream functions might partly contribute to the effect of circSMAD2 on regulating the EMT of HCC cells. However, the detailed mechanism requires further investigation.
The main limitation of our study is that our experiments were done in vitro and the biological function of circSMAD2 remains to be validated in vivo. Besides, we only verified the relationship between miR-629 and circSMAD2. The potential relationships between other predicted target miRNAs and circSMAD2 remain to be further investigated in HCC.
Conclusion
We found that circSMAD2 was downregulated in HCC tissues and markedly associated with the differentiation degree of tumors. In vitro experiments revealed that circSMAD2 inhibited the migration, invasion, and EMT of HCC cells. Furthermore, we demonstrated that miR-629 directly binds circSMAD2 and circSMAD2 inhibits the migration, invasion, and EMT of HCC cells by suppressing miR-629 expression.
Acknowledgment
This study was supported by the National Basic Research Program of China (973 Program; 2012CB720605).
Disclosure
The authors report no conflicts of interest in this work.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90. | ||
El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118–1127. | ||
Zhong JH, Ke Y, Gong WF, et al. Hepatic resection associated with good survival for selected patients with intermediate and advanced-stage hepatocellular carcinoma. Ann Surg. 2014;260(2):329–340. | ||
Dimitroulis D, Damaskos C, Valsami S, et al. From diagnosis to treatment of hepatocellular carcinoma: an epidemic problem for both developed and developing world. World J Gastroenterol. 2017;23(29):5282–5294. | ||
Memczak S, Jens M, Elefsinioti A, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495(7441):333–338. | ||
Jeck WR, Sorrentino JA, Wang K, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA. 2013;19(2):141–157. | ||
Guo JU, Agarwal V, Guo H, Bartel DP. Expanded identification and characterization of mammalian circular RNAs. Genome Biol. 2014;15(7):409. | ||
Salzman J, Gawad C, Wang PL, Lacayo N, Brown PO. Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PLoS One. 2012;7(2):e30733. | ||
Li Z, Huang C, Bao C, et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat Struct Mol Biol. 2015;22(3):256–264. | ||
Hansen TB, Jensen TI, Clausen BH, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495(7441):384–388. | ||
Meng S, Zhou H, Feng Z, et al. CircRNA: functions and properties of a novel potential biomarker for cancer. Mol Cancer. 2017;16(1):94. | ||
Zhang J, Liu H, Hou L, et al. Circular RNA_LARP4 inhibits cell proliferation and invasion of gastric cancer by sponging miR-424-5p and regulating LATS1 expression. Mol Cancer. 2017;16(1):151. | ||
Zhang Y, Liu H, Li W, et al. CircRNA_100269 is downregulated in gastric cancer and suppresses tumor cell growth by targeting miR-630. Aging. 2017;9(6):1585–1594. | ||
Liang G, Liu Z, Tan L, Su AN, Jiang WG, Gong C. HIF1alpha-associated circDENND4C promotes proliferation of breast cancer cells in hypoxic environment. Anticancer Res. 2017;37(8):4337–4343. | ||
Kong Z, Wan X, Zhang Y, et al. Androgen-responsive circular RNA circSMARCA5 is up-regulated and promotes cell proliferation in prostate cancer. Biochem Biophys Res Commun. 2017;493(3):1217–1223. | ||
Han D, Li J, Wang H, et al. Circular RNA MTO1 acts as the sponge of miR-9 to suppress hepatocellular carcinoma progression. Hepatology. 2017;66(4):1151–1164. | ||
Li F, Zhang L, Li W, et al. Circular RNA ITCH has inhibitory effect on ESCC by suppressing the Wnt/beta-catenin pathway. Oncotarget. 2015;6(8):6001–6013. | ||
Heldin CH, Moustakas A. Role of Smads in TGFbeta signaling. Cell Tissue Res. 2012;347(1):21–36. | ||
Conn SJ, Pillman KA, Toubia J, et al. The RNA binding protein quaking regulates formation of circRNAs. Cell. 2015;160(6):1125–1134. | ||
Liang D, Wilusz JE. Short intronic repeat sequences facilitate circular RNA production. Genes Dev. 2014;28(20):2233–2247. | ||
Hatziapostolou M, Polytarchou C, Aggelidou E, et al. An HNF4alpha-miRNA inflammatory feedback circuit regulates hepatocellular oncogenesis. Cell. 2011;147(6):1233–1247. | ||
Jingushi K, Ueda Y, Kitae K, et al. miR-629 targets TRIM33 to promote TGFbeta/Smad signaling and metastatic phenotypes in ccRCC. Mol Cancer Res. 2015;13(3):565–574. | ||
Chen J, Li Y, Zheng Q, et al. Circular RNA profile identifies circPVT1 as a proliferative factor and prognostic marker in gastric cancer. Cancer Lett. 2017;388:208–219. | ||
Giannelli G, Koudelkova P, Dituri F, Mikulits W. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol. 2016;65(4):798–808. | ||
Dupont S, Zacchigna L, Cordenonsi M, et al. Germ-layer specification and control of cell growth by Ectodermin, a Smad4 ubiquitin ligase. Cell. 2005;121(1):87–99. | ||
He W, Dorn DC, Erdjument-Bromage H, Tempst P, Moore MA, Massague J. Hematopoiesis controlled by distinct TIF1gamma and Smad4 branches of the TGFbeta pathway. Cell. 2006;125(5):929–941. | ||
Dupont S, Mamidi A, Cordenonsi M, et al. FAM/USP9x, a deubiquitinating enzyme essential for TGFbeta signaling, controls Smad4 monoubiquitination. Cell. 2009;136(1):123–135. | ||
Ding ZY, Jin GN, Wang W, et al. Reduced expression of transcriptional intermediary factor 1 gamma promotes metastasis and indicates poor prognosis of hepatocellular carcinoma. Hepatology. 2014;60(5):1620–1636. |
Supplementary materials
  | Table S1 List of primers in this study |
  | Table S2 Sequences of siRNA and miRNA mimics in this study |
  | Figure S2 Transfection of miR-629 mimics significantly upregulated the expression of miR-629 in HepG2 cells. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.