Back to Journals » Journal of Pain Research » Volume 13
Chronic Pain Patients’ Kinesiophobia and Catastrophizing are Associated with Activity Intensity at Different Times of the Day
Authors Miller MB , Roumanis MJ, Kakinami L, Dover GC
Received 6 September 2019
Accepted for publication 24 December 2019
Published 31 January 2020 Volume 2020:13 Pages 273—284
DOI https://doi.org/10.2147/JPR.S230039
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Michael A Ueberall
Matthew B Miller,1 Melissa J Roumanis,1 Lisa Kakinami,2,3 Geoffrey C Dover1,3,4
1Department of Health, Kinesiology, and Applied Physiology, Concordia University, Montreal, Canada; 2Department of Mathematics & Statistics, Concordia University, Montreal, Canada; 3PERFORM Centre, Concordia University, Montreal, Canada; 4Centre de Recherché Interdisciplinaire en Réadaptation du Montréal Metropolitain, Montreal, Canada
Correspondence: Matthew B Miller
Department of Health, Kinesiology, and Applied Physiology, Concordia University, L - SP 165.28, Richard J. Renaud Science Complex, 7141 Sherbrooke W, Montreal, Canada
Tel +1 514-848-2424 ext. 3304
Fax +1 514-848-8681
Email [email protected]
Purpose: To examine the relationship between baseline kinesiophobia and baseline pain catastrophizing with the 4-day average activity intensity at different times of the day while accounting for different wake and sleep-onset times in chronic pain patients.
Methods: Twenty-one participants suffering from idiopathic chronic pain completed baseline questionnaires about kinesiophobia, catastrophizing, disability, depression, and pain. We measured the participants’ activity using accelerometers and calculated activity intensity in the morning, afternoon, and evening. We performed a 2-way repeated measures ANOVA to compare activity levels at different times of the day, and multiple linear regressions.
Results: Baseline kinesiophobia was significantly associated with 4-day average evening light activity and sedentary activity at all time periods while baseline catastrophizing was significantly associated with increased 4-day average light activity in the evening and more moderate to vigorous activity in the morning. Our participants engaged in more light activity on average than sedentary activity, and very little moderate-vigorous activity. Participants were most active in the afternoon.
Conclusion: Baseline kinesiophobia and baseline catastrophizing were not associated with the 4-day average total daily activity; however, they were associated with 4-day average activity intensities at different times throughout the day. Segmenting daily activity into morning, afternoon, evening may influence the relationship between daily activity, and kinesiophobia and pain catastrophizing. Individuals with chronic pain are less sedentary than previously thought which may affect future interventions.
Keywords: accelerometer, physical activity, pain, Tampa Scale, sedentary, daily activity
Introduction
Chronic pain is a complex condition that can negatively impact many aspects of an individual's life.1 Chronic pain has physical consequences (such as loss of independence, reliance on pain medication, reduced physical capacity), and psychological consequences (such as emotional distress, insomnia, and impaired social and work-related function).2–4 Sedentary behaviour resulting from chronic pain is linked to increased depressive symptoms. For example, inactive individuals have double the chance of developing depressive symptoms compared to healthy individuals.5,6 People who experience chronic pain live a more sedentary lifestyle compared to the general population, leading to several health conditions such as cardiovascular disease, obesity, type II diabetes, cancer, and decreased quality of life.7–9
Most multidisciplinary clinics that treat chronic pain focus on improving function; therefore, accurately measuring function is critical. Assessments of physical activity or physical function can be self-reported questionnaires, clinical tests, or accelerometers. Questionnaire-based assessments of physical function, such as the Pain Disability Index (PDI)10 and the 36-Item Short Form Health Survey (SF-36),11 are often used as a cost-effective strategy to assess subjective physical function in the chronic pain population. Questionnaire-based assessments are a qualitative method of assessment and are open to interpretation or bias from the patient.8,12 An objective, but more expensive alternative, is the free-living assessment of physical activity and function using wearable accelerometers.13 Increasing function through activity participation and enhancing quality of life are treatment goals for the chronic pain population, but fear of movement or re-injury (kinesiophobia) can hinder rehabilitation outcomes.14
Kinesiophobia and pain catastrophizing are related to rehabilitation outcomes in chronic pain, but the relationship between kinesiophobia and pain catastrophizing, and activity is unclear.14–17 In the chronic pain population, kinesiophobia is more disabling than pain itself,15 thus, prolonging rehabilitation and worsening the chronic pain experience.15,18,19 Due to the association between kinesiophobia and disability,20 treatment has focused on increasing activity levels, quantifying activity goals, and changing patients’ behaviours and perceptions towards pain, rather than solely focused on decreasing pain levels.21–23 Changing patients’ behaviour is challenging and involves, for example, explaining to the patient that just because they are in pain, it does not mean they should limit their activity.24 The association between kinesiophobia and physical activity implies that people with chronic pain are less likely to participate in an active lifestyle.25 However, the relationship between kinesiophobia and pain catastrophizing, and daytime movement is inconsistent in the literature. The relationship between kinesiophobia, pain catastrophizing, disability, and depression and total activity is complex. Song et al (2012) and Elfving et al (2007) indicate there is a relationship between kinesiophobia, pain catastrophizing, disability, and depression and activity levels,5,26,27 while Carvalho et al (2017) suggest there is no association.26 The inconsistency in these results may be due to the methodology of activity measurement. Individuals in chronic pain experience sleep disturbances in the form of varying wake times, several night-time awakenings, poor sleep efficiency, and general insomnia.28,29 Therefore, collecting activity data during the time each participant is awake may be more accurate than standard time periods (ie, morning from 6am to 12pm). Previous authors suggest future projects should investigate the influence of perceived disability and kinesiophobia on the activity levels of people with chronic pain using both questionnaires and accelerometers to assess activity intensity and disability.13,30 To date, there are no studies that associate kinesiophobia and pain catastrophizing to daily activity segmented by time of day and activity intensity using an accelerometer to assess the activity habits of individuals with chronic pain.
The purpose of our study is to determine if measuring activity based on wake and sleep times and by separating activity into the morning, afternoon, and evening would influence the relationship between baseline kinesiophobia and baseline pain catastrophizing, with activity in chronic pain patients. The a priori hypotheses include: 1) People who suffer from chronic pain will participate in more activity in the afternoon compared to the morning and evening, and 2) Baseline kinesiophobia or baseline catastrophizing and self-reported disability is associated with sedentary, light, and moderate-vigorous physical activity in the morning, afternoon, and evening.
Materials and Methods
Participants
We recruited 40 participants from a waiting list for a multidisciplinary chronic pain program, which has occurred in other studies.31,32 All participants suffered from idiopathic chronic pain, which is characterized as pain not related to any known disease or injury and pain that cannot better be explained by another chronic pain condition, experienced at least once a week during the last 3 months.33 Participants were included in this study if they were on the waiting list for a chronic pain treatment program which includes being diagnosed with idiopathic chronic pain. The chronic pain treatment program requires patients to participate for five days a week for at least 6 hrs a day, so participants were typically unemployed or on disability leave. Exclusion criteria included the consumption of recreational drugs, being clinically diagnosed with a sleep disorder or seasonal affective disorder, and a current prescription of a sleep aid regimen. Our study was part of a larger study evaluating the relationship between sleep and chronic pain. This study was approved by the Centre de Recherché Interdisciplinaire en Réadaptation du Montréal metropolitain (CRIR-759-0812).
Outcome Measures
Tampa Scale of Kinesiophobia (TSK)
The TSK is a 17-item questionnaire designed to assess a patient’s fear of movement or (re)injury. The total score ranges from 17 to 68, participants with higher scores are rated as having greater fear of movement. Score categories for the TSK include: 13–22, subclinical; 23–32, mild; 33–42, moderate; 43–52, severe.34
Pain Catastrophizing Scale (PCS)
The PCS is a 14-item questionnaire. Participants are asked to rate the thoughts and feelings they experience when in pain using a 5-point Likert scale. The PCS total score ranges from 0–52 and is calculated by summing all items. Pain catastrophizing scores can be categorized into rumination, magnification, and helplessness, but for the purposes of this study total scores were used in all analyses. The TSK and PCS have been used in chronic pain populations,35,36 and have been correlated to poor rehabilitation outcomes, missed work, and decreased activity in chronic pain patients.14
Beck Depression Inventory (BECK)
The BECK is a 21-item questionnaire designed to measure the severity of a person’s depression. Scores range from 0–63, with a score of 10–18 indicating mild depression, a score of 19–29 indicating moderate depression, and a score greater than 30 indicating severe depression. The BECK has demonstrated good validity and internal consistency in the chronic pain population.37
Pain Disability Index (PDI)
The PDI is a 7-item questionnaire designed to measure perceived disability. Participants rate how pain interferes with their functioning in family/home, responsibilities, recreation, social activities, occupation, sexual behaviors, self-care, and life-support activities. Scores range from 0 to 70, a higher score suggests a higher level of disability due to pain.38
McGill Pain Questionnaire (MPQ)
The MPQ consists of 78 words that describe qualities of the pain experience. Participants describe the quality and intensity of their pain by selecting the words that best describe their pain experience. Scores range from 0–78, with higher scores indicating higher levels of pain.14
Activity
We assessed activity using the Actiwatch Score (Respironics, Oregon, USA), a wrist-worn activity monitor with a built-in accelerometer. The Actiwatch Score continually detects and logs movement as activity counts. The Actiwatch was configured to collect activity counts each minute with a one-minute epoch length. The wake threshold (at what intensity the accelerometer will record movement) was set at 20 activity counts per minute.39 We placed the Actiwatch on the wrist to measure activity in our participants similar to previous studies.40 Some authors suggest that measuring activity in an older or more physically limited population the wrist is the preferred location compared to the waist. As indicated above, our study is part of a larger study that examined sleep habits in the chronic pain population. Participants accelerometer data were included if they completed four non-consecutive days of 24 hrs wear time (3 weekdays and 1 weekend day).
Procedures
All participants provided written informed consent, and that this study was conducted in accordance with the Declaration of Helsinki. The participants completed the questionnaires described above, once, on the first day of the study. Age and sex were self-reported, and height and weight were measured by study staff to calculate body mass index (BMI). After completing the questionnaires, the participants received the accelerometer watch and were instructed to wear the watch on their non-dominant wrist for 24 hrs a day for seven consecutive days, and to only remove the watch for bathing purposes. Participants were instructed to follow their regular daily routines even if it included exercise for the entire seven-day period. Caffeine and tobacco consumption were permitted; however, alcohol and illicit drugs were not permitted.
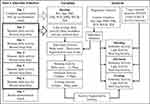
Participants were asked to keep a sleep diary recording the approximate time they woke up and fell asleep each day. The participants were informed that the accelerometer would evaluate their sleep at night but were not told that the accelerometer would always be on and measure movement data during the day. Therefore, the participants were partially blinded to the activity data collection during the day, which we hoped would contribute to a true measure of activity for the participants. See Figure 1 for a research timeline.
Data Analysis
All data from the Actiwatch were downloaded and raw activity data were exported from the Actiware software (Respironics, Bend, Oregon) to a Microsoft Office Excel spreadsheet and arranged by day. Daily activity was divided into three segments. The morning segment started from each individual's wake-onset to 12:00pm, the afternoon segment was 12:00pm–6:00pm, and the evening was 6:00pm – sleep-onset of each participant. Wake and sleep-onset times were calculated based on the sleep diaries and cross-referenced with the readings from the accelerometer.
Activity data were segmented into the three categories based on the number of activity counts.41 In accordance with previous studies, four non-consecutive days of data were included (three weekdays and one weekend day) therefore activity was averaged over 4 days.13 Sedentary time was defined as ≤100 activity counts per minute, light activity was defined as between 101 and 1535 activity counts per minute, and moderate-vigorous activity was defined as any activity counts above 1535 counts per minute.41 We included naps in the daily activity because napping behaviour is prominent in the chronic pain population and we wanted to gather data on a full day’s activity. Questionnaire data for the TSK, PCS, MPQ, and PDI were transferred from the questionnaire recording sheet to a Microsoft Office Excel spreadsheet for analysis.
Statistical Analysis
In this study, we conducted one 2-way repeated measures ANOVA to evaluate the differences between morning, afternoon, and evening activity, and sedentary, light, and moderate-vigorous activity intensity. Normality was tested with Mauchly’s test of sphericity; if normality was violated, the Greenhouse-Geisser correction was used. When significance was identified, Tukey’s post hoc test was used to identify any significant differences among the nine means.
Multiple linear regressions were used to predict the daily average (1) sedentary, (2) light, (3) moderate-vigorous activity, and (4) total overall activity in separate models. All models controlled for age, sex, BMI, and Beck depression scores. Models additionally included TSK and PDI, or PCS and PDI. For all models, the variance inflation factor (VIF) was checked to confirm that multicollinearity was not present.
Results
A total of 19 participants were excluded due to incomplete questionnaires or missing accelerometer data. One person requested to be withdrawn from the study. Therefore, a total of 21 participants completed our research study (males = 6, females = 15, age = 45.6±11.7 years, BMI = 26.9±7.7 kg/m2). There were no statistically significant differences between male and female groups in age, sex, height, weight, or BMI, nor were there statistically significant sex differences between TSK, PCS, BECK, PDI, or MPQ scores. Due to the different group sizes and the similarities between male and female groups, we have selected to pool both males and females in all subsequent analyses. See Table 1 for full demographic information.
 |
Table 1 Demographics, Questionnaire Results (TSK, PSC, BECK, PDI, MPQ), Minutes of Activity, and Sleep Variables in a Group of Chronic Pain Patients Awaiting Treatment in a Multidisciplinary Clinic |
The results of the 2-way repeated measures ANOVA using time of day and activity data violated the assumption of normality; thus, results incorporating the Greenhouse-Geisser correction are exclusively presented here. The 2-way repeated measures ANOVA indicated there was a statistically significant interaction between time of day and activity intensity [F (2.54,50.96) = 10.26, p <0.001], and a statistically significant main effect of time of day [F (1.12,22.49) = 5.79, p =0.02] and activity intensity [F (1.22,24.55) = 110.20, p <0.001]. Overall, individuals with chronic pain participated in more average light activity than sedentary activity over a 4-day period, and very little moderate-vigorous activity. Of note, chronic pain patients were most active in the afternoon, in which they participated in more minutes of light activity than other activity intensities at all times of the day. See Table 2 for all comparisons between 4-day average activity intensity at different times of the day.
 |
Table 2 A 2-Way Repeated Measures ANOVA Between 4-Day Average Activity Intensity (Sedentary, Light, and Moderate-Vigorous) and Time of Day (Morning, Afternoon, and Evening) |
Further exploration of activity intensities and time of day in multiple linear regression models indicated that the associations between baseline TSK or PCS with activity intensity also varied by time of day. All analyses adjusted for sex, age, BMI, PDI and Beck depression scores as previously mentioned. Greater baseline TSK was associated with significantly less 4-day average sedentary activities in the morning, afternoon, and evening (B = −4.59, B = −3.35, and B = −4.19, respectively, all p <0.05). In contrast, greater baseline TSK was associated with greater 4-day average light activity in the evening only (B = 6.18, p = 0.02) and greater moderate-vigorous activity in the morning and evening (B = 0.19 and B = 0.16, respectively, both p <0.05) (Table 3).
Greater baseline PCS was associated with significantly less 4-day average sedentary activity in the morning (B = −4.30, p = 0.03), and greater light activity in the evening (B = 4.2, p = 0.02), and greater moderate-vigorous activity in the morning (B = 0.14, p = 0.03) (Table 4). Neither the TSK model nor PCS model were significant predictors of total daily activity (Table 5)
Discussion
The results of our regression analysis indicated that baseline kinesiophobia was associated with 4-day average evening light activity, but not morning or afternoon light activity and baseline pain catastrophizing was associated with 4-day average sedentary and moderate-to-vigorous activity in the morning, and light evening activity. In addition, baseline kinesiophobia was associated with 4-day average sedentary activity in the morning, afternoon, and evening, and neither baseline kinesiophobia nor baseline pain catastrophizing were associated with 4-day average total daily activity. Our findings indicate that chronic pain patients’ baseline kinesiophobia and baseline pain catastrophizing measured once at beginning of the study are associated with the 4-day average of different activity intensities at different times of the day. Future research should investigate the significant relationship between baseline kinesiophobia and the 4-day average of light activity in the afternoon and the influence this relationship has on rehab interventions. Of interest is the finding that the most activity our chronic pain patients participated in was light activity in the afternoon, but the regression results suggest that baseline levels of kinesiophobia and baseline pain catastrophizing are not associated with the 4-day average of light activity in the afternoon. This may be explained by the amount of activity in the afternoon our participants engaged in, it is possible that our participants were more active in the afternoon, leading to an exacerbation of their pain and more sedentary activity in the evening. Chronic pain has been linked to changes in the brain (specifically in the anterior cingulate cortex) suggesting that pain may cause avoidance behaviours.42 It is possible that the chronic pain population may become mentally and physically tired during the evening time leading to increased avoidance behaviours as a result of increased pain in the evening, especially if these patients engage in more activity in the afternoon.
The regression results are similar to other studies that found no association between baseline kinesiophobia and baseline pain catastrophizing, and 4-day average total daily activity,26 but our results indicate a significant negative relationship between baseline kinesiophobia and 4-day average sedentary activity at all times of the day. This important finding suggests that chronic pain patients’ activity habits are affected by their baseline level of kinesiophobia and baseline pain catastrophizing. Baseline kinesiophobia was positively associated with more minutes of light activity suggesting that individuals who participate in higher intensity activities may be more aware of their chronic pain and experience increased fear of movement while engaging in higher intensity activities. Individuals who choose higher intensity activities may understand the importance of physical activity in the treatment of their pain and engage in these activities despite their kinesiophobia. In contrast, individuals who choose sedentary activity are not engaging in any activity and as a result have less fear of movement because they are sedentary. Our results suggest that it is important to investigate activity intensity at different times of the day since the relationship between baseline kinesiophobia, baseline pain catastrophizing and 4-day average activity changes depending on the time of day.
Our chronic pain population engaged in more 4-day average light activity than sedentary activity, contradicting previous work indicating that individuals with chronic pain live a primarily sedentary lifestyle.8 In addition, our chronic pain population engaged in more 4-day average afternoon light activity compared to the morning and evening activity, agreeing with previous work suggesting people with chronic pain have higher movement intensity in the afternoon.13 Another study which assessed acceleration (via accelerometers) as their dependent variable found that chronic pain patients participate in higher average acceleration in the morning than the afternoon and evening.43 Comparing activity of chronic pain participants across studies is difficult due to methodological differences in the measurement of activity, but most authors agree that the activity level of a person suffering from chronic pain varies in intensity throughout the day.8,13,43
Our chronic pain population participated in 354.1±111 mins of sedentary activity averaged over a 4-day period, in contrast, sedentary activity from a robust sample of Canadians was reported to be 570–588 mins, highlighting our implication that individuals with chronic pain live a less sedentary lifestyle than previously reported.41 See Table 6 for study comparisons. This result was surprising given the severe health status of our participants, who had higher scores for kinesiophobia, catastrophizing, and depression compared to other studies.44–47 Our chronic pain population engaged in, on average, 484.4±118.6 mins of light activity over a 4-day period, whereas the large Canadian sample reported light activity as 245–258 mins, suggesting that our chronic pain population participated in more light activity than the Canadian population.41 Another study suggested that individuals with chronic pain participate in similar levels of sedentary and light activity compared to the general population.48 However, individuals with chronic pain are presumed to avoid movement so they do not aggravate their pain. Our finding that individuals in chronic pain are less sedentary compared to a healthy population is surprising, but this may be more of a statement about physical activity levels in the general population. One explanation may be that a healthy population is typically employed in a sedentary job,49,50 whereas our study participants were generally unemployed, and thus may have participated in more frequent light activity (ie, housework, errands, etc.). There is evidence to supporting little or no relationship between physical function and pain.51 Therefore, future interventions encouraging physical activity in chronic pain patients may be influenced by the quantity, intensity, and the time of the day this population participates in physical activity. Perhaps interventions aimed at increasing activity in the afternoon are better tolerated by the chronic pain population than originally thought.
 |
Table 6 Study Comparison Between Age, Height, Weight, BMI, and Daily Activity between the Current Study and a Study by Colley et al (Health Reports, 2011)37 in the General Canadian Population |
Measuring activity based on individual wake and sleep-onset times, and across three separate time periods during the day is a strength of our research. Some previous authors measured morning activity, for example, from 6am to 12pm, without accounting for variability in wake-onset time.13,52 Our chronic pain population experienced wake and sleep-onset times with high variability, which is consistent with previous research indicating the chronic pain population often experiences disrupted sleep and inconsistent sleep patterns.4 In our study, average wake time was 7:31am, so segmenting morning data using a 6am–12pm window would inflate the amount of sedentary activity in the morning because 90 mins of activity would have been recorded when the participants were asleep. Napping is different than sleeping because naps may be planned or unplanned, and sleeping is intentional behaviour to go to sleep. Further, the standard deviation around the average wake time was plus or minus 90 mins indicating there was a large variation in the wake times of our participants, this high variability also suggests that using individual wake and sleep-onset times would better capture the activity habits of the chronic pain population.
It is important to note the severe health status of our participants. For example, our participants’ kinesiophobia scores fell into the severe category (43–52 on the TSK) and were markedly higher compared to another study of 912 chronic pain sufferers whose scores fell into the moderate category (33–43).34 Further, our chronic pain population suffered from more pain catastrophizing (higher PCS scores than the referenced study) than a group of 60 subjects from an outpatient chronic pain treatment group.14,44,45 Additionally, our participants had higher depression scores (higher BECK scores than the referenced study), reported more severe subjective ratings of pain (higher MPQ scores than the referenced study), and more severe subjective perceptions of disability (higher PDI scores than the referenced study) than the previous literature in chronic pain populations.14,45–47 Nevertheless, our participants were most active in the afternoon engaging in light activity, and they participated in less sedentary activity compared to the general Canadian population.41
Our findings have important clinical implications, 1) Practitioners who encourage chronic pain patients to participate in activity must be aware that these patients will have higher kinesiophobia and pain catastrophizing, even when participating in higher intensity activity, and 2) our pain patients participated in the most light activity in the afternoon, so clinicians may be more successful at scheduling activity interventions in the afternoon.
Limitations
The severe health status of the participants in our study limits the generalizability of the results. Our study also included more women than men, but women tend to experience more chronic pain, especially fibromyalgia, so the demographic characteristics of our sample are similar to the general chronic pain population.2,53 We used a low wake threshold of 20 counts per minute to collect activity data from our participants. The low wake threshold was a feature of the Actiwatch Score designed to measure sleep in this particular population, which may affect the movement counts. Although the study sample of 21 participants is relatively small, the previous literature suggests a minimum of 12 subjects would be needed in these models to minimize bias in the regression coefficients.54 Recruiting subjects from a wait list may affect the results since this would be a sample of participants that are seeking treatment. Previous studies have approached subjects on a wait list in order not to withhold treatment for research purposes.31,32 However, most chronic pain subjects have interacted with multiple clinicians and are often seeking additional treatment options but their choice of seeking treatment may affect the results of this study. We did not conduct a power calculation a priori so these results must be interpreted with caution.
Conclusion
By evaluating 4-day average activity based on time of day (morning, afternoon, and evening) and by incorporating individual wake-sleep onset times we were able to demonstrate that baseline kinesiophobia is associated with 4-day average evening light activity and sedentary activity during the day. In addition, baseline catastrophizing was significantly correlated with more 4-day average light activity in the morning and more moderate to vigorous activity in the evening. Another key finding was that the participants with chronic pain are less sedentary than previously thought. Despite the severe health status of the chronic pain population in our research, our participants engaged in more 4-day average light activity than sedentary activity. The different results with total daily activity and daily activity categorized by time of day and activity intensity demonstrate the importance of separating daily activity by wake and sleep times, time of day, and activity intensity in the chronic pain population in future studies.
Acknowledgment
Matthew B. Miller and Melissa J. Roumanis have contributed equally to the research.
Funding
This research was partially supported by Centre de Recherché Interdisciplinaire en Réadaptation du Montréal metropolitain (CRIR), CRIR-759-0812, and La Corporation des Thérapeutes du Sport du Québec (CTSQ).
Disclosure
The authors report no conflicts of interest in this work.
References
1. De Souza LH, Frank AO. Experiences of living with chronic back pain: the physical disabilities. Disabil Rehabil. 2007;29(7):587–596. doi:10.1080/09638280600925852
2. Kurklinsky S, Perez RB, Lacayo ER, Sletten CD. The efficacy of interdisciplinary rehabilitation for improving function in people with chronic pain. Pain Res Treat. 2016;2016:7217684.
3. Duque I, Parra JH, Duvallet A. Maximal aerobic power in patients with chronic low back pain: a comparison with healthy subjects. Eur Spine J. 2011;20(1):87–93. doi:10.1007/s00586-010-1561-0
4. Smith MT, Haythornthwaite JA. How do sleep disturbance and chronic pain inter-relate? Insights from the longitudinal and cognitive-behavioral clinical trials literature. Sleep Med Rev. 2004;8(2):119–132. doi:10.1016/S1087-0792(03)00044-3
5. Song MR, Lee YS, Baek JD, Miller M. Physical activity status in adults with depression in the National Health and Nutrition Examination Survey, 2005–2006. Public Health Nurs. 2012;29(3):208–217. doi:10.1111/j.1525-1446.2011.00986.x
6. Werneck AO, Oyeyemi AL, Silva DR. Physical activity and depression: is 150 min/week of moderate to vigorous physical activity a necessary threshold for decreasing risk of depression in adults? Different views from the same data. Soc Psych Psych Epid. 2018;53(3):323–324. doi:10.1007/s00127-018-1490-5
7. Biswas A, Oh PI, Faulkner GE, Bajaj RR, Silver MA, Mitchell MS. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults (vol 162, pg 123, 2015). Ann Intern Med. 2015;163(5):400.
8. Perruchoud C, Buchser E, Johanek LM, Aminian K, Paraschiv-Ionescu A, Taylor RS. Assessment of physical activity of patients with chronic pain. Neuromodulation. 2014;17(Suppl 1):42–47. doi:10.1111/ner.2014.17.issue-s1
9. Herman KM, Hopman WM, Vandenkerkhof EG, Rosenberg MW. Physical activity, body mass index, and health-related quality of life in Canadian adults. Med Sci Sport Exer. 2012;44(4):625–636. doi:10.1249/MSS.0b013e31823a90ae
10. Chibnall JT, Tait RC. The pain disability index: factor structure and normative data. Arch Phys Med Rehabil. 1994;75(10):1082–1086. doi:10.1016/0003-9993(94)90082-5
11. Ware JE
12. Peppin JF, Marcum S, Kirsh KL. The chronic pain patient and functional assessment: use of the 6-minute walk test in a multidisciplinary pain clinic. Curr Med Res Opin. 2014;30(3):361–365. doi:10.1185/03007995.2013.828587
13. Raijmakers BG, Nieuwenhuizen MG, Beckerman H, de Groot S. Differences in the course of daily activity level between persons with and without chronic pain. Am J Phys Med Rehabil. 2015;94(2):
14. Sullivan MJ, Adams H, Horan S, Maher D, Boland D, Gross R. The role of perceived injustice in the experience of chronic pain and disability: scale development and validation. J Occup Rehabil. 2008;18(3):249–261. doi:10.1007/s10926-008-9140-5
15. Crombez G, Vlaeyen JWS, Heuts PHTG, Lysens R. Pain-related fear is more disabling than pain itself: evidence on the role of pain-related fear in chronic back pain disability. Pain. 1999;80(1–2):329–339. doi:10.1016/S0304-3959(98)00229-2
16. Schutze R, Rees C, Smith A, Slater H, Campbell JM, O’Sullivan P. How can we best reduce pain catastrophizing in adults with chronic noncancer pain? A systematic review and meta-analysis. J Pain. 2018;19(3):233–256. doi:10.1016/j.jpain.2017.09.010
17. Turk DC, Robinson JP, Burwinkle T. Prevalence of fear of pain and activity in patients with fibromyalgia syndrome. J Pain. 2004;5(9):483–490. doi:10.1016/j.jpain.2004.08.002
18. Susan H, Picavet J, Vlaeyen JWS, Schouten JSAG. Pain catastrophizing and kinesiophobia: predictors of chronic low back pain. Am J Epidemiol. 2002;156(11):1028–1034. doi:10.1093/aje/kwf136
19. Damsgard E, Thrane G, Anke A, Fors T, Roe C. Activity-related pain in patients with chronic musculoskeletal disorders. Disabil Rehabil. 2010;32(17):1428–1437. doi:10.3109/09638280903567877
20. Kindermans HPJ, Roelofs J, Goossens MEJB, Huijnen IPJ, Verbunt JA, Vlaeyen JWS. Activity patterns in chronic pain: underlying dimensions and associations with disability and depressed mood. J Pain. 2011;12(10):1049–1058. doi:10.1016/j.jpain.2011.04.009
21. Haldorsen EMH, Grasdal AL, Skouen JS, Risa AE, Kronholm K, Ursin H. Is there a right treatment for a particular patient group? Comparison of ordinary treatment, light multidisciplinary treatment, and extensive multidisciplinary treatment for long-term sick-listed employees with musculoskeletal pain. Pain. 2002;95(1–2):49–63. doi:10.1016/S0304-3959(01)00374-8
22. Liddle SD, Baxter GD, Gracey JH. Chronic low back pain: patients’ experiences, opinions and expectations for clinical management. Disabil Rehabil. 2007;29(24):1899–1909. doi:10.1080/09638280701189895
23. Gordon D, Eaton L, Rue T, et al. Measuring activity goals important to individuals with chronic pain. J Pain. 2014;15(4):S100–S100. doi:10.1016/j.jpain.2014.01.410
24. Main CJ, George SZ. Psychologically informed practice for management of low back pain: future directions in practice and research. Phys Ther. 2011;91(5):820–824. doi:10.2522/ptj.20110060
25. Vlaeyen JWS, Kolesnijders AMJ, Boeren RGB, Vaneek H. Fear of movement (re)injury in chronic low-back-pain and its relation to behavioral performance. Pain. 1995;62(3):363–372. doi:10.1016/0304-3959(94)00279-N
26. Carvalho FA, Maher CG, Franco MR, et al. Fear of movement is not associated with objective and subjective physical activity levels in chronic nonspecific low back pain. Arch Phys Med Rehab. 2017;98(1):96–104. doi:10.1016/j.apmr.2016.09.115
27. Elfving B, Andersson T, Grooten WJ. Low levels of physical activity in back pain patients are associated with high levels of fear-avoidance beliefs and pain catastrophizing. Physiother Res Int. 2007;12(1):14–24. doi:10.1002/(ISSN)1471-2865
28. Blagestad T, Pallesen S, Lunde LH, Sivertsen B, Nordhus IH, Gronli J. Sleep in older chronic pain patients a comparative polysomnographic study. Clin J Pain. 2012;28(4):277–283. doi:10.1097/AJP.0b013e3182313899
29. Stiefel F, Stagno D. Management of insomnia in patients with chronic pain conditions. CNS Drugs. 2004;18(5):285–296. doi:10.2165/00023210-200418050-00002
30. Skender S, Ose J, Chang-Claude J, et al. Accelerometry and physical activity questionnaires – a systematic review. BMC Public Health. 2016;16:515.
31. Spenkelink CD, Hutten MM, Hermens HJ, Greitemann BO. Assessment of activities of daily living with an ambulatory monitoring system: a comparative study in patients with chronic low back pain and nonsymptomatic controls. Clin Rehabil. 2002;16(1):16–26. doi:10.1191/0269215502cr463oa
32. Zhang QJS, Lufei Y. The effectiveness of group-based physiotherapy-led behavioural psychological interventions on adults with chronic low back pain. A systematic review and meta-analysis. Am Jour Phys Med Rehab. 2019;93(3):215–225. doi:10.1097/PHM.0000000000001053
33. Hoftun GB, Romundstad PR, Zwart JA, Rygg M. Chronic idiopathic pain in adolescence – high prevalence and disability: the young HUNT study 2008. Pain. 2011;152(10):2259–2266. doi:10.1016/j.pain.2011.05.007
34. Neblett R, Hartzell MM, Mayer TG, Bradford EM, Gatchel RJ. Establishing clinically meaningful severity levels for the Tampa Scale for Kinesiophobia (TSK-13). Eur J Pain. 2016;20(5):701–710. doi:10.1002/ejp.795
35. Cook AJ, Brawer PA, Vowles KE. The fear-avoidance model of chronic pain: validation and age analysis using structural equation modeling. Pain. 2006;121(3):195–206. doi:10.1016/j.pain.2005.11.018
36. Hirsh AT, George SZ, Bialosky JE, Robinson ME. Fear of pain, pain catastrophizing, and acute pain perception: relative prediction and timing of assessment. J Pain. 2008;9(9):806–812. doi:10.1016/j.jpain.2008.03.012
37. Harris CA, D’Eon JL. Psychometric properties of the Beck Depression Inventory-Second Edition (BDI-II) in individuals with chronic pain. Pain. 2008;137(3):609–622. doi:10.1016/j.pain.2007.10.022
38. Tait RC, Pollard CA, Margolis RB, Duckro PN, Krause SJ. The Pain Disability Index: psychometric and validity data. Arch Phys Med Rehabil. 1987;68(7):438–441.
39. Respironics. Actiware and Actiware CT Software and Hardware Manual: Actiwatch Communication and Sleep Analysis Software with ActiReader. Respironics, Inc.; 2009.
40. Migueles JH, Cadenas-Sanchez C, Ekelund U, et al. Accelerometer data collection and processing criteria to assess physical activity and other outcomes: a systematic review and practical considerations. Sports Med. 2017;47(9):1821–1845. doi:10.1007/s40279-017-0716-0
41. Colley RC, Garriguet D, Janssen I, Craig CL, Clarke C, Tremblay MS. Physical activity of Canadian adults: accelerometer results from the 2007 to 2009 Canadian health measures survey. Health Rep. 2011;22(1):1–7.
42. Rainville P. Brain mechanisms of pain affect and pain modulation. Curr Opin Neurobiol. 2002;12(2):195–204. doi:10.1016/S0959-4388(02)00313-6
43. van Weering MGH, Vollenbroek-Hutten MMR, Tonis TM, Hermens HJ. Daily physical activities in chronic lower back pain patients assessed with accelerometry. Eur J Pain. 2009;13(6):649–654. doi:10.1016/j.ejpain.2008.07.005
44. Osman A, Barrios FX, Gutierrez PM, Kopper BA, Merrifield T, Grittmann L. The pain catastrophizing scale: further psychometric evaluation with adult samples. J Behav Med. 2000;23(4):351–365. doi:10.1023/A:1005548801037
45. Nicholas MK, Asghari A, Blyth FM. What do the numbers mean? Normative data in chronic pain measures. Pain. 2008;134(1–2):158–173. doi:10.1016/j.pain.2007.04.007
46. Orenius T, Koskela T, Koho P, et al. Anxiety and depression are independent predictors of quality of life of patients with chronic musculoskeletal pain. J Health Psychol. 2013;18(2):167–175. doi:10.1177/1359105311434605
47. Richardson EJ, Ness TJ, Doleys DM, Banos JH, Cianfrini L, Richards JS. Depressive symptoms and pain evaluations among persons with chronic pain: catastrophizing, but not pain acceptance, shows significant effects. Pain. 2009;147(1–3):147–152. doi:10.1016/j.pain.2009.08.030
48. Dansie EJ, Turk DC, Martin KR, Van Domelen DR, Patel KV. Association of chronic widespread pain with objectively measured physical activity in adults: findings from the national health and nutrition examination survey. J Pain. 2014;15(5):507–515. doi:10.1016/j.jpain.2014.01.489
49. JO WH H, Reed GW, Peters JC. Obesity and the environment: where do we go from here? Science. 2003;299(5608):853–855. doi:10.1126/science.1079857
50. Brown WJ, Miller YD, Miller R. Sitting time and work patterns as indicators of overweight and obesity in Australian adults. Int J Obesity. 2003;27(11):1340–1346. doi:10.1038/sj.ijo.0802426
51. Grotle M, Vollestad NK, Brox JI. Clinical course and impact of fear-avoidance beliefs in low back pain: prospective cohort study of acute and chronic low back pain: II. Spine (Phila Pa 1976). 2006;31(9):1038–1046. doi:10.1097/01.brs.0000214878.01709.0e
52. Spenkelink CD, Hutten MMR, Hermens HJ, Greitemann BOL. Assessment of activities of daily living with an ambulatory monitoring system: a comparative study in patients with chronic low back pain and nonsymptomatic controls. Clin Rehabil. 2002;16(1):16–26. doi:10.1191/0269215502cr463oa
53. Latorre-Roman P, Santos-Campos M, Heredia-Jimenez J, Delgado-Fernandez M, Soto-Hermoso V. Analysis of the performance of women with fibromyalgia in the six-minute walk test and its relation with health and quality of life. J Sports Med Phys Fitness. 2014;54(4):511–517.
54. Austin PC, Steyerberg EW. The number of subjects per variable required in linear regression analyses. J Clin Epidemiol. 2015;68(6):627–636. doi:10.1016/j.jclinepi.2014.12.014
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.