Back to Journals » Neuropsychiatric Disease and Treatment » Volume 17
Characteristics of TCM Constitution and Related Biomarkers for Mild Cognitive Impairment
Authors Deng X, Teng J, Nong X, Yu B, Tang L, Liang J, Zou Z, Liu Q, Zhou L, Li Q, Zhao L
Received 5 November 2020
Accepted for publication 22 March 2021
Published 20 April 2021 Volume 2021:17 Pages 1115—1124
DOI https://doi.org/10.2147/NDT.S290692
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Xiangming Deng,1 Jinlong Teng,1 Xiucheng Nong,1 Bihan Yu,1 Liying Tang,2,* Jinsong Liang,3 Zhuocheng Zou,1,* Qiang Liu,2,* Lu Zhou,4 Qirong Li,5 Lihua Zhao1
1Department of Acupuncture and Moxibustion, The First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning, 530023, People’s Republic of China; 2The Xinhu Outpatient Clinic of the People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning, 530021, People’s Republic of China; 3Department of Clinical Laboratory of the First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning, 530023, People’s Republic of China; 4Medical Examination Center of the First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning, 530023, People’s Republic of China; 5Renai Branch of the First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning, 530023, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lihua Zhao
Department of Acupuncture and Moxibustion, The First Affiliated Hospital of Guangxi University of Chinese Medicine, Dongge Road & 89-9, Nanning, 530023, People’s Republic of China
Tel +86 130 06913108
Email [email protected]
Qirong Li
Department of Acupuncture and Moxibustion, Renai Branch of the First Affiliated Hospital of Guangxi University of Chinese Medicine, Mingxiu East Road & 179, Nanning, 530023, People’s Republic of China
Tel +86 136 07717755
Email [email protected]
Introduction: The incidence of Alzheimer’s disease is on the rise, early detection of cognitive impairment of the elderly is very important. In traditional Chinese medicine, constitution is related to the susceptibility of the human body to diseases. Based on the theory of constitution of traditional Chinese medicine (TCM), the human population can be classified into 9 constitutions. However, little is known about the characteristics of medical constitution and related biomarkers in subjects with mild cognitive impairment (MCI).
Methods: We measured the TCM Constitution of 214 subjects by using the Constitution in Chinese Medicine Questionnaire (CCMQ). MMSE and MoCA were used to assess cognitive function. The subjects were divided into mild cognitive impairment group (MCI, n = 152) and normal control group (NC, n = 62). The levels of serum Hcy and serum/urine 8-iso-PGF 2α were determined.
Results: 1) It was found that there was a significant difference in constitution types between MCI and NC. There were significant differences in MMSE and MoCA score, serum Hcy and serum/urine 8-iso-PGF 2a levels between the two groups. 2) In logistic regression analysis, the variables with statistical significance were TCM Constitution of Yang-Deficient, Phlegm-Dampness, Blood-Stasis and abnormal increase of Hcy (OR> 1). 3) The MoCA scores had a positive correlation with the MMSE. A statistically significant inverse association was found between serum Hcy, blood and urine 8-iso-PGF 2a and scores of cognitive assessment in MCI.
Conclusion: Constitution types (Yang-Deficient, Phlegm-Dampness and Blood-Stasis) and abnormal serum Hcy elevation can be used as risk factors for MCI. MoCA scores can serve to detect MCI at early stage. Serum/urine 8-iso-PGF 2α has a certain relationship with MCI. Higher levels of serum/urine 8-iso–PGF 2α are more likely to be associated with MCI risk.
Keywords: cognitive impairment, constitutions of traditional Chinese medicine, biomarkers, MoCA score
Introduction
Mild cognitive impairment (MCI) is commonly used to refer to patients with objective cognitive impairment and normal daily living abilities that do not meet the criteria for dementia.1–4 MCI is an important risk factor for dementia and in some cases may represent the prodromal phase of AD or other neurodegenerative diseases. Approximately 35% of MCI patients progress to AD dementia during a 3-year follow-up with an annual conversion rate of 5%-10%.5,6 However, compared with healthy subjects, the conversion rate of AD was higher.7 Therefore, the identification of MCI is very important in clinic. Currently, diagnostic aids for MCI and AD include cerebrospinal fluid (CSF) examination and blood biomarkers,8,9 functional MRI,10 and PET imaging.11 However, CSF is invasive. Functional MRI and PET imaging are limited by equipment and expensive examination costs, thus, we need more simple and accurate diagnostic tests to identify MCI patients whose AD may be a potential cause in the early stages of the disease. The most widely used measure of cognitive impairment is the mini–mental state examination (MMSE) scale. However, there may be a ceiling effect in MMSE, which can lead to false negative results. The MoCA is a brief cognitive screening tool with high sensitivity and specificity for detecting MCI as currently conceptualized in patients performing in the normal range on the MMSE.12–18 The use of biomarkers also contributes to the early diagnosis and early treatment of MCI. Studies have shown that Homocysteine (Hcy), 8-iso-prostaglandin F2α (8-iso-PGF2α) is a potential biomarker for cognitive impairment.19,20 Hcy is a sulfur–containing amino acid. Current studies have shown that Hcy is associated with cognitive impairment, and its possible ways of inducing cognitive impairment include increasing glutamate excitotoxicity, reducing DNA repair ability of neurons, accelerating oxidative stress and Aβ protein formation, and damaging the ability of hippocampal neurons.21–23 On the other hand, Hcy can induce vascular endothelial cell dysfunction, which may lead to cerebrovascular cognitive impairment.24 8-iso-PGF 2α is the end product of endogenous lipid peroxidation and the product of free radical catalyzed arachidonic acid. Autopsy showed that patients with Alzheimer’s disease had obvious oxidative damage, and the content of 8-iso-PGF 2α in temporal lobe and frontal lobe was significantly increased.25 In addition, studies have shown that plasma 8-iso-PGF 2α concentration is related to cognitive impairment in non-dementia elderly.26,27 8-iso-PGF 2α has been accepted as a reliable biomarker for evaluating oxidative stress.28,29 In the theory of traditional Chinese medicine (TCM), the body constitution is an individual character compatible with nature and the social environment, there are nine constitutions: Neutral, Qi-Deficient, Yang-Deficient, Yin-Deficient, Phlegm-Dampness, Dampness-Heat, Blood-Stasis, Qi–Depression, and Special.30,31 The difference in individual constitution makes the individual susceptible to certain pathogenic factors, and even has an impact on physical and mental health.32–34 In April 2009, The Constitution in Chinese Medicine Questionnaire (CCMQ) was published and recommended as the constitution measurement standard by Chinese Traditional Medicine Association.35 The reliability and construct validity of the scale were thoroughly evaluated.36 More than 20,000 surveys have been completed in China and other Asian countries to help the respondents understand their TCM Constitution and get health care advice. With the wide application of CCMQ in recent years, more and more research on TCM constitution has appeared, such as clinical research related to metabolic syndrome (MetS), dyslipidemia, etc,37,38 as well as research on the relationship between biomarkers of type 2 diabetes and TCM syndromes.39 All of these have positive significance for understanding the Constitution and the diagnosis and treatment of diseases. Based on previous studies,40 we know that constitutions are associated with vascular cognitive impairment. A recent study showed that there is an interaction between blood-stasis constitution and atherosclerosis in cognitive impairment in the elderly.41 However, few studies have systematically explored the relationship between TCM Constitution and biomarkers related to cognitive decline. This study aimed to investigate the MCI characteristics from the perspective of TCM constitution and biomarkers, to reveal the distribution of constitutional types of patients with MCI.
Materials and Methods
Subjects
A total of 214 subjects (71 males, 143 females) were recruited from the health examination center and the memory outpatient clinic of the First Affiliated Hospital of Guangxi University of Traditional Chinese Medicine and community center in Guangxi Nanning in the period between January 2014 to December 2017. Inclusion criteria were: 1) 55≤age≤82 years, 2) MCI group needs to meet the MCI diagnostic criteria of Mayo Clinic-revised defined by Petersen et al,42,43 with 24≤MMSE score≤27, 18≤MoCA score≤30,15 3) NC group should be complied with noncognitive impairment, with MMSE score > 27 points, MoCA score >18 points.15 Exclusion criteria: 1) age below 55 years, 2) other diseases with advanced, severe primary diseases such as liver and kidney, 3) severe hearing and visual impairment, unable to cooperate with assessment, 4) history of neurological diseases (such as macrovascular stroke, dementia, Parkinson’s disease, seizures), 5) mental illness (such as schizophrenia, bipolar disorder), 6) subjects who are known to have used other drugs that may affect serum Hcy, serum and urine 8-iso-PGF 2a.
TCM Constitution Assessment
The TCM constitution classification table adopts Wang Qi’s nine-point method of constitution classification, which divides human constitution into Neutral, Qi-Deficient, Yang-Deficient, Yin-Deficient, Phlegm-Dampness, Dampness-Heat, Blood-Stasis, Qi–Depression, and Special constitution.44 The TCM constitutions were measured and classified with Constitution in Chinese Medicine Questionnaire (CCMQ).45 Neutral type is a balanced one with higher score indicating a better constitution status, while the other eight types are pathological types with higher score indicating worse constitution status. There are 66 items in CCMQ. According to the constitution classification, it is divided into 9 sub scales, each sub scale contains 7–8 items, and each item is divided into 1–5 points. As the subjects are 55 years old and above, in order to avoid wrong answers caused by differences in education level, knowledge composition and understanding ability, all items in the CCMQ are asked and filled in one by one by trained researchers. After filling in, researchers need to check the scale again to ensure the integrity of the data and then take it back on the spot. After taking back, according to the scoring method, the scores of each item of the nine subscales were added up, and the total score calculated by each subscale was the original score of each constitution, and then the transformation score was calculated according to the formula, the transformation score = [(Original Score - number of items)/(number of items × 4)] × 100%. When the transformation score of Neutral constitution≥60, and the transformation score of the other eight constitutions≤40, it is judged as Neutral constitution. When only one of the biased constitutions’ transformation score is ≥40, we judge it as biased constitution. For a certain biased constitution, if there are multiple constitutions at the same time and the transformation score≥40, the biased constitution with the highest transformation score is used as the judgment constitution.
Neuropsychological Assessment
Cognitive assessments include Mini Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA). The MMSE consists of 10 tasks, testing direction, memory, attention, calculation, language (naming, repetition, auditory comprehension, reading and writing) and visual special ability, with a total score of 30.
MoCA tests 8 cognitive areas, visual spatial ability, attention, executive function, immediate memory, delayed memory, language, abstraction, calculation and orientation, the highest total score is 30. If the subjects’ education years are less than 12 years, the measured total score is the test score plus 1 point.15 All tests were administered and graded by professionals trained in neuropsychological testing.
Blood Biochemical Tests
Serum and Urine 8-Iso-PGF 2a Testing
The level of serum and urine 8-iso-PGF 2a was measured by enzyme-linked immunosorbent assay (ELISA). In the morning, 5 mL of fasting venous blood and 5 mL of urine were taken from the subjects; serum was separated and kept below −20°C until analysis; urine was immediately kept under −80°C until analysis. Enzyme-linked immunosorbent assay (ELISA) kits were purchased from Shanghai Lianshuo Biotechnology Co., Ltd. (Batch No.: 201605, 201610, 201612, 201703, 201707, 201710, 201712).
Hcy Test
On an empty stomach the next morning after a normal dinner, 3 mL of elbow venous blood was taken by a professional and collected with 5 mL vacuum blood collection tube (without anticoagulant) and sent to the laboratory for testing. The instrument was Hitachi 7600. Automatic biochemical analyzer and its supporting reagents were used. 0 < Hcy ≤ 10 μmol/L is the normal value.
Data Analysis
The results were reported by using professional statistical software (Statistical Package for Social Sciences 23.0, SPSS). All data were expressed as mean ± standard deviation (M±SD). Two independent sample t-tests were used for comparison between measurement data sets. Chi-squared test was used for counting data. Pearson correlation coefficient (r) was used to correlate MMSE, MoCA, and serum biological variables analysis; logistic regression analysis was used to analyze the binary dependent variables MCI and independent variables including nine TCM constitutions, abnormal Hcy elevation, occupation, age, education level. Multiple linear variables regression analysis was used for MMSE, MoCA, urine 8-iso-PGF 2αlevel, serum 8-iso-PGF 2α level. The ratio of risk to odds (OR), regression coefficient (B) and P value were analyzed. The difference was statistically significant at P<0.05.
Results
Sociodemographic and Medical Characteristics at Inclusion
A total of 214 subjects were included in the final sample, including 152 subjects (50 males and 102 females) in the cognitive impairment group and 62 controls (21 males and 41 females). The ratio of gender, age, education, occupation status and blood pressure of the two groups were shown in Table 1. There was no difference in gender, age, education level, occupation, blood pressure between the two groups. There were significant differences in MMSE and MoCA score, serum Hcy and serum/urine 8-iso-PGF 2a levels between the two groups (P < 0.001), see Table 2.
 |
Table 1 Sociodemographic and Medical Characteristics at Inclusion (MCI=152, NC=62) |
 |
Table 2 Comparison Between MCI and NC Subjects Regarding Mean Scores of Cognitive Assessment Scales and Urine/Serum 8-Iso-PGF2α Level |
TCM Constitution Distribution of MCI Patients and Normal Control Group
There was statistical significance between the two groups in terms of the proportion of Neutral type and unneutral types’ distribution (P < 0.01). In NC group, the proportion of Neutral type was 64.5%, and the unneutral type was 35.5%. In MCI group, the proportion of Neutral type was only 33.6%. Compared with NC group, the unneutral type was 66.4%. Yang-Deficient was the first, followed by Qi-Deficient, Phlegm-Dampness and Blood-Stasis, which indicates that the proportion of unneutral types in MCI patients was higher than that in NC group except Qi-Deficient. There was no special constitution in both groups, Table 3.
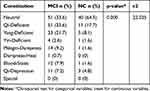 |
Table 3 Characteristics of Traditional Chinese Medical Constitution |
Logistic Regression Analysis of Common TCM Constitution and Related Abnormal Hcy Elevation in Two Groups
The binary variables logistic regression analysis was carried out on non-frequency covariables including age, occupation, education level, TCM constitutions, and binary response variable MCI cases or not. The overall percentage for MCI classification was 71.5% (cut-off value of 0.5). It showed TCM constitution covariables, wald=17.485, P=0.015 (Table 4). There were significant differences in the three types of Yang-Deficient, Phlegm-Dampness, Blood-Stasis and the abnormal Hcy elevation (P<0.05), and three constitutions OR (95% CI) of Yang-Deficient, Phlegm-Dampness, Blood-Stasis and the abnormal Hcy elevations were 1.853 (1.893,15.082), 11.006 (1.376,88.02), 11.006 (1.376,88.02) and 1.973 (1.01, 3.855) respectively (Table 5).
 |
Table 4 Logistic Regression Analysis: Non-Frequency Covariables (N=214) |
 |
Table 5 Logistic Regression Analysis of 7 Common TCM Constitutions and Abnormal Hcy in Two Groups |
Multiple Linear Variables Regression Analysis of MoCA Scores, Urinary 8-Iso-Prostaglandin F2α (8-Iso-PGF2α), and Serum 8-Iso-PGF2α
MoCA, urine/serum 8-iso-PGF2α had significant influence on the dependent variable MMSE by multiple linear variables regression analysis (R Square=0.50, p=0.0001) (Table 6). It suggested that the level of serum and urine 8-iso-PGF2α has a certain relationship with MCI.
 |
Table 6 Coefficients of Multiple Linear Regression Analysis |
Correlation Between Cognitive Scores and Changes of Serum Hcy, Urinary 8-Iso-Prostaglandin F2α (8-Iso-PGF2α), and Serum 8-Iso-PGF2α
The MoCA scores had a positive correlation with MMSE (r =0.564, P <0.001), there was negative correlation between urine/serum 8-iso-PGF 2a (r =−0.445 P <0.001/r =−0.561, P <0.001) and MMSE. Scatter plots are shown in Figure 1.
Discussion
According to the constitution theory of traditional Chinese medicine, the determination of constitution can provide reference for the prevention and treatment of diseases. In recent years, a series of evaluations has been carried out on the correlation between TCM Constitution and diseases, eg, Phlegm-Dampness, Qi-Deficient and Dampness-Heat are the main constitution types of MetS patients, in which phlegm dampness and Qi deficiency are related to the occurrence of MetS.37 Neutral constitution is the protective factor of dyslipidemia, while Phlegm-Dampness is the risk factor of dyslipidemia.38 We studied the relationship between TCM constitutional characteristics and related biomarkers and cognitive abilities in patients with cognitive impairment to help with early identification and intervention of MCI.
Due to the relative stability of constitution, each living individual presents different physical characteristics. Neutral constitution is an ideal type. With the increase of age, the body’s Qi gradually decreases. Qi-Deficiency prevails in old age.30 This study found that the proportion of Qi-Deficiency in MCI group was similar to that in normal NC group, indicating that Qi-Deficiency should be the common feature of unneutral type in this age stage. Our study showed that the ORs of three unneutral constitutions of Yang-Deficient, Phlegm-Dampness and Blood-Stasis and abnormal Hcy elevation (>10 μmol/L) were significantly greater than 1, which resulted in significantly higher risk of MCI than in other constitutions and normal Hcy level (≤10 μmol/L), indicating that people both with Yang-Deficient, Phlegm-Dampness, Blood-Stasis, and abnormal Hcy elevation were more likely to suffer from MCI. The performance of Yang-deficiency: cold hands and feet, fear of cold, pale face, prefer hot food and drink, fat and tender tongue with tooth print and white coating, deep and weak pulse. The performance of Phlegm-dampness: overweight with abdomen fatness, more phlegm and sticky sensation in mouth, slippery and fat tongue with thick white coating, tightness in the chest, laziness prone to lying down, light yellow complexion, more oil on the face, greasy and fat tongue with white coating. The performance of Blood stasis: dark complexion, lips and eye sockets, roughness of skin, all kinds of pain.46
Measurement of relevant biomarkers can help to intervene in early cognitive impairment.47,48 Hcy is a sulfur-containing nonessential amino acid, which participates in the metabolism of cysteine and methionine.49 Methionine or cysteine metabolic dysfunction can cause hyperhomocysteinemia, lead to small vessel damage, lead to leukoaraiosis or asymptomatic cerebral infarction, and leukoaraiosis and asymptomatic cerebral infarction are important risk factors for cognitive impairment.50 Lu et al51 found that hyperhomocysteinemia promoted the appearance of white matter damage symptoms and was closely related to the degree of lesions. Studies have shown that hyperhomocysteinemia is a risk factor for cognitive dysfunction.52–55 Annerbo et al found that plasma homocysteine levels were significantly correlated with the risk of conversion to AD in patients with MCI. The plasma homocysteine levels in patients with MCI conversion to AD were higher than those in untransformed patients,56 suggesting that the conversion of MCI to AD may depend on the deposition of the toxic metabolite homocysteine. At the same time, this study found that Hcy abnormality is related to the onset of MCI, similar to previous studies.
Several studies57–61 have shown that in the process of AD and oxidative stress, the accumulation of oxidative damage to proteins, DNA and fat is considered to be an important factor in the pathogenesis of AD. Reactive oxygen species cause non-enzymatic peroxidation of polyunsaturated fatty acids, producing different peroxidation products. Many lipid hydroperoxides, including oxidized phospholipids (oxPLs), are considered to be biologically relevant lipid signaling molecules. 8-isoprostane (8-IsoP) is used for high-throughput analysis of oxPLs and is a biomarker of oxidative stress.62 Pratico et al63 found that cerebral oxidative stress was enhanced in patients with MCI, and levels of 8,12-iso-iPF (2α)-VI were detected in plasma, cerebrospinal fluid, and urine, indicating a progression to AD trend. This study found that MMSE, MoCA scores were negatively correlated with serum Hcy and serum/urine 8-iso-PGF 2α, among which MMSE scores were moderately correlated with serum/urine 8-iso-PGF 2α, while MMSE scores were weakly correlated with serum Hcy. MoCA scores were weakly correlated with serum Hcy and serum/urine 8-iso-PGF 2α. It is suggested that the level of serum and urine 8-iso-PGF2α has a certain relationship with MCI. Most patients with AD were diagnosed during dementia, but the irreversible damage was found in the neurons of the brain. In the MCI stage, biomarkers can be used to identify potential clinical causes and to evaluate the severity of the disease. In the MCI period, timely and accurate diagnosis and treatment will hopefully delay or prevent further development of AD. Therefore, it is very important to detect biomarkers. At the same time, the idea and practice of combining TCM Constitution with modern medicine is forming a medical and health innovation system.64 Because the two medical systems are complementary, the identification of TCM constitution of MCI is helpful for the subsequent development of personalized treatment.
Conclusion
We found that MCI patients were dominated by the constitution of Yang-Deficient, followed by Phlegm-Dampness, Blood-Stasis, and abnormal Serum Hcy elevation. Among MCI patients, there was a negative correlation between urine/serum 8-iso-PGF 2a and the MMSE, the MoCA scores had a positive correlation with the MMSE. Higher levels of serum/urine 8-iso-PGF2α were more likely to be associated with risk of MCI. From the perspective of the combination of disease and TCM Constitution, combined with modern research methods and objective indicators, we searched for MCI susceptible constitution and investigated biomarkers to provide the basis for the identification of MCI and the possibility of specific individual diagnosis and treatment. However, there are still many problems to be further verified, such as whether there is a corresponding relationship between clinical research and basic research. Because the research on constitution of traditional Chinese medicine is still a relatively new subject, the research on the correlation between constitution and diseases outside China is still very little. The MCI patients in this study were all from China. Therefore, it is not clear whether the conclusions drawn in this study can be extended to other populations.
Data Sharing Statement
All participants provided informed consent. The data analyzed here have not been made publicly available yet. The data analyzed will be made available from the corresponding author on reasonable request in 2-3 years. Corresponding author Lihua Zhao, E-mail: [email protected].
Ethical Consideration
This study was conducted in accordance with the Declaration of Helsinki. All the subjects provided informed consent according to a protocol approved by the Research Ethics Committee of the First Affiliated Hospital of Guangxi University of Traditional Chinese Medicine (Lot number: [2016] 009). The study was registered in http://www.chictr.org.cn. The Clinical Trial Registration Number was ChiCTR-IPR-16009144.
Funding
This work was supported by the National Natural Science Foundation of China, (Grant No. 81360561); The Science and Technology plan of Guangxi (Gui, Grant No. 14124004-1-27), and the Science and Technology plan of Guangdong (Grant No. 2016A020215202).
Disclosure
The authors declare no conflicts of interest.
References
1. Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):270–279. doi:10.1016/j.jalz.2011.03.008
2. Petersen RC, Caracciolo B, Brayne C, Gauthier S, Jelic V, Fratiglioni L. Mild cognitive impairment: a concept in evolution. J Intern Med. 2014;275(3):214–228. doi:10.1111/joim.12190
3. Winblad B, Palmer K, Kivipelto M, et al. Mild cognitive impairment—beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med. 2004;256(3):240–246. doi:10.1111/j.1365-2796.2004.01380.x
4. Bowen J, Teri L, Kukull W, McCormick W, McCurry SM, Larson EB. Progression to dementia in patients with isolated memory loss. Lancet. 2007;349(9054):763–765. doi:10.1016/S0140-6736(96)08256-6
5. Mitchell AJ, Shiri-Feshki M. Rate of progression of mild cognitive impairment to dementia—meta-analysis of 41 robust inception cohort studies. Acta Psychiatr Scand. 2009;119(4):252–265. doi:10.1111/j.1600-0447.2008.01326.x
6. Grundman M, Petersen RC, Ferris SH, et al. Mild cognitive impairment can be distinguished from Alzheimer disease and normal aging for clinical trials. Arch Neurol. 2004;61(1):59–66. doi:10.1001/archneur.61.1.59
7. Devanand DP, Bansal R, Liu J, et al. MRI hippocampal and entorhinal cortex mapping in predicting conversion to Alzheimer’s disease. Neuroimage. 2012;60(3):1622–1629. doi:10.1016/j.neuroimage.2012.01.075
8. Olsson B, Lautner R, Andreasson U, et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: a systematic review and meta-analysis. Lancet Neurol. 2016;15(7):673–684. doi:10.1016/S1474-4422(16)00070-3
9. Zetterberg H. Blood-based biomarkers for Alzheimer’s disease-an update. J Neurosci Methods. 2019;319:2–6. doi:10.1016/j.jneumeth.2018.10.025
10. Rombouts SARB, Barkhof F, Goekoop R, et al. Altered resting state networks in mild cognitive impairment and mild Alzheimer’s disease: an fMRI study. Hum Brain Mapp. 2010;26(4):231–239. doi:10.1002/hbm.20160
11. Valotassiou V, Malamitsi J, Papatriantafyllou U, et al. SPECT and PET imaging in Alzheimer’s disease. Ann Nucl Med. 2018;32(9):583–593. doi:10.1007/s12149-018-1292-6
12. Lu PH, Lee GJ. The role of neuropsychology in the assessment of the cognitively impaired elderly. Neurol Clin. 2017;35(2):191–206. doi:10.1016/j.ncl.2017.01.002
13. Yong SS, Dong WY, Kim HJ, Park YH, Kim SY. Characteristic differences in the mini-mental state examination used in Asian countries. BMC Neurol. 2017;17(1):141–150. doi:10.1186/s12883-017-0925-z
14. Devenney E, Hodges JR. The Mini-Mental State Examination: pitfalls and limitations. Pract Neurol. 2016;17(1):79–80. doi:10.1136/practneurol-2016-001520
15. Paula TT, Helen H, Shufang W, Brett W, Andrew JS, Andrew JS. Relationship between the Montreal Cognitive Assessment and Mini-mental State Examination for assessment of mild cognitive impairment in older adults. BMC Geriatr. 2015;15(1):107. doi:10.1186/s12877-015-0103-3
16. Espino DV, Lichtenstein MJ, Palmer RF, Hazuda HP. Ethnic differences in mini-mental state examination (MMSE) scores: where you live makes a difference. J Am Geriatr Soc. 2001;49(5):538–548. doi:10.1046/j.1532-5415.2001.49111.x
17. Nasreddine ZS, Phillips NA, Bedirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699. doi:10.1111/j.1532-5415.2005.53221.x
18. Giebel CM, Challis D. Sensitivity of the mini-mental state examination, montreal cognitive assessment and the addenbrooke’s cognitive examination III to everyday activity impairments in dementia: an exploratory study. Int J Geriatr Psychiatry. 2016;32(10):1085–1093. doi:10.1002/gps.4570
19. García-Blanco A, Baquero M, Vento M, Gil E, Bataller L, Cháfer-Pericás C. Potential oxidative stress biomarkers of mild cognitive impairment due to Alzheimer disease. J Neurol Sci. 2017;373(15):295–302
20. Minagawa H, Watanbe A, Akastu H, et al. Homocysteine, another risk factor for Alzheimer disease, impairs apolipoprotein E3 function. J Biol Chem. 2010;285(49):1473–1474. doi:10.1074/jbc.M110.146258
21. Zhuo JM, Portugal GS, Kruger WD, et al. Diet-induced hyperhomocysteinemia increases amyloid-beta formation and deposition in a mouse model of Alzheimer’s disease. Curr Alzheimer Res. 2010;7(2):140–149. doi:10.2174/156720510790691326
22. Kinoshita M, Numata S, Tajima A, et al. Plasma total homocysteine is associated with DNA methylation in patients with schizophrenia. Epigenetics. 2013;8(6):584–590. doi:10.4161/epi.24621
23. Dietrich-Muszalska A, Malinowska J, Olas B, et al. The oxidative stress may be induced by the elevated homocysteine in schizophrenic patients. Neurochem Res. 2012;37(5):1057–1062. doi:10.1007/s11064-012-0707-3
24. Sreckovic B, Sreckovic VD, Soldatovic I, et al. Homocysteine is a marker for metabolic syndrome and atherosclerosis. Diabetes Metab Syndr. 2017;11(3):179–182. doi:10.1016/j.dsx.2016.08.026
25. Pratico D, Lee VM, Trojanowski JQ, Rokach J, FitzGerald GA. Increased F2-isoprostanes in Alzheimer’s disease: evidence for enhanced lipid peroxidation in vivo. FASEB J off Publ Fed Am Soc Exp Biol. 1998;12(15):1777–1783. doi:10.1096/fasebj.12.15.1777
26. García-Blancoa A, Miguel B, Máximo V, et al. Esperanza Gila Luis Batallerb Consuelo Cháfer-Pericása. Potential oxidative stress biomarkers of mild cognitive impairment due to Alzheimer disease. J Neurol Sci. 2017;373(15):295–302.
27. Pratico D. The neurobiology of isoprostanes and Alzheimer’s disease. Biochimica Et Biophysica Acta Mol Cell Biol Lipids. 2010;1801(8):930–933. doi:10.1016/j.bbalip.2010.01.009
28. Milne GL, Yin H, Hardy KD, Davies SS, Roberts LJ. Isoprostane generation and function. Chem Rev. 2011;111(10):5973–5996. doi:10.1021/cr200160h
29. Basu S. F2-isoprostanes in human health and diseases: from molecular mechanisms to clinical implications. Antioxid Redox Signal. 2008;10(8):1405–1434. doi:10.1089/ars.2007.1956
30. Wang Q, Bai M, Yang Y, et al. Application of TCM constitution in lifetime health maintenance. J Tradit Chin Med. 2018;5(1):6–15.
31. Zhu Y, Shi H, Wang Q, et al. Association between Nine Types of TCM constitution and five chronic diseases: a correspondence analysis based on a sample of 2,660 participants. Evid Based Complement Alternat Med. 2017;2017(4):1–7.
32. Wang Q, Yao S. Molecular basis for cold-intolerant yang-deficient constitution of traditional Chinese medicine. Am J Chin Med. 2008;36(5):827–834. doi:10.1142/S0192415X08006272
33. Wu HC, Chen KH, Hwang JS. Association of menopausal symptoms with different constitutions in climacteric women. Complement Med Res. 2018;25(6):398–405. doi:10.1159/000491389
34. Lv HL, Zhu L, Chen ZQ, Jin HJ, Jin L. Physical and mental health conditions of young college students with different Traditional Chinese Medicine constitutions in Zhejiang Province of China. J Tradit Chin Med. 2015;35(6):703–708. doi:10.1016/S0254-6272(15)30163-1
35. Qi W, Minghua B, Yin Y, et al. Application of TCM constitution in lifetime health maintenance. J Tradit Chin Med Sci. 2018;5(1):6–15.
36. Zhu YB, Wang Q, Hideki O. Evaluation on reliability and validity of the constitution in Chinese medicine questionnaire (CCMQ). Chin J Behavioral Med Sci. 2007;16(7):651–654.
37. Luo H, Li T, Liao X, et al. Association between metabolic syndrome and body constitution of traditional Chinese medicine: a systematic review and meta-analysis. J Tradit Chin Med Sci. 2020;7(4):355–365.
38. Ma YL, Yao H, Yang WJ, et al. Correlation between traditional chinese medicine constitution and dyslipidemia: a systematic review and meta-analysis. Evid Based Complement Alternat Med. 2017;2017:1896746. doi:10.1155/2017/1896746
39. Luo CM, Song YL, Huang LH, et al. The correlation of lab data, hormone peptides, quality of life, and different traditional chinese medicine syndrome groups in Type 2 diabetes patients. J Tradit Complement Med. 2013;3(2):126–133. doi:10.4103/2225-4110.110409
40. Fu C, Zhang NL, Chen BX, et al. Identification and classification of traditional Chinese medicine syndrome types among senior patients with vascular mild cognitive impairment using latent tree analysis. J Integr Med. 2017;15(3):186–200. doi:10.1016/S2095-4964(17)60335-2
41. Liu Z, Yang H, Zhang M, et al. The interaction effect between blood stasis constitution and atherosclerotic factors on cognitive impairment in elderly people. Evid Based Complement Alternat Med. 2018;1–8.
42. Petersen RC. Mild cognitive impairment as a diagnostic entity. J Int Med. 2004;256(3):183–189. doi:10.1111/j.1365-2796.2004.01388.x
43. Petersen RC, Morris JC. Mild cognitive impairment as a clinical entity and treatment target. Arch Neurol. 2005;62(7):1160–1163. doi:10.1001/archneur.62.7.1160
44. Wang J, Li Y, Ni C, Zhang H, Li L, Wang Q. Cognition research and constitutional classification in Chinese medicine. Am J Chin Med. 2011;39(4):651–660. doi:10.1142/S0192415X11009093
45. Wang Q, Zhu YB, Xue HS, Li S. Primary compiling of constitution in Chinese medicine questionnaire. Chin J Clin Rehabil. 2006;10(3):12–14.
46. Sun Y, Zhao Y, Xue SA, Chen J. The theory development of traditional Chinese medicine constitution: a review. J Tradit Chin Med Sci. 2018;5(1):16–28.
47. Irizarry MC. Biomarkers of Alzheimer disease in plasma. NeuroRx. 2004;1(2):226–234. doi:10.1602/neurorx.1.2.226
48. Song F, Poljak A, Smythe GA, Sachdeu P. Plasma biomarkers for mild cognitive impairment and Alzheimer’s disease. Brain Res Rev. 2009;61(2):69–80. doi:10.1016/j.brainresrev.2009.05.003
49. Kumar BH, Kumar BD, Diwan PV. Hesperidin, a citrus flavonoid, protects against l-methionine-induced hyperhomocysteinemia by abrogation of oxidative stress, endothelial dysfunction and neurotoxicity in Wistar rats. Pharm Biol. 2017;55(1):146–155. doi:10.1080/13880209.2016.1231695
50. Shankle WR, Hara J, Barrentine LW, et al. CerefolinNAC therapy of hyperhomocysteinemia delays cortical and white matter atrophy in Alzheimer’s disease and cerebrovascular disease. J Alzheimers Dis. 2016;54(3):1073–1084. doi:10.3233/JAD-160241
51. Lu D, Li P, Zhou Y, et al. Association between serum non-high-density lipoprotein cholesterol and cognitive impairment in patients with acute ischemic stroke. BMC Neurol. 2016;16(1):154. doi:10.1186/s12883-016-0668-2
52. Price BR, Wilcock DM, Weekman EM. Hyperhomocysteinemia as a risk factor for vascular contributions to cognitive impairment and dementia. Front Aging Neurosci. 2018;10:350. doi:10.3389/fnagi.2018.00350
53. Beydoun MA, Beydoun HA, Gamaldo AA, et al. Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis. BMC Public Health. 2014;14:643.
54. Seshadri S, Beiser A, Selhub J, et al. Plasma homocysteine as risk factor for dementia and Alzheimer’s disease. N Engl J Med. 2002;346(7):476–483. doi:10.1056/NEJMoa011613
55. Anello G, Guéantrodríguez RM, Bosco P, et al. Homocysteine and methylenetetrahydrofolate reductase polymorphism in Alzheimer’s disease. Neuroreport. 2004;15(5):859–861. doi:10.1097/00001756-200404090-00025
56. Annerbo S, Wahlund LO, Lökk J. The relation between homocysteine levels and devel opment of Alzheimer’s disease in mild cognitive impairment patients. Dement Geriatr Cogn Disord. 2005;20:209–214. doi:10.1159/000087297
57. Galasko G, Montine TJ. Biomarkers of oxidative damage and inflammation in Alzheimer’s disease. Biomark Med. 2010;4(1):27–36. doi:10.2217/bmm.09.89
58. Mecocci P, Polidori MC. Antioxidant clinical trials in mild cognitive impairment and Alzheimer’s disease. Biochim Biophys Acta. 2010;20(18):631–638.
59. López N, Tormo C, Blas ID, Llinares I, Alom J. Oxidative stress in Alzheimer’s disease and mild cognitive impairment with high sensitivity and specificity. J Alzheimers Dis. 2013;33(3):823–829. doi:10.3233/JAD-2012-121528
60. Padurariu M, Ciobica A, Hritcu L, Stoica B. Changes of some oxidative stress markers in the serum of patients with mild cognitive impairment and Alzheimer’s disease. Neurosci Lett. 2010;469(1):6–10. doi:10.1016/j.neulet.2009.11.033
61. Nunomura A, Perry G, Aliev G, et al. Oxidative damage is the earliest event in Alzheimer disease. J Neuropathol Exp Neurol. 2001;60(8):759–767. doi:10.1093/jnen/60.8.759
62. Sirin FB, Kumbul DD, Vural H, et al. Plasma 8-isoPGF2α and serum melatonin levels in patients with minimal cognitive impairment and Alzheimer disease. Turk J Med Sci. 2015;45(5):1073–1077. doi:10.3906/sag-1406-134
63. Pratico D, Clark CM, Liun F, Lee YM, Trojanowski JQ. Increase of brain oxidative stress in mild cognitive impairment: a possible predictor of Alzheimer disease. Arch Neurol. 2002;59(6):972–977. doi:10.1001/archneur.59.6.972
64. Jan VDG, Wietmarschen HV, Schroën J, et al. Systems biology-based diagnostic principles as pillars of the bridge between chinese and western medicine. Planta Med. 2010;76(17):2036–2047. doi:10.1055/s-0030-1250450
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

