Back to Journals » Cancer Management and Research » Volume 10
CD8+ tumor-infiltrating lymphocytes as a novel prognostic biomarker in lung sarcomatoid carcinoma, a rare subtype of lung cancer
Authors Chen J, He Q, Liu J, Xiao Y, Xiao C, Chen K, Xie D, Zhang X
Received 6 April 2018
Accepted for publication 5 June 2018
Published 13 September 2018 Volume 2018:10 Pages 3505—3511
DOI https://doi.org/10.2147/CMAR.S169074
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lu-Zhe Sun
Jiewei Chen,1,2,* Qingmei He,2,* Jun Liu,1,2,* Yongbo Xiao,1 Canhua Xiao,3 Keming Chen,1 Dan Xie,1,2 Xinke Zhang1,2
1Department of Pathology, Sun Yat-sen University Cancer Center, Guangzhou, 510060, People’s Republic of China; 2Sun Yat-sen University Cancer Center, State Key Laboratory of Oncology in South China, Collaborative Innovation Center for Cancer Medicine, Guangzhou, 510060, People’s Republic of China; 3Department of Pathology, Qingyuan People’s Hospital, Qingyuan, 511581, People’s Republic of China
*These authors contributed equally to this work
Purpose: The aim of this study was to investigate the degree of infiltration of CD8+ tumor-infiltrating lymphocytes (TILs) including high and low density in lung sarcomatoid carcinoma (LSC) and their clinicopathological significance.
Patients and methods: The density of CD8+ TILs in paraffin-embedded tissue sections from 100 LSC patients was detected by immunohistochemical staining, and the relationship of CD8+ TILs with clinicopathological features and prognosis was analyzed.
Results: The chi-squared test showed that the degree of infiltration of CD8+ TILs was significantly correlated with the clinicopathological stage and T stage of LSC (P<0.05). The univariate analysis demonstrated that tumor size, clinicopathological stage, T stage, N stage, M stage, and CD8+ TILs are risk factors that affect prognosis of the patients (P<0.05). The mean overall survival (OS) of LSC patients with a high density of CD8+ TILs was 92.3 months, which was significantly higher than 31.2 months in patients with a low density of CD8+ TILs (P<0.05). Cox regression multivariate analysis confirmed that the density of CD8+ TILs was an independent prognostic factor for OS time of LSC patients (hazard ratio=0.455, P<0.05).
Conclusion: CD8+ TILs could be used as an effective prognostic index for LSC patients, and a high density of CD8+ TILs in tumor tissue may predict a better outcome.
Keywords: lung sarcomatoid carcinoma, CD8+ TILs, immunohistochemistry, prognosis
Introduction
Lung cancer is a serious global public health problem. The data of the National Central Cancer Registry showed that the incidence of lung cancer and mortality rate ranks first in the year 2012 in China.1 Lung sarcomatoid carcinoma (LSC) is a group of rare, poorly differentiated, non-small-cell lung cancer (NSCLC) containing sarcoma or sarcomatoid differentiation. Incidence of lung cancer accounted for 0.1%–0.4% of the total number, which is more common in older male smokers.2,3 Compared with other histological subtypes, LSC was more aggressive and had a higher recurrence rate.4 Surgical treatment of LSC is the main therapeutic strategy.5 LSC has a resistance to conventional first-line chemotherapy, and treatment effect is not ideal and has a poor prognosis.6 To date, the study of LSC prognostic indicators is very rare; prior literatures revealed that PD-L1 expression was significantly correlated with a worse overall survival (OS) in LSC using immunohistochemistry (IHC).7,8 However, the cutoff value of PD-L1 expression remained to be unidentified in different studies due to the different clone numbers of this antibody. In addition, the molecular biomarker of LSC has been performed, including EGFR and Kras mutation, although the study showed that the associated targeted inhibitors alone may not be an effective therapeutic approach.9 However, the molecular detection is too expensive to limiting its application. Therefore, in the light of a certain degree of limitation for the clinical application of these prior biomarkers, exploring the other prognostic indicators is very urgent for LSC, and today, we focus on the study of relationship between tumor-infiltrating lymphocytes (TILs) and tumor stromal and clinical outcomes.
To the best of our knowledge, tumor local immune response is mainly involved by tumor-infiltrating immune cells, and TILs are the main types of tumor-infiltrating immune cells and play a key role in immune surveillance of abnormal malignant cells.10 Among the different TILs subgroups, the most valuable is the CD8+ cytotoxic T-cell (CTL), and the cytotoxicity of CD8+ T cells is one of the mechanisms of antitumor immunity.11 It mediates the type 1 immune response and plays an important role in destroying the tumor.12 TILs kill tumor cells and mainly rely on the effective activation of CD8+ T cells. T cells are transformed into effector cells and then play an antitumor effect.13 CD8+ TILs can identify endogenous antigenic peptides presented by major histocompatibility complex (MHC) I molecules, which mediate the organism to kill the tumor cells through the immune response.14 High-density infiltration of CD8+ TILs in tumor tissue is closely related to better prognosis in breast cancer, NSCLC, colorectal cancer, oral squamous cell carcinoma and hepatocellular carcinoma.15–19 However, the clinical significance and mechanism of CD8+ TILs in LSC have not been reported. This study was used to detect the level of CD8+ TILs in patients with LSC by IHC. The correlation between CD8+ TILs and the prognosis of LSC was evaluated by statistical analysis.
Patients and methods
Patients and tissue specimens
A total of 100 cases of paraffin-embedded LSC tissue samples were collected from the Department of Pathology, Sun Yat-sen University Cancer Center. All samples of paraffin were pretreated by standard. The retrospective analysis data of 100 patients with LSC who had pneumonectomy and/or lymphadenectomy came from Sun Yat-sen University Cancer Center, and the specimens were collected between December 2000 and June 2016 by removing the patients with neoadjuvant therapy. In all, eight of 100 cases had the synchronous metastases. All cases were diagnosed according to the WHO classification criteria in 2000, 2002 US Joint Commission and the International Joint Cancer TNM Classification System. The study was approved by the Sun Yat-sen University Cancer Center Medical Ethics Committee, and the written informed consent was obtained from all the patients who donated their specimens.
IHC
The immunohistochemical staining of CD8 protein was detected according to standard EnVision™ procedure. The paraffin-embedded tissue blocks were cut into 3 µm thick sequential sections. The slides were dried and deparaffinized in xylene, rehydrated through graded alcohol, immersed in 3% hydrogen peroxide for 10 minutes to block endogenous peroxidase activity and antigen retrieved by pressure cooking for 3 minutes in citrate buffer (pH=6). Then, the slides were incubated with 5% BSA for 15 minutes to reduce nonspecific reaction. Subsequently, the slides were incubated with the mouse monoclonal antibody anti-CD8 (ab8105, dilution 1:100; Abcam, Beijing, People’s Republic of China) for 50 minutes at 37°C. The slides were sequentially incubated with a secondary antibody (EnVision, Dako, Denmark) for 30 minutes in the incubator at 37°C and stained with 3,3-diaminobenzidine. Finally, the sections were counterstained with Mayer’s hematoxylin, dehydrated and mounted. A negative control was obtained by replacing the primary antibody with a normal rabbit IgG.
IHC evaluation
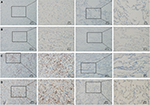
CD8 is mainly located in the cell membrane and cytoplasm. Using the high-power microscope, five visual fields with the most abundant infiltration of LSC were selected from each slice to calculate the percentage of CD8+ T lymphocytes in the total lymphocytes and take the mean value of five fields as the density of CD8+ TILs (%).20 The expression of CD8 was double-blindly evaluated by two experienced pathologists. The final score was the average value of percentages that the two pathologists assessed.
Statistical analyses
Statistical analysis was performed using SPSS 20.0. The cutoff values were defined by the median.21,22 Correlation between density of CD8+ TILs and clinicopathological parameters in patients with LSC was analyzed using the chi-squared test. The survival analysis of LSC patients was evaluated by the Kaplan–Meier method with log-rank test. Multivariate analyses were performed using Cox proportional hazard model. All P-values were reported by two-sided analyses, and P<0.05 represented the statistical significance level. All data in our study have been recorded at the Sun Yat-sen University Cancer Center for future reference.
Results
Patients’ characteristics
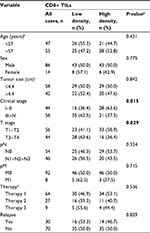
The clinicopathological characteristics of LSC patients are detailed in Table 1. This LSC cohort consisted of 86 (86.0%) men and 14 (14.0%) women with mean age of 57 years. The average follow-up period was 26.2 months (median, 27.4 months; range, 1.0–129.0 months). In all, 56 (56.0%) patients were diagnosed at late stages (III and IV) and the other 44 (44.0%) patients at early stages (I and II).
Expression of the level of CD8+TILs in LSC tissues by IHC
The results of IHC showed that CD8+ TILs were differentially expressed in LSC. In all, 49% (49/100) of LSC had high-density infiltration of CD8+ TILs. The variable density of CD8+ TIL infiltration of different LSC tissues and adjacent tissues was randomly selected as shown in Figure 1.
Selection of cutoff value for different degrees of infiltration of CD8+ TILs
Statistical analysis showed that the median score of CD8+ TILs was 7.60%. Therefore, the median count (7.60%) was considered as the cutoff value for different degrees of infiltration of CD8+ TILs. In all, ≤7.60% was defined as a low-density infiltration and >7.60% as a high-density infiltration.
Association of different degrees of infiltration of CD8+TILs with LSC patients’ clinicopathological features
The chi-squared test showed that the degree of infiltration of CD8+ TILs was significantly correlated with clinical stage (P=0.015) and T stage (P=0.029), but there was no significant correlation with other clinical pathological parameters such as age, sex, lymph node metastasis, treatment and recurrence (P>0.05). All the details are given in Table 1.
Relationship between clinicopathological features, expression of CD8+ TILs and LSC patients’ survival: univariate survival analysis
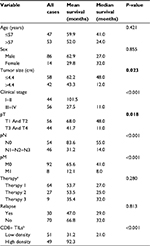
The 10 clinical pathologic parameters of patients with LSC, including CD8+ TILs, were analyzed by univariate analysis. The univariate analysis demonstrated that tumor size (P=0.023), clinicopathological stage (P<0.001), T stage (P=0.018), N stage (P<0.001), M stage (P<0.001) and CD8+ TILs (P<0.001) were the risk factors of survival time. The detailed data are shown in Table 2.
Survival analysis showed that the OS time and progression-free survival (PFS) time of CD8+ TILs high-density infiltration group were higher than those of low-density infiltration group. The mean survival time of patients with LSC in the high-density infiltration group was 92.3 months, which was higher than that in the low-density infiltration group (31.2 months, P<0.05; Figure 2A); The mean PFS time of CD8+ TILs high-density infiltration group was 71.6 months in patients with LSC, and the mean PFS of the low-density infiltration group was 20.2 months (P<0.05; Figure 2B).
Independent prognostic factors of LSC: multivariate Cox regression analysis
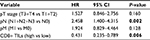
The significant risk factors (pT, pN, pM, CD8+ TILs) were enrolled into Cox regression model for multivariate analysis. The results showed that T stage and M stage were not independent prognostic risk factors (P>0.05), while N stage (P=0.002) and infiltration of CD8+ TILs (P=0.006) could be used as an independent prognosis factor for patients with LSC. The detailed data are given in Table 3.
Relationship between the expression level of CD8+TILs in LSC and the 3-year and 5-year OS rate
Chi-squared test analysis showed that the 3-year and 5-year OS rates of CD8+ TILs low-density infiltration group was 15.7% (8/51) and 3.9% (2/51), respectively, compared with 36.7% (18/49) and 22.4% (11/49), respectively, in CD8+ TILs high-density infiltration group. The 3-year and 5-year OS rates were statistically significant between two groups (P<0.05). All the details are given in Tables 4 and 5.
Discussion
The study suggests that the patient’s immune status is an important factor for influencing the development and outcome of the tumor.23 TILs are heterogeneous lymphocyte population predominantly found in tumor microenvironment with CD4+ and CD8+ T cells. In recent years, two studies reported that high- and low-density infiltration of CD8+ TILs could be used as an important indicator of prognosis of patients with NSCLC.24,25
In our study, we showed that the degree of infiltration of tumor CD8+ TILs was significantly correlated with the clinicopathological stage and T stage of LSC, suggesting that a high density of CD8+ TILs is significantly associated with the early stage of LSC. However, the partial or complete regression of LSC is extremely rare despite of infiltration of TILs, and the tumor remained to continue development and progression under the circumstance of presence of CD8+ TILs, which indicated that host immune response had a certain degree of role during killing tumors. Infiltration of CD8+ TILs could be inversely associated with better clinical outcomes in patients with LSC, independent of conventional predictive and prognostic factors, such as T stage and tumor metastasis status. So far, no studies have explored the prognostic value of infiltration of tumor CD8+ TILs in LSC. Our results indicated that the local immune response cells had a critical impact on development of the LSC, which is consistent with the studies of Teng et al21 and Al-Shibli26 who showed that CD8+ TILs could be used as independent prognostic factors in NSCLC.,
In our study, Kaplan–Meier survival analysis showed that the mean OS of LSC patients with a high density of CD8+ TILs was 92.3 months, which was significantly higher than 31.2 months in patients with a low density of CD8+ TILs. The mean PFS time was 71.6 months and 20.2 months, respectively. The 3-year and 5-year survival rates of infiltration of CD8+ TILs with a high density were significantly higher than those of infiltration of CD8+ TILs with a low density. These results suggested that high-density CD8+ T lymphocytes have antitumor benefits and play an important role in improving the survival of patients with LSC. The high-density infiltration of CD8+ TILs is associated with better prognosis, which is in agreement with the findings of Al-Shibli et al26 in NSCLC and Miyashita et al27 in breast cancer. We found that 51% (51/100) of LSC patients harboring low-density infiltration of CD8+ TILs had the worse prognosis, and these partial patients also had not ideal response to traditional chemotherapy, thus, whether we could transfer the designed specific T cell (tumor-reactive T cells) to patients with LSC that harbor low-density infiltration for better therapeutic effect due to tumor-reactive T cells that could be increased in vitro by flow cytometry cell sorting technology.28
To the best of our knowledge, CD8 is an important surface marker of cytotoxic T cells (CTL) and effective functioning of CD8+ CTL is the main antitumor effector. T cells transformed into effector cells require two stimulatory signals. T cell receptors specifically recognize and bind to pMHC (antigen peptide–MHC complex) of antigen-presenting cells (APCs), and this process provides the first signal of T-cell activation. Combining with APC surface, costimulatory molecules B7 and CD28 of T-cell surface receptor provide a second signal.29 CD8+ TILs are activated to play the antitumor effect, mainly through three ways to kill tumor cells, including death receptor pathway,30 degranulation pathway24 and secretion of TNF-α, IFN-γ and other cytokines.31,32 Therefore, we speculated that CD8+ TILs in LSC may play a role in inhibiting tumor growth through the abovementioned molecular mechanisms, furthermore increasing the survival time of patients.
Our study demonstrated that prognostic significance of the cytotoxic T-cell in LSC, which also provide evidence that CD8+ TILs have antitumor activity for LSC, and potential immunotherapeutic strategy could be applied in the clinical practice in future. In view of the complex effects of tumor microenvironment and the diversity of immune cell connections, the mechanism of CD8+ TILs in antitumor immune effects in LSC will be a meaningful attempt.
Since this was a single-center retrospective study, there were several limitations: first, the number of patients with LSC was small due to these extremely rare cases. Second, in our study, we detected the expression of CD8 IHC for infiltration of TILs by conventional tissue samples instead of tissue microarray. However, these samples are still not completely reflecting the tumor immunomicroenvironmental status. Therefore, the question of the heterogeneity of infiltration of TILs required us to determine the minimal tissue sample size for evaluation of TILs. However, to date, this question remained to be debated.33 Thus, we only preliminarily investigated the correlation of different degrees of infiltration of CD8+ TILs and clinical prognosis, and we have first reported the prognostic significance of the cytotoxic T-cell in LSC. Further multi-institution investigation would be warranted to define the prognostic implication of the cytotoxic T-cell in LSC.
Conclusion
Infiltration of CD8+ TILs was closely related to the occurrence and development of LSC. CD8+ TILs could be used as an effective prognostic index for LSC patients, and high infiltration of CD8+ TILs in tumor tissue may predict a better outcome.
Acknowledgment
This work was supported by the National Key R&D Program of China (2017YFC1309000).
Disclosure
The authors report no conflicts of interest in this work.
References
Chen W, Zheng R, Zeng H, Zhang S. The incidence and mortality of major cancers in China, 2012. Chin J Cancer. 2016;35(1):73. | ||
Ung M, Rouquette I, Filleron T, et al. Characteristics and Clinical Outcomes of Sarcomatoid Carcinoma of the Lung. Clin Lung Cancer. 2016;17(5):391–397. | ||
Pelosi G, Sonzogni A, de Pas T, et al. Review article: pulmonary sarcomatoid carcinomas: a practical overview. Int J Surg Pathol. 2010;18(2):103–120. | ||
Martin LW, Correa AM, Ordonez NG, et al. Sarcomatoid carcinoma of the lung: a predictor of poor prognosis. Ann Thorac Surg. 2007;84(3):973–980. | ||
Steuer CE, Behera M, Liu Y, et al. Pulmonary Sarcomatoid Carcinoma: An Analysis of the National Cancer Data Base. Clin Lung Cancer. 2017;18(3):286–292. | ||
Vieira T, Girard N, Ung M, et al. Efficacy of first-line chemotherapy in patients with advanced lung sarcomatoid carcinoma. J Thorac Oncol. 2013;8(12):1574–1577. | ||
Yvorel V, Patoir A, Casteillo F, et al. PD-L1 expression in pleomorphic, spindle cell and giant cell carcinoma of the lung is related to TTF-1, p40 expression and might indicate a worse prognosis. PLoS One. 2017;12(7):e0180346. | ||
Chang YL, Yang CY, Lin MW, Wu CT, Yang PC. High co-expression of PD-L1 and HIF-1α correlates with tumour necrosis in pulmonary pleomorphic carcinoma. Eur J Cancer. 2016;60:125–135. | ||
Jiang X, Liu Y, Chen C, et al. The value of biomarkers in patients with sarcomatoid carcinoma of the lung: molecular analysis of 33 cases. Clin Lung Cancer. 2012;13(4):288–296. | ||
Verdegaal EM, Hoogstraten C, Sandel MH, et al. Functional CD8+ T cells infiltrate into nonsmall cell lung carcinoma. Cancer Immunol Immunother. 2007;56(5):587–600. | ||
Usó M, Jantus-Lewintre E, Bremnes RM, et al. Analysis of the immune microenvironment in resected non-small cell lung cancer: the prognostic value of different T lymphocyte markers. Oncotarget. 2016;7(33):52849–52861. | ||
Stanton SE, Disis ML. Clinical significance of tumor-infiltrating lymphocytes in breast cancer. J Immunother Cancer. 2016;4:59. | ||
Tamada K, Shimozaki K, Chapoval AI, et al. Modulation of T-cell-mediated immunity in tumor and graft-versus-host disease models through the LIGHT co-stimulatory pathway. Nat Med. 2000;6(3):283–289. | ||
Cao X. Regulatory T cells and immune tolerance to tumors. Immunol Res. 2010;46(1-3):79–93. | ||
Mahmoud SM, Paish EC, Powe DG, et al. Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer. J Clin Oncol. 2011;29(15):1949–1955. | ||
Kawai O, Ishii G, Kubota K, et al. Predominant infiltration of macrophages and CD8(+) T Cells in cancer nests is a significant predictor of survival in stage IV nonsmall cell lung cancer. Cancer. 2008;113(6):1387–1395. | ||
Galon J, Costes A, Sanchez-Cabo F, et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science. 2006;313(5795):1960–1964. | ||
Fang J, Li X, Ma D, et al. Prognostic significance of tumor infiltrating immune cells in oral squamous cell carcinoma. BMC Cancer. 2017;17(1):375. | ||
Giuşcă SE, Wierzbicki PM, Amălinei C, Căruntu ID, Avădănei ER. Comparative analysis of CD4 and CD8 lymphocytes - evidences for different distribution in primary and secondary liver tumors. Folia Histochem Cytobiol. 2015;53(3):272–281. | ||
Wang Q, Lou W, di W, Wu X. Prognostic value of tumor PD-L1 expression combined with CD8+ tumor infiltrating lymphocytes in high grade serous ovarian cancer. Int Immunopharmacol. 2017;52:7–14. | ||
Teng F, Meng X, Wang X, et al. Expressions of CD8+TILs, PD-L1 and Foxp3+TILs in stage I NSCLC guiding adjuvant chemotherapy decisions. Oncotarget. 2016;7(39):64318–64329. | ||
Tokito T, Azuma K, Kawahara A, et al. Predictive relevance of PD-L1 expression combined with CD8+ TIL density in stage III non-small cell lung cancer patients receiving concurrent chemoradiotherapy. Eur J Cancer. 2016;55:7–14. | ||
Prendergast GC, Jaffee EM. Cancer immunologists and cancer biologists: why we didn’t talk then but need to now. Cancer Res. 2007;67(8):3500–3504. | ||
Jackute J, Zemaitis M, Pranys D, et al. The prognostic influence of tumor infiltrating Foxp3(+)CD4(+), CD4(+) and CD8(+) T cells in resected non-small cell lung cancer. J Inflamm. 2015;12:63. | ||
Bremnes RM, Donnem T, Busund LT. Importance of tumor infiltrating lymphocytes in non-small cell lung cancer? Ann Transl Med. 2016;4(7):142. | ||
Al-Shibli KI, Donnem T, Al-Saad S, Persson M, Bremnes RM, Busund LT. Prognostic effect of epithelial and stromal lymphocyte infiltration in non-small cell lung cancer. Clin Cancer Res. 2008;14(16):5220–5227. | ||
Miyashita M, Sasano H, Tamaki K, et al. Prognostic significance of tumor-infiltrating CD8+ and FOXP3+ lymphocytes in residual tumors and alterations in these parameters after neoadjuvant chemotherapy in triple-negative breast cancer: a retrospective multicenter study. Breast Cancer Res. 2015;17:124. | ||
Turcotte S, Gros A, Tran E, et al. Tumor-reactive CD8+ T cells in metastatic gastrointestinal cancer refractory to chemotherapy. Clin Cancer Res. 2014;20(2):331–343. | ||
Michel F, Attal-Bonnefoy G, Mangino G, Mise-Omata S, Acuto O. CD28 as a molecular amplifier extending TCR ligation and signaling capabilities. Immunity. 2001;15(6):935–945. | ||
Blok EJ, van den Bulk J, Dekker-Ensink NG, et al. Combined evaluation of the FAS cell surface death receptor and CD8+ tumor infiltrating lymphocytes as a prognostic biomarker in breast cancer. Oncotarget. 2017;8(9):15610–15620. | ||
Bertrand F, Rochotte J, Colacios C, et al. Targeting TNF alpha as a novel strategy to enhance CD8+ T cell-dependent immune response in melanoma? Oncoimmunology. 2016;5(1):e1068495. | ||
Shao S, Risch E, Burner D, Lu L, Minev B, Ma W. IFNγ enhances cytotoxic efficiency of the cytotoxic T lymphocytes against human glioma cells. Int Immunopharmacol. 2017;47:159–165. | ||
Schalper KA, Brown J, Carvajal-Hausdorf D, et al. Objective measurement and clinical significance of TILs in non-small cell lung cancer. J Natl Cancer Inst. 2015;107(3). |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.