Back to Journals » International Journal of General Medicine » Volume 15
Causes and Risk Factors for Male Infertility: A Scoping Review of Published Studies
Authors Okonofua FE , Ntoimo LFC, Omonkhua A , Ayodeji O , Olafusi C, Unuabonah E, Ohenhen V
Received 25 February 2022
Accepted for publication 16 May 2022
Published 4 July 2022 Volume 2022:15 Pages 5985—5997
DOI https://doi.org/10.2147/IJGM.S363959
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Friday Ebhodaghe Okonofua,1– 3 Lorretta Favour Chizomam Ntoimo,3,4 Akhere Omonkhua,2,5 Oladiran Ayodeji,6 Celestina Olafusi,6 Emmanuel Unuabonah,7 Victor Ohenhen8
1Department of Obstetrics and Gynecology, University of Benin, Benin, Edo State, Nigeria; 2Center of Excellence in Reproductive Health Innovation, University of Benin, Benin, Edo State, Nigeria; 3Women’s Health and Action Research Centre, Benin City, Edo State, Nigeria; 4Department of Demography and Social Statistics, Federal University, Oye-Ekiti, Ekiti State, Nigeria; 5Department of Medical Biochemistry, School of Basic Medical Sciences, University of Benin, Benin, Edo State, Nigeria; 6Department of Obstetrics and Gynaecology, University of Medical Sciences, Ondo, Ondo State, Nigeria; 7Department of Chemistry, Redeemer University, Ede, Osun State, Nigeria; 8Department of Obstetrics and Gynaecology, Central Hospital, Benin, Edo State, Nigeria
Correspondence: Friday Ebhodaghe Okonofua, Department of Obstetrics and Gynecology, University of Benin, Benin, Edo State, Nigeria, Tel +234 8023347828, Email [email protected]
Background: Despite the evidence of declining sperm counts worldwide and the increasing prevalence of male infertility, there has been limited documentation of the potential causes and risk factors for male infertility. With limited methods of primary treatment of male infertility, understanding the causes and risk factors will pinpoint specific lines of prevention.
Objective: We reviewed published studies on causes and risk factors for male infertility and identify gaps in the literature enabling more focused research and innovations.
Design/Setting: The study was a scoping review. The review included studies on the causes and risk factors of male infertility published in scientific/academic publications. It was not limited to any region or date. The following databases were searched: Cumulative Index to Nursing and Allied Health Literature, Wiley online, HINARI, Science Direct, PubMed/Medline, Google Scholar, African Journals Online, and the WHO Repository. In total, 1295 articles were identified. After removing 357 duplicates, 938 articles were screened, and 71 articles were identified for inclusion, while 60 articles met the inclusion criteria.
Results: Four broad causes and risk factors of male infertility identified were 1) biological/physiological/genetic causes, 2) behavioral/lifestyle risk factors, 3) environmental factors, and 4) socio-demographic risk factors. However, no substantive results on actual causes under each category were identified, leaving gaps for further research and experimentation.
Conclusion: The results indicate limited knowledge of the actual causes of male infertility in published studies. The gaps in knowledge that need to be bridged to enable a fuller understanding of the actual causes of male infertility were highlighted.
Keywords: male infertility, causes, risk factors, scoping review, spermatozoa defects
Plain Language Summary
This was a scoping review designed to identify the plausible causes of male infertility based on published evidence and to suggest lines of new research or interventions to better inform the prevention and primary treatment of male infertility. We identified 60 published studies from the literature that address the objective of the study. The results of this review permit us to categorize the reported causes and risk factors of male infertility into four broad causes. These include 1) biological/physiological/genetic causes, 2) behavioral/lifestyle risk factors, 3) environmental factors, and 4) socio-demographic risk factors. The major biological factors associated with male infertility identified in this review were genetic factors, uro-genital infections, and varicoceles. The behavioral risk factors associated with male infertility in this review included smoking, alcohol intake, inappropriate body mass index, sexual behavior, and exposure to drugs. The major environmental risk factors for male infertility identified in this review were exposures to chemicals, various pesticides, and mycotoxins. As for socio-demographic factors, increasing age featured as the most common among those reported in the papers under review. The results of this review indicate that more information is required to confirm specific causes for male infertility. While some associations have been identified, there is a need to establish causation with purposefully designed studies such as longitudinal cohort studies or large randomized control trials.
Introduction
In several publications, defects in spermatozoa due to inadequate numbers (azoospermia/oligospermia), poor motility, and abnormal structure/morphology have been reported as leading causes of male infertility worldwide.1–4 Indeed, numerous publications have reported systematic reductions in sperm counts over the past decades,3,5 a situation that accounts for the increasing contribution of the male factor to the increasing prevalence of infertility worldwide.6 The decline in counts and quality of spermatozoa has been recognized by the World Health Organization, which gave rise to its revision of the criteria for normal versus abnormal sperm counts to be used by andrology laboratories worldwide.7
Furthermore, male infertility has been described as a form of infertility that is poorly responsive to primary treatment. Reports indicate that only a small proportion of male factor infertility is resolvable with primary methods of treatment.8 By contrast, male factor infertility is mostly resolved with secondary measures, such as artificial insemination,9 intra-uterine insemination,10 in vitro fertilization and embryo transfer,11 intra-cytoplasmic sperm transfer,12 and child fostering/adoption.13,14
Despite the increasing incidence and the difficulty in treating male infertility, there has been limited documentation of the leading causes and risk factors for male infertility. Understanding the causes and risk factors will enable the identification of primary prevention methods, as well as effective methods for the primary treatment of male infertility. Such understanding may also foster the process of determining innovative secondary and tertiary approaches for dealing with the problem.
Several causes and risk factors have been speculated and/or proven for male infertility and published in the literature. Some of these include smoking, alcohol intake, drugs, obesity, past or present testicular infections, exposure to environmental toxins, exposure of the testicles to excessive heat, hormonal disorders, testicular trauma and ejaculatory/erectile disorders among several others.15–17 However, to date, there has been limited documentation of studies that aggregate and rank these potential causes and risk factors in ways to enable the development of a consensus of the causal pathway for male infertility, and the identification of simple preventive and treatment strategies. While the identified risk factors and causes have been more broadly defined, the segregation of patterns of causes and risk factors will enable the specification of more lines of research and innovations that will pinpoint the actual mechanisms and processes leading to the current decline being experienced in male fertility.
It is within this context that this scoping review of published studies on the causes and risk factors for male infertility in the international literature has been conducted. The objectives of the study are twofold: 1) to identify published studies on causes and risk factors for male infertility and pinpoint the leading causes reported worldwide; and 2) to identify gaps in the literature relating to the causes and risk factors for male infertility enabling more focused research and innovations. We hypothesize that climatic and environmental factors would feature as leading causes and risk factors for male infertility, due to the enormous changes that have taken place in these domains in recent years, with the resulting impact on sperm production and viability. We believe that this exploratory research will enable the identification of novel approaches for prevention and the primary treatment of male infertility.
Methods
Study Design
This study is a scoping review of published articles. This design was chosen instead of a systematic review because of the wide range of studies that have been conducted on the subject.18 The methodological approach developed by Arksey & O’Malley (2005)19 was followed. It involves five steps: 1) identifying the question, 2) identifying relevant studies, 3) study selection, 4) charting the data – recording key themes emerging from the literature, and 5) collating, summarising, and reporting the results. A protocol was developed for the review following the principles of PRISMA Extension for Scoping Reviews.20
Eligibility Criteria
The review included studies on the causes and risk factors of male primary and secondary infertility published in scientific/academic publications. Grey literature was excluded. Quantitative studies of any design, systematic reviews and meta-analysis, published in English were eligible for inclusion in the review. There was no limitation on the setting and date of publication as we intended to review a wide range of possible causes and risk factors for male infertility.
Search Strategy
Following the protocol designed for the review, relevant studies were identified through a database search. The following databases were searched: Cumulative Index to Nursing and Allied Health Literature (CINAHL), Wiley online, HINARI, Science Direct, PubMed/Medline, Google Scholar, African Journals Online (AJOL), and the WHO Repository. The reference lists of the included articles were reviewed to identify additional eligible articles. The search terms were developed from the existing literature and recommendations from health librarians, and included controlled vocabulary and keyword terms related to causes, risk factors, and male infertility. The search terms were combined in diverse ways and used following the peculiarities of each database. Causes and Risks factors: Cause OR causes Predictor OR predictors OR Risk factors OR risk factor OR determinants OR determinant OR associated factors. Male: Male partners OR men OR male OR males. Infertility: Infertile OR sterile OR infertility OR sterility OR impotent OR barren. Other male infertility-related words: Oligospermia OR oligozoospermia OR Aspermia OR Azoospermia; Low sperm count AND no sperm count OR abnormal sperm OR abnormal semen OR seminal fluid OR semen profile OR impaired semen OR seminal fluid indices OR semen; Testicular function OR testicular malfunction OR erectile dysfunction OR male reproductive function OR dysfunction OR malfunction.
Study Selection
A total of 1295 articles were identified from the various databases and reference lists (Figure 1). After removing 357 duplicates, 938 articles were screened. We retrieved and reviewed the abstracts of 550 potentially relevant articles. Two reviewers independently screened the titles and abstracts of the retrieved publications. After this initial screening, 71 articles were identified for inclusion and their full texts were retrieved. The full text of the 71 articles was screened, and 60 met the inclusion criteria. Disagreements about inclusion and exclusion were resolved through consensus.
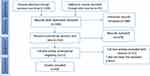 |
Figure 1 Framework for the process of inclusion and exclusion of articles. |
Data Extraction and Result Presentation
A Microsoft Excel spreadsheet was used for data extraction. Two reviewers independently extracted data from the included articles. Extracted data included year of publication, authors, publication title, research objectives, study design, and findings. A third reviewer checked the extracted data for accuracy and completeness and discrepancies were resolved by consensus. The results of the review are presented narratively using four broad themes following the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist (see Supplementary Material 2).
Results
Sixty (60) articles were included in the review. The methodological design varied (see Supplementary Material 1). The majority of the articles were case–control studies (n = 18), review articles (n = 18; 4 were systematic reviews and meta-analysis), and cross-sectional studies (n = 11). Others were cohort studies (n = 2), prospective studies (n = 3), study/review of case notes (n = 4), and 4 observational studies. The majority of the articles were from the developing countries (Supplementary Material 1). The subjects of the articles were categorized into five broad themes as shown in Figure 2. Most of the studies focused on more than one subject area; thus, the total number of articles per theme is not equal to the 60 articles. More than half of the articles presented findings on behavioral/lifestyle risk factors. See Supplementary Material 1 for a detailed description of the articles.
 |
Figure 2 Themes of the articles by the number of articles. |
Causes and Risk Factors of Male Infertility
The causes and risk factors of male infertility as identified in the reviewed studies are presented using four broad themes: biological/physiological causes, behavioral/lifestyle risk factors, environmental risk factors, and socio-demographic risk factors.
Biological/ Physiological Causes
The major causes of male infertility – oligozoospermia, asthenozoospermia, azoospermia, or a combination as identified in the study – were specific biological or physiological conditions. A 9-year prospective study of 1737 patients in Estonia categorized the observed primary causal factors into absolute (secondary hypogonadism, genetic causes, seminal tract obstruction), severe (oncological diseases, severe sexual dysfunction), and plausible causal factors (congenital anomalies in the urogenital tract, acquired or secondary testicular damage).3
In a retrospective review of case notes of all patients who were treated at the Urology Clinic of the Ahmadu Bello University Teaching Hospital, Zaria, Nigeria, between January 1991 and December 2005 all the patients (537) with histories of infertility between 2 and 13 years had normal female partners. The major cause of male infertility in this study was primary testicular insufficiency, which included congenital hypoplastic testes and testicular atrophy resulting from torsion, cryptorchidism, and post-mumps orchitis.21
In a case–control study in Southern Nigeria, male infertility was associated with biologically and physiologically lower sperm concentration (<5 million), poorer sperm motility, and a lower percentage of viable forms of spermatozoa as compared to fertile men. Infertile men were significantly more likely to have bacterial organisms, such as Staphylococcus aureus, Streptococcus faecalis, Trichomonas vaginalis, and Candida albicans in semen culture than fertile men were. The study also showed that previous penile discharge, painful micturition, and previous genital ulcer were associated with infertility in men.22
Similarly, Osazuwa et al 23 reported a higher prevalence of Chlamydia trachomatis antibodies among infertile men and its relationship with abnormal semen features. C. trachomatis antibodies were identified in 19.5% of the 215 infertile males in the study. A significant association was observed between C. trachomatis infection and male infertility, and poor semen characteristics. This study showed that C. trachomatis antibodies were found in all types of seminal infertility, particularly in oligozoospermia and azoospermia where significant association was observed. Also, a systematic review and meta-analysis in the Middle East and North Africa reported a high current C trachomatis infection prevalence in infertile patients.24
Other review articles on the physiological causes of male infertility identified genito-urinary tract infections, inflammatory testicular or prostate conditions, hormonal imbalance, concurrent medical illnesses, and surgical procedures, such as hernia repairs.1,5,25–27
In 2016, Jeje et al28 conducted an audit of 70 cases of male infertility in Lagos, Nigeria. The most common pathology of male infertility identified in the study was varicocele in 53 out of the 70 cases. Of these, 51.4% had bilateral varicocele, and 24.3% had unilateral varicocele. In 30 patients (42.9%), unilateral varicocele occurred alone, in 22 patients, it occurred in combination with hormonal imbalance, and vas occlusion in 1.4%. Other aetiological factors were hormonal imbalance, vassal obstruction, chromosomal abnormality (XXY), dysfunction (retrograde ejaculation), and idiopathic.
Similarly, Garba-Alkali et al29 evaluated the semen profiles of male partners of women attending infertility clinics; 154 semen samples were analyzed. Less than half of the clients (46.8%) had normal semen (normozoospermia), whereas 53.2% had abnormal semen profiles. Of the 82 abnormal cases, asthenozoospermia was 41.5%, oligoasthenoteratozoospermia 29.3%, azoospermia 19.5%, and oligozoospermia 9.7%, while aspermia was not seen. When the semen samples were cultured, the majority (85.7%) did not grow any organism. The most common organism in the cultured semen was Staphylococcus aureus. The semen analysis (normal and abnormal) was statistically related to risk factors of infertility with the following categories: Inguinoscrotal problems, past penile discharge, past STD treatment, and erectile dysfunction. Except for alcohol intake, the proportion of abnormal semen was higher for all the categories than normal semen and the association between risk factors and semen analysis was significant.
In a study of 704 male patients with identifiable male factors contributing to infertility conducted by Idrisa et al30 in Maiduguri, Nigeria, to determine the extent of the male contribution to infertility, azoospermia, oligozoospermia, and a combination of oligo/asthenozoospermia or oligo/terato/asthenozoospermia were the most common abnormalities identified. The most cultured pathogenic organisms recorded in this study were Staphylococcus aureus, Escherichia coli, and Klebsiella. In the clinical findings, there was no significant finding in about 58% of the patients’ semen, but the major findings in the seminal fluid of the remaining 42% were varicocoele, herniorrhaphy, hydrocoelectomy, accessory gland infection, chronic epididymo-orchitis, trauma to the groin, and orchidopexy. Testicular biopsy was done on some patients because of azoospermia and severe oligozoospermia; the histology results showed obstructive azoospermia, tubular sclerosis, and seminiferous tubular hyalinization as common. The limitation of this study was that it was only descriptive and no major hypothesis was tested. These findings were similar to the results of a case–control study in Lagos by Enwurua et al.31
In Emokpae et al32, an inverse relationship was observed between seminal fluid infection and sperm density, motility, and morphology. A decrease in sperm density, motility, and morphology was associated with an increase in seminal fluid infection. The pathogenic organisms detected in the semen fluid were S. aureus, E. coli, candida species, streptococcus, and a mixed growth of S. aureus and E. coli. Similar bacterial growth was identified in Ugwuja et al33, and a study of 311 men with infertility problems in Zimbabwe by Folkvord et al34.
In a study to examine the seroprevalence of Chlamydia trachomatis infection and its relationship with semen quality of men undergoing assisted reproduction therapy in Benin, Nigeria, 24% of the 156 patients were seropositive for C. trachomatis antibody. The authors, however, concluded that C. trachomatis may be an independent risk factor affecting the quality of semen and male infertility.35
Osegbe & Amaku36 conducted a prospective study in Nigeria on the causes of male infertility using 504 patients. They concluded that the major causes were varicocele, infection, bilateral testicular failure, idiopathic oligospermia, multifactorial, cryptorchidism, sexual problems, and surgical injury. The organisms isolated in this study were S. aureus, Trichomonas vaginalis, Proteus mirabilis, Streptococcus faecalis, and enterococcus.
After controlling for multiple risk factors such as testicular volume and a history of STIs, unilateral and bilateral epididymitis, and history of pathology causing testicular damage, Bayasgalan et al37 reported that Grade 2 varicocele were significantly associated with sperm abnormality, and azoospermia. High DNA fragmentation in spermatozoa, caused majorly by oxidative stress, is a major risk factor in male infertility.38 According to Mahboubi et al39, varicocele, a history of hernia surgery, and consumption of ranitidine were significantly associated with male infertility.
In an experiment that investigated the correlation between oxidative stress and incidence of idiopathic infertility and subfertility in Egyptian men,40 there were no significant differences in glutathione-S-transferase (GST) activities in azoospermic and normospermic patients relative to fertile men, but GST activity was significantly lower in oligospermic men. Compared to fertile men, the glutathione peroxidase (GPx) activity increased significantly by up to two times in oligospermic and azoospermic patients, whereas there was a sharp decrease (about three times) for normospermic patients. There was a significant decrease in superoxide dismutase (SOD) activities in semen samples of oligospermic and azoospermic men, and an increase in normospermic males compared to fertile patients. The seminal protein contents significantly increased in oligospermic men but decreased in normospermic men relative to the fertile men.
In a case–control study in South Africa, Leisegang et al41 assessed the effect of metabolic syndrome (MetS) on testosterone, progesterone and semen parameters. A significantly lower sperm concentration was observed in the MetS group, as well as total sperm count, total motility, sperm vitality, mitochondrial membrane potential (MMP), free testosterone, and free progesterone.
Causes associated with Gene/Genotype
Male infertility is associated with genetic risk factors, such as chromosomal abnormalities, microdeletions in the Y chromosome, gene mutation, and polymorphisms in genes affecting spermatogenesis.42–44
A statistically significant difference was observed between the infertile and fertile control groups in sperm count, sperm motility, serum FSH level, and CAG repeats. In fertile men, CAG repeat length was significantly lower than those of infertile oligozoospermics and asthenospermics. The authors conclude that long stretches of CAG repeat may be associated with lower androgen receptor function and with imbalances in sperm production, which may cause male infertility.45
A case–control study to investigate the role of glutathione S-transferase Mu-1 gene polymorphism (GSTM1) in a sample of Egyptian males with idiopathic infertility observed a higher frequency of GSTM1 null genotype in infertile men compared to fertile men. Significantly lower sperm concentration and sperm count were observed in patients with the GSTM1 null genotype compared to patients with the GSTM1-positive genotype.46
Omran et al47 investigated the relationship between urinary bisphenol A (BPA) and semen quality. There was a negative association between total BPA levels, semen quality, antioxidant levels, and a positive correlation with DNA damage.
Eloualid et al48 related pathogenic c.144delC mutation in the AURKC gene to male infertility in Morocco.
In a case–control study that involved 241 infertile and 115 fertile men to examine the different types of chromodomain protein, Y-linked 1 (CDY1) gene deletions, their effect on male infertility and spermatogenesis in Tunisia, and deletion of CDY1b were significantly more frequent in infertile patients than in fertile men.49 Their analysis showed that independent of sperm concentration, deletion of the CDY1b copy gene was a significant risk factor for male infertility.
Behavioral/Lifestyle Risk Factors
Existing evidence points to cigarette or tobacco smoking, alcohol intake, poor health-seeking behavior among men, untreated or poorly sexually transmitted infections, sexual promiscuity, overweight/obesity, medication, and coital frequency as some of the significant behavioural/lifestyle risk factors for male infertility.
Smoking, Alcohol Intake, STD, Medication
Garba-Alkali et al29 examined the statistical relationship between semen analysis and selected variables. The semen analysis (normal and abnormal) was statistically related to the following risk factors for male infertility: past STD treatment, smoking history, and alcohol intake. Except for alcohol intake, the proportion of abnormal semen was higher for all the categories than normal semen and the association between risk factors and semen analysis was significant. A significant association was also found for medication used for the treatment of peptic ulcer disease and for HIV/AIDS. A higher percentage of cases of abnormal semen used medication (14.3%) compared to 7.8% in cases with normal semen. However, this study tested the significant relationship using chi-square, which was unable to detect and disaggregate the magnitude of relationship by the categories, as well as show the direction of effect.
Also, a cross-sectional examination of male infertility in southeastern Nigeria pointed to genital tract infection which the authors associated with sexual promiscuity, and poorly treated sexually transmitted infections as risk factors.50 A large proportion of the men in a study in Zimbabwe had or ever treated sexually transmitted diseases, syphilis, gonorrhea, chlamydia, chancroids, and other urinary tract/genital tract infections, including unspecified urethritis.34 Emeghe & Ekeke51 also identified sexually transmitted disease as a risk factor. In a review by Ness et al52, gonorrhea and/or chlamydia were identified as possible causes of male infertility. Clinical and pathologic evidence links them to urethritis and links urethritis to epididymo-orchitis, and epididymo-orchitis is linked to male infertility. Gonorrhoea and other STIs were significant risk factors for sperm abnormality, and azoospermia in Bayasgalan et al37.
Cigarette smoking lowers the quality of sperm.53 Okonofua et al22 found that male infertility was associated with a longer duration of smoking and heavy drinking of alcohol. Specifically, the quantity of alcohol intake of (1–2 glasses of alcohol per day and weekly) were positive predictors of male infertility compared to occasional alcohol intake. Concerning health-seeking behavior in Okonofua et al, use of referral hospitals and chemists for treatment of STIs, and use of native medications were reported as possible predictors of male infertility. In a retrospective study conducted by Benbella et al54 in Morocco, sexually transmitted diseases, tuberculosis, obesity, cigarette smoking, and alcohol consumption were identified as common infertility risk factors.
In a report of seminal fluid analysis conducted by Meri et al55 for 1438 men, a significant effect of smoking on sperm motility was found. Among smokers, lower sperm concentration and a higher percentage of abnormal sperms were found among heavy smokers (20 or more cigarettes per day) relative to non-heavy smokers. Leukocyte infiltration was also higher in the semen of smokers, particularly heavy smokers. In a systematic review of the relationship between smoking and male infertility, the majority of the studies reported that smoking significantly decreased sperm production, sperm motility, sperm normal forms, and sperm fertilizing capacity through increased seminal oxidative stress and DNA damage; only a few articles reported no significant relationship.56 The analysis of the clinical and seminal data obtained from 1726 suspected infertile men conducted in South Africa over 6 years found behavioural risk factors, such as extramarital sexual contact at least once a week, taking of alcohol at least once a week, and a history of smoking as risk factors for male infertility.57 On the contrary, the duration of smoking had no significant effects on the motility and morphology of sperm in a study in Iran; however, an increase of 1 cigarette per day decreased sperm motility by 1%.39
Body Mass Index (BMI)
Abnormal (low and elevated) body mass index (BMI) was identified in many studies as a significant risk factor for male infertility.58–62 Low sperm count and poor spermatozoa motility were associated with abnormal BMI in Oghagbon et al62. In Abayomi et al58 semen volume, sperm count, and motility were significantly lower in obese men than men with normal weight. Egwurugwu et al63 presented a significant positive correlation between body mass index and infertility among men; as body mass increases, sperm count decreases but progesterone (ng/mL) and oestradiol are positively correlated with body mass index. The limitation of this study was the use of correlation, which did not specify the BMI cut-offs and did not control for confounders. In a prospective study of 1000 males attending the infertility clinics conducted by Amah et al64, it was observed that men with a BMI greater than 25 kg/m2 had fewer sperm cells per ejaculate. The mean percentage motility of spermatozoa, sperm cell count, and normal morphology was significantly lower in infertile men compared to fertile subjects. In another study conducted in Ilorin, Nigeria, to evaluate the level of seminal fluid and body mass index of infertile men, BMI was significantly associated with infertility. Low and high BMIs were associated with abnormal sperm count and motility. The highest mean sperm count and motility (39.35 million/mL) was found among men with normal BMI (20–24Kg/M Square), relative to those who had low or high BMI.62 Controlling the effects of sexual dysfunction, overweight and obese men were more likely to be infertile, according to a Norwegian study.61 Similarly, in a study in Iran, obese men were 3.5 times more likely to have oligospermia than men with normal BMI.59
However, BMI was not found to be a significant factor in Garba-Alkali et al29; there were more cases with abnormal semen who had normal BMI (18.5–24.9) than cases with normal semen. According to Alshahrani et al65, sperm concentration was the only sperm parameter that was significantly lower in men whose BMI was high compared to normal BMI.
A review showed that hypotestosteronaemia, a hormonal defect in obese men, is the primary mechanism that lowers their fecundity, and the causes of infertility in overweight and obese men are reversible.66
Other Behavioural/Lifestyle Factors
Other risk factors for male infertility included excessive intake of antioxidants, previous exposure to drugs, and the use of native medications, and infections.1,5,25–27,67–69 Illicit drugs such as marijuana, cocaine, anabolic–androgenic steroids, opiates (narcotics), and methamphetamines, psychological stress, caffeine and unhealthy diet were identified as lifestyle risk factors for male infertility in a review conducted by Durairajanayagam.53 Another significant factor was coital frequency.29
Environmental Risk Factors
Review articles on the risk factors of male infertility identified exposure to toxic metals, such as lead, zinc deficiency, pesticides, industrial chemicals, and mycotoxins. Other risk factors for male infertility identified in the reviews included chemotherapeutic agents, radiation exposure and pharmaceutical agents that act either as direct spermatotoxins or through a steroidal pathway.1,5,25–27
In a study in Lebanon, after adjusting for confounders, exposure to chemical and physical agents in the workplace such as solvents, pesticides, fuel, cement increased the risk of male infertility two-fold, but metal pollutants were not significantly related to male infertility.70 In another study conducted in Saudi Arabia, occupational exposure to high temperature increased the risk of male infertility among bakers.71
Occupational exposure to pesticides caused testicular dysfunction and male infertility.72 Oliva et al73 studied 225 male partners from consecutively recruited couples who presented for infertility treatment in Argentina. After adjusting potential confounders such as age, BMI, and smoking, a significant association was observed between exposure to pesticides and solvents and low sperm count. Also, exposure to pesticides increased serum oestradiol concentrations, and LH concentrations were lower in men who were exposed to solvents compared to the unexposed men.
Indoor spraying of the pesticide DDT [1, 1,1-trichloro-2,2-bis (chlorodiphenyl) ethane] was associated with male infertility. An assessment of data on seminal parameters of 311 young men in Limpopo, South Africa, by Aneck-Hahn et al74 points to the effect of DDT exposure on male infertility. Evidence from the study indicates a significant association between nonoccupational exposure to p, p9-DDT and p, p9-dichlorodiphenyl-dichloroethylene (p, p9-DDE), its metabolite, and the seminal parameters of the young men. Both p, p’-DDT and p, p’-DDE were significantly higher in men who reside in in-sprayed houses compared to men from non-sprayed houses. Oligozoospermia was significantly associated with the lipid-adjusted p, p9-DDE, and asthenozoospermia was associated with the lipid-adjusted p, p9-DDT. The incidence of oligozoospermia and asthenozoospermia increased with higher p, p9- DDE concentration.
In another case–control study by Akinloye et al75 in Nigeria, exposure to cadmium toxicity was significantly associated with male infertility. Aflatoxin is associated with male infertility. In a case–control study, a higher percentage of spermatozoal abnormalities was observed in infertile men with aflatoxin in their semen than the fertile men.76 In a systematic review and meta-analysis of evidence for decreasing sperm count in African population, the authors reported that infertile men with aflatoxin in their semen showed a 50% higher percentage of spermatozoa abnormalities than the fertile men (10–15%)5. This suggests that pesticides, industrial chemicals and mycotoxins like aflatoxins may be implicated in the declining fertility of African men.
Socio-Demographic Risk Factors
Male infertility was significantly higher among men who had not fathered a child with their present wife or another wife than those who had.22 Age was significantly associated with male infertility.77 A systematic review and meta-analysis of alterations in sperm concentration in the African population from 1965 to 2015 identified age as a risk factor in 13 of the 14 studies. There was a significant decrease in sperm concentration with age.5 In Ugwuja et al33, a higher percentage of semen abnormalities were found in the age group 31–40 years and men aged 40 years and over in Emeghe & Ekeke51. The least sperm count and motility were among infertile men who were 30–39 years old in Oghagbon et al62. Ugwuja et al submitted that men aged 31–40 represent the active reproductive age group who may be harbouring residual infections acquired before marital life33.
In Osazuwa et al23, a significantly higher prevalence of C. trachomatis antibodies was observed in men aged 20–29 and 30–39 years. Abayomi et al58 attempted to establish a synergistic relationship between age and BMI in sperm parameters to document alterations in volume, liquefaction time, motility, and sperm count at age less than 45 years and at age 45 years and above. The study observed a significantly higher mean semen volume in men aged <45 years compared with men aged 45 years and above. Age and abnormal BMI together pose a higher risk of infertility. Also, the highest mean BMI was recorded in the 30–39 years old group, which recorded the worst spermogram.62 Men who were obese and above 45 years old were nearly 2 and half times more likely to produce semen <1.5 mL in Abayomi et al58.
Also, semen abnormalities are more common among civil servants than other professionals. The authors deduced that the high prevalence of abnormal semen in civil servants could be explained by involvement in some social activities such as smoking and excessive alcohol consumption that may interfere with semen qualities.33 In another study in Iran, manual workers were more at risk of infertility than office workers. The authors attributed it to strenuous physical work and testicular damage and its effect on the quality of sperm. Compared to respondents who had office jobs, varicocele and hernia were, respectively, 2.4 and 1.6 times more in the manual workers.39
Discussion
The study was designed to identify the plausible causes of male infertility based on published evidence and to suggest lines of new research or interventions to better inform the prevention and primary treatment of male infertility. To our surprise, despite the rising prevalence of male infertility, our results showed limited international publications that systematically address the causation and risk factors for male infertility. We could identify only 60 published studies from the international literature that address the objectives of the study, with many addressing only the superficial aspects without the deeper interrogation of the causal pathway to male infertility. Of greater surprise was that many of the studies on the causes of male infertility emanated from low- and middle-income countries, with few such studies coming from high-income countries. This may be due to the use of the widely effective secondary treatment for male infertility in high-income countries, such as in-vitro fertilization and intracytoplasmic sperm insemination (ICSI),78,79 which may preclude the need for investigation of the causes of male infertility in those countries.
The results of this review permit us to categorize the reported causes of male infertility into four broad causes. These include 1) biological/physiological/genetic causes, 2) behavioural/lifestyle risk factors, 3) environmental factors, and 4) socio-demographic risk factors.
The major biological factors associated with male infertility identified in this review were genetic factors, uro-genital infections, and varicoceles. While the role of chromosomal abnormalities such as deletions or additional X-chromosomes has been well established, it is not well known whether genetic alterations in the presence of normal chromosomal composition are associated with male infertility. The question as to whether male infertility runs in families has not been adequately addressed, and if so, the exact genetic or epi-genetic mechanisms that may be associated.
Similarly, while infections of various types as cultured from semen samples were associated with male infertility in the studies reported, the exact mechanisms through which these infections cause infertility were not fully elucidated. Merely finding bacterial or chlamydia in semen samples of infertile men is not sufficient to attribute those infections to male infertility. There has to be research to pinpoint the exact mode of acquisition, the sites of action and the mechanisms for any associated compromise of male fertility.
Over the years, varicocele has been reported as a cause of infertility,28,37,39 but several other publications have also refuted this finding.80,81 Indeed, the role of varicocelectomy as a treatment for male infertility remains a moot point. There is therefore a need for a randomized control trial, or meta-analysis to confirm the effectiveness of varicocelectomy, and the exact role of varicocele in the causation of male infertility.
The behavioural risk factors associated with male infertility in this review included smoking, alcohol intake, inappropriate body mass index, sexual behavior, and exposure to drugs. These risk factors were elucidated in cross-sectional or case–control studies, which suggest a low level of evidence that supports causation. Studies that are more robust are required to establish causation, and to determine the possible mechanisms of action. Of interest is that although diabetes mellitus is a metabolic disease that is associated with increased body mass index, no studies were identified from this study that investigated the relationship between diabetes mellitus or pre-diabetes and diabetic medications associated with male infertility. This is a gap in the literature that warrants investigation, especially as diabetes mellitus may be associated with vascular disease that may limit penile erection and ejaculation.
The major environmental risk factors for male infertility identified in this review were exposures to chemicals, various pesticides, and mycotoxins. Unfortunately, the evidence for these in the literature is based on limited evidence without systematic research to substantiate their involvement as causes of male infertility. Also, lacking is the elucidation of the possible causative pathway through which these toxins may lead to infertility. Based on the increasing use of these chemicals for household and agricultural purposes in many countries, it is critical that more systematic research is conducted to confirm possible causes of infertility. Several authors have presented evidence that rising environmental temperatures due to the prevailing climate changes may compromise the process of spermatogenesis and therefore reduce sperm counts.71,82 While this argument is understandable, there has as yet not been any research purposefully conducted to identify the relationship of environmental temperature attributable to climate changes to the increasing rate of male infertility.
As for socio-demographic factors, increasing age featured as the most common among those reported in the papers under review. However, the exact mechanisms through which age mediates declining semen quality is lacking in many of these publications – a gap that requires more systemic investigation. Additionally, some papers examined the role of polygamy – a phenomenon that is highly prevalent in African countries, and its association with male infertility. The question as to whether men in polygamous marriages compared to those in monogamous marriages are more or less likely to be infertile has not been adequately addressed in the papers reviewed. This is clearly an important gap in the literature that needs to be further elucidated.
Overall, the results of this review indicate a substantial deficit of information required to confirm specific causes for male infertility. While some associations have been identified, there is a need to establish causation with purposefully designed studies. These should consist of longitudinal cohort studies or large randomized control trials that investigate the effectiveness of interventions that remedy some of the risk factors to determine the extent to which they reduce male infertility. Interventions such as behavioral changes, reduction of exposures to environmental risk factors and treatment of infections would be relevant to investigate in clinical trials as primary prevention methods for male infertility.
Additionally, a robust investigation of male infertility would be needed to deplore high-tech and scientific methods, such as genetic and epigenetic studies, metabolomics, chemical and micronutrient studies, as well as microbiological studies. Only a scientific clarification of the specific causes of male infertility will help the process of identifying relevant primary prevention and treatment methods.
Strengths and Limitations
The study reviewed studies published in the English language literature that investigated the causes and risk factors for male infertility. Publications not based on empirical research were excluded. This enabled the paper to focus on actual causes rather than those based on anecdotes that are highly prevalent in the literature for male infertility. The fact that publications from both high-income and low-income countries were reviewed further strengthens the potential impact and relevance of this study.
The major limitation is the fact that the study was based on a scoping review rather than a systematic review of the literature. A cursory preliminary assessment showed that there were limited publications that focused on the specific research question. Different studies that aimed at identifying the causes of male infertility attempted to do so in diverse ways – many focused on “prevalence” and “risk factors” rather than causes. The risk factors were identified through examination of semen parameters only, and in many cases, even the socio-demographic characteristics of the male participants were not included in the studies. As such, it was difficult to pinpoint studies that addressed the specific research question posed by this study to enable a more complete assessment. The review was limited to studies published in English; thus, this limited the scope of the review as publications in other languages were excluded.
Conclusion
The results of this scoping review indicate limited knowledge of the actual causes of male infertility in published studies. The gaps in knowledge that need to be bridged to have a fuller understanding of the actual causes of male infertility have been highlighted. We believe that a better understanding of the causes of male infertility will help identify more effective primary prevention and treatment methods for male infertility.
Acknowledgment
We acknowledge Austin Iroghama Aruomaren for his contribution in the initial search for the articles.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The Tertiary Education Trust Fund (TETFUND) of Nigeria funded the study, under a National Research Fund grant referenced “TETFund/DR&D/CE/NRF/STI/44/VOL1” to FEO.
Disclosure
The authors report no competing interests in this work.
References
1. Abarikwu SO. Causes and risk factors for male-factor infertility in Nigeria: a review. Afr J Reprod Health. 2013;17:150–166.
2. Kolesnikova LI, Kolesnikov SI, Kurashova NA, Bairova TA. Causes and factors of male infertility. Annals Russ acad med sci. 2015;70(5):579–584. doi:10.15690/vramn.v70.i5.1445
3. Punab M, Poolamets O, Paju P, et al. Causes of male infertility: a 9-year prospective monocentre study on 1737 patients with reduced total sperm counts. Hu Reprod. 2017;32(1):18–31. doi:10.1093/humrep/dew284
4. Sharma A. Male infertility; evidences, risk factors, causes, diagnosis and management in human. Ann Clin Lab Sci. 2017;5(3):188. doi:10.21767/2386-5180.1000188
5. Sengupta P, Nwagha U, Dutta S, Krajewska-Kulak E, Izuka E. Evidence for decreasing sperm count in African population from 1965 to 2015. Afr Health Sci. 2017;17(2):418–427. doi:10.4314/ahs.v17i2.16
6. Kumar N, Singh AK. Trends of male factor infertility, an important cause of infertility: a review of literature. J Hum Reprod Sci. 2015;8(4):191–196. doi:10.4103/0974-1208.170370
7. World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen.
8. Dabaja AA, Schlegel PN. Medical treatment of male infertility. Transl Androl Urol. 2014;3(1):9–16. doi:10.3978/j.issn.2223-4683.2014.01.06
9. Ombelet W, Van Robays J. Artificial insemination history: hurdles and milestones. Facts Views Vis Obgyn. 2015;7(2):137–143.
10. Abdelkader AM, Yeh J. The potential use of intrauterine insemination as a basic option for infertility: a review for technology-limited medical settings. Obstet Gynecol Int. 2009;2009:1–11. Article ID 584837. doi:10.1155/2009/584837
11. Iketubosin F. In vitro fertilization embryo transfer processes and pathway: a review from practice perspective. Trop J Obstet Gynaecol. 2018;35(3):227–232. doi:10.4103/TJOG.TJOG_83_18
12. Sutcliffe AG. Intracytoplasmic sperm injection and other aspects of new reproductive technologies. Arch Dis Child. 2000;83(2):98–101. doi:10.1136/adc.83.2.98
13. Chandy A. Adoption: an option in infertility. Curr Med Issues. 2016;14(4):104. doi:10.4103/0973-4651.194474
14. Shiraishi E, Takae S, Faizal AM, Sugimoto K, Okamoto A, Suzuki N. The scenario of adoption and foster care in relation to the reproductive medicine practice in Asia. Int J Environ Res Public Health. 2021;18(7):3466. doi:10.3390/ijerph18073466
15. Barak S, Baker HG. Clinical management of male infertility. Endotext [internet]; 2016. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279160/.
16. Barratt CL, Björndahl L, De Jonge CJ, et al. The diagnosis of male infertility: an analysis of the evidence to support the development of global WHO guidance—challenges and future research opportunities. Hum Reprod Update. 2017;23(6):660–680. doi:10.1093/humupd/dmx021
17. Fainberg J, Kashanian JA. Recent advances in understanding and managing male infertility. F1000Res. 2019;8:670. doi:10.12688/f1000research.17076.1
18. Munn Z, Peters MDJ, Stern C, Tufanaru C, McArthur A, Aromataris E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med Res Methodol. 2018;18(1):143. doi:10.1186/s12874-018-0611-x
19. Arksey H, O’Malley L. Scoping studies: towards a methodological framework. Int J Soc Res Methodol. 2005;8(1):19–32. doi:10.1080/1364557032000119616
20. PRISMA - Transparent reporting of systematic reviewe and meta-analysis. Available from https://prisma-statement.org/Extensions/ScopingReviews
21. Ahmed A, Bello A, Mbibu NH, Maitama HY, Kalayi GD. Epidemiological and aetiological factors of male infertility in northern Nigeria. Niger J Clin Pract. 2010;13:205–209.
22. Okonofua F, Omo-Aghoja LO, Menakaya U, Onemu SO, Bergstrom S. A case-control study of risk factors for male infertility in Southern Nigeria. Trop J Obstet Gynaecol. 2005;22:136–143. doi:10.4314/tjog.v22i2.14513
23. Osazuwa F, Aiguobarueghian OI, Alekwe L, Imade PE, Ibadin KO. The prevalence of Chlamydia trachomatis infection among infertile males and its association with abnormal semen characteristics in Delta State, Nigeria. Tanzan J Health Res. 2013;15. doi:10.4314/thrb.v15i2.3
24. Smolak A, Chemaitelly H, Hermez JG, Low N, Abu-Raddad LJ. Epidemiology of Chlamydia trachomatis in the Middle East and north Africa: a systematic review, meta-analysis, and meta-regression. Lancet Glob. 2019;7(9):e1197–e1225. doi:10.1016/S2214-109X(19)30279-7
25. Olayemi F. Review on some causes of male infertility. Afr J Biotechnol. 2010;9:20.
26. Olooto WE. Infertility in male; risk factors, causes and management-A review. J Microbiol Biotechnol Res. 2012;2(4):641–645.
27. Uadia PO, Emokpae AM. Male infertility in Nigeria: a neglected reproductive health issue requiring attention. J Basic Clin Reprod Sci. 2015;4(2):45–53. doi:10.4103/2278-960X.161042
28. Jeje EA, Alabi TO, Ojewola RW, Ogunjimia MA, Osunkoya SA. Male infertility: an audit of 70 cases in a single centre. Arab J Urol. 2016;22(3):223–226. doi:10.1016/j.afju.2015.10.003
29. Garba-Alkali AE, Adesiyun AG, Randawa AJ. Semen profile of male partners of women attending infertility clinic in Zaria, Nigeria. Trop J Obstet Gynaecol. 2018;35(3):256–260. doi:10.4103/TJOG.TJOG_18_18
30. Idrisa A, Ojiyi E, Tomfafi O, Kamara TB, Pindiga HU. Male contribution to infertility in Maiduguri, Nigeria. Trop J Obstet Gynaecol. 2001;18(2):87–90. doi:10.4314/tjog.v18i2.14437
31. Enwurua CA, Iwalokuna B, Enwuru VN, Ezechi O, Oluwadun A. The effect of presence of facultative bacteria species on semen and sperm quality of men seeking fertility care. Afr J Urol. 2016;22(3):213–222. doi:10.1016/j.afju.2016.03.010
32. Emokpae MA, Uadia PO, Sadiq NM. Contribution of bacterial infection to male infertility in Nigerians. Online J Health Allied Sci. 2009;8(1).https://www.researchgate.net/publication/26627176_Contribution_of_Bacterial_Infection_to_Male_Infertility_in_Nigerians
33. Ugwuja E, Ugwu N, Ejikeme B. Prevalence of low sperm count and abnormal semen parameters in male partners of women consulting at infertility clinic in Abakaliki, Nigeria. Afr J Reprod Health. 2008;12(1):67–73.
34. Folkvord S, Odegaard OA, Sundby J. Male infertility in Zimbabwe. Patient Educ Couns. 2005;59(3):239–243. doi:10.1016/j.pec.2005.08.003
35. Ibadin KO, Enabulele OI, Eghafona NO, Osemwenkha AP. Seroevidence of chlamydia trachomatis infection in infertile male and its association with semen quality In University of Benin Teaching Hospital, Benin City, Nigeria. Benin J Postgrad Med. 2009;11. doi:10.4314/bjpm.v11i1.48820
36. Osegbe D, Amaku E. The causes of male infertility in 504 consecutive Nigerian patients. Int Urol Nephrol. 1985;17(4):349–358. doi:10.1007/BF02083505
37. Bayasgalan G, Naranbat D, Radnaabazar J, Lhagvasuren T, Rowe PJ. Male infertility: risk factors in Mongolian men. Asian J Androl. 2004;6(4):305–311.
38. Wright C, Milne S, Leeson H. Sperm DNA damage caused by oxidative stress: modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod Biomed Online. 2014;28(6):684–703. doi:10.1016/j.rbmo.2014.02.004
39. Mahboubi M, Foroughi F, Ghahramani F, Shahandeh H, Moradi S, Shirzadian T. A case–control study of the factors affecting male infertility. Turk J Med Sci. 2014;44(5):862–865. doi:10.3906/sag-1304-35
40. Khalil AA, Hussien HM, Sarhan EM. Oxidative stress induces idiopathic infertility in Egyptian males. Afr J Biotechnol. 2012;11(6):1516–1522. doi:10.4314/ajb.v11i6
41. Leisegang K, Udodong A, Bouic PJD, Henkel RR. Effect of the metabolic syndrome on male reproductive function: a case-controlled pilot study. Andrologia. 2014;46(2):167–176. doi:10.1111/and.12060
42. Ferlin A, Raicu F, Gatta V, Zuccarello D, Palka G, Foresta C. Male infertility: role of genetic background. Reprod Biomed Online. 2007;14(6):734–745. doi:10.1016/S1472-6483(10)60677-3
43. Ferlin A, Foresta C. New genetic markers for male infertility. Curr Opin Obstet Gynecol. 2014;26(3):193–198. doi:10.1097/GCO.0000000000000061
44. Krausz C, Giachini C. Genetic risk factors in male infertility. Arch Androl. 2007;53(3):125–133. doi:10.1080/01485010701271786
45. Mosaad YM, Shahin D, Elkholy AAM, Mosbah A, Badawy W. CAG repeat length in androgen receptor gene and male infertility in Egyptian patients. Andrologia. 2012;44(1):26–33. doi:10.1111/j.1439-0272.2010.01100.x
46. Roshdy OH, Hussein TM, Zakaria NH, Sabry AA. Glutathione S-transferase Mu-1 gene polymorphism in Egyptian patients with idiopathic male infertility. Andrologia. 2015;47(5):587–593. doi:10.1111/and.12306
47. Omran GA, Gaber HD, Mostafa NAM, Abdel-Gaber RM, Salah EA. Potential hazards of bisphenol A exposure to semen quality and sperm DNA integrity among infertile men. Reprod Toxicol. 2018;81:188–195. doi:10.1016/j.reprotox.2018.08.010
48. Eloualid A, Rouba H, Rhaissi H, et al. Prevalence of the Aurora kinase C c. 144delC mutation in infertile Moroccan men. Fertil Steril. 2014;101(4):1086–1090. doi:10.1016/j.fertnstert.2013.12.040
49. Ghorbel M, Baklouti-Gargouri S, Keskes R, et al. Deletion of CDY1b copy of Y chromosome CDY1 gene is a risk factor of male infertility in Tunisian men. Gene. 2014;548(2):251–255. doi:10.1016/j.gene.2014.07.042
50. Ikechebelu JI, Adinma JIB, Orie EF, Ikegwuonu SO. High prevalence of male infertility in southeastern Nigeria. J Obstet Gynaecol. 2003;23(6):657–659. doi:10.1080/01443610310001604475
51. Emeghe IN, Ekeke ON. Sero-Prevalence of anti-sperm antibodies in infertile males in Port Harcourt, Nigeria. East Afr Med J. 2017;94(2):125–130.
52. Ness RB, Markovic N, Carlson CL, Coughlin MT. Do men become infertile after having sexually transmitted urethritis? An epidemiologic examination. Fertil Steril. 1997;68(2):205–213. doi:10.1016/S0015-0282(97)81502-6
53. Durairajanayagam D. Lifestyle causes of male infertility. Arab J Urol. 2018;16(1):10–20. doi:10.1016/j.aju.2017.12.004
54. Benbella A, Aboulmakarim S, Hardizi H, Zaidouni A, Bezad R. Infertility in the Moroccan population: major risk factors encountered in the reproductive health centre in Rabat. Pan Afr Med J. 2018;30:195.
55. Meri ZB, Irshid IB, Migdadi M, Irshid AB, Mhanna SA. Does cigarette smoking affect seminal fluid parameters? A comparative study. Oman Med J. 2013;28(1):12–15. doi:10.5001/omj.2013.03
56. Mostafa T. Cigarette smoking and male infertility. J Adv Res. 2010;1(3):179–186. doi:10.1016/j.jare.2010.05.002
57. Bornman MS, Schulenburg GW, Boomker D, Chauke TR, Reif S. Observations in infertile African males at an andrology clinic in South Africa. Arch Androl. 1994;33(2):101–104. doi:10.3109/01485019408987810
58. Abayomi BA, Afolabi BM, Victor DA, Oyetunji I. Semen parameters associated with male infertility in a Sub-Saharan black population: the effect of age and body mass index. J Obstet Gynecol Infertil. 2018;1:1–8.
59. Hajshafiha M, Ghareaghaji R, Salemi S, Sadegh-Asadi N, Sadeghi-Bazargani H. Association of body mass index with some fertility markers among male partners of infertile couples. Int J Gen Med. 2013;6:447. doi:10.2147/IJGM.S41341
60. Keszthelyi M, Gyarmathy VA, Kaposi A, Kopa Z. The potential role of central obesity in male infertility: body mass index versus waist to Hip ratio as they relate to selected semen parameters. BMC Public Health. 2020;20(1):1–10. doi:10.1186/s12889-020-8413-6
61. Nguyen RH, Wilcox AJ, Skjærven R, Baird DD. Men’s body mass index and infertility. Hum Reprod. 2007;22(9):2488–2493. doi:10.1093/humrep/dem139
62. Oghagbon EK, Jimoh AAG, Adebisi SA. Seminal fluid analysis and biophysical profile: findings and relevance in infertile males in Ilorin, Nigeria. Afri J Clin Exp Microbiol. 2004;5(3):280–284. doi:10.4314/ajcem.v5i3.7392
63. Egwurugwu JN, Nwafor A, Chike CPR, et al. The relationship between body mass index, semen and sex hormones in adult male. Niger J Physiol Sci. 2011;26:1.
64. Amah H, Nwachukwu C, Obiajuru I, Amah C. Studies on the male partners of couples presenting with infertility. Int j Biol Chem Sci. 2012;6(6):6062–6068.
65. Alshahrani S, Ahmed AF, Gabr AH, Abalhassan M, Ahmad G. The impact of body mass index on semen parameters in infertile men. Andrologia. 2016;48(10):1125–1129. doi:10.1111/and.12549
66. Katib A. Mechanisms linking obesity to male infertility. Cent European J Urol. 2015;68(1):79. doi:10.5173/ceju.2015.01.435
67. Henkel R, Sandhu IS, Agarwal A. The excessive use of antioxidant therapy: a possible cause of male infertility? Andrologia. 2019;51(1):e13162. doi:10.1111/and.13162
68. Henkel R, Offor U, Fisher D. The role of infections and leukocytes in male infertility. Andrologia. 2020;53:e13743. doi:10.1111/and.13743
69. Leisegang K, Sengupta P, Agarwal A, Henkel R. Obesity and male infertility: mechanisms and management. Andrologia. 2020;53:e13617. doi:10.1111/and.13617
70. Inhorn MC, King L, Nriagu JO, et al. Occupational and environmental exposures to heavy metals: risk factors for male infertility in Lebanon? Reprod Toxicol. 2008;25(2):203–212. doi:10.1016/j.reprotox.2007.10.011
71. Al-Otaibi ST. Male infertility among bakers associated with exposure to high environmental temperature at the workplace. J Taibah Univ Medical Sci. 2018;13(2):103. doi:10.1016/j.jtumed.2017.12.003
72. Saalu LC, Osinubi AA. Environmental endocrine disruptors of testicular function. Afri J Endocrinol Metabol. 2009;8(1):15–25.
73. Oliva A, Spira A, Multigner L. Contribution of environmental factors to the risk of male infertility. Hu Reprod. 2001;16(8):1768–1776. doi:10.1093/humrep/16.8.1768
74. Aneck-Hahn NH, Schulenburg GW, Bornman MS, Farias P, de Jager C. Impaired semen quality associated with environmental DDT exposure in young men living in a malaria area in the Limpopo Province, South Africa. J Androl. 2007;28(3):423–434. doi:10.2164/jandrol.106.001701
75. Akinloye O, Arowojolu AO, Shittu OB, Anetor JI. Cadmium toxicity: a possible cause of male infertility in Nigeria. Reprod Biol. 2006;6(1):17–30.
76. Ibeh IN, Uraih N, Ogonar JI. Dietary exposure to aflatoxin in human male infertility in Benin City, Nigeria. Int J Fertil Menopausal Stud. 1994;39(4):208–214.
77. Dunson DB, Baird DD, Colombo B. Increased infertility with age in men and women. Obstet Gynecol. 2004;103(1):51–56. doi:10.1097/01.AOG.0000100153.24061.45
78. Drakopoulos P, Garcia-Velasco J, Bosch E, et al. ICSI does not offer any benefit over conventional IVF across different ovarian response categories in non-male factor infertility: a European multicenter analysis. J Assist Reprod Genet. 2019;36(10):2067–2076. doi:10.1007/s10815-019-01563-1
79. Merchant R, Gandhi G, Allahbadia GN. In vitro fertilization/intracytoplasmic sperm injection for male infertility. Indian J Urol. 2011;27(1):121–132. doi:10.4103/0970-1591.78430
80. Alsaikhan B, Alrabeeah K, Delouya G, Zini A. Epidemiology of varicocele. Asian J Androl. 2016;18(2):179–181. doi:10.4103/1008-682X.172640
81. Jarow JP. Effects of varicocele on male fertility. Hum Reprod Update. 2001;7(1):59–64. doi:10.1093/humupd/7.1.59
82. Sharpe RM. Lifestyle and environmental contribution to male infertility. Br Med Bull. 2000;56(3):630–642. doi:10.1258/0007142001903436
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Exploring Factors Associated with Falls in Multiple Sclerosis: Insights from a Scoping Review
Kaddoura R, Faraji H, Othman M, Abu Hijleh A, Loney T, Goswami N, T S Benamer H
Clinical Interventions in Aging 2024, 19:923-938
Published Date: 23 May 2024
