Back to Journals » Cancer Management and Research » Volume 11
Carbon-ion radiotherapy in accelerated hypofractionated active raster-scanning technique for malignant lacrimal gland tumors: feasibility and safety
Authors Akbaba S, Lang K, Held T, Herfarth K, Rieber J, Plinkert P, Auffarth GU , Rieken S, Debus J , Adeberg S
Received 7 October 2018
Accepted for publication 27 December 2018
Published 1 February 2019 Volume 2019:11 Pages 1155—1166
DOI https://doi.org/10.2147/CMAR.S190051
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Sati Akbaba,1–3 Kristin Lang,1,2 Thomas Held,1,2 Klaus Herfarth,1–3 Juliane Rieber,1–3 Peter Plinkert,4 Gerd U Auffarth,5 Stefan Rieken,1–3 Juergen Debus,1–3 Sebastian Adeberg1–3
1Department of Radiation Oncology, University Hospital Heidelberg, Heidelberg 69120, Germany; 2Heidelberg Institute of Radiation Oncology (HIRO), National Center for Radiation Research in Oncology (NCRO), University Hospital Heidelberg, Heidelberg 69120, Germany; 3Heidelberg Ion-Beam Therapy Center (HIT), University Hospital Heidelberg, Heidelberg 69120, Germany; 4Department of Otorhinolaryngology, Head and Neck Surgery, University Hospital Heidelberg, Heidelberg 69120, Germany; 5Department of Ophthalmology, University Hospital Heidelberg, Heidelberg 69120, Germany
Introduction: We evaluated treatment outcomes of CIRT in an active raster-scanning technique alone or in combination with IMRT for lacrimal gland tumors.
Methods: A total of 24 patients who received CIRT for a malignant lacrimal gland tumor at the HIT between 2009 and 2018 were analyzed retrospectively for LC, OS, and distant progression-free survival (DPFS) using Kaplan–Meier estimates. Toxicity was assessed according to the CTCAE version 5.
Results: Median follow-up was 30 months and overall median LC, OS, and DPFS 24 months, 36 months, and 31 months, respectively. Two-year LC, OS, and DPFS of 93%, 96%, and 87% with CIRT was achieved for all patients. Local failure occurred only in patients with ACC and after a median follow-up of 30 months after the completion of RT (n=5, 21%; P=0.09). We identified a significant negative impact of a macroscopic tumor disease, which was diagnosed on planning CT or MRI before RT, on LC (P=0.026). In contrast, perineural spread (P=0.661), T stage (P=0.552), and resection margins in operated patients (P=0.069) had no significant impact on LC. No grade ≥3 acute or grade >3 chronic toxicity occurred. Late grade 3 side effects were identified in form of a wound-healing disorder 3 months after RT in one patient and temporal lobe necrosis 6 months after RT in another (n=2, 8%).
Conclusion: Accelerated hypofractionated active raster-scanning CIRT for relative radioresistant malignant lacrimal gland tumors results in adequate LC rates and moderate acute and late toxicity. Nevertheless, LC for ACC histology remains challenging and risk factors for local recurrence are still unclear. Further follow-up is necessary to evaluate long-term clinical outcome.
Keywords: carbon-ion radiotherapy, bimodal RT, malignant lacrimal gland tumor, adenoid cystic carcinoma, local control
Introduction
Lacrimal gland tumors account for 6%–12% of all orbital space-occupying lesions and are one of the rarest malignancies in the head and neck region, with an estimated incidence of one per 1,000,000 annually.1,2 Tumors of the lacrimal gland consist of a heterogeneous group of different histology, which can be divided into nonepithelial lesions in 70%–80% (lymphoid tumors, plasmocytoma, histiocytoma, lipoma, hemangioma, inflammation) and in epithelial lesions in 20%–30% (50% benign pleomorphic adenoma, 50% malignant tumors) of cases.1–6 Malignant epithelial tumors consist of ACC in 60%, adenocarcinoma in 20%–30%, and mucoepidermoid carcinoma in 5% of cases.1,5
Guidelines defining therapeutic approaches are lacking, due to the rarity of these tumors. Surgery followed by postoperative RT in the majority of cases seems to be the mainstay of treatment to date, while the type of surgery and postoperative RT are widely discussed and remain challenging.7–11 Several studies have shown equal survival outcome for patients treated with globe-sparing surgery compared to exenteration.9,12,13 Some authors have pleaded for postoperative external-beam RT with photons or protons after globe-sparing surgery, while others advocate brachytherapy or neoadjuvant plus adjuvant intracarotid chemotherapy with cisplatin and doxorubicin in combination with exenteration and postoperative RT.9–11,14
Due to morphological and embryological similarities, lacrimal gland tumors are mostly considered and treated analogously to salivary gland tumors.15,16 Since November 2009, both lacrimal and salivary gland tumors have been treated bimodally with CIRT in combination with IMRT or CIRT alone in our center, based on prior experiences with high-LET RT for malignant salivary gland tumors.17–21 In the current analysis, we present our first experiences with CIRT in active raster scanning, which is known for more accurate tumor targeting due to superior dose distribution compared with photons or protons and increased biological effectiveness, as well as better preservation of surrounding tissue, for lacrimal gland tumors in a case series of 24 patients, regarding LC, survival outcome, and toxicity.
Methods
Evaluation
Medical records of 24 patients with a lacrimal gland tumor who were treated with bimodal RT, including IMRT and CIRT, or CIRT alone between 2009 and 2018 at the HIT were analyzed retrospectively regarding OS, LC, distant progression-free survival (DPFS), acute and late toxicity, and prognostic factors for LC and survival events.
LC, OS, and DPFS were calculated with the Kaplan–Meier method from first diagnosis to last follow-up or time of event (death/distant progression). LC was assessed from beginning of RT to local progression. Kaplan–Meier estimates of potential prognostic factors were compared using the log-rank test for univariate analysis. P<0.05 was considered statistically significant. Multivariate analyses were not performed, due to our low patient population (n=24). Statistical tests were all conducted with SPSS version 24 (IBM Corporation, Armonk, NY, USA).
Toxicity was assessed according to the CTCAE version 5. Acute toxicity was defined as toxicity occurring within 3 months after completion of RT, and late toxicity as toxicity ≥3 months after RT. Tumor response was evaluated according to RECIST on the basis of regularly performed MRI. Follow-up examinations were planned for every 3 months during the first 2 years after the treatment, every half year during the third and fourth years after treatment, and then once a year, including a clinical examination by a head and neck surgeon and a current MRI. Additionally, a CT scan of the chest was performed annually to screen for pulmonary metastases.
Treatment planning and features
For treatment planning, a CT scan (native and contrast-enhanced) in head-first supine position was performed and patients immobilized with thermoplastic head masks. Target delineation was based on actual MRI, which was matched with the CT scan in irradiation position for better tumor demarcation. Treatment planning was performed with Syngo RT planning system (VC13; Siemens Healthcare). Two CTVs (CTV1 and CTV2) were delineated, CTV1 including the macroscopic tumor or tumor bed and CTV2 including CTV1 and typical pathways of tumor spread and the ipsilateral cervical nodal levels (I–II). The planning TV (PTV) included the CTV, and a 3 mm margin and was covered by the 95% prescription dose. Critical structures like optic chiasma, optic nerves, brain stem, spinal cord, and eyes were spared according to the Quantitative Analyses of Normal Tissue Effects in the Clinic guidelines.22
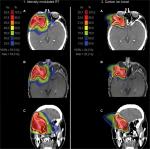
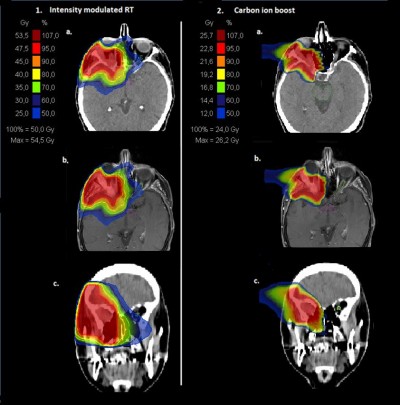
Photon RT was performed using TomoTherapy (Accuray, Sunnyvale, CA, USA) in five fractions per week (2 Gy each), and carbon ions were applied with intensity-controlled active raster-scanning in five to six fractions per week (3 Gy relative biological effectiveness [RBE]) fractions (table/gantry angle 90°/230° and 90°/175°, two horizontal beams, initial full width at half maximum 6 mm, scan grid 2 mm, range step 3 mm, maximum beam size 20×20 cm). The equivalent dose to two Gy single-dose fractions (EQD2) to compare the different fractionation regimes was calculated with a tumor α:β of 2 according to the local effectiveness model 1.23 Treatment features are depicted in Table 1. Overall, 88% of patients (n=21) received bimodal RT with IMRT to CTV2 and CIRT to CTV1, and 12% (n=3) received RT with CIRT alone to CTV1. The median prescribed C12 dose to CTV1 was 24 Gy (RBE) in eight fractions (range 18–66 Gy RBE), corresponding to a median EQD2 dose of 30 Gy (range 22.5–82.5 Gy), and median IMRT dose to CTV2 was 50 Gy in 25 fractions (range 50–54 Gy), which corresponded to a median cumulative total dose of 74 Gy RBE (total median EQD2 80 [72.5–82.5] Gy) to CTV1. CTVs were moderate, with a median CTV1 of 52 cm3 (range 7–353 cm3) and a median CTV2 of 130 cm3 (range 19–645 cm3). Figure 2 shows a combined treatment plan with IMRT and CIRT for patient 20 with an inoperable cT4N0M0-stage ACC of the right lacrimal gland.
Patient characteristics
The median age of our patient collective was 51 years (range 13–70 years). Histology differed from ACC (n=18, 75%) to adenocarcinoma (n=3, 13%), mucoepidermoid carcinoma (8%, n=2), and adenosquamous carcinoma (n=1, 4%). Patient and tumor characteristics are listed in Table 1. Most patients had advanced tumors (T4 stage, n=14, 58%) without perineural spread histopathologically (Pn0, n=12 of 20, 60%). Two patients showed suspicious lymph nodes on staging MRI before RT: one patient ipsilateral regional lymph nodes (N1, 4%) and another patient bilateral cervical lymph nodes (cN2c, 4%). No patient had distant metastases on the first presentation. RT was assessed predominantly in a primary setting (n=18, 75%) and postoperatively in the majority of patients (n=16, 67%). Enucleation of the eye globe before RT was assessed in 21% of patients (n=5). Overall, 79% of patients received RT for a macroscopic tumor disease (n=19): 17% for R1 (n=4) and 4% for R0 (n=1).
Ethics approval and consent to participate
The final protocol was approved by the ethics committee of the University of Heidelberg, Heidelberg, Germany (S-421/2015). Written consent to review their medical records was required from each patient. Analysis of the patient data was assessed in accordance with the Declaration of Helsinki.
Results
The median follow-up was 30 months (range 6–102 months). At last follow-up, 88% were still alive (n=21), of whom 71% were free of progression (15 of 21). CR occurred in 21% (5 of 24), all in patients who had been irradiated postoperatively, PR in 29% (7 of 24), and SD in 29% (7 of 24). In Figure 2, the treatment response of patient 20 with an ACC (T4N0M0, Pn1), who was treated in a primary setting with bimodal RT including IMRT and CIRT. Overall, five patients showed local relapse (overall 21%, one of five out of field, two of five in area where critical structures were spared, two of five in field) and one patient had a locoregional relapse into the bilateral cervical lymph nodes (4%). Distant relapse was identified in two patients: in one case into the lungs (4%) and in the other into the brain (4%). Median LC, OS, and DPFS were 24 months (range 6–59 months), 36 months (range 9–102 months), and 31 months (range 6–102 months), respectively. The corresponding 2-year LC, OS, and DPFS were 93%, 96%, and 87% and the estimated 5-year LC, OS, and DPFS 44%, 68%, and 58% for all patients (Figure 3A).
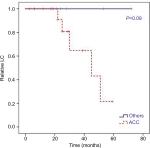
Local failure occurred a median of 30 months after the completion of RT (range 22–51 months) and only in patients with ACC (Figure 4). The corresponding 2-year, 3-year, and estimated 5-year LC/OS/DPFS for ACC patients were 90%/94%/93%, 65%/94%/93%, and 21%/71%/70%, respectively (Figure 3B). Univariate analysis showed the presence of a macroscopic tumor in the planning CT or MRI scans before RT as a significant negative prognostic factor for LC (HR 0.04, 95% CI 0–361.22; P=0.026; Figure 5).
In contrast, perineural spread (Pn0 vs Pn1, P=0.661), T stage (T2/3 vs T4, P=0.552), resection margins (R0/1 vs R2, P=0.069), and the RT setting (postoperative RT vs definitive RT, P=0.635) had no significant impact on LC, OS, or DPFS. Local recurrence occurred only in patients who had been irradiated postoperatively (n=1 with R1, n=4 with R2), and three of these five patients showed perineural spread (Pn1, 60%). Two patients received salvage surgery for recurrence (8%), two patients more RT (8%), and one enucleation of the eye globe in combination with more RT (4%).
Therefore, among patients who received eye-sparing surgery and postoperative RT as well as primary RT, eye preservation was achieved in 95% (18 of 19). Treatment results are shown in Table 2.
  | Table 2 Treatment results Abbreviations: DPFS, distant progression-free survival; LC, local control; LN, lymph node; NA, not applicable; OS, overall survival; Pul, pulmonary. |
Toxicity analysis
The treatment was tolerated well. Overall, 13% of patients (n=3) complained of acute grade 3 and 8% (n=2) chronic grade 3 toxicity at last follow-up. No grade 4 or 5 toxicity occurred. Treatment-related acute and late toxicity are shown in Table 3. The majority of patients reported acute dermatitis (88%, n=21) and acute xerophthalmia (50%, n=12), which had resolved for the most part at last follow-up. At that time, only 17% of patients still suffered from xerophthalmia grade 1–2 (n=4). Only one patient showed a motoric disorder of the facial nerve under RT, which remained for 3 months after completion of RT (4%). As for late grade 3 toxicity, a wound-healing disorder appeared in one patient 3 months after RT in the RT field, and they underwent multiple wound revisions as a consequence (4%). Additionally, radionecrosis of the temporal lobe occurred in two patients where 10%–30% of the total prescribed dose involved the temporal lobe, each with T4 tumors and orbital conus involvement (8%). In one patient, radionecrosis was asymptomatic and diagnosed during follow-up with MRI 3 years after RT (CTCAE grade 1). In another patient, radionecrosis appeared 6 months after RT and led to progressive dizziness, such that surgical revision was necessary (CTCAE grade 3). In this case, radionecrosis was proven histopathologically, and symptoms disappeared after surgery. During follow-up, no case of visual impairment or visual loss was reported by patients.
  | Table 3 Treatment-related acute and late toxicity (n=24) |
Discussion
Patients analyzed in the current study showed preferentially unfavorable tumor characteristics with ACC histology, which is known for its radioresistance and high local relapse rates (75%, n=18), advanced T stages (T3/4, 92%, n=22), and perineural spread (40%, 8 of 20). Nevertheless, dose-escalated CIRT led to adequate treatment results, with 2-year LC, OS, and DPFS of 93%, 96%, and 87% and estimated 5-year LC, OS, and DPFS of 44%, 68%, and 58%. Within 30 months’ follow-up, five patients developed local recurrence, all with ACC histology (21%). Treatment was tolerated well, with acute grade 3 toxicity in three (13%) and late grade 3 toxicity in two patients only (8%). No visual loss or visual impairment occurred within the follow-up, and enucleation of the eye globe due to local failure was obligatory in only one patient (5%).
Malignant epithelial tumors of the lacrimal gland have a low incidence.1,2 Therefore, therapy standards are lacking as a consequence of poor evidence. For malignant lacrimal gland tumors, several treatment modalities have been described in the literature.7–13,24 Traditionally, the treatment of choice consists of orbital exenteration.25 Nowadays, eye-sparing treatment modalities are gaining in importance, as no survival benefit has been shown for orbital exenteration.25,26 Surgery (eg, globe-sparing vs exenteration) is mostly followed by postoperative RT. Primary RT is necessary in cases of tumor inoperability.7 Thus, RT in the form of external-beam RT with photons, protons, or carbon ions as well as brachytherapy is used in >50% of all diagnosed malignant lacrimal gland malignancies.14,24,27,28
Studies reporting high-LET RT for radioresistant intraorbital tumors are rare. While postoperative 3-D RT and IMRT can decrease the local relapse rate in comparison with conventional RT techniques, neutron RT, one kind of high-LET RT, has resulted in high rates of ipsilateral vision loss, despite a 5-year LC of 80%.29–31 Concerning CIRT for lacrimal gland tumors, a comprehensive literature research by the authors resulted in few case series. In one study consisting of 21 patients with lacrimal gland tumors who were treated with primary CIRT or CIRT postoperatively, 3-year LC and OS of 82% and 79% were achieved.32 Additionally, Mizoe et al described in a dose-escalation study (phase I/II) with CIRT alone a 5-year LC rate of 50% for locally advanced ACC of the head and neck (four of nine overall ACC patients in this study had lacrimal gland tumors).33 For combined bimodal RT with IMRT and CIRT boost, as used in the current analysis, data are lacking to date. Therefore, the current study will be the first analysis reporting the outcome of this therapy regime. Generally, poor prognosis with a 10-year OS of 20%–39% and a 10-year distant control of 50% has been reported for lacrimal gland ACC by several authors.1,7,8,28 Andreoli et al showed a 10-year OS of 38.7% for lacrimal gland ACC, 40.3% for lacrimal gland mucoepidermoid carcinoma, and 27.7% for lacrimal gland adenocarcinoma, without statistically significant OS differences among the histological subgroups.28
Radioresistant choroidal melanoma-treatment results have shown excellent LC and eye-retention rates, as well as lower toxicity for CIRT compared with other RT modalities.34–36 Tsuji et al identified superior 3-year LC and enucleation rates of 97% and 5% for CIRT in choroidal melanoma patients when compared with proton RT.34,37 In the current study, enucleation of the eye globe due to a local relapse was necessary in only one patient, 46 months posttreatment, resulting in a comparable outcome, with an equal eye-retention rate compared to CIRT for choroidal melanoma (5%).
Due to the lack of evidence and similarity in histological subtypes, malignant lacrimal gland tumors are mostly considered and treated in accordance with malignant salivary gland tumors. First results using CIRT in the treatment of salivary gland tumors were promising regarding LC and toxicity.38–41 In a prospective phase II trial (COSMIC), Jensen et al identified superior 3-year LC and OS of 82% and 78% with less toxicity for CIRT vs photon RT for salivary gland tumors.17,42–44 In a further prospective carbon-ion pilot project, Jensen et al even showed superior outcomes for bimodal treatment of IMRT and CIRT vs IMRT alone for head and neck ACC, with 5-year LC, PFS, and OS of 60%, 48%, and 77% vs 40%, 27%, and 59%.18
T stage and perineural spread have been assessed as negative prognostic factors regarding LC for head and neck ACC in multiple studies.32,45–48 For lacrimal gland tumors, perineural invasion, positive resection margins, and larger tumors were identified as negative prognostic factors for decreased LC in recent studies.1,7,8,28 Additionally, increased LC after postoperative RT when R1 could be achieved by surgery has been shown.49 In the current analysis, 80% of the identified local recurrences occurred in patients with T3/4 tumors who were operated at R2 (four of five), and 60% showed Pn1 (three of five) without significant relevance, probably due to the low patient numbers. We were able to identify only presence of macroscopic tumor disease before RT start as a negative prognostic factor (P=0.026). Although ACC is the most common tumor in the head and neck area treated by CIRT, many studies have also reported on similarly good outcomes in non-ACC tumors of the salivary glands, ie, adenocarcinomas and mucoepidermoid carcinomas.20,45,46,50,51 Jensen et al identified 3-year LC, PFS, and OS of 82%, 67%, and 73% at follow-up of 26 months for non-ACC malignant salivary gland tumors of the head and neck treated with bimodal RT.20
Toxicity in our cohort was low, with 13% acute grade 3 and 8% chronic grade 3 toxicity, comparable to previous experiences with particle therapy in our center.17,18,20,21,52–55 Overall, CIRT tends to decreased toxicity compared with photon therapy, but prospective studies comparing photons and particles for head and neck tumors are missing to date.56 Although modern RT techniques like IMRT show an improved toxicity profile after RT of tumors in the head and neck region compared with 3-D RT, RT of tumors in the orbit keep challenging the radiation oncologist, because of the anatomic proximity of these tumors to the eye globe, optic system, and the brain.57–59
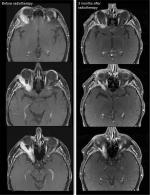
Besides a grade 3 wound-healing disorder, we identified two cases of a temporary brain-barrier disorder of the temporal lobe. In one case, an asymptomatic brain-barrier disorder in the temporal lobe occurred 3 years post-RT and remained stable up to the last follow-up. In another case, increasing T2-hyperintense changes in the temporal lobe were diagnosed at follow-up MRI 6 months after RT and the patient was treated with surgery for progressive dizziness. Temporal lobe necrosis is known as one of the most serious late side effects after RT for skull-base-infiltrating tumors besides osteoradionecrosis and cranial nerve palsy, and occurs predominantly within a few years after treatment.60–64
It is also known that the probability of developing a brain-barrier disorder depends on the prescribed RT dose and the volume, affected by high RT doses.65,66 Schlampp et al identified temporal lobe necrosis in 5% of patients who received heavy ions with RT doses of 68.8 Gy RBE on the skull base vs 50% irradiated with 87.3 Gy RBE.65 In the current analysis, the entire cohort received RT up to doses of 74 Gy, RBE corresponding to a median EQD2 of 80 Gy. Nevertheless, during a median follow-up of 30 months, we did not observe any osteoradionecrosis or severe effect on the cranial nerves. In addition, no case of visual loss after CIRT for lacrimal gland tumors was identified in the current study or in the current literature.
Although superior results for CIRT in the treatment of malignant salivary gland tumors of the head and neck have been shown in recent years, LC for malignant lacrimal gland tumors seems to remain challenging, even after dose-escalated, active raster-scanning CIRT (5-year LC, OS, and DPFS 44%, 68%, and 58% in the current analysis), possibly due to histopathological differences and differing locoregional challenges regarding surrounding organs at risk, when compared with malignant salivary gland tumors of other locations in the head and neck region. Further follow-up will be necessary to make a clearer statement in this regard and to assess late toxicity clearly, as severe chronic side effects, eg, osteoradionecrosis, brain-barrier disorder, visual impairment, and visual loss, can occur years after treatment. Additionally, higher patient numbers are required for a better determination of risk factors, especially for LC, in these tumors. Therefore, multicenter studies are required to recruit a sufficient number of patients.
Conclusion
First experiences regarding the use of active raster-scanning CIRT for malignant lacrimal gland tumors resulted in adequate LC and low toxicity. Nevertheless, in accordance with prior results concerning salivary gland tumors, LC in ACC of the lacrimal gland remains challenging, while RT for a macroscopic tumor disease influences LC negatively.
Abbreviations
ACC, adenoid cystic carcinoma; CIRT, carbon-ion radiotherapy; CR, complete remission; CT, computed tomography; CTV, clinical target volume; CTCAE, Common Toxicity Terminology Criteria for Adverse Events; EQD2, equivalent dose in 2 Gy per fraction; HIT, Heidelberg Ion-Beam Therapy Center; IMRT, intensity-modulated radiotherapy; LC, local control; LET, linear energy transfer; MRI, magnetic resonance imaging; OS, overall survival; PR, partial remission; PTV, planning target volume; RECIST, Response Evaluation Criteria in Solid Tumors; RT, radiotherapy; SD, stable disease.
Acknowledgments
We thank our Head and Neck Cancer Research Group members for their great effort. The study was financed by the Department of Radiation Oncology of the University Hospital Heidelberg. There was no external funding source.
Author contributions
SaA and SeA developed and planned the current retrospective analysis. All authors read and approved the final manuscript. Additionally, all authors made substantial contributions to conception and design, data acquisition, or data analysis and interpretation, drafting the article or critically revising it for important intellectual content, final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of the work are appropriately investigated and resolved.
Disclosure
The authors report no conflicts of interest in this work.
References
Shields JA, Shields CL, Epstein JA, Scartozzi R, Eagle RC. Review: primary epithelial malignancies of the lacrimal gland: the 2003 Ramon L. Font lecture. Ophthalmic Plast Reconstr Surg. 2004;20(1):10–21. | ||
von Holstein SL, Therkildsen MH, Prause JU, Stenman G, Siersma VD, Heegaard S. Lacrimal gland lesions in Denmark between 1974 and 2007. Acta Ophthalmol. 2013;91(4):349–354. | ||
Cl S, Shields JA. Review of lacrimal gland lesions. Trans Pa Acad Ophthalmol Otolaryngol. 1990;42:925–930. | ||
Gao Y, Moonis G, Cunnane ME, Eisenberg RL. Lacrimal gland masses. AJR Am J Roentgenol. 2013;201(3):W371–W381. | ||
Ni C, Kuo PK, Dryja TP. Histopathological classification of 272 primary epithelial tumors of the lacrimal gland. Chin Med J (Engl). 1992;105(6):481–485. | ||
Zeng J, Shi JT, Li B, et al. Epithelial tumors of the lacrimal gland in the Chinese: a clinicopathologic study of 298 patients. Graefes Arch Clin Exp Ophthalmol. 2010;248(9):1345–1349. | ||
von Holstein SL, Rasmussen PK, Heegaard S. Tumors of the lacrimal gland. Semin Diagn Pathol. 2016;33(3):156–163. | ||
Woo KI, Yeom A, Esmaeli B. Management of lacrimal gland carcinoma: lessons from the literature in the past 40 years. Ophthalmic Plast Reconstr Surg. 2016;32(1):1–10. | ||
Wright JE, Rose GE, Garner A. Primary malignant neoplasms of the lacrimal gland. Br J Ophthalmol. 1992;76(7):401–407. | ||
Pommier P, Liebsch NJ, Deschler DG, et al. Proton beam radiation therapy for skull base adenoid cystic carcinoma. Arch Otolaryngol Head Neck Surg. 2006;132(11):1242–1249. | ||
Meldrum ML, Tse DT, Benedetto P. Neoadjuvant intracarotid chemotherapy for treatment of advanced adenocystic carcinoma of the lacrimal gland. Arch Ophthalmol. 1998;116(3):315–321. | ||
Lee DA, Campbell RJ, Waller RR, Ilstrup DM. A clinicopathologic study of primary adenoid cystic carcinoma of the lacrimal gland. Ophthalmology. 1985;92(1):128–134. | ||
Skinner HD, Garden AS, Rosenthal DI, et al. Outcomes of malignant tumors of the lacrimal apparatus: the University of Texas MD anderson cancer center experience. Cancer. 2011;117(12):2801–2810. | ||
Shields JA, Shields CL, Freire JE, Brady LW, Komarnicky L. Plaque radiotherapy for selected orbital malignancies: preliminary observations: the 2002 Montgomery lecture, part 2. Ophthalmic Plast Reconstr Surg. 2003;19(2):91–95. | ||
von Holstein SL. Tumours of the lacrimal gland. epidemiological, clinical and genetic characteristics. Acta Ophthalmol. 2013;91(Pt 1):1–28. | ||
Le Tourneau C, Razak AR, Levy C, et al. Role of chemotherapy and molecularly targeted agents in the treatment of adenoid cystic carcinoma of the lacrimal gland. Br J Ophthalmol. 2011;95(11):1483–1489. | ||
Jensen AD, Nikoghosyan AV, Lossner K, et al. COSMIC: a regimen of intensity modulated radiation therapy plus dose-escalated, raster-scanned carbon ion boost for malignant salivary gland tumors: results of the prospective phase 2 trial. Int J Radiat Oncol Biol Phys. 2015;93(1):37–46. | ||
Jensen AD, Nikoghosyan AV, Poulakis M, et al. Combined intensity-modulated radiotherapy plus raster-scanned carbon ion boost for advanced adenoid cystic carcinoma of the head and neck results in superior locoregional control and overall survival. Cancer. 2015;121(17):3001–3009. | ||
Jensen AD, Poulakis M, Nikoghosyan AV, et al. High-LET radiotherapy for adenoid cystic carcinoma of the head and neck: 15 years’ experience with raster-scanned carbon ion therapy. Radiother Oncol. 2016;118(2):272–280. | ||
Jensen AD, Poulakis M, Vanoni V, et al. Carbon ion therapy (C12) for high-grade malignant salivary gland tumors (MSGTs) of the head and neck: do non-ACCs profit from dose escalation? Radiat Oncol. 2016;11(1):90. | ||
Jensen AD, Nikoghosyan AV, Ecker S, et al. Raster-scanned carbon ion therapy for malignant salivary gland tumors: acute toxicity and initial treatment response. Radiat Oncol. 2011;6(1):149. | ||
Bentzen SM, Constine LS, Deasy JO, et al. Quantitative analyses of normal tissue effects in the clinic (QUANTEC): an introduction to the scientific issues. Int J Radiat Oncol Biol Phys. 2010;76(3):S3–S9. | ||
Karger CP, Peschke P. RBE and related modeling in carbon-ion therapy. Phys. Med. Biol. 2017;63(1):01TR02. | ||
Woo KI, Kim YD, Sa HS, Esmaeli B. Current treatment of lacrimal gland carcinoma. Curr Opin Ophthalmol. 2016;27(5):449–456. | ||
Esmaeli B, Golio D, Kies M, Demonte F. Surgical management of locally advanced adenoid cystic carcinoma of the lacrimal gland. Ophthalmic Plast Reconstr Surg. 2006;22(5):366–370. | ||
Polito E, Leccisotti A. Epithelial malignancies of the lacrimal gland: survival rates after extensive and conservative therapy. Ann Ophthalmol. 1993;25(11):422–426. | ||
Douglas JG, Laramore GE, Austin-Seymour M, Koh W, Stelzer K, Griffin TW. Treatment of locally advanced adenoid cystic carcinoma of the head and neck with neutron radiotherapy. Int J Radiat Oncol Biol Phys. 2000;46(3):551–557. | ||
Andreoli MT, Aakalu V, Setabutr P. Epidemiological trends in malignant lacrimal gland tumors. Otolaryngol Head Neck Surg. 2015;152(2):279–283. | ||
Roshan V, Pathy S, Mallick S, Chander S, Sen S, Chawla B. Adjuvant radiotherapy with three-dimensional conformal radiotherapy of lacrimal gland adenoid cystic carcinoma. J Clin Diagn Res. 2015;9(10):XC05–XC07. | ||
Tao R, Ma D, Takiar V, et al. Orbital carcinomas treated with adjuvant intensity-modulated radiation therapy. Head Neck. 2016;38(S1):E580–E587. | ||
Gensheimer MF, Rainey D, Douglas JG, et al. Neutron radiotherapy for adenoid cystic carcinoma of the lacrimal gland. Ophthalmic Plast Reconstr Surg. 2013;29(4):256–260. | ||
Mizoguchi N, Tsuji H, Toyama S, et al. Carbon-ion radiotherapy for locally advanced primary or postoperative recurrent epithelial carcinoma of the lacrimal gland. Radiother Oncol. 2015;114(3):373–377. | ||
Mizoe JE, Tsujii H, Kamada T, et al. Dose escalation study of carbon ion radiotherapy for locally advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2004;60(2):358–364. | ||
Tsuji H, Ishikawa H, Yanagi T, et al. Carbon-ion radiotherapy for locally advanced or unfavorably located choroidal melanoma: a phase I/II dose-escalation study. Int J Radiat Oncol Biol Phys. 2007;67(3):857–862. | ||
Toyama S, Tsuji H, Mizoguchi N, et al. Long-term results of carbon ion radiation therapy for locally advanced or unfavorably located choroidal melanoma: usefulness of CT-based 2-port orthogonal therapy for reducing the incidence of neovascular glaucoma. Int J Radiat Oncol Biol Phys. 2013;86(2):270–276. | ||
Wang Z, Nabhan M, Schild SE, et al. Charged particle radiation therapy for uveal melanoma: a systematic review and meta-analysis. Int J Radiat Oncol Biol Phys. 2013;86(1):18–26. | ||
Fuss M, Loredo LN, Blacharski PA, Grove RI, Slater JD. Proton radiation therapy for medium and large choroidal melanoma: preservation of the eye and its functionality. Int J Radiat Oncol Biol Phys. 2001;49(4):1053–1059. | ||
Kamada T, Tsujii H, Blakely EA, et al. Carbon ion radiotherapy in Japan: an assessment of 20 years of clinical experience. Lancet Oncol. 2015;16(2):e93–e100. | ||
Tsujii H, Kamada T. A review of update clinical results of carbon ion radiotherapy. Jpn J Clin Oncol. 2012;42(8):670–685. | ||
Schulz-Ertner D, Nikoghosyan A, Thilmann C, et al. Results of carbon ion radiotherapy in 152 patients. Int J Radiat Oncol Biol Phys. 2004;58(2):631–640. | ||
Mizoe JE, Hasegawa A, Jingu K, et al. Results of carbon ion radiotherapy for head and neck cancer. Radiother Oncol. 2012;103(1):32–37. | ||
Vikram B, Strong EW, Shah JP, Spiro RH. Radiation therapy in adenoid-cystic carcinoma. Int J Radiat Oncol Biol Phys. 1984;10(2):221–223. | ||
Sur RK, Donde B, Levin V, et al. Adenoid cystic carcinoma of the salivary glands: a review of 10 years. Laryngoscope. 1997;107(9):1276–1280. | ||
Mendenhall WM, Morris CG, Amdur RJ, Werning JW, Hinerman RW, Villaret DB. Radiotherapy alone or combined with surgery for adenoid cystic carcinoma of the head and neck. Head Neck. 2004;26(2):154–162. | ||
Shirai K, Saitoh JI, Musha A, et al. Prospective observational study of carbon-ion radiotherapy for non-squamous cell carcinoma of the head and neck. Cancer Sci. 2017;108(10):2039–2044. | ||
Koto M, Hasegawa A, Takagi R, et al. Definitive carbon-ion radiotherapy for locally advanced parotid gland carcinomas. Head Neck. 2017;39(4):724–729. | ||
Holtzman A, Morris CG, Amdur RJ, Dziegielewski PT, Boyce B, Mendenhall WM. Outcomes after primary or adjuvant radiotherapy for salivary gland carcinoma. Acta Oncol. 2017;56(3):484–489. | ||
Orlandi E, Iacovelli NA, Bonora M, Cavallo A, Fossati P, Gland S. Salivary gland. Photon beam and particle radiotherapy: present and future. Oral Oncol. 2016;60:146–156. | ||
Noh JM, Lee E, Ahn YC, et al. Clinical significance of post-surgical residual tumor burden and radiation therapy in treating patients with lacrimal adenoid cystic carcinoma. Oncotarget. 2016;7(37):60639–60646. | ||
Koto M, Hasegawa A, Takagi R, et al. Feasibility of carbon ion radiotherapy for locally advanced sinonasal adenocarcinoma. Radiother Oncol. 2014;113(1):60–65. | ||
Shirai K, Koto M, Demizu Y, et al. Multi-institutional retrospective study of mucoepidermoid carcinoma treated with carbon-ion radiotherapy. Cancer Sci. 2017;108(7):1447–1451. | ||
Mohr A, Chaudhri N, Hassel JC, et al. Raster-scanned intensity-controlled carbon ion therapy for mucosal melanoma of the paranasal sinus. Head Neck. 2016;38(S1):E1445–E1451. | ||
Jensen AD, Uhl M, Chaudhri N, Herfarth KK, Debus J, Roeder F. Carbon ion irradiation in the treatment of grossly incomplete or unresectable malignant peripheral nerve sheaths tumors: acute toxicity and preliminary outcome. Radiat Oncol. 2015;10(1):109. | ||
Jensen AD, Nikoghosyan AV, Ecker S, Ellerbrock M, Debus J, Münter MW. Carbon ion therapy for advanced sinonasal malignancies: feasibility and acute toxicity. Radiat Oncol. 2011;6(1):30. | ||
Uhl M, Mattke M, Welzel T, et al. High control rate in patients with chondrosarcoma of the skull base after carbon ion therapy: first report of long-term results. Cancer. 2014;120(10):1579–1585. | ||
Ramaekers BL, Pijls-Johannesma M, Joore MA, et al. Systematic review and meta-analysis of radiotherapy in various head and neck cancers: comparing photons, carbon-ions and protons. Cancer Treat Rev. 2011;37(3):185–201. | ||
Bhide SA, Newbold KL, Harrington KJ, Nutting CM. Clinical evaluation of intensity-modulated radiotherapy for head and neck cancers. Br J Radiol. 2012;85(1013):487–494. | ||
Thariat J, Bolle S, Demizu Y, et al. New techniques in radiation therapy for head and neck cancer: IMRT, CyberKnife, protons, and carbon ions. Improved effectiveness and safety? Impact on survival? Anticancer Drugs. 2011;22(7):596–606. | ||
Pauloski BR, Rademaker AW, Logemann JA, Discekici-Harris M, Mittal BB. Comparison of swallowing function after intensity-modulated radiation therapy and conventional radiotherapy for head and neck cancer. Head Neck. 2015;37(11):1575–1582. | ||
Chew NK, Sim BF, Tan CT, Goh KJ, Ramli N, Umapathi P. Delayed post-irradiation bulbar palsy in nasopharyngeal carcinoma. Neurology. 2001;57(3):529–531. | ||
Lam TC, Wong FC, Leung TW, Ng SH, Tung SY. Clinical outcomes of 174 nasopharyngeal carcinoma patients with radiation-induced temporal lobe necrosis. Int J Radiat Oncol Biol Phys. 2012;82(1):e57–e65. | ||
Hsiao KY, Yeh SA, Chang CC, Tsai PC, Wu JM, Gau JS. Cognitive function before and after intensity-modulated radiation therapy in patients with nasopharyngeal carcinoma: a prospective study. Int J Radiat Oncol Biol Phys. 2010;77(3):722–726. | ||
Teo PML, Leung SF, Chan AT, et al. Final report of a randomized trial on altered-fractionated radiotherapy in nasopharyngeal carcinoma prematurely terminated by significant increase in neurologic complications. Int J Radiat Oncol Biol Phys. 2000;48(5):1311–1322. | ||
Yeh SA, Ho JT, Lui CC, Huang YJ, Hsiung CY, Huang EY. Treatment outcomes and prognostic factors in patients with supratentorial low-grade gliomas. Br J Radiol. 2005;78(927):230–235. | ||
Schlampp I, Karger CP, Jäkel O, et al. Temporal lobe reactions after radiotherapy with carbon ions: incidence and estimation of the relative biological effectiveness by the local effect model. Int J Radiat Oncol Biol Phys. 2011;80(3):815–823. | ||
Pehlivan B, Ares C, Lomax AJ, et al. Temporal lobe toxicity analysis after proton radiation therapy for skull base tumors. Int J Radiat Oncol Biol Phys. 2012;83(5):1432–1440. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.