Back to Journals » International Journal of Nanomedicine » Volume 15
Cancer-Derived Exosomes: Their Role in Cancer Biology and Biomarker Development
Received 16 July 2020
Accepted for publication 29 September 2020
Published 19 October 2020 Volume 2020:15 Pages 8019—8036
DOI https://doi.org/10.2147/IJN.S272378
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Linlin Sun
Victor C Kok,1,2 Cheng-Chia Yu3,4
1Division of Medical Oncology, Kuang Tien General Hospital Cancer Center, Taichung, Taiwan; 2Department of Bioinformatics and Medical Engineering, Asia University, Taichung, Taiwan; 3Institute of Oral Sciences, Chung Shan Medical University, Taichung, Taiwan; 4School of Dentistry, Chung Shan Medical University, Taichung, Taiwan
Correspondence: Victor C Kok
Division of Medical Oncology, Kuang Tien General Hospital Cancer Center, 117 Shatien Road, Shalu, Taichung 43303, Taiwan
Tel +886 4 2662 5111 Ext 2263
Fax +886 4 2665 5050
Email [email protected]
Abstract: Exosomes are a subset of tiny extracellular vesicles manufactured by all cells and are present in all body fluids. They are produced actively in tumor cells, which are released and utilized to facilitate tumor growth. Their characteristics enable them to assist major cancer hallmarks, leveraged by cancer cells in fostering cancer growth and spread while implementing ways to escape elimination from the host environment. This review updates on the latest progress on the roles of cancer-derived exosomes, of 30– 100 nm in size, in deregulating paracrine trafficking in the tumor microenvironment and circulation. Thus, exosomes are being exploited in diagnostic biomarker development, with its potential in clinical applications as therapeutic targets utilized in exosome‐based nanoparticle drug delivery strategies for cancer therapy. Ongoing studies were retrieved from PubMed® and Scopus database and ClinicalTrials.gov registry for review, highlighting how cancer cells from entirely different cell lines rely on genetic information carried by their exosomes for homotypic and heterotypic intercellular communications in the microenvironment to favor proliferation and invasion, while establishing a pre-metastatic niche in welcoming cancer cells’ arrival. We will elaborate on the trafficking of tumor-derived exosomes in fostering cancer proliferation, invasion, and metastasis in hematopoietic (leukemia and myeloma), epithelial (breast cancer), and mesenchymal (soft tissue sarcoma and osteosarcoma) cancers. Cancer-derived exosomal trafficking is observed in several types of liquid or solid tumors, confirming their role as cancer hallmark enabler. Their enriched genetic signals arising from their characteristic DNA, RNA, microRNA, and lncRNA, along with specific gene expression profiles, protein, or lipid composition carried by the exosomal cargo shed into blood, saliva, urine, ascites, and cervicovaginal lavage, are being studied as a diagnostic, prognostic, or predictive cancer biomarker. We reveal the latest research efforts in exploiting the use of nanoparticles to improve the overall cancer diagnostic capability in the clinic.
Keywords: tumor-released exosomes, carcinoma-associated fibroblasts, exosome cargo, exosome-induced chemoresistance, hallmarks of cancer, tumor-stromal communications
Introduction
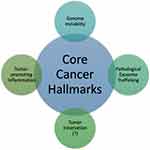
There exist six well-researched cancer hallmarks, and they are as follows: (1) sustaining proliferative signaling, (2) evading growth suppressors, (3) enabling replicative immortality, (4) activating invasion and metastasis, (5) inducing angiogenesis, and (6) resisting cell death. However, two emerging hallmarks were identified and added by Hanahan and Weinberg:1,2 (7) deregulating cellular metabolism and (8) avoiding immune destruction. Cancer cells typically acquire these core capabilities through sustaining selective pressures and adopting alterations in specific and ubiquitous cellular function, defined as “enabling characteristics,” to permit cancer hallmark capabilities. Enabling characteristics, which foster cancer growth and spread, are typically adopted by cancer cells to survive, proliferate, and escape elimination by the host environment. In the next-generation review by Hanahan and Weinberg on cancer hallmarks’ molecular mechanisms, they proposed two enabling characteristics that are present in cancer cells across the board, which are genome instability/mutation and tumor-promoting inflammation.1
In this review, we propose pathological trafficking of genetic materials carried by cancer-derived exosomes (CDE) cargo as a new enabling characteristic that fits the proposed criteria (Table 1). This CDE cargo phenomenon has been demonstrated to be present in virtually all kinds of cancer cell types, fostering the core hallmarks of cancer by effective homotypic or heterotypic intercellular (tumor-to-microenvironment) communications, which facilitate cancer invasion and metastasis as well as evasion from immune destruction. The second part of the review is dedicated to the updated status of CDE being evaluated as a valid diagnostic cancer biomarker in human studies.
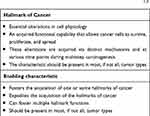 |
Table 1 Criteria for Classification as a Hallmark of Cancer or an Enabling Characteristic, According to Hanahan and Weinberg1,2 |
This study aims to present a comprehensive overview of the roles of CDE in cancer progression and development, assimilated from the current literature and translational cancer research to stimulate cancer biologists, scientists, and oncologists who are interested in the involvement of CDE in cancer pathogenesis, cancer microenvironment, molecular mechanisms on cancer progression and who plan to apply the knowledge in developing more effective diagnostic strategies for various types of cancer. Ongoing research studies evaluating CDE as a cancer biomarker registered in the ClinicalTrials.gov were selected to enrich the discussion on the clinical application of CDE as a biological cancer marker and to indicate future opportunities for cross-disciplinary collaborations.
Literature Search
To ensure a comprehensive and unbiased literature review, we performed both electronic and manual literature search in the PubMed® and Scopus database to retrieve relevant original articles. We leveraged the use of PubMed Advanced Search Builder, Medical Subject Headings (MeSH), and Boolean logic to add terms or combine search terms using connector words, such as AND, OR, or NOT, as well as truncate terms. We used a controlled vocabulary to produce highly relevant search results. The search terms included exosome, exosomal cargo, cancer-derived or tumor-derived, cancer biology, proteasome, and cancer biology. Subsequently, we surveyed the ClinicalTrials.gov registry for clinical studies conducted in the United States and around the world.
Cancer-Derived Exosomes in Cancer Biology
The tumor microenvironment (TME) surrounding cancer cells is identified to be comprised of cancer-associated fibroblasts, blood vessels, nerve fibers, immune cells, other stromal cells, and extracellular vesicles containing various kinds of genetic signals. Considering all are functional in anticancerous immunosuppressive cells, the TME is known to create a milieu that prevents the free spread of the malignant cells.3 The cancer cells, however, communicate with the neighboring stromal and immune cells, promoting immune evasion, and could also activate angiogenesis, tumor innervation, and epithelial-mesenchymal transition (EMT), in order to facilitate neoplastic growth. Recently, a phenomenon involving tumor-infiltrating innervation in the TME has also been proposed as a prerequisite for cancer cells of many types such as in prostate, gastric, pancreatic, and rectal cancers.4–6 Tumors are capable of recruiting nerves via the release of neurotrophic factors and axonal guidance molecules, and, with the contribution of CDEs, induction of axonogenesis is initiated, whereby the communication between the tumor and potentially innervating nerves work in concert to promote tumor innervation.4 Thus, it has been proposed that tumor innervation with neurite outgrowth (axonogenesis), just like angiogenesis, might be considered a new emerging hallmark of cancer.7–13 Although research has demonstrated that angiogenesis is frequently associated with axonogenesis, more studies are eagerly required to elucidate the roles of tumor-infiltrating innervation in tumor initiation, growth, and spread for this phenomenon to be widely accepted as a hallmark of cancer. Recent studies were able to confirm that the neurite recruitment/outgrowth and tumor innervation were promoted by the release of exosomes in the head and neck squamous cell carcinomas model5 and human papillomavirus-positive uterine cervical cancer cell lines.6
The name “tumor-derived exosomes” was coined in 1981, and this phenomenon has received extensive research attention over the past decade.14 Exosomes are extracellular, membranous, cup-shaped microvesicles 30 to 100 nm in size, which are produced by most types of cells.15–17 They originate from intracellular multivesicular bodies and are released by exocytosis into the extracellular microenvironment.18 Exosome typically consists of a variety of genetic messengers such as DNA, mRNA, microRNA, cytosolic proteins, and lipids.19–23 Exosomal markers such as tetraspanin proteins CD63, CD9, and CD81 allow sorting, selective recruitment, capturing, or profiling of CDEs.24,25 Once the recipient cells internalize tumor-derived exosomes, the ensuing biological response is determined explicitly by the dedicated trafficking routes, the exosomal internalization pathway, and the complex surface molecules on the membrane of both the extracellular vesicle and the recipient cell. With the advent of theranostic nanotechnologies such as differential ultracentrifugation, nanofluidic technology, and the exosome total isolation chip (ExoTIC), a size-based extracellular vesicle isolation apparatus, researchers nowadays are now able to capture nano-sized CDE for further analyses.26–28 The latest biosensing technologies, such as afterglow sensors with aptamer-based signal amplification, improve the limit of detection (LOD) that is nearly two orders of magnitude lower than that of fluorescence methods.29 With the advent of these sensitive biosensors, the LOD can practically be improved to 102 exosomes per milliliter.
These exosomes, particularly those that are tumor-derived, act as signal transducers or messengers in the cell-cell communication.5,30–33 The recipient cells respond to the exosomal contents (such as microRNA) by changing their phenotypes. microRNAs are considered an evolutionarily conserved family of molecules that bind to complementary sequences in the 3ʹ-untranslated region (3ʹUTR) of their target mRNAs, post-transcriptionally repressing gene expression.34 It has been demonstrated that in high-grade bladder cancer cell line, TCC-SUP, for example, exosomes promoted angiogenesis and migration of both cancer and endothelial cells.35 In another study in prostate cancer, the malicious CDEs induced differentiation of the stromal mesenchymal stem cells toward alpha-smooth muscle actin-positive myofibroblasts, which secreted high levels of proangiogenic VEGF-A, pro-invasive HGF, MMP-1, MMP−3, and MMP−13.36
The role of CDEs as characteristic enablers of cancer hallmarks to facilitate organ-specific metastasis has been demonstrated by the proof-of-principle study conducted by Hoshino et al,37 In their peripheral blood study of mouse and human cell lines, they claimed that during the metastatic cascade, organ-specific metastasis took place not by a random process but by somewhat predictable and trackable events. This happened through distinct integrin expression patterns contained in the CDEs, a phenomenon that now elucidates the mechanism of specific cancer organotropism adequately. The exosomal integrin αvβ5 was associated with hepatic metastasis, while exosomal integrins α6β4 and α6β1 were linked to lung metastasis.37
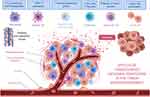
In the following sections, we will use three different cancer types: hematopoietic, epithelial, and sarcomatous malignancies (leukemia/myeloma, breast cancer, soft tissue sarcoma, and osteosarcoma) to prove, using compelling evidence, that cancer cells across the board leverage the pathological trafficking of exosomes to promote neoplastic growth, facilitate cancer spread through tumor-stromal interaction, and evade destruction by the host (Figure 1).
Exosomal Trafficking in Leukemia Pathogenesis
Although leukemia can reach every part of the host body through the ever-reaching blood vessels, recent research has reported that leukemic cells also employ paracrine exosome trafficking to achieve leukemogenesis, maintain leukemic persistence by shaping the leukemic niche and its progression, suppress hematopoiesis, modify anti-leukemic immunity, and evade destruction by chemotherapy. Table 2 illustrates these aspects taking acute myeloid leukemia (AML) as an example.
 |
Table 2 Paracrine Exosome Trafficking Employed by Acute Myeloid Leukemia (AML) as an Example and Its Specific Functional Outcomes |
Patients with chronic lymphocytic leukemia (CLL) have been identified to have decreased T-cell immunity. A recent study showed that CLL induced myeloid-derived suppressor cells (MDSCs), which, in turn, suppressed T-cell activation and induced suppressive regulatory T cells (Treg) through exosomal miR-155 transfer.38,39 This exosome-mediated transfer of microRNAs to monocytes could significantly contribute to CLL-related immune escape via PD-L1 expression. Colleagues from the University of Liverpool verified that CLL-derived exosomes encapsulate small RNAs, and the encapsulated microRNA miR-202-3p enhanced the expression of a Hedgehog signaling intermediate.40 An enriching body of evidence shows that the TME created by the bone marrow significantly favors the survival, growth, and proliferation of leukemic cells. For example, CLL leukemic cells can establish their favorable leukemic niche in the TME. Paggetti et al have discovered that CLL-derived exosomes could affect bone marrow stromal cells in adopting a cancer-associated fibroblast phenotype, which would contribute to a tumor-supportive microenvironment.41 In the chronic myeloid leukemia (CML) model, CML-derived exosomal paracrine trafficking stimulated bone marrow stromal cells to produce interleukin (IL)-8.42,43 Further, another study demonstrated that exosomes released from CML cells affect the endothelium directly to modify the neovascularization process.44
Exosomal Trafficking in Multiple Myeloma (MM) Pathogenesis
In a murine MM model, the myeloma exosomes were identified to have a proangiogenic function to enhance the viability of bone marrow endothelial cells; besides, an in vivo experiment demonstrated that these exosomes increased the presence of bone marrow MDSCs and changed their subsets to a more tumorigenic profile.55 MM-derived exosomes could modify the bone marrow microenvironment to facilitate myeloma progression. Conversely, the bone marrow stromal cells could, reciprocally, also release certain exosomes to be taken up by MDSCs through the STAT3 and STAT1 pathways, which leads to increased immunosuppression, thereby inducing MM expansion.56 Initially, investigators from the Dana-Farber Cancer Institute demonstrated that there were significant differences in microRNA profiling between normal and bone marrow mesenchymal stromal cell-derived exosomes in MM.57 A recent study in patients with MM using small RNA sequencing of circulating exosomes from ten patient samples confirms that microRNAs are the most predominant small RNAs present in MM exosomes.58 Meanwhile, investigators from the Karolinska Institute have examined the human bone marrow stromal cell line L88 and verified that caspase-3 is activated by the stroma cell–released exosomes, which can cleave the anti-apoptotic protein Bcl-xL, localized on the outer exosomal membrane. Through the cleavage of Bcl-xL, these exosomes could then be internalized using plasma cell myeloma, which led to their increased proliferation.59
Finally, in another study performed at Tokyo Medical University, researchers established a hypoxia-resistant MM cell model to mimic the in vivo hypoxic microenvironment induced by the rapid proliferation of MM in the bone marrow. Their experiment showed that under normoxic or acute hypoxic settings, the hypoxia-resistant MM cells produced more exosomes than the parental cells, and the major functional protein in the exosomal cargo was identified to be miR-135b. This protein directly suppressed factor-inhibiting hypoxia-inducible factor 1 (FIH-1) in bone marrow endothelial cells.60 Hence, further studies are needed to test if miR-135b could be used as a target for therapeutically avert angiogenesis in MM.
Exosomal Trafficking in Breast Cancer Pathogenesis
A study using plasma exosomal microRNAs as a diagnostic biomarker in breast cancer patients demonstrated that these molecules have outstanding power to distinguish breast cancer patients from normal counterparts. Zhai et al used a nucleic acid-functionalized Au nanoflare probe, which are known to have the ability to directly enter plasma exosomes and generate quantitative fluorescent signals for successful in situ detection of exosome-located microRNA-1246. At its best cutoff point, the in situ detection of the exosomal miRNA-1246 in the peripheral blood was able to distinguish 46 breast cancer patients from 28 healthy controls with 100% sensitivity and 93% specificity.61 Another clinical study on the exosomal microRNA signatures of 20 healthy women and 435 breast cancer patients discovered that 10 miRNAs in the entire breast cancer patient cohort, 13 in the HER2-positive subgroup (211 patients), and 17 in the triple-negative subgroup (224 patients) were significantly deregulated in comparison to those in healthy women, indicating different underlying aspects of cancer biology in different breast cancer types.62 These different exosomal microRNA signatures are associated with the clinicopathological features of each subgroup. In addition, exosomes that are released by breast cancer cells could modify TME through direct suppression of T-cell proliferation and inhibition of NK cell cytotoxicity, thus dampening the anticancer immune response in pre-metastatic organs.63
Another hallmark of cancer is the transfer of chemoresistant or hormone-resistant propensity from breast cancer stem cells to the daughter cells, explored in the study of Santos et al, who demonstrated that miR-155 was upregulated in breast cancer stem cells and chemoresistant cells and was involved in the EMT. An enrichment in miR-155 was noted in exosomes isolated from stem-like breast cancer stem and chemoresistant cells. Moreover, the experiments demonstrated the capability of the horizontal transfer of miR-155 from the chemoresistant cells’ exosomal cargo to the recipient sensitive cells.64 This study supports the presence of exosome-mediated chemoresistance and EMT in refractory cancer. Estrogen receptor (ER)-positive cancers are found to transition from an endocrine sensitive/dormant state to a resistant one, acquiring host mitochondrial DNA, which promoted oxidative phosphorylation (OXPHOS) and signaled the transition from metabolic quiescence toward hormonal therapy resistance.65 Further, functional studies have identified cancer-associated fibroblast-derived extracellular vesicles containing whole genomic mitochondrial DNA in patients and xenograft models.
In a breast cancer cell line, recipient cells treated with exosomes from stemness-related breast cancer CXCR4-positive cells showed an increase in the same oncogenic abilities.66 This experiment has also demonstrated that inoculating exosomes derived from CXCR4-positive cells into immunocompromised mice can stimulate primary tumor proliferation and metastatic potential. The same investigators also discovered a “stemness and metastatic” signature in the exosomes of patients with worse prognoses after comparing exosomal nucleic acid contents.66
Exosomal Trafficking in the Pathogenesis of Soft Tissue Sarcoma
In 2013, for the first time, a study has showed that exosome-mediated pathogenesis, similar to epithelial carcinoma and hematopoietic malignancy, was also present in Ewing sarcoma.67 Microarray analysis of exosomes shed by the Ewing sarcoma cell line revealed that their exosomal content shared a transcriptional signature potentially involved in the modification of the surrounding microenvironment via G-protein-coupled signaling, neurotransmitter signaling, and stemness.67
A recently published study used both patient plasma samples and cell lines to demonstrate that liposarcoma cells secreted miR-25-3p and miR-92a-3p in exosomes. Subsequently, it stimulated the secretion of the proinflammatory cytokine, interleukin (IL)-6, in tumor-associated macrophages through a TLR7/8-dependent mechanism, which can ultimately cause liposarcoma progression.68 In another study using patient-derived Ewing sarcoma cells, miR-34a, an inhibitor of Notch-NFκB signaling, was enriched and secreted through exosomes shed by CD99-silenced (by small interfering RNA) cells.69 CD99 has been identified as a cell surface molecule involved in cell differentiation, migration, and death. In Ewing sarcoma cells, it is pro-oncogenic due to its effect on the prevention of NFκB-mediated neural differentiation and is continuously present at high levels. The horizontal transfer of miR-34a through exosomes to recipient cells enhanced neural differentiation in Ewing sarcoma.69
Moreover, another study demonstrated that the membrane-type 1 matrix metalloproteinase (MT1-MMP, MMP14) was released by exosomes of cultured human fibrosarcoma (HT-1080) cells.70 MT1-MMP is a crucial metalloproteinase that facilitates tumor invasion by remodeling the extracellular matrix. Pathological sarcomatous exosomal trafficking carrying MT1-MMP could be detrimental to the host by providing a favorable microenvironment for sarcoma.
Exosomal Trafficking in Osteosarcoma Pathogenesis
Osteosarcomas are known as malignant mesenchymal-derived bone tumors and the most common bone cancers in children and adolescents. Emerging evidence has also shed light on the exosomal trafficking employed by osteosarcoma cells to shape its supporting TME and facilitate growth, as well as hematogenous spread. Among the specific exosomal contents, miR-148a and miR-21-5p are known to help shape the TME.71 The microRNA, miR-21, is a common oncological molecule taking part in the pathogenesis of various types of malignancies.71–80 Take esophageal cancer as an example; it has been demonstrated in a human esophageal carcinomas cell line co-cultivation experiment that miR-21 in the CDE shuttled from donor cells significantly promoted the migration and invasion capability of recipient cells by activating c-Jun N-terminal kinase signaling pathway.74 A recent multi-omics study observed that the progression from localized to metastatic osteosarcoma was accompanied by an elevation of the levels of urokinase plasminogen activator (uPA) and uPA receptor in the metastatic cells’ exosomal cargo.81 The impact of abundant miR-25-3p in the liposarcoma-derived exosomes on the surrounding microenvironment was similar to what was observed in osteosarcoma cases.82 Jerez et al conducted a gene ontology analysis of predicted targets for the miRNAs present in osteosarcoma-derived extracellular vesicles. Their bioinformatics analysis indicated that miRNAs derived from osteosarcoma cell lines might regulate metastatic potential by inhibiting a network of genes involved in apoptosis and cell adhesion.83 Further research is needed to provide more evidence on the details and importance of exosomal trafficking in osteosarcoma pathogenesis and to determine it as a core hallmark of cancer.
Other Molecules (Proteins, Enzymes, Receptors, Ligands, or Signaling Molecules) That Can Exert Neoplastic Functional Activities Carried by Exosomes
Certainly, exosomal cargoes are not limited by miRNAs only but also by a lot of other candidate molecules such as proteins, lipids, enzymes, signaling molecules, which can exert their functional activities far from the exosome-producing cells.84 Table 3 demonstrates that CDE cargoes could contain basically any purpose-built loaded nano-molecules for its ultimately release from the parental cells. It is evident that horizontal or paracrine transfer of these molecules, when received by the specific recipient cells in the TME or any distant metastatic niches, could facilitate the progression, invasion, and metastatic spread of cancer cells. Some of these exosomal molecules have the potential to serve as valid biomarkers, and, thus, there should be worthwhile testing for cancer detection and/or diagnosis.
In summary, it is evident that cancer cells from entirely different lineages, such as those from leukemia to osteosarcoma, rely on their exosomes to carry the genetic information for homotypic and heterotypic intercellular communications in the TME (Figure 2). This communication creates a favorable environment for cell proliferation and invasion and further establishes a pre-metastatic niche that is readily welcoming for the arrival of cancer cells when they carry the correct form of exosomal integrins. Therefore, these CDEs and their pathological trafficking capabilities should be considered as an emerging enabling characteristic for the well-established hallmarks of cancer.
Cancer-Derived Exosomes in Biomarker Development
The first half of the review has exemplified the role of exosome in specific cancer types. Shifting gear to discuss the potential clinical application, this review will examine and discuss how we can develop cancer biomarkers based on characteristics such as exosomal cargo contents, detection methods, and localization of these exosomes in peripheral blood, pleural effusions, ascites, or urine. Further clinical studies are eagerly awaited to establish and validate the usefulness of specific CDE biomarkers in the different clinical setting during the cancer management. For example, in breast cancer, we can leverage the characteristics of exosomal microRNA signatures and exosomal nucleic acid contents in assisting in breast cancer subtyping or discovering a stemness and metastatic signature, as mentioned previously. Similarly, in the case of soft tissue sarcoma, in recognition of the role of exosomal cargo transcriptional signatures or microRNA profiling including miR-25-3p, miR-92a-3p, miR-34a, MT1-MMP, and MMP-14 playing in the promotion of sarcoma progression, remodeling extra-sarcomatous matrix to facilitate tumor invasion, and establishing a favorable TME for sarcoma growth, we can develop a practical analysis to investigate these soft tissue sarcoma-derived exosomes in assisting diagnosis or monitoring of disease along with the treatment milestones for a patient.
Although the technology for developing exosome-encapsulated therapeutics as targeted drug delivery is still in infancy, with the help of improving detection methods, rapid application of the analytic tests for specific exosomal cargoes for diagnostic purposes has become feasible, facilitating exosomal biomarker development. Several characteristics of CDEs, previously discussed in the cancer biology section, such as analyzable cancer-specific and stage-specific genetic contents in the cargo of CDEs, allow us to capture, profile and quantify using the current nanoanalytical technology. The phenomenon of pathological exosomal trafficking during cancer development and progression can be utilized in cancer diagnosis, prognostication, and treatment strategies. Studies have demonstrated that CDEs containing enriched genetic signals involved in cancer initiation and progression are shed by cancer cells into the blood, saliva, urine, ascites, and even cervicovaginal lavage. In clinical oncology, a cancer biomarker can be used for a diagnostic purpose, for example, in differentiating cancer from the non-cancer conditions. It can also be used for disease monitoring during antineoplastic therapy or follow-ups, for prognosticating a patient’s survival, and for predicting a tumor response after anticancer treatment.
Current sophisticated purification techniques offer an opportunity to utilize isolated exosomal cargoes to assist in differentiating the type of cancer and high tumor grade from low-grade cancer.61,75,100–102 In a recent study, using a urinary exosome 3-gene signature obtained from the ExoDx Prostate IntelliScore urine exosome assay, the investigators can differentiate high-grade (Gleason’s score > 7) vs low-grade prostate cancer and benign prostatic hyperplasia.100 This noninvasive urine testing implies that many unnecessary invasive transrectal biopsies could be avoided. As was aforementioned in the review, in patients with multiple myeloma, serum exosomal miRNAs could add to the risk stratification in identifying newly diagnosed multiple myeloma with particularly poor outcomes.58 Table 3 presents the select representations of exosomal cargo other than miRNAs, namely, proteins, lipids, signaling molecules, DNA, mitochondrial DNAs, circRNAs, lncRNAs, integrin, and enzyme, which potentially serve as a biomarker in various cancer types. Laboratory analytic methods to measure these contents in the research included triple SILAC quantitative proteomic analysis, mass spectrometry (MS)-based proteomic assays, lectin blotting, NP-HPLC analysis, ultrahigh-resolution Fourier transform MS, shotgun and targeted molecular quantitative lipidomic assays, capillary liquid chromatography-MS, MALDI MS, electrospray ionization MS, dsDNA-specific shrimp DNase and atomic force microscopy, RNA-seq analysis, transmission electron microscopy, nanoparticle tracking analysis, etc., depending on the study design (Table 3).
One of the advantages of investigating the CDEs as either a diagnostic, prognostic, or predictive biomarker is that physicians can obtain a specimen for CDE testing from a patient via relatively noninvasive methods. The shed CDEs into body secretion or discharges such as saliva, ascites, and cervicovaginal lavage can now be noninvasively or microinvasively assessed. In the past 5 years, clinical studies on exploiting CDEs as a clinical biomarker reported some promising results in various types of cancer (Table 4). The CDE cargo tested include lncRNAs, microRNAs, exosomal phosphatidylserine, urinary 3-gene expression profile, shuttle RNA pattern, RNA cargo, exosomal cancer stem cell-like marker CD133, exosomal EpCAM protein, and exosomal glutathione S-transferase P1 (Table 4).
 |
Table 4 Published Clinical Studies on Exploiting Exosomes as Diagnostic, Prognostic, or Predictive Biomarkers in Various Types of Cancer |
As of this writing, there have been several dozens of prospective observational studies being carried out to investigate the role of specific exosomal cargo as a cancer biomarker in various cancers and their diagnostic performance in a particular clinical setting (Table 5).
 | 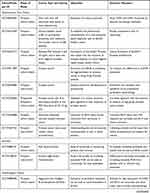 |  |
Table 5 Ongoing Human Studies Investigating Exosomes as a Biomarker in Various Types of Cancer, as Registered in ClinicalTrials.gov |
Rapidly evolving nanotechnologies provide an opportunity to exploit and engineer exosomes for therapeutic purposes, which is gradually becoming a new class of cell-free nanomedicine. Therapeutic blockade of the exosome biogenesis to halt cancer progression at specific stages of the disease could be enticing in the development of cancer therapeutics.80,114,115 The potential application of responsive exosome nano‐bioconjugates for cancer therapy has also been confirmed in a recent study; the nano‐bioconjugates can actively target tumors through the specific recognition on the surface of tumor cell and abolished signaling and improved phagocytosis of macrophages.116 There are growing interests in investigating engineered exosomes as potential therapeutic vehicles or an active drug delivery system.117–123 Making use of the exosomal organotropic characteristics, exosomes loaded with therapeutic compounds could be employed to target a recipient cell to carry out gene therapy selectively.
The following examples shall illustrate how the application of exosomal engineering technology may enhance cancer therapeutics. Targeting the immune cells in the TME as an adjunct of anticancer treatment has been becoming a hot research area. In the application of nanomedicine, various forms of nanoparticles-bioconjugate exosomes have been synthesized and tested to target specific immune cells in the acidic TME. Recent research has demonstrated that anti-tumoral M1 macrophages-derived exosomes conjugated with CD47 and SIRPα antibodies effectively reprogrammed the macrophages from M2 to M1 phenotype in the TME.116 In another study in the living mice, cancer-associated fibroblasts in the TME can be specifically targeted by activated fibroblasts whose cell membrane was coated with semiconducting polymer nanoagents aiming to enhance multimodal cancer theranostics.124
MicroRNA-21 is a well-known microRNA that overexpresses in almost all cancer types, where its upregulation promotes cell proliferation, invasion, and metastasis.69–78 MiR-21 derived from the exosomes of MSCs regulates the death and differentiation of neurons in patients with spinal cord injury. Recent efforts involve utilizing an exosomal transfer of miRNAs or anti-miRNAs to tumor cells as a new approach for the therapeutic application of miRNAs to combat the most aggressive form of glioma, glioblastoma multiforme. Monfared and coworkers recently attempted to down-regulate miR-21 expression in glioma cell lines, U87-MG and C6, and rat glioblastoma models treated with miR-21-sponge exosomes and demonstrated a decline in tumor cell proliferation, a dramatic enhancement of apoptotic rate, and a significant reduction in tumor volume.125
Conclusion
Cancer-derived exosomal trafficking is observed in almost all types of liquid or solid tumors, including leukemia, soft tissue sarcoma, and osteosarcoma, which supports its role as an enabling characteristic for cancer hallmarks. The cargoes carried by CDEs contain enriched genetic signals in the form of DNA, RNA, microRNA, lncRNA, protein, lipid composition, or specific gene expression profiles, which are shed into blood, saliva, urine, effusions, ascites, and cervicovaginal lavage. There are a growing number of studies that investigate CDE as either a diagnostic, prognostic, or predictive nano-biomarker in various kinds of cancer. Out of the published clinical studies on exploiting CDE as a cancer biomarker, 70% of them were looking at the CDE as a diagnostic biomarker. In contrast, the rest of the studies were testing the role of CDE as a prognostic or predictive biomarker. Not surprisingly, only a few of them have reached the state of validation trials. In the near future, we shall expect to see more prospective clinical trials to validate the performance of these nanoparticle biomarkers aiming to improve the overall cancer diagnostic capability in the clinic.
Acknowledgments
We are grateful that part of this review has been critically reviewed by Prof. Ed Harlow of Harvard Medical School, Boston, in the High-Impact Cancer Research: Cancer Biology and Therapeutics program, where Prof. Robert Weinberg was also a faculty member. A significant amount of time was dedicated to the hallmarks of cancer. The authors of this review took part in the program as students and this review is a result of these activities.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article and revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
This work received no external fundings.
Disclosure
The authors report no conflicts of interest.
References
1. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi:10.1016/j.cell.2011.02.013
2. Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70. doi:10.1016/S0092-8674(00)81683-9
3. Xu B, Wang T. Intimate cross-talk between cancer cells and the tumor microenvironment of B-cell lymphomas: the key role of exosomes. Tumour Biol. 2017;39(6):1010428317706227. doi:10.1177/1010428317706227
4. Vermeer PD. Exosomal induction of tumor innervation. Cancer Res. 2019;79(14):3529–3535. doi:10.1158/0008-5472.CAN-18-3995
5. Madeo M, Colbert PL, Vermeer DW, et al. Cancer exosomes induce tumor innervation. Nat Commun. 2018;9(1):4284. doi:10.1038/s41467-018-06640-0
6. Lucido CT, Wynja E, Madeo M, et al. Innervation of cervical carcinoma is mediated by cancer-derived exosomes. Gynecol Oncol. 2019. doi:10.1016/j.ygyno.2019.04.651
7. Hosoi J, Murphy GF, Egan CL, et al. Regulation of Langerhans cell function by nerves containing calcitonin gene-related peptide. Nature. 1993;363(6425):159–163. doi:10.1038/363159a0
8. Magnon C, Hall SJ, Lin J, et al. Autonomic nerve development contributes to prostate cancer progression. Science. 2013;341(6142):1236361. doi:10.1126/science.1236361
9. Dubeykovskaya Z, Si Y, Chen X, et al. Neural innervation stimulates splenic TFF2 to arrest myeloid cell expansion and cancer. Nat Commun. 2016;7:10517. doi:10.1038/ncomms10517
10. Hayakawa Y, Sakitani K, Konishi M, et al. Nerve growth factor promotes gastric tumorigenesis through aberrant cholinergic signaling. Cancer Cell. 2017;31(1):21–34. doi:10.1016/j.ccell.2016.11.005
11. Monje M. Settling a nervous stomach: the neural regulation of enteric cancer. Cancer Cell. 2017;31(1):1–2. doi:10.1016/j.ccell.2016.12.008
12. Zahalka AH, Arnal-Estape A, Maryanovich M, et al. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science. 2017;358(6361):321–326. doi:10.1126/science.aah5072
13. Renz BW, Takahashi R, Tanaka T, et al. beta2 adrenergic-neurotrophin feedforward loop promotes pancreatic cancer. Cancer Cell. 2018;33(1):75–90.e77.
14. Trams EG, Lauter CJ, Salem N
15. Thery C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2(8):569–579. doi:10.1038/nri855
16. Vlassov AV, Magdaleno S, Setterquist R, Conrad R. Exosomes: current knowledge of their composition, biological functions, and diagnostic and therapeutic potentials. Biochim Biophys Acta. 2012;1820(7):940–948. doi:10.1016/j.bbagen.2012.03.017
17. Shah R, Patel T, Freedman JE. Circulating extracellular vesicles in human disease. N Engl J Med. 2018;379(10):958–966. doi:10.1056/NEJMra1704286
18. Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750.
19. Simona F, Laura S, Simona T, Riccardo A. Contribution of proteomics to understanding the role of tumor-derived exosomes in cancer progression: state of the art and new perspectives. Proteomics. 2013;13(10–11):1581–1594. doi:10.1002/pmic.201200398
20. Hannafon BN, Ding WQ. Intercellular communication by exosome-derived microRNAs in cancer. Int J Mol Sci. 2013;14(7):14240–14269. doi:10.3390/ijms140714240
21. Thakur BK, Zhang H, Becker A, et al. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 2014;24(6):766–769. doi:10.1038/cr.2014.44
22. Li Y, Zheng Q, Bao C, et al. Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res. 2015;25(8):981–984. doi:10.1038/cr.2015.82
23. Kalluri R, LeBleu VS. Discovery of double-stranded genomic DNA in circulating exosomes. Cold Spring Harb Symp Quant Biol. 2016;81:275–280. doi:10.1101/sqb.2016.81.030932
24. Bilen MA, Pan T, Lee YC, et al. Proteomics profiling of exosomes from primary mouse osteoblasts under proliferation versus mineralization conditions and characterization of their uptake into prostate cancer cells. J Proteome Res. 2017;16(8):2709–2728. doi:10.1021/acs.jproteome.6b00981
25. Khushman M, Bhardwaj A, Patel GK, et al. Exosomal markers (CD63 and CD9) expression pattern using immunohistochemistry in resected malignant and nonmalignant pancreatic specimens. Pancreas. 2017;46(6):782–788. doi:10.1097/MPA.0000000000000847
26. Mizutani K, Terazawa R, Kameyama K, et al. Isolation of prostate cancer-related exosomes. Anticancer Res. 2014;34(7):3419–3423.
27. Ko J, Bhagwat N, Yee SS, et al. Combining machine learning and nanofluidic technology to diagnose pancreatic cancer using exosomes. ACS Nano. 2017;11(11):11182–11193. doi:10.1021/acsnano.7b05503
28. Liu F, Vermesh O, Mani V, et al. The exosome total isolation chip. ACS Nano. 2017;11(11):10712–10723. doi:10.1021/acsnano.7b04878
29. Lyu Y, Cui D, Huang J, Fan W, Miao Y, Pu K. Near-infrared afterglow semiconducting nano-polycomplexes for the multiplex differentiation of cancer exosomes. Angew Chem Int Ed Engl. 2019;58(15):4983–4987. doi:10.1002/anie.201900092
30. Dioufa N, Clark AM, Ma B, Beckwitt CH, Wells A. Bi-directional exosome-driven intercommunication between the hepatic niche and cancer cells. Mol Cancer. 2017;16(1):172.
31. Milane L, Singh A, Mattheolabakis G, Suresh M, Amiji MM. Exosome mediated communication within the tumor microenvironment. J Control Release. 2015;219:278–294. doi:10.1016/j.jconrel.2015.06.029
32. Sung BH, Weaver AM. Exosome secretion promotes chemotaxis of cancer cells. Cell Adh Migr. 2017;11(2):187–195. doi:10.1080/19336918.2016.1273307
33. Bobrie A, Thery C. Exosomes and communication between tumours and the immune system: are all exosomes equal? Biochem Soc Trans. 2013;41(1):263–267. doi:10.1042/BST20120245
34. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215–233. doi:10.1016/j.cell.2009.01.002
35. Beckham CJ, Olsen J, Yin PN, et al. Bladder cancer exosomes contain EDIL-3/Del1 and facilitate cancer progression. J Urol. 2014;192(2):583–592. doi:10.1016/j.juro.2014.02.035
36. Chowdhury R, Webber JP, Gurney M, Mason MD, Tabi Z, Clayton A. Cancer exosomes trigger mesenchymal stem cell differentiation into pro-angiogenic and pro-invasive myofibroblasts. Oncotarget. 2015;6(2):715–731. doi:10.18632/oncotarget.2711
37. Hoshino A, Costa-Silva B, Shen TL, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527(7578):329–335. doi:10.1038/nature15756
38. Bruns H, Bottcher M, Qorraj M, et al. CLL-cell-mediated MDSC induction by exosomal miR-155 transfer is disrupted by vitamin D. Leukemia. 2017;31(4):985–988. doi:10.1038/leu.2016.378
39. Jitschin R, Braun M, Buttner M, et al. CLL-cells induce IDOhi CD14+HLA-DRlo myeloid-derived suppressor cells that inhibit T-cell responses and promote TRegs. Blood. 2014;124(5):750–760. doi:10.1182/blood-2013-12-546416
40. Farahani M, Rubbi C, Liu L, Slupsky JR, Kalakonda N. CLL exosomes modulate the transcriptome and behaviour of recipient stromal cells and are selectively enriched in miR-202-3p. PLoS One. 2015;10(10):e0141429. doi:10.1371/journal.pone.0141429
41. Paggetti J, Haderk F, Seiffert M, et al. Exosomes released by chronic lymphocytic leukemia cells induce the transition of stromal cells into cancer-associated fibroblasts. Blood. 2015;126(9):1106–1117. doi:10.1182/blood-2014-12-618025
42. Corrado C, Raimondo S, Saieva L, Flugy AM, De Leo G, Alessandro R. Exosome-mediated crosstalk between chronic myelogenous leukemia cells and human bone marrow stromal cells triggers an interleukin 8-dependent survival of leukemia cells. Cancer Lett. 2014;348(1–2):71–76. doi:10.1016/j.canlet.2014.03.009
43. Gao X, Wan Z, Wei M, et al. Chronic myelogenous leukemia cells remodel the bone marrow niche via exosome-mediated transfer of miR-320. Theranostics. 2019;9(19):5642–5656. doi:10.7150/thno.34813
44. Taverna S, Flugy A, Saieva L, et al. Role of exosomes released by chronic myelogenous leukemia cells in angiogenesis. Int J Cancer. 2012;130(9):2033–2043. doi:10.1002/ijc.26217
45. Barrera-Ramirez J, Lavoie JR, Maganti HB, et al. Micro-RNA profiling of exosomes from marrow-derived mesenchymal stromal cells in patients with acute myeloid leukemia: implications in leukemogenesis. Stem Cell Rev Rep. 2017;13(6):817–825. doi:10.1007/s12015-017-9762-0
46. Hong CS, Danet-Desnoyers G, Shan X, Sharma P, Whiteside TL, Boyiadzis M. Human acute myeloid leukemia blast-derived exosomes in patient-derived xenograft mice mediate immune suppression. Exp Hematol. 2019;76:60–66.e62.
47. Hong CS, Sharma P, Yerneni SS, et al. Circulating exosomes carrying an immunosuppressive cargo interfere with cellular immunotherapy in acute myeloid leukemia. Sci Rep. 2017;7(1):14684. doi:10.1038/s41598-017-14661-w
48. Boyiadzis M, Whiteside TL. Exosomes in acute myeloid leukemia inhibit hematopoiesis. Curr Opin Hematol. 2018;25(4):279–284. doi:10.1097/MOH.0000000000000439
49. Hornick NI, Doron B, Abdelhamed S, et al. AML suppresses hematopoiesis by releasing exosomes that contain microRNAs targeting c-MYB. Sci Signal. 2016;9(444):ra88. doi:10.1126/scisignal.aaf2797
50. Chen T, Zhang G, Kong L, Xu S, Wang Y, Dong M. Leukemia-derived exosomes induced IL-8 production in bone marrow stromal cells to protect the leukemia cells against chemotherapy. Life Sci. 2019;221:187–195. doi:10.1016/j.lfs.2019.02.003
51. Wang B, Wang X, Hou D, et al. Exosomes derived from acute myeloid leukemia cells promote chemoresistance by enhancing glycolysis-mediated vascular remodeling. J Cell Physiol. 2019;234(7):10602–10614. doi:10.1002/jcp.27735
52. Huan J, Hornick NI, Goloviznina NA, et al. Coordinate regulation of residual bone marrow function by paracrine trafficking of AML exosomes. Leukemia. 2015;29(12):2285–2295. doi:10.1038/leu.2015.163
53. Javidi-Sharifi N, Martinez J, English I, et al. FGF2-FGFR1 signaling regulates release of leukemia-protective exosomes from bone marrow stromal cells. eLife. 2019;8.
54. Kumar B, Garcia M, Weng L, et al. Acute myeloid leukemia transforms the bone marrow niche into a leukemia-permissive microenvironment through exosome secretion. Leukemia. 2018;32(3):575–587. doi:10.1038/leu.2017.259
55. Wang J, De Veirman K, Faict S, et al. Multiple myeloma exosomes establish a favourable bone marrow microenvironment with enhanced angiogenesis and immunosuppression. J Pathol. 2016;239(2):162–173. doi:10.1002/path.4712
56. Wang J, De Veirman K, De Beule N, et al. The bone marrow microenvironment enhances multiple myeloma progression by exosome-mediated activation of myeloid-derived suppressor cells. Oncotarget. 2015;6(41):43992–44004. doi:10.18632/oncotarget.6083
57. Roccaro AM, Sacco A, Maiso P, et al. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J Clin Invest. 2013;123(4):1542–1555. doi:10.1172/JCI66517
58. Manier S, Liu CJ, Avet-Loiseau H, et al. Prognostic role of circulating exosomal miRNAs in multiple myeloma. Blood. 2017;129(17):2429–2436. doi:10.1182/blood-2016-09-742296
59. Vardaki I, Sanchez C, Fonseca P, et al. Caspase-3-dependent cleavage of Bcl-xL in the stroma exosomes is required for their uptake by hematological malignant cells. Blood. 2016;128(23):2655–2665. doi:10.1182/blood-2016-05-715961
60. Umezu T, Tadokoro H, Azuma K, Yoshizawa S, Ohyashiki K, Ohyashiki JH. Exosomal miR-135b shed from hypoxic multiple myeloma cells enhances angiogenesis by targeting factor-inhibiting HIF-1. Blood. 2014;124(25):3748–3757. doi:10.1182/blood-2014-05-576116
61. Zhai LY, Li MX, Pan WL, et al. In situ detection of plasma exosomal microRNA-1246 for breast cancer diagnostics by a au nanoflare probe. ACS Appl Mater Interfaces. 2018;10(46):39478–39486. doi:10.1021/acsami.8b12725
62. Stevic I, Muller V, Weber K, et al. Specific microRNA signatures in exosomes of triple-negative and HER2-positive breast cancer patients undergoing neoadjuvant therapy within the GeparSixto trial. BMC Med. 2018;16(1):179. doi:10.1186/s12916-018-1163-y
63. Wen SW, Sceneay J, Lima LG, et al. The biodistribution and immune suppressive effects of breast cancer-derived exosomes. Cancer Res. 2016;76(23):6816–6827. doi:10.1158/0008-5472.CAN-16-0868
64. Santos JC, Lima NDS, Sarian LO, Matheu A, Ribeiro ML, Derchain SFM. Exosome-mediated breast cancer chemoresistance via miR-155 transfer. Sci Rep. 2018;8(1):829. doi:10.1038/s41598-018-19339-5
65. Sansone P, Savini C, Kurelac I, et al. Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancer. Proc Natl Acad Sci U S A. 2017;114(43):E9066–E9075. doi:10.1073/pnas.1704862114
66. Rodriguez M, Silva J, Herrera A, et al. Exosomes enriched in stemness/metastatic-related mRNAS promote oncogenic potential in breast cancer. Oncotarget. 2015;6(38):40575–40587. doi:10.18632/oncotarget.5818
67. Miller IV, Raposo G, Welsch U, et al. First identification of Ewing’s sarcoma-derived extracellular vesicles and exploration of their biological and potential diagnostic implications. Biol Cell. 2013;105(7):289–303. doi:10.1111/boc.201200086
68. Casadei L, Calore F, Creighton CJ, et al. Exosome-Derived miR-25-3p and miR-92a-3p Stimulate Liposarcoma Progression. Cancer Res. 2017;77(14):3846–3856. doi:10.1158/0008-5472.CAN-16-2984
69. Ventura S, Aryee DN, Felicetti F, et al. CD99 regulates neural differentiation of Ewing sarcoma cells through miR-34a-Notch-mediated control of NF-kappaB signaling. Oncogene. 2016;35(30):3944–3954. doi:10.1038/onc.2015.463
70. Hakulinen J, Sankkila L, Sugiyama N, Lehti K, Keski-Oja J. Secretion of active membrane type 1 matrix metalloproteinase (MMP-14) into extracellular space in microvesicular exosomes. J Cell Biochem. 2008;105(5):1211–1218. doi:10.1002/jcb.21923
71. Raimondi L, De Luca A, Gallo A, et al. Osteosarcoma cell-derived exosomes affect tumor microenvironment by specific packaging of microRNAs. Carcinogenesis. 2019.
72. Goto T, Fujiya M, Konishi H, et al. An elevated expression of serum exosomal microRNA-191, - 21, −451a of pancreatic neoplasm is considered to be efficient diagnostic marker. BMC Cancer. 2018;18(1):116. doi:10.1186/s12885-018-4006-5
73. Zheng P, Chen L, Yuan X, et al. Exosomal transfer of tumor-associated macrophage-derived miR-21 confers cisplatin resistance in gastric cancer cells. J Exp Clin Cancer Res. 2017;36(1):53. doi:10.1186/s13046-017-0528-y
74. Ragusa M, Barbagallo C, Statello L, et al. miRNA profiling in vitreous humor, vitreal exosomes and serum from uveal melanoma patients: pathological and diagnostic implications. Cancer Biol Ther. 2015;16(9):1387–1396. doi:10.1080/15384047.2015.1046021
75. Liu W, Chen S, Liu B. Diagnostic and prognostic values of serum exosomal microRNA-21 in children with hepatoblastoma: a Chinese population-based study. Pediatr Surg Int. 2016;32(11):1059–1065. doi:10.1007/s00383-016-3960-8
76. Liao J, Liu R, Shi YJ, Yin LH, Pu YP. Exosome-shuttling microRNA-21 promotes cell migration and invasion-targeting PDCD4 in esophageal cancer. Int J Oncol. 2016;48(6):2567–2579. doi:10.3892/ijo.2016.3453
77. Hannafon BN, Trigoso YD, Calloway CL, et al. Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res. 2016;18(1):90. doi:10.1186/s13058-016-0753-x
78. Foj L, Ferrer F, Serra M, et al. Exosomal and non-exosomal urinary miRNAs in prostate cancer detection and prognosis. Prostate. 2017;77(6):573–583. doi:10.1002/pros.23295
79. Bhome R, Goh RW, Bullock MD, et al. Exosomal microRNAs derived from colorectal cancer-associated fibroblasts: role in driving cancer progression. Aging. 2017;9(12):2666–2694. doi:10.18632/aging.101355
80. Bahrami A, Aledavood A, Anvari K, et al. The prognostic and therapeutic application of microRNAs in breast cancer: tissue and circulating microRNAs. J Cell Physiol. 2018;233(2):774–786. doi:10.1002/jcp.25813
81. Endo-Munoz L, Cai N, Cumming A, et al. Progression of osteosarcoma from a non-metastatic to a metastatic phenotype is causally associated with activation of an autocrine and paracrine uPA axis. PLoS One. 2015;10(8):e0133592. doi:10.1371/journal.pone.0133592
82. Fujiwara T, Uotani K, Yoshida A, et al. Clinical significance of circulating miR-25-3p as a novel diagnostic and prognostic biomarker in osteosarcoma. Oncotarget. 2017;8(20):33375–33392. doi:10.18632/oncotarget.16498
83. Jerez S, Araya H, Hevia D, et al. Extracellular vesicles from osteosarcoma cell lines contain miRNAs associated with cell adhesion and apoptosis. Gene. 2019;710:246–257. doi:10.1016/j.gene.2019.06.005
84. Wu G, Zhang J, Zhao Q, et al. Molecularly engineered macrophage-derived exosomes with inflammation tropism and intrinsic heme biosynthesis for atherosclerosis treatment. Angew Chem Int Ed Engl. 2020;59(10):4068–4074. doi:10.1002/anie.201913700
85. Clark DJ, Fondrie WE, Yang A, Mao L. Triple SILAC quantitative proteomic analysis reveals differential abundance of cell signaling proteins between normal and lung cancer-derived exosomes. J Proteomics. 2016;133:161–169. doi:10.1016/j.jprot.2015.12.023
86. Sinha A, Ignatchenko V, Ignatchenko A, Mejia-Guerrero S, Kislinger T. In-depth proteomic analyses of ovarian cancer cell line exosomes reveals differential enrichment of functional categories compared to the NCI 60 proteome. Biochem Biophys Res Commun. 2014;445(4):694–701. doi:10.1016/j.bbrc.2013.12.070
87. Gangoda L, Liem M, Ang CS, et al. Proteomic profiling of exosomes secreted by breast cancer cells with varying metastatic potential. Proteomics. 2017;17:23–24. doi:10.1002/pmic.201600370
88. Giampieri R, Piva F, Occhipinti G, et al. Clinical impact of different exosomes’ protein expression in pancreatic ductal carcinoma patients treated with standard first line palliative chemotherapy. PLoS One. 2019;14(5):e0215990. doi:10.1371/journal.pone.0215990
89. Blomme A, Fahmy K, Peulen O, et al. Myoferlin is a novel exosomal protein and functional regulator of cancer-derived exosomes. Oncotarget. 2016;7(50):83669–83683. doi:10.18632/oncotarget.13276
90. Fan TWM, Zhang X, Wang C, et al. Exosomal lipids for classifying early and late stage non-small cell lung cancer. Anal Chim Acta. 2018;1037:256–264. doi:10.1016/j.aca.2018.02.051
91. Yang JS, Lee JC, Byeon SK, Rha KH, Moon MH. Size dependent lipidomic analysis of urinary exosomes from patients with prostate cancer by flow field-flow fractionation and nanoflow liquid chromatography-tandem mass spectrometry. Anal Chem. 2017;89(4):2488–2496.
92. Roberg-Larsen H, Lund K, Seterdal KE, et al. Mass spectrometric detection of 27-hydroxycholesterol in breast cancer exosomes. J Steroid Biochem Mol Biol. 2017;169:22–28. doi:10.1016/j.jsbmb.2016.02.006
93. Skotland T, Ekroos K, Kauhanen D, et al. Molecular lipid species in urinary exosomes as potential prostate cancer biomarkers. Eur J Cancer. 2017;70:122–132. doi:10.1016/j.ejca.2016.10.011
94. Mao L, Li X, Gong S, et al. Serum exosomes contain ECRG4 mRNA that suppresses tumor growth via inhibition of genes involved in inflammation, cell proliferation, and angiogenesis. Cancer Gene Ther. 2018;25(9–10):248–259. doi:10.1038/s41417-018-0032-3
95. Harada T, Yamamoto H, Kishida S, et al. Wnt5b-associated exosomes promote cancer cell migration and proliferation. Cancer Sci. 2017;108(1):42–52. doi:10.1111/cas.13109
96. Kitai Y, Kawasaki T, Sueyoshi T, et al. DNA-containing exosomes derived from cancer cells treated with topotecan activate a STING-dependent pathway and reinforce antitumor immunity. J Immunol. 2017;198(4):1649–1659. doi:10.4049/jimmunol.1601694
97. Pan L, Liang W, Fu M, et al. Exosomes-mediated transfer of long noncoding RNA ZFAS1 promotes gastric cancer progression. J Cancer Res Clin Oncol. 2017;143(6):991–1004. doi:10.1007/s00432-017-2361-2
98. Singh A, Fedele C, Lu H, Nevalainen MT, Keen JH, Languino LR. Exosome-mediated transfer of alphavbeta3 integrin from tumorigenic to nontumorigenic cells promotes a migratory phenotype. Mol Cancer Res. 2016;14(11):1136–1146. doi:10.1158/1541-7786.MCR-16-0058
99. Yang SJ, Wang DD, Li J, et al. Predictive role of GSTP1-containing exosomes in chemotherapy-resistant breast cancer. Gene. 2017;623:5–14. doi:10.1016/j.gene.2017.04.031
100. McKiernan J, Donovan MJ, O’Neill V, et al. A novel urine exosome gene expression assay to predict high-grade prostate cancer at initial biopsy. JAMA Oncol. 2016;2(7):882–889. doi:10.1001/jamaoncol.2016.0097
101. Samsonov R, Burdakov V, Shtam T, et al. Plasma exosomal miR-21 and miR-181a differentiates follicular from papillary thyroid cancer. Tumour Biol. 2016;37(9):12011–12021. doi:10.1007/s13277-016-5065-3
102. Li M, Zhou Y, Xia T, et al. Circulating microRNAs from the miR-106a-363 cluster on chromosome X as novel diagnostic biomarkers for breast cancer. Breast Cancer Res Treat. 2018;170(2):257–270. doi:10.1007/s10549-018-4757-3
103. Zhang J, Liu SC, Luo XH, et al. Exosomal long noncoding RNAs are differentially expressed in the cervicovaginal lavage samples of cervical cancer patients. J Clin Lab Anal. 2016;30(6):1116–1121. doi:10.1002/jcla.21990
104. Lea J, Sharma R, Yang F, Zhu H, Ward ES, Schroit AJ. Detection of phosphatidylserine-positive exosomes as a diagnostic marker for ovarian malignancies: a proof of concept study. Oncotarget. 2017;8(9):14395–14407.
105. McKiernan J, Donovan MJ, Margolis E, et al. A prospective adaptive utility trial to validate performance of a novel urine exosome gene expression assay to predict high-grade prostate cancer in patients with prostate-specific antigen 2-10ng/mL at initial biopsy. Eur Urol. 2018;74(6):731–738. doi:10.1016/j.eururo.2018.08.019
106. De Palma G, Sallustio F, Curci C, et al. The three-gene signature in urinary extracellular vesicles from patients with clear cell renal cell carcinoma. J Cancer. 2016;7(14):1960–1967. doi:10.7150/jca.16123
107. Yoshida A, Fujiwara T, Uotani K, et al. Clinical and functional significance of intracellular and extracellular microRNA-25-3p in Osteosarcoma. Acta Med Okayama. 2018;72(2):165–174.
108. Sedlarikova L, Bollova B, Radova L, et al. Circulating exosomal long noncoding RNA PRINS-First findings in monoclonal gammopathies. Hematol Oncol. 2018;36(5):786–791. doi:10.1002/hon.2554
109. Jin X, Chen Y, Chen H, et al. Evaluation of tumor-derived exosomal miRNA as potential diagnostic biomarkers for early-stage non-small cell lung cancer using next-generation sequencing. Clin Cancer Res. 2017;23(17):5311–5319. doi:10.1158/1078-0432.CCR-17-0577
110. Sharma R, Huang X, Brekken RA, Schroit AJ. Detection of phosphatidylserine-positive exosomes for the diagnosis of early-stage malignancies. Br J Cancer. 2017;117(4):545–552. doi:10.1038/bjc.2017.183
111. Sakaue T, Koga H, Iwamoto H, et al. Glycosylation of ascites-derived exosomal CD133: a potential prognostic biomarker in patients with advanced pancreatic cancer. Med Mol Morphol. 2019;52(4):198–208. doi:10.1007/s00795-019-00218-5
112. Shi M, Jiang Y, Yang L, Yan S, Wang YG, Lu XJ. Decreased levels of serum exosomal miR-638 predict poor prognosis in hepatocellular carcinoma. J Cell Biochem. 2018;119(6):4711–4716. doi:10.1002/jcb.26650
113. Lin Y, Dong H, Deng W, et al. Evaluation of salivary exosomal chimeric GOLM1-NAA35 RNA as a potential biomarker in esophageal carcinoma. Clin Cancer Res. 2019;25(10):3035–3045. doi:10.1158/1078-0432.CCR-18-3169
114. Sung JS, Kang CW, Kang S, et al. ITGB4-mediated metabolic reprogramming of cancer-associated fibroblasts. Oncogene. 2019.
115. Pando A, Reagan JL, Quesenberry P, Fast LD. Extracellular vesicles in leukemia. Leuk Res. 2018;64:52–60. doi:10.1016/j.leukres.2017.11.011
116. Nie W, Wu G, Zhang J, et al. Responsive exosome nano-bioconjugates for synergistic cancer therapy. Angew Chem Int Ed Engl. 2020;59(5):2018–2022. doi:10.1002/anie.201912524
117. Chen G, Huang AC, Zhang W, et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature. 2018;560(7718):382–386. doi:10.1038/s41586-018-0392-8
118. Cheng Q, Shi X, Han M, Smbatyan G, Lenz HJ, Zhang Y. Reprogramming exosomes as nanoscale controllers of cellular immunity. J Am Chem Soc. 2018;140(48):16413–16417. doi:10.1021/jacs.8b10047
119. Gomari H, Forouzandeh Moghadam M, Soleimani M, Ghavami M, Khodashenas S. Targeted delivery of doxorubicin to HER2 positive tumor models. Int J Nanomedicine. 2019;14:5679–5690.
120. Gorji-Bahri G, Hashemi A, Moghimi HR. ExomiRs: a novel strategy in cancer diagnosis and therapy. Curr Gene Ther. 2018;18(6):336–350. doi:10.2174/1566523218666181017163204
121. Kumar S, Michael IJ, Park J, Granick S, Cho YK. Cloaked exosomes: biocompatible, durable, and degradable encapsulation. Small. 2018;14(34):e1802052. doi:10.1002/smll.201802052
122. Vazquez-Rios AJ, Molina-Crespo A, Bouzo BL, Lopez-Lopez R, Moreno-Bueno G, de la Fuente M. Exosome-mimetic nanoplatforms for targeted cancer drug delivery. J Nanobiotechnology. 2019;17(1):85. doi:10.1186/s12951-019-0517-8
123. Yu M, Gai C, Li Z, et al. Targeted exosome-encapsulated erastin induced ferroptosis in triple negative breast cancer cells. Cancer Sci. 2019;110(10):3173–3182.
124. Li J, Zhen X, Lyu Y, Jiang Y, Huang J, Pu K. Cell membrane coated semiconducting polymer nanoparticles for enhanced multimodal cancer phototheranostics. ACS Nano. 2018;12(8):8520–8530. doi:10.1021/acsnano.8b04066
125. Monfared H, Jahangard Y, Nikkhah M, Mirnajafi-Zadeh J, Mowla SJ. Potential therapeutic effects of exosomes packed with a miR-21-sponge construct in a rat model of glioblastoma. Front Oncol. 2019;9:782. doi:10.3389/fonc.2019.00782
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.