Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
Bronchoscopic lung volume reduction by endobronchial valve in advanced emphysema: the first Asian report
Authors Park TS ![]() , Hong Y, Lee JS, Oh SY, Lee SM
, Hong Y, Lee JS, Oh SY, Lee SM ![]() , Kim N
, Kim N ![]() , Seo J
, Seo J ![]() , Oh YM
, Oh YM ![]() , Lee SD, Lee SW
, Lee SD, Lee SW ![]()
Received 31 March 2015
Accepted for publication 16 May 2015
Published 29 July 2015 Volume 2015:10(1) Pages 1501—1511
DOI https://doi.org/10.2147/COPD.S85744
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Richard Russell
Tai Sun Park,1 Yoonki Hong,2 Jae Seung Lee,1 Sang Young Oh,3 Sang Min Lee,3 Namkug Kim,3 Joon Beom Seo,3 Yeon-Mok Oh,1 Sang-Do Lee,1 Sei Won Lee1
1Department of Pulmonary and Critical Care Medicine and Clinical Research Center for Chronic Obstructive Airway Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea; 2Department of Internal Medicine, College of Medicine, Kangwon National University, Chuncheon, Korea; 3Department of Radiology and Research Institute of Radiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
Purpose: Endobronchial valve (EBV) therapy is increasingly being seen as a therapeutic option for advanced emphysema, but its clinical utility in Asian populations, who may have different phenotypes to other ethnic populations, has not been assessed.
Patients and methods: This prospective open-label single-arm clinical trial examined the clinical efficacy and the safety of EBV in 43 consecutive patients (mean age 68.4±7.5, forced expiratory volume in 1 second [FEV1] 24.5%±10.7% predicted, residual volume 208.7%±47.9% predicted) with severe emphysema with complete fissure and no collateral ventilation in a tertiary referral hospital in Korea.
Results: Compared to baseline, the patients exhibited significant improvements 6 months after EBV therapy in terms of FEV1 (from 0.68±0.26 L to 0.92±0.40 L; P<0.001), 6-minute walk distance (from 233.5±114.8 m to 299.6±87.5 m; P=0.012), modified Medical Research Council dyspnea scale (from 3.7±0.6 to 2.4±1.2; P<0.001), and St George’s Respiratory Questionnaire (from 65.59±13.07 to 53.76±11.40; P=0.028). Nine patients (20.9%) had a tuberculosis scar, but these scars did not affect target lobe volume reduction or pneumothorax frequency. Thirteen patients had adverse events, ten (23.3%) developed pneumothorax, which included one death due to tension pneumothorax.
Conclusion: EBV therapy was as effective and safe in Korean patients as it has been shown to be in Western countries. (Trial registration: ClinicalTrials.gov: NCT01869205).
Keywords: bronchoscopy, chronic obstructive pulmonary disease, collateral ventilation, fissure integrity, treatment outcome
Introduction
Chronic obstructive pulmonary disease (COPD) is a leading cause of disability and death worldwide.1 It is characterized by chronic airflow limitation that is caused by a mixture of small airway disease and parenchymal destruction.2 Lung parenchyma destruction, also known as emphysema, results in decreased elastic recoil, progressive hyperinflation, and air trapping, which in turn lead to dyspnea and decreased lung function. Currently, bronchodilator is the mainstay of COPD treatment, and it can improve dyspnea and lung function. However, these treatments are less effective in emphysema-dominant phenotype.3
Lung volume reduction surgery (LVRS) entails resection of lung areas that are particularly hyperinflated. It was developed as a palliative treatment for severe emphysema4,5 and clearly improves lung function, exercise capacity, and survival in appropriately selected patients.6 However, it also associated with substantial procedure-related morbidity and mortality. Therefore, less invasive bronchoscopic approaches for treating hyperinflation in advanced emphysema have been proposed. Several bronchoscopic lung volume reduction (BLVR) methods have shown the similar physiological effects of LVRS with lower morbidity and mortality.7
However, the efficacies of BLVR have been documented mostly in the studies of Western countries. It is unclear whether these effects are still consistent in Asian population because the COPD phenotypes of Asian and Western patients may differ.8–10 Along with tobacco smoking, previous exposure to biomass fuels or dust-bearing jobs, and treated pulmonary tuberculosis (TB) are also major causes of COPD in Asia.11,12 The prevalence of TB is relatively higher in the Asia-Pacific area,13 and it is significant because the history of TB is associated with airway and parenchymal scarring causing airflow limitation, a characteristics of COPD.11,14,15
The COPD heterogeneity indicates that the feasibility of BLVR in this population should be assessed. We previously reported the results of a small pilot study on the short-term feasibility and safety of BLVR with endobronchial valve (EBV) in the Koreans.16 However, the long-term efficacy and safety of EBV treatment of severe emphysema in Asia have not been assessed. This study is an extension of the previous pilot study. Its aim was to prospectively assess the clinical efficacy and safety of BLVR with EBV in Asians with different medical environment and distinct clinical phenotype from those of Westerns.
Materials and methods
Study design and patients
This is a prospective open-label single-arm clinical trial. Its main objective was to evaluate the effectiveness of BLVR with EBVs in patients with severe emphysema who had “complete target lobar exclusion”.17 The primary outcome measurements were mean changes in pulmonary function, exercise capacity, and quality of life after the procedure. The secondary outcomes included clinical response predictors and procedure-related complications.
Patients with advanced emphysema who did not respond sufficiently to the standard treatment for COPD were considered to be eligible. In July 2012–December 2013, consecutive patients were recruited from the pulmonary clinic at Asan Medical Center, Seoul, Korea. Written informed consent was obtained from each patient. The study received approval from The Institutional Review Board of Asan Medical Center.
Patient selection criteria were based on previous studies.18–20 Patient inclusion criteria were an age of ≥40 years, a smoking history of >10 pack year, the presence of advanced heterogeneous emphysema on chest computed tomography (CT), a forced expiratory volume in 1 second (FEV1) of ≤45% of predicted value, a total lung capacity (TLC) of >100% of predicted value, a residual volume (RV) of >150% of predicted value, persistent dyspnea despite optimal medical treatment and pulmonary rehabilitation for more than 3 months, and negative collateral ventilation of the target lobe. Patients with homogeneous or incomplete fissure of the target lobe on CT, collateral ventilation of the target lobe, a diffusing capacity for carbon monoxide corrected by alveolar ventilation (DLCO/VA) of <20% of predicted value, giant bullae (>5 cm) at the ipsilateral lobe, previous thoracotomy, excessive sputum, severe pulmonary hypertension, an active respiratory infection, and/or an unstable cardiac condition were excluded.
BLVR procedure
BLVR was performed with a flexible bronchoscope (BF-1T60® or BF-1T 260®, Olympus, Tokyo, Japan) by placing Zephyr® EBVs (Pulmonx, Inc., Redwood City, CA, USA) in the bronchi of the target lobe. Patients were given intravenous anesthesia using midazolam (0.05–0.08 mg/kg) under spontaneous ventilation. Before placing the EBVs, the Chartis™ system (Pulmonx, Inc.) was used to confirm that the target lobe lacked collateral ventilation with other lobes.19 Collateral ventilation was deemed to be absent when the air flow declined gradually over time after occlusion of the target lobe airway with a balloon catheter. However, if persistent airflow was observed after occlusion for more than 5 minutes, significant collateral ventilation was suspected.21 After the procedure, daily chest X-ray was performed until 5 days after the procedure to ensure the EBVs were adequately placed and to detect possible atelectasis or pneumothorax. Chest CT was performed before the procedure by using a dual-source CT system (SOMATOM Definition; Siemens Healthcare, Forchiheim, Germany). Fissure integrity was assessed by experienced thoracic radiologists who were unaware of the clinical information. The fissure was defined as “complete” when it could separate two adjacent lobes in more than 90% on at least one plane (axial and coronal planes) on thin-slice CT.20 Heterogeneity was screened by lobar emphysema and heterogeneity scoring, which was used in the previous study.18 The extent of emphysema was graded on a lobar level on a scale of 0–4 (called emphysema score). Heterogeneity was defined as ≥2 of heterogeneity score, which was derived by subtracting the best emphysema score for the worst emphysema score in single lung. Additionally, heterogeneity was assessed by the ratio of perfusion and volume of each lobe per whole lung after lobe segmentation. The perfusion-to-volume ratio of each lobe was calculated by quantitatively measuring the increased CT density caused by iodine in each voxel of each lobe using the in-house software.22,23 The target lobe was selected on the basis of these CT data (ie, the most hyperinflated and the least perfused lobe) after discussions between a pulmonologist and two radiologists (Figure 1).
Assessment of outcomes
Baseline evaluations were performed before the procedure. These included detailed history taking, physical examination, chest radiography, CT, laboratory examination, pulmonary function tests, and the 6-minute walk distance (6MWD). Health-related quality of life was documented by using the modified Medical Research Council (mMRC) dyspnea scale and St George’s Respiratory Questionnaire (SGRQ).
Three and six months after the procedure, clinical effectiveness was assessed by measuring FEV1, 6MWD, mMRC, and SGRQ. Target lobe volume reduction (TLVR) was defined as >50% volume reduction of the target lobe on chest X-ray at 3 months and was measured on the basis of visual assessments of the two thoracic radiologists.
Adverse events were recorded, including death, pneumothorax, pneumonia, empyema, hemoptysis, and acute COPD exacerbations.
Statistical analysis
All descriptive continuous variables were presented as the mean ± SD. For all study variables, the follow-up data were compared to the baseline data by using paired t-test (the Wilcoxon signed rank test). Minimum clinically important differences (MCID) for outcome variables were used as recommended,24 and they were ≥100 mL for FEV1,25 ≥26 m for 6MWD,26 and ≥4 units for SGRQ.27 Groups were compared by using Pearson’s chi-squared test or Mann–Whitney U-test. For key variables, the study data were compared to the data of previous trials by using Student’s t-test. P-values <0.05 were considered to indicate statistical significance; all P-values were two tailed. Statistical analysis was performed by using SPSS 21.0 software for Windows (SPSS, Chicago, IL, USA).
Results
Patients
In total, 83 consecutive patients were screened during the study period for inclusion in the study. Six patients were excluded because of collateral ventilation of target lobe on Chartis™ system, although they had complete fissure of target lobe on CT scan. The remaining 43 patients underwent BLVR with EBVs. Their baseline characteristics are shown in Table 1. Of these patients, 35 and 27 were followed up for 3 months and 6 months, respectively (Figure 2).
Procedural details
The right upper lobe was most frequently targeted for treatment (n=18, 41.9%), followed by the left lower (n=15, 34.9%), right lower (n=6, 14.0%), and left upper (n=1, 2.3%) lobes. Both the right upper and middle lobes were selected as the target lobes in six patients (14.0%) because of collateral ventilation between those lobes. The median number of EBVs inserted per patient was three (range: 2–5). A TB scar was present in the target lobe of five patients (11.6%) or in the adjacent lobe of five patients (11.6%). TLVR after the procedure was achieved in 36 of the 43 patients (83.7%). The mean hospital days of patients without and with pneumothorax were 7.4±3.2 and 14.7±13.2, respectively.
Efficacy outcomes
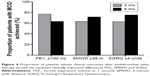
In the patients who were followed up at 3 months and 6 months, the FEV1 increased from 0.68±0.26 L to 0.89±0.37 L and 0.92±0.40 L, respectively (both P<0.001), the 6MWD improved from 233.5±114.8 m to 283.7±121.6 m (P=0.001) and 299.6±87.5 m (P=0.012), respectively, the dyspnea scale (mMRC) dropped from 3.7±0.6 to 2.5±1.2 and 2.4±1.2 (both P<0.001), respectively, and the SGRQ total scores fell from 65.59±13.07 to 55.70±13.79 (P=0.019) and 53.76±11.40 (P=0.028), respectively (Figure 3). Of the patients who were followed up at 3 months and 6 months, the FEV1 MCID (≥100 mL) was achieved by 27/35 (77.1%) and 17/27 (63.0%), respectively, the 6MWD MCID (26 m) was achieved by 19/30 (63.3%) and 15/21 (71.4%), and the SGRQ MCID (total score ≤4 points) was achieved by 17/27 (63.0%) and 15/20 (75.0%), respectively (Figure 4). RV was decreased from 208.7±47.9 L to 165.0%±47.7% predicted at 3 months and 155.9%±42.6% predicted at 6 months (both P<0.001). Other pulmonary function test data are shown in Table S1.
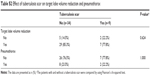
Although all 43 patients had complete exclusion of target lobe (as confirmed by CT scan and the Chartis™ system), TLVR was not achieved in seven patients (16.3%). Compared to patients without TLVR, the patients with TLVR had significantly greater improvements in FEV1 relative to baseline at both 3 (0.26±0.22 L vs –0.02±0.17 L, P=0.001) and 6 months (0.29±0.23 L vs –0.02±0.13 L, P=0.002). Similarly, the patients with TLVR tended to have greater improvements in 6MWD and SGRQ total score at 3 months and 6 months, although these differences did not achieve statistical significance (Table 2).
Regarding the ten patients (23.3%) with a history of previously treated pulmonary TB, nine patients had TB scars in the target or adjacent lobe, as determined by CT. The patients with and without a TB scar did not differ significantly in terms of TLVR achievement or development of pneumothorax (Table S2).
Safety outcomes
All follow-up patients were monitored for adverse events. Thirteen of the 43 patients (30.2%) developed complications. Four patients died within 6 months of the procedure. In the one procedure-related mortality case, tension pneumothorax developed 2 days after BLVR and the patient expired due to sudden-onset ventricular fibrillation. The other three patients did not exhibit clinical effectiveness after BLVR and died of chronic respiratory failure. Ten of the 43 patients (23.3%) developed pneumothorax. The median time to pneumothorax onset after BLVR was two (range: 1–26) days. Eight of the ten pneumothorax cases required chest tube insertion. Four patients exhibited prolonged air leakage (≥7 days). Some or all inserted EBVs were removed from three of these four patients for lung expansion (Table 3). Two patients whose pneumothorax was successfully treated with a chest tube developed a recurrence of pneumothorax. Of the ten patients who developed pneumothorax after BLVR, six and four were followed up at 3 months and 6 months, respectively. The FEV1 MCID (>100 mL) was achieved by four (66.7%) and three (75%) of these patients, respectively. Three studies have examined the outcomes of BLVR with EBVs in Western countries, namely, the USA VENT study (NCT00129584),28 the European VENT study (NCT00129584),20 and the Chartis™ study (NCT01101958).19 The incidence of pneumothorax was significantly higher in our study than in these three studies (23.3% vs 10.6%, P=0.03).29 Comparison of baseline characteristics indicated that the patients in our study were older and had a lower body mass index (BMI) and a poorer exercise capacity than those in the preceding studies. Moreover, although the baseline FEV1 in our cohort was lower, our patients also had less hyperinflation (as measured by RV) (Table 4).
  | Table 3 Adverse events in the 6 months after bronchoscopic lung volume reduction with endobronchial valve. (n=43) |
Discussion
In the past decade, BLVR has become recognized as a therapeutic option for advanced emphysema. However, its clinical utility in Asian populations has not been assessed yet. Our study investigated the relatively long-term outcomes of BLVR with EBV in Koreans with severe emphysema and found that BLVR with EBV is effective and acceptably safe in this population, and there were significant improvements in FEV1, 6MWD, and SGRQ that were sustained for 6 months. These observations are comparable with those of other studies.19,20,28,30
In our cohort, BLVR yielded definite clinical effects in terms of primary outcomes at 3 months, including FEV1 (absolute change 0.21±0.24 L, percent change 33.1%±33.2%), SGRQ (difference –6.7±10.5), and 6MWD (absolute change 55.1±92.3 m, percent change 54.4%±120.5%). The improvements in these variables at 3 months were higher than the improvements detected by previous studies.19,20 The outcomes at 6 months were similar to the 3 months outcomes (FEV1 absolute change: 0.22±0.25 L, percent change: 32.4%±34.2%; SGRQ difference points: –8.0±15.7; 6MWD absolute change: 54.3±97.5 m, percent change: 49.7%±107.5%). The greater improvements in our study relative to those in other studies could reflect the judicious selection of the patients, which was based on the results of the previous clinical trials. In particular, of the eligible patients with heterogeneous emphysema with complete fissure on CT, patients were only selected for BLVR if the Chartis™ system confirmed that there was no collateral ventilation. Moreover, the target lobe was carefully selected on the basis of quantitative CT analysis that indicated the degree of perfusion and hyperinflation of the lobes.
It is important to ensure that there is no collateral ventilation between the target lobe and other lobes because it promotes the achievement of TLVR, which is an important predictor of clinical improvement after BLVR.31 Two methods, namely, CT and the Chartis™ system have been used to predict the presence of collateral ventilation.32 CT can detect collateral ventilation with high sensitivity, but its specificity (44%) is low.33 This has been clearly demonstrated by post hoc analyses of the previous trials: TLVR (volume reduction of target lobe >50%) was achieved in only 32.5% of patients with complete fissure on CT.31 Assessment with the Chartis™ system can improve TLVR prediction: 70.6% achieved TLVR among the patients who lacked collateral ventilation defined by the Chartis™ system.19 In our study, we chose patients with complete fissure and lacking collateral ventilation (as shown by the Chartis™ system) in consideration of cost-effectiveness, and 83.7% (36/43) has achieved TLVR.
In about 15% of patients, the Chartis™ system does not clearly indicate whether collateral ventilation is present because of blockage of the catheter by secretions, apposition of the catheter against the airway wall, or anatomical factors.34,35 Indeed, in seven of our 43 patients (16.3%), we could not come to a clear conclusion regarding collateral ventilation of the target lobe. These patients exhibited dynamic expiratory airway collapse or close bifurcation to place the catheter. In these patients, collateral ventilation of the adjacent lobe was tested. Since collateral ventilation in those adjacent lobes was not detected, the patients were deemed eligible for EBV insertion. In these seven patients, four achieved TLVR. Thus, in cases of inconclusive results regarding target lobe collateral ventilation, the results of the adjacent lobes could serve as a surrogate.
The prevalence of tuberculosis is higher in Korea than in Western countries. Together with smoking, it is a major risk factor for COPD in Asia. In our study, a relatively large proportion of patients (23.3%) had a past history of tuberculosis. Patients with emphysema in tuberculosis-prevalent countries such as Korea can have more heterogeneous phenotypes, and they exhibit hyperinflation at once, and their lung volume is reduced due to the presence of the inflammatory scar. Since the main effects of BLVR are due to the reduction of hyperinflation, its efficacy can be decreased by the presence of other pathogeneses such as fibrosis or previous inflammation associated with dyspnea. It is also possible that these pathogeneses elevate the risk of pneumothorax because lobes that bear scars can resist expansion. However, our data showed that the tuberculosis scar did not increase the achievement of TLVR or the development of pneumothorax. This may reflect the fact that in all nine patients with a tuberculosis scar, the scar affected lesser than 10% of the relevant lobes. This suggests that old inflammatory scars should not contraindicate BLVR if they are not widespread.
The number of adverse events in our study was modest compared to other studies.19,20,28 However, the pneumothorax rate was higher than that seen in previous trials (23.3% vs 10.6%).29 This can be explained by several points. First, strict selection criteria were applied for patients who were expected to achieve TLVR. We excluded not only patients with incomplete fissure on CT, but also patients with collateral ventilation on Chartis™ system even though they had complete fissure. In previous studies, patients were selected without confirmation of collateral ventilation20,28,29 or complete fissure.19 Our strict strategy resulted in a higher rate of TLVR, followed by a higher rate of pneumothorax. In a recent review with lobar exclusion, 30% of a pneumothorax rate was also reported, which is consistent with our report.36 Second, the differences between the patient cohorts in terms of baseline characteristics such as the patients in our study were older and had a lower BMI and a poorer exercise capacity. Moreover, although our patients had a lower baseline FEV1 than those in the other cohorts, they also had less hyperinflation, as indicated by the RV. This together with the patient demographics suggests that the patients in our study could be particularly vulnerable to complications, such as pneumothorax. EBV is quite expensive compared to other medical cost in Korea and is not yet covered by the national insurance system. This may have caused patients to select BLVR as the last resort. This may reflect the fact that Asians tend to have smaller body sizes, including height or thoracic cage dimensions, than Westerners.
Our study has some limitations. First, the sample size was relatively small. This reflects the expense of BLVR with EBV in Korea, which caused many patients and their guardians to decide against the procedure during screening. It also reflects the fact that the physicians had to be very constrained in their selection of patients to maximize cost-effectiveness. Second, many patients were lost to follow-up during the study period. This study was performed during the introduction of BLVR in Korea and, thus, the patients were referred from all around the country. Many of the patients who resided far from our center had difficulty in visiting regularly. Third, a significant number of patients were not assessed by 6MWD and SGRQ, although patients were followed up and were tested by spirometry and chest X-ray at 3 and 6 months. Spirometry, 6MWD, and SGRQ were performed on the same day. Because even spirometry was hard to perform for advanced emphysema patients, many patients refused to undertake 6MWD sequentially after spirometry. Furthermore, response to SGRQ was poor because of its lengthy questions in many patients.
Conclusion
The efficacy and safety of BLVR with EBV in Korea, an Asian country, were acceptable and consistent with the results of previous studies in Western countries. Thus, ethnicity and geographical area do not seem to affect the outcomes of BLVR as long as the clinical predictors of a response are present. However, the socioeconomic and medical environments can shape patient selection, which in turn could affect the results of procedure. Thus, the effort to reduce economic burden by reimbursement or cost-saving measures can expand the benefits of BLVR, especially in countries that have recently imported this treatment.
Acknowledgments
This study was supported by a grant from the National Strategic Coordinating Center for Clinical Research (HI10C2020) and the Korean Health Technology R&D Project (HI14C3339), Ministry for Health and Welfare Affairs, Republic of Korea. We would like to thank Yee Hyung Kim (Kyung Hee University Hospital at Gangdong, Seoul, Korea) and Junghee Lee (Asan Medical Center, Seoul, Korea) for their support in the statistical analyses.
Disclosure
The author reports no conflicts of interest in this work.
References
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442. | ||
Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187(4):347–365. | ||
Lee JH, Lee YK, Kim EK, et al. Responses to inhaled long-acting beta-agonist and corticosteroid according to COPD subtype. Respir Med. 2009;104(4):542–549. | ||
Cooper JD, Patterson GA, Sundaresan RS, et al. Results of 150 consecutive bilateral lung volume reduction procedures in patients with severe emphysema. J Thorac Cardiovasc Surg. 1996;112(5):1319–1329. discussion 1329–1330. | ||
Cooper JD, Trulock EP, Triantafillou AN, et al. Bilateral pneumectomy (volume reduction) for chronic obstructive pulmonary disease. J Thorac Cardiovasc Surg. 1995;109(1):106–116. discussion 116–109. | ||
Fishman A, Martinez F, Naunheim K, et al; National Emphysema Treatment Trial Research Group. A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med. 2003;348(21):2059–2073. | ||
Herth FJ, Gompelmann D, Ernst A, Eberhardt R. Endoscopic lung volume reduction. Respiration. 2010;79(1):5–13. | ||
Salvi SS, Barnes PJ. Chronic obstructive pulmonary disease in non-smokers. Lancet. 2009;374(9691):733–743. | ||
Bhome AB, Brashier B. Profiles of chronic obstructive lung disease: characteristics of stable chronic obstructive lung disease in different parts of Asia. Curr Opin Pulm Med. 2014;20(2):165–172. | ||
Eisner MD, Anthonisen N, Coultas D, et al; Committee on Nonsmoking COPD, Environmental and Occupational Health Assembly. An official American Thoracic Society public policy statement: novel risk factors and the global burden of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2010;182(5):693–718. | ||
Lee SW, Kim YS, Kim DS, Oh YM, Lee SD. The risk of obstructive lung disease by previous pulmonary tuberculosis in a country with intermediate burden of tuberculosis. J Korean Med Sci. 2011;26(2): 268–273. | ||
Oh YM, Bhome AB, Boonsawat W, et al. Characteristics of stable chronic obstructive pulmonary disease patients in the pulmonology clinics of seven Asian cities. Int J Chron Obstruct Pulmon Dis. 2013;8: 31–39. | ||
Wu J, Dalal K. Tuberculosis in Asia and the pacific: the role of socioeconomic status and health system development. Int J Prev Med. 2012;3(1):8–16. | ||
Menezes AM, Hallal PC, Perez-Padilla R, et al; Latin American Project for the Investigation of Obstructive Lung Disease (PLATINO) Team. Tuberculosis and airflow obstruction: evidence from the PLATINO study in Latin America. Eur Respir J. 2007;30(6):1180–1185. | ||
Elkington PT, Friedland JS. Matrix metalloproteinases in destructive pulmonary pathology. Thorax. 2006;61(3):259–266. | ||
Park TS, Hong Y, Lee JS, et al. Efficacy of bronchoscopic lung volume reduction by endobronchial valves in patients with heterogeneous emphysema: report on the first Asian cases. J Korean Med Sci. 2014; 29(10):1404–1410. | ||
Flandes Aldeyturriaga J. Bronchoscopic lung volume reduction: 7 lessons learned. Arch Bronconeumol. 2012;48(7):221–222. | ||
Strange C, Herth FJ, Kovitz KL, et al. Design of the endobronchial Valve for Emphysema Palliation Trial (VENT): a non-surgical method of lung volume reduction. BMC Pulm Med. 2007;7:10. | ||
Herth FJ, Eberhardt R, Gompelmann D, et al. Radiological and clinical outcomes of using Chartis to plan endobronchial valve treatment. Eur Respir J. 2013;41(2):302–308. | ||
Herth FJ, Noppen M, Valipour A, et al; International VENT Study Group. Efficacy predictors of lung volume reduction with Zephyr valves in a European cohort. Eur Respir J. 2012;39(6):1334–1342. | ||
Gompelmann D, Eberhardt R, Michaud G, Ernst A, Herth FJ. Predicting atelectasis by assessment of collateral ventilation prior to endobronchial lung volume reduction: a feasibility study. Respiration. 2010;80(5):419–425. | ||
Chae EJ, Kim N, Seo JB, et al. Prediction of postoperative lung function in patients undergoing lung resection: dual-energy perfusion computed tomography versus perfusion scintigraphy. Invest Radiol. 2013;48(8):622–627. | ||
Chae EJ, Seo JB, Jang YM, et al. Dual-energy CT for assessment of the severity of acute pulmonary embolism: pulmonary perfusion defect score compared with CT angiographic obstruction score and right ventricular/left ventricular diameter ratio. AJR Am J Roentgenol. 2010;194(3):604–610. | ||
Jones PW, Beeh KM, Chapman KR, Decramer M, Mahler DA, Wedzicha JA. Minimal clinically important differences in pharmacological trials. Am J Respir Crit Care Med. 2014;189(3):250–255. | ||
Donohue JF. Minimal clinically important differences in COPD lung function. COPD. 2005;2(1):111–124. | ||
Puhan MA, Chandra D, Mosenifar Z, et al; National Emphysema Treatment Trial (NETT) Research Group. The minimal important difference of exercise tests in severe COPD. Eur Respir J. 2011;37(4):784–790. | ||
Jones PW. St George’s respiratory questionnaire: MCID. COPD. 2005;2(1):75–79. | ||
Sciurba FC, Ernst A, Herth FJ, et al; VENT Study Research Group. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med. 2010;363(13):1233–1244. | ||
Gompelmann D, Herth FJ, Slebos DJ, et al. Pneumothorax following endobronchial valve therapy and its impact on clinical outcomes in severe emphysema. Respiration. 2014;87(6):485–491. | ||
Venuta F, Anile M, Diso D, et al. Long-term follow-up after bronchoscopic lung volume reduction in patients with emphysema. Eur Respir J. 2012;39(5):1084–1089. | ||
Valipour A, Herth FJ, Burghuber OC, et al; VENT Study Group. Target lobe volume reduction and COPD outcome measures after endobronchial valve therapy. Eur Respir J. 2014;43(2):387–396. | ||
Shah PL, Herth FJ. Current status of bronchoscopic lung volume reduction with endobronchial valves. Thorax. 2014;69(3):280–286. | ||
Reymond E, Jankowski A, Pison C, et al. Prediction of lobar collateral ventilation in 25 patients with severe emphysema by fissure analysis with CT. AJR Am J Roentgenol. 2013;201(4):W571–W575. | ||
Gompelmann D, Eberhardt R, Herth FJ. Collateral ventilation. Respiration. 2013;85(6):515–520. | ||
Shah PL, Herth FJ. Dynamic expiratory airway collapse and evaluation of collateral ventilation with Chartis. Thorax. 2014;69(3):290–291. | ||
Valipour A, Slebos DJ, de Oliveira HG, et al. Expert statement: pneumothorax associated with endoscopic valve therapy for emphysema – potential mechanisms, treatment algorithm, and case examples. Respiration. 2014;87(6):513–521. |
Supplementary materials
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.