Back to Journals » OncoTargets and Therapy » Volume 13
Bioinformatics Analysis to Reveal Potential Differentially Expressed Long Non-Coding RNAs and Genes Associated with Tumour Metastasis in Lung Adenocarcinoma
Authors Yao X, Zhang H, Tang S, Zheng X, Jiang L
Received 17 December 2019
Accepted for publication 3 April 2020
Published 16 April 2020 Volume 2020:13 Pages 3197—3207
DOI https://doi.org/10.2147/OTT.S242745
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sanjeev K. Srivastava
Xiaojun Yao, 1, 2 Hongwei Zhang, 2 Shujun Tang, 2 Xinglong Zheng, 2 Liangshuang Jiang 1
1Department of Thoracic Surgery, The Public Health Clinical Center of Chengdu, Chengdu, Republic of China; 2Department of Thoracic Surgery, Meishan Cancer Hospital, Chengdu, People’s Republic of China
Correspondence: Liangshuang Jiang
Department of Thoracic Surgery, The Public Health Clinical Center of Chengdu, No. 18, Jingjusi Road, Jinjiang District, Chengdu 610061, People’s Republic of China
Tel +86-18980070581
Fax +86-28-84537477
Email [email protected]
Background: Due to the onset of metastases, the survival rate of lung adenocarcinoma (LUAD) is still low. In view of this, we performed this study to screen metastasis-associated genes and lncRNAs in LUAD.
Methods: The mRNA and lncRNA expression profiles of 185 metastatic LUAD and 217 non-metastatic LAUD samples were retrieved from the TCGA database and included in this study. The differentially expressed mRNAs (DEmRNAs) and lncRNAs (DElncRNAs) between metastatic samples and non-metastatic samples of LAUD, as well as the cis nearby-targeted DEmRNAs of DElncRNAs and the DElncRNA-DEmRNA co-expression network, were obtained. Quantitative real-time polymerase chain reaction (qRT-PCR) was used to detect the expression levels of selected DEmRNAs. Survival analysis of selected DElncRNAs and DEmRNAs was performed.
Results: In total, 1351 DEmRNAs and 627 DElncRNAs were screened between the LUAD primary tissue samples and metastatic samples. Then, 194 DElncRNA-nearby-targeted DEmRNA pairs and 191 DElncRNA-DEmRNA co-expression pairs were detected. Except for RHCG and KRT81, the expression of the other six DEmRNAs in the qRT-PCR results generally exhibited the same pattern as that in our integrated analysis. The expression of CRHR2, FAM83A-AS1, FAM83A and Z83843.1 was significantly correlated with the overall survival time of patients with metastatic LUAD.
Conclusion: We speculate that two interaction pairs (FAM83A-AS1-FAM83A and Z83843.1-MATR3) and four genes (CRHR2, UGT2B15, CHGB and NEFL) are closely associated with the metastasis of LUAD.
Keywords: lung adenocarcinoma, LUAD, metastasis, TCGA, long non-coding RNA, lncRNA
Corrigendum for this paper has been published
Introduction
Lung cancer is a prevalent malignancy worldwide and is divided into two main histological subtypes: small-cell lung carcinoma (SCLC) and non-small-cell lung carcinoma (NSCLC); the latter includes large cell lung cancer, lung squamous cell carcinoma (LSCC) and lung adenocarcinoma (LUAD), and LUAD is one of the most common types of lung cancer.1 LUAD is difficult to diagnose due to its small tumour size, rapid invasion and metastasis in the early stage.2 Despite the use of chemotherapy and radiotherapy for LUAD, the survival rate is still low, and LUAD accounts for 90% of cancer-related deaths.3 Importantly, the underlying mechanisms of LUAD cell invasion and metastasis are still not fully understood.4 Hence, a better understanding of metastatic progression in LUAD may result in improved treatment for patients.
Recently, long non-coding RNAs (lncRNAs) have become novel and popular topics in the field of cancer treatment. Genome-wide transcriptomic studies over the past decade have generated a large quantity of information about lncRNAs, facilitating the understanding of the aetiology of diverse cancers, including LUAD, at the molecular level.5 It has been reported that highly expressed SPRY4-IT1 promotes tumour cell migration and invasion in LUAD.6 A group of seven-lncRNAs was reported to predict survival in the early stage of LUAD.7 DKFZP434 L187 and LOC285548 may serve as prognostic markers for LUAD patients.8 Investigation of disease-associated genes can improve the understanding of disease aetiology and development, thereby facilitating the design and development of novel preventive and treatment strategies.
In this work, we performed a study, involving 185 metastatic LUAD and 217 non-metastatic LAUD samples collected from the TCGA database, to investigate metastasis-associated genes and lncRNAs in LUAD. In addition to the acquisition of differentially expressed mRNAs (DEmRNAs) and lncRNAs (DElncRNAs), identification of cis nearby-targeted DEmRNAs of DElncRNAs and construction of DElncRNA-DEmRNA co-expression network were performed. In doing this work, we expected to contribute to expanding the knowledge on LUAD.
Materials and Methods
Data Acquisition and Analysis of mRNA and lncRNA Expression Profiles
The mRNA and lncRNA expression data, including data for 185 metastatic LUAD and 217 non-metastatic LAUD samples, were collected from the TCGA database (http://tcga-data.nci.nih.gov/). Table 1 displays the clinical characteristics of all these patients. With DESeq2 (http://bioconductor.org/packages/DESeq2/) in R version 3.5.1, both DEmRNAs and DElncRNAs between metastatic LUAD and non-metastatic LAUD were acquired with false discovery rate (FDR) < 0.01. By using the R package “pheatmap”, hierarchical clustering analysis of DEmRNAs and DElncRNAs was conducted.
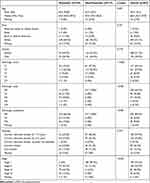 |
Table 1 Clinical Characteristics of LUAD Patients |
Functional Annotation and Protein–Protein Interaction (PPI) Network Construction of DEmRNAs
Gene Ontology (GO) classification and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis of DEmRNAs were conducted with CPDB (http://cpdb.molgen.mpg.de/CPDB). A value of p < 0.01 was set as the cut-off for significance. The top 100 up- and down-regulated DEmRNAs were searched with BioGrid (http://www.uniprot.org/database/DB-0184). Then, PPI networks were constructed with Cytoscape software (version 3.6.1, http://www.cytoscape.org).
Cis Nearby-Targeted DEmRNAs of the DElncRNAs
DEmRNAs transcribed within a 100-kb window upstream or downstream of DElncRNAs, which were defined as cis nearby-targeted DEmRNAs of DElncRNAs, were searched to obtain the targeted DEmRNAs of DElncRNAs with cis-regulatory effects. GO classification and KEGG enrichment analysis of cis nearby-targeted DEmRNAs of DElncRNAs were performed by using CPDB with p-value < 0.01.
DElncRNA-DEmRNA Co-Expression Network
Pearson’s correlation coefficient (PCC) of each DElncRNA-DEmRNA pair was calculated. DElncRNA-DEmRNA pairs with an absolute value of PCC > 0.7 and p-value < 0.01 were defined as co-expressed DElncRNA-DEmRNA pairs. By using Cytoscape, DElncRNA-DEmRNA co-expression networks were constructed.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Validation
Thirteen samples were collected from 7 patients with non-metastatic LUAD and 6 patients with metastatic LUAD. We obtained written informed consent from every participant. The present study was approved by the Medical Ethics Committee of Meishan Cancer Hospital (201,908) and in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Total RNA was isolated with Trizol reagent (Invitrogen, USA). The qRT-PCR reactions were performed based on SuperReal PreMix Plus (Invitrogen, USA) in an ABI 7500 Real-time PCR Detection System. With the 2-ΔΔCt method, relative gene expression was determined. Human GAPDH and ACTB were treated as endogenous controls in the analysis.
Survival Analysis of Selected DElncRNAs and DEmRNAs
To further investigate the prognostic value of selected DElncRNAs and DEmRNAs, survival analysis was performed by using the survival package (https://cran.r-project.org/web/packages/survival/index.html) in R.
Results
Identification of DEmRNAs and DElncRNAs in Tumour Tissues Between Metastatic LUAD and Non-Metastatic LAUD
In total, 1351 DEmRNAs (882 up- and 469 down-regulated DEmRNAs) and 627 DElncRNAs (493 up- and 134 down-regulated DElncRNAs) were screened out between the LUAD primary tissue samples and metastatic samples. The top 10 up- and down-regulated DEmRNAs and DElncRNAs are shown in Tables 2 and 3, respectively. Hierarchical clustering analysis of top 100 up- and down-regulated DEmRNAs and DElncRNAs is shown in Figure 1A and B, respectively.
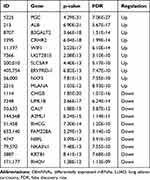 |
Table 2 Top 10 Up- and Down-Regulated DEmRNAs in Tumor Tissues Between Metastatic LUAD and Non-Metastatic LUAD |
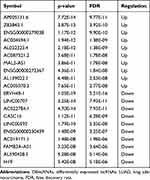 |
Table 3 Top 10 Up- and Down-Regulated DElncRNAs in Tumor Tissues Between Metastatic LUAD and Non-Metastatic LUAD |
Functional Annotation and PPI Network of DEmRNAs
Regulation of cellular process (p = 8.84E-06), aromatic compound biosynthetic process (p = 1.05E-04), extracellular matrix (p = 4.31E-07), ion binding (p = 4.86E-12) and cation binding (p = 4.56E-10) were the significantly enriched GO terms in metastatic samples (Figure 2A–C). Glycolysis/Gluconeogenesis (p = 7.04E-04), p53 signaling pathway (p = 2.29E-03) and DNA replication (p = 2.77E-03) were significantly enriched KEGG pathways in metastatic samples (Figure 2D). The PPI network included 102 nodes and 81 edges. ALB (degree = 19), MATR3 (degree = 12) and CYLD (degree = 6) were three hub proteins of the PPI network (Figure 3).
Cis Nearby-Targeted DEmRNAs of the DElncRNAs
A total of 194 DElncRNA-nearby-targeted DEmRNA pairs, involving in 156 DElncRNAs and 166 DEmRNAs, were detected (Figure 4). Modification by symbiont of host morphology or physiology (p = 5.42E-04), regulation of cellular metabolic process (p = 9.76E-04), nucleus (p = 2.10E-04), SUMO ligase activity (p = 6.86E-04) and DNA binding (p = 1.01E-03) were the significantly enriched GO terms (Figure S1A–C). Cholinergic synapse (p = 2.40E-03) and Aldosterone synthesis and secretion (p = 8.35E-03) were the significantly enriched KEGG pathways (Figure S1D).
DElncRNA-DEmRNA Co-Expression Network
A total of 191 DElncRNA-DEmRNA co-expression pairs including 56 DElncRNAs and 108 DEmRNAs were obtained with an absolute value of PCC > 0.7 and p < 0.01 (Figure 5). Chromosome segregation (p = 4.43E-14), mitotic cell cycle process (p = 1.52E-13), chromosomal region (p = 3.90E-09), heterocyclic compound binding (p = 4.38E-05) and organic cyclic compound binding (p = 6.63E-05) were the significantly enriched GO terms (Figure S2A–C). P53 signaling pathway (p = 5.59E-04), Cell cycle (p = 6.39E-04) and Amphetamine addiction (p = 6.48E-03) were the significantly enriched KEGG pathways (Figure S2D).
QRT-PCR Validation
Eight DEGs, including CHGB, CRHR2, A2ML1, RHCG, UGT2B15, NEFL, KRT81 and RHOV, were selected for qRT-PCR validation. In our integrated analysis, CRHR2 and UGT2B15 were up-regulated while the other six DEGs were down-regulated in metastatic LUAD. Except for RHCG and KRT81, the expression of the others in the qRT-PCR results generally exhibited the same pattern as that in our integrated analysis (Figure 6).
 |
Figure 6 The qRT-PCR results of selected DEmRNAs. The x-axis represents the DEmRNAs and the y-axis represents log2 (fold change). |
Survival Analysis
Survival analysis was performed to evaluate the prognostic value of two DElncRNAs (FAM83A-AS1 and Z83843.1) and six DEmRNAs (FAM83A, MATR3, CRHR2, UGT2B15, CHGB and NEFL). On the basis of the results of survival analysis, the expression of CRHR2 (p = 0.046), FAM83A-AS1 (p =0.001), FAM83A (p =0.0021) and Z83843.1 (p =0.036) was significantly correlated with the overall survival time of patients with metastatic LUAD (Figure S3A–D), while the expression of CHGB (p = 0.0051), CRHR2 (p = 0.047), FAM83A (p = 0.044) and NEFL (p < 0.0001) was significantly correlated with the overall survival time of patients with non-metastatic LUAD (Figure S3E–H). Of note, CRHR2 and FAM83A were involved not only in metastatic LUAD but also in non-metastatic LUAD.
Discussion
Although LUAD is a common type of lung cancer, its mechanisms underlying its oncogenesis and progression remain unclear. This present work utilized an analysis to identify key dysregulated lncRNAs and genes associated with metastatic progression in LUAD.
Family with sequence similarity 83 member A (FAM83A), a member of the FAM83 family, is located on chromosome 8, locus q24.13.9 Numerous studies have linked FAM83A to lung cancer in recent years. A previous study revealed that FAM83A was associated with metastasis and recurrence in lung cancer.10 Li et al identified that FAM83A was overexpressed in both smoking and nonsmoking groups of patients with LUAD.11 Zhang et al indicated that overexpression of FAM83A may predict poor prognosis in LUAD patients.9 Wang et al demonstrated that increased FAM83A was negatively associated with poor survival in LUAD, which was consistent with our survival analysis result.12 FAM83A-AS1, a natural antisense transcript (NAT) RNA, was reported to promote cell proliferation, migration, invasion and the epithelial-mesenchymal transition (EMT) in LUAD.13 Shi et al reported that FAM83A-AS1 could promote LUAD by elevating FAM83A expression.14 In this analysis, FAM83A-AS1 was significantly down-regulated in the metastatic LUAD group compared with the non-metastatic LUAD group, and FAM83A was a nearby-targeted and co-expressed DEmRNA of FAM83A-AS1. These results may indicate the critical role of the FAM83A-AS1-FAM83A interaction pair in the metastatic progression of LUAD.
Matrin 3 (MATR3) encodes a nuclear matrix protein that is widely expressed in various tissues and can bind to DNA and RNA via zinc finger domains and RNA recognition motifs.15 Mutation in MATR3 was associated with amyotrophic lateral sclerosis (ALS).16 Yang et al demonstrated that highly expressed MATR3 was associated with poor survival of patients with neuroblastoma and that it may enhance neuroblastoma progression by interacting with lncRNA SNHG1.17 Nho et al suggested that MATR3 was involved in carcinogenesis progression by regulating apoptosis and colony formation in oral squamous cell carcinoma (OSCC).15 MATR3 was a hub gene in the PPI network in our study, although there is no previous report linking MATR3 with lung cancer. On the other hand, Z83843.1 was significantly up-regulated in the metastatic LUAD group compared with the non-metastatic LUAD group, which was also an lncRNA that covered most DEmRNAs, including MATR3, in the DElncRNA-DEmRNA co-expression network. The discovery of the Z83843.1-MATR3 interaction pair reminders us to focus on its role in the development of LUAD.
Corticotropin-releasing hormone receptor 2 (CRHR2), located at 7p21-p15, consists of 12 exons, and alternative splicing of the first exon gives rise to three isoforms.18 The protein encoded by this gene is a G-protein coupled receptor that belongs to the subfamily of corticotropin releasing hormone receptors, with nearly 71% amino acid sequence similarity with CRHR1, and is known as a receptor of corticotropin-releasing hormone (CRH).18 A correlation between the rs2267716 polymorphism in CRHR2 and hepatocellular carcinoma (HCC) in patients with chronic hepatitis C virus (HCV) or HBV infection was proposed.18 Rodriguez et al reported that decreased CRHR2 promotes tumour growth and EMT in colorectal cancer (CRC).19 UDP glucuronosyltransferase family 2 member B15 (UGT2B15) encodes a member of the uridine diphosphate-glucuronosyltransferase (UGT) family, and is involved in several metabolic pathways, such as, pentose and glucuronate interconversion, steroid hormone biosynthesis, and retinol metabolism.20 Chen et al reported that UGT2B15 was up-regulated in gastric cancer (GC), which may be an oncogene in GC.21 Decreased expression levels of UGT2B15 were detected in prostate cancer, and were associated with lymph node metastases.22 In this study, both CRHR2 and UGT2B15 were significantly up-regulated in metastatic LUAD samples, which may suggest that high expression of CRHR2 and UGT2B15 is involved LUAD metastasis.
Chromogranin B (CHGB) encodes a tyrosine-sulfated secretory protein that is expressed abundantly in peptidergic endocrine cells and neurons and was first described in pheochromocytoma in 1987.23,24 Decreased cytoplasmic CHGB expression was found in primary tumours of patients with the pancreatic neuroendocrine tumour (PNET) phenotype in Von Hippel-Lindau (VHL) syndrome with metastatic disease compared with those without metastatic disease.25 CHGB was reported to be associated with prolactin pituitary (PRL) tumour metastasis.26 Stenman et al suggested that CHGB may serve as a possible preoperative and postoperative marker for pheochromocytomas (PCCs) and abdominal paragangliomas (PGLs) with potential for aggressive behaviour.23 Neurofilament light (NEFL) encodes the light chain neurofilament protein, which functionally maintains the neuronal caliber and plays a role in intracellular transport to axons and dendrites.27 It is located at chromosome 8p21, a region reported to be enriched with tumour suppressor genes (TSGs).28 Lower NEFL expression was found in lymph node metastases than in paired primary breast cancer tissues.29 Oh et al reported that the expression level of NEFL was greatly decreased and may be associated with the course of colorectal cancer (CRC) progression and liver metastasis.30 In addition, methylation of NEFL was reported to be a novel mechanism for NSCLC invasion and metastasis.31 Notedly, both CHGB and NEFL were significantly down-regulated in metastatic LUAD samples. Hence, we speculated that CHGB and NEFL may contribute to LUAD progression and are involved in the metastasis of LUAD.
In conclusion, a total of 1351 DEmRNAs and 627 DElncRNAs were detected in LUAD metastatic samples compared with non-metastatic samples. We emphasized the crucial roles of two interaction pairs (FAM83A-AS1-FAM83A and Z83843.1-MATR3) and four genes (CRHR2, UGT2B15, CHGB and NEFL) in the metastasis of LUAD. Our study had the limitation of a small sample size for validation. Due to this limitation, although the qRT-PCR results suggested that our integrated analysis results were generally convincing, the validation results were not significant. More samples will be collected and more in deep research on functional experiments will be included in our future work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Qin Y, Zhou X, Huang C, et al. Lower miR-340 expression predicts poor prognosis of non-small cell lung cancer and promotes cell proliferation by targeting CDK4. Gene. 2018;675:278–284. doi:10.1016/j.gene.2018.06.062
2. Spiro SG, Tanner NT, Silvestri GA, et al. Lung cancer: progress in diagnosis, staging and therapy. Respirology. 2010;15(1):44–50. doi:10.1111/j.1440-1843.2009.01674.x
3. Zhang P, Sun J, Kai J, et al. ASAP3 is a downstream target of HIF-1alpha and is critical for progression of lung adenocarcinoma. Onco Targets Ther. 2019;12:5793–5803. doi:10.2147/OTT.S199603
4. Ren W, Mi D, Yang K, et al. The expression of hypoxia-inducible factor-1alpha and its clinical significance in lung cancer: a systematic review and meta-analysis. Swiss Med Wkly. 2013;143:w13855.
5. Wang Y, Wu N, Liu J, Wu Z, Dong D. FusionCancer: a database of cancer fusion genes derived from RNA-seq data. Diagn Pathol. 2015;10:131. doi:10.1186/s13000-015-0310-4
6. Zhang X, Chi Q, Zhao Z. Up-regulation of long non-coding RNA SPRY4-IT1 promotes tumor cell migration and invasion in lung adenocarcinoma. Oncotarget. 2017;8(31):51058–51065. doi:10.18632/oncotarget.16918
7. Chen M, Liu B, Xiao J, Yang Y, Zhang Y. A novel seven-long non-coding RNA signature predicts survival in early stage lung adenocarcinoma. Oncotarget. 2017;8(9):14876–14886. doi:10.18632/oncotarget.14781
8. Li L, Feng T, Qu J, et al. LncRNA expression signature in prediction of the prognosis of lung adenocarcinoma. Genet Test Mol Biomarkers. 2018;22(1):20–28. doi:10.1089/gtmb.2017.0194
9. Zhang JT, Lin YC, Xiao BF, Yu BT. Overexpression of family with sequence similarity 83, member A (FAM83A) predicts poor clinical outcomes in lung adenocarcinoma. Med Sci Monit. 2019;25:4264–4272. doi:10.12659/MSM.910804
10. Li Y, Dong X, Yin Y, et al. BJ-TSA-9, a novel human tumor-specific gene, has potential as a biomarker of lung cancer. Neoplasia. 2005;7(12):1073–1080. doi:10.1593/neo.05406
11. Li Y, Xiao X, Ji X, Liu B, Amos CI. RNA-seq analysis of lung adenocarcinomas reveals different gene expression profiles between smoking and nonsmoking patients. Tumor Biol. 2015;36(11):8993–9003. doi:10.1007/s13277-015-3576-y
12. Wang Y, Lu T, Wo Y, et al. Identification of a putative competitive endogenous RNA network for lung adenocarcinoma using TCGA datasets. Peer J. 2019;7:e6809. doi:10.7717/peerj.6809
13. Xiao G, Wang P, Zheng X, Liu D, Sun X. FAM83A-AS1 promotes lung adenocarcinoma cell migration and invasion by targeting miR-150-5p and modifying MMP14. Cell Cycle. 2019;18:2972–2985.
14. Shi R, Jiao Z, Yu A, Wang T. Long noncoding antisense RNA FAM83A-AS1 promotes lung cancer cell progression by increasing FAM83A. J Cell Biochem. 2019;120(6):10505–10512. doi:10.1002/jcb.28336
15. Nho SH, Yoon G, Seo JH, et al. Licochalcone H induces the apoptosis of human oral squamous cell carcinoma cells via regulation of matrin 3. Oncol Rep. 2019;41(1):333–340. doi:10.3892/or.2018.6784
16. Johnson JO, Pioro EP, Boehringer A, et al. Mutations in the Matrin 3 gene cause familial amyotrophic lateral sclerosis. Nat Neurosci. 2014;17(5):664–666. doi:10.1038/nn.3688
17. Yang TW, Sahu D, Chang YW, Hsu CL, Hsieh CH. RNA-binding proteomics reveals MATR3 interacting with lncRNA SNHG1 to enhance neuroblastoma progression. J Proteome Res. 2019;18(1):406–416.
18. Gu X, Qi P, Zhou F, et al. An intronic polymorphism in the corticotropin-releasing hormone receptor 2 gene increases susceptibility to HBV-related hepatocellular carcinoma in Chinese population. Hum Genet. 2010;127(1):75–81. doi:10.1007/s00439-009-0750-6
19. Rodriguez JA, Huerta-Yepez S, Law IK, et al. Diminished expression of CRHR2 in human colon cancer promotes tumor growth and EMT via persistent IL-6/Stat3 signaling. Cell Mol Gastroenterol Hepatol. 2015;1(6):610–630. doi:10.1016/j.jcmgh.2015.08.001
20. Kudryavtseva AV, Lukyanova EN, Kharitonov SL, et al. Bioinformatic identification of differentially expressed genes associated with prognosis of locally advanced lymph node-positive prostate cancer. J Bioinform Comput Biol. 2019;17(1):1950003. doi:10.1142/S0219720019500033
21. Chen X, Li D, Wang N, et al. Bioinformatic analysis suggests that UGT2B15 activates the HippoYAP signaling pathway leading to the pathogenesis of gastric cancer. Oncol Rep. 2018;40(4):1855–1862. doi:10.3892/or.2018.6604
22. Grosse L, Paquet S, Caron P, et al. Androgen glucuronidation: an unexpected target for androgen deprivation therapy, with prognosis and diagnostic implications. Cancer Res. 2013;73(23):6963–6971. doi:10.1158/0008-5472.CAN-13-1462
23. Stenman A, Svahn F, Hojjat-Farsangi M, et al. Molecular profiling of pheochromocytoma and abdominal paraganglioma stratified by the PASS algorithm reveals chromogranin B as associated with histologic prediction of malignant behavior. Am J Surg Pathol. 2019;43(3):409–421. doi:10.1097/PAS.0000000000001190
24. Benedum UM, Lamouroux A, Konecki DS, et al. The primary structure of human secretogranin I (chromogranin B): comparison with chromogranin A reveals homologous terminal domains and a large intervening variable region. EMBO J. 1987;6(5):1203–1211. doi:10.1002/j.1460-2075.1987.tb02355.x
25. Weisbrod AB, Zhang L, Jain M, Barak S, Quezado MM, Kebebew E. Altered PTEN, ATRX, CHGA, CHGB, and TP53 expression are associated with aggressive VHL-associated pancreatic neuroendocrine tumors. Horm Cancer. 2013;4(3):165–175. doi:10.1007/s12672-013-0134-1
26. Zhang W, Zang Z, Song Y, Yang H, Yin Q. Co-expression network analysis of differentially expressed genes associated with metastasis in prolactin pituitary tumors. Mol Med Rep. 2014;10(1):113–118. doi:10.3892/mmr.2014.2152
27. Leermakers FA, Zhulina EB. How the projection domains of NF-L and alpha-internexin determine the conformations of NF-M and NF-H in neurofilaments. Eur Biophys J. 2010;39(9):1323–1334. doi:10.1007/s00249-010-0585-z
28. Huang Z, Zhuo Y, Shen Z, et al. The role of NEFL in cell growth and invasion in head and neck squamous cell carcinoma cell lines. J Oral Pathol Med. 2014;43(3):191–198. doi:10.1111/jop.12109
29. Feng Y, Sun B, Li X, et al. Differentially expressed genes between primary cancer and paired lymph node metastases predict clinical outcome of node-positive breast cancer patients. Breast Cancer Res Treat. 2007;103(3):319–329. doi:10.1007/s10549-006-9385-7
30. Oh BY, Cho J, Hong HK, et al. Exome and transcriptome sequencing identifies loss of PDLIM2 in metastatic colorectal cancers. Cancer Manag Res. 2017;9:581–589. doi:10.2147/CMAR.S149002
31. Shen Z, Chen B, Gan X, et al. Methylation of neurofilament light polypeptide promoter is associated with cell invasion and metastasis in NSCLC. Biochem Biophys Res Commun. 2016;470(3):627–634. doi:10.1016/j.bbrc.2016.01.094
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.