Back to Journals » International Journal of General Medicine » Volume 14
Bicarbonate Therapy in Renally Compromised Critically Ill Patients with Metabolic Acidosis: Study of Clinical Outcomes and Mortality Rate
Authors Rafique Z, Tariq MH , Khan A, Farrukh MJ , Khan N, Burki AM, Mehmood K
Received 7 December 2020
Accepted for publication 8 March 2021
Published 24 June 2021 Volume 2021:14 Pages 2817—2826
DOI https://doi.org/10.2147/IJGM.S296095
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Zakia Rafique,1 Muhammad Haseeb Tariq,2,3 Arif-ullah Khan,1 Muhammad Junaid Farrukh,4 Nida Khan,5 Ahmed Mujadid Burki,6 Khalid Mehmood7
1Riphah Institute of Pharmaceutical Sciences, Riphah International University, Islamabad, Pakistan; 2Department of Clinical Pharmacy, School of Pharmaceutical Sciences, Universiti Sains Malaysia, Gelugor Penang, Malaysia; 3Division of Pharmaceutical Evaluation & Registration, Drug Regulatory Authority of Pakistan (DRAP), Islamabad, Pakistan; 4Faculty of Pharmaceutical Sciences, UCSI University, Kuala Lumpur, Malaysia; 5Advanced Educational Institute & Research Center (AEIRC), Karachi, Pakistan; 6Combined Military Hospital (CMH), Rawalpindi, Pakistan; 7Pak Emirates Military Hospital (PEMH), Rawalpindi, Pakistan
Correspondence: Muhammad Haseeb Tariq
Division of Pharmaceutical Evaluation & Registration, Drug Regulatory Authority of Pakistan (DRAP), 4th Floor T.F Complex G-9/4, Islamabad, Pakistan
Email [email protected]
Muhammad Junaid Farrukh
Faculty of Pharmaceutical Sciences, UCSI University, Kuala Lumpur, Malaysia
Email [email protected]
Background: Metabolic acidosis is the most frequent medical condition occurring in critically ill renally compromised patients. This study was aimed to determine clinical outcomes of bicarbonate therapy in renally compromised critically ill patients having metabolic acidosis.
Methods: A prospective longitudinal cohort study was undertaken in three military hospitals in Rawalpindi, Pakistan. All patients fulfilling the inclusion criteria who were admitted to the ICU of any of the three study hospitals from July 2019 to March 2020 were studied for clinical outcomes of bicarbonate therapy using an evidence-based clinical checklist. Outcome measures include changes in blood pH, serum potassium, and sodium levels, blood pressure and weight, along with other clinically significant laboratory parameters.
Results: Eighty-one patients fulfilling the inclusion criteria were evaluated. The mean age of the patients was 55.61± 19.5 years, while the mean weight was 63.43± 14.19 Kg. A mortality rate of 45.7% was observed. Disease-related complications including hypoxia, cardiac failure, multiple organ failure, elevated blood pressure, and ischemic heart disease (IHD) were found to be associated with a higher mortality rate (P< 0.005). Whereas using Fisher’s exact test, concomitant administration of sodium chloride, along with bicarbonate therapy was associated with a low mortality rate and had no significant impact on sodium loading or weight gain. Moreover, various drug–drug interactions were found to be associated with a higher mortality rate (P< 0.05).
Conclusion: Bicarbonate therapy was not found to affect the mortality rate in critically ill renally compromised patients with metabolic acidosis.
Keywords: metabolic acidosis, chronic kidney disease, bicarbonate therapy, clinical outcomes, mortality rate
Introduction
Metabolic acidosis has become a significant ailment that is represented as inequity within the body’s acid-base equilibrium. It frequently occurs in critically ill patients with an overall prevalence of 8–64% where about 14–42% is due to acute acidemia.1–3 The significantly higher mortality rate of up to 57% has been reported among patients with persistent acidemia with poor prognosis.4 It affects 15% of Chronic Kidney Disease (CKD) patients with serum HCO3 level below 22 mEq/L and the percentage increases with the worsening of kidney function. As studied, acidosis has been observed in 7% of subjects in CKD stage 2, 13% in stage 3, and 37% in stage 4.5,6
Acidemia is defined as depletion in blood pH from 7.02 leading to low bicarbonate (-HCO3) ions concentration in the blood accompanied by reduced arterial pressure of CO2 production.7 Metabolic acidosis refers to bicarbonate loss through the kidneys (during impaired renal function) or sometimes during diarrhea. Forms of acidosis other than metabolic encompass lactic acidosis, respiratory and diabetic ketoacidosis.8,9 Sodium bicarbonate is the treatment of choice for metabolic acidosis in critically ill patients, which has been practiced in ICU patients for better clinical outcomes.7 Due to the lack of evidence based clinical studies, the use of bicarbonate therapy in acidosis patients is still controversial with little evidence to support its use. Furthermore, the therapy is proved to be insignificant in patients with lactic acidosis, hemodynamic and septic shocks.10
Some noticeable adverse effects of this therapeutic strategy include a decline in levels of calcium and potassium, but a surge in lactate production and sodium levels, prolonged QT interval, calcium accumulation in the vessels, and carbon dioxide generation.11 Among all, production of carbon dioxide is one of the harmful effects which leads to hypocalcaemia and becomes fatal to patients with decreased cardiac output and respiratory failure.11,12 However, use of this therapy may act as supportive therapy; for example, it may reduce the need for the renal transplant in most of the CKD patients, detrimental effects of acidosis on cardiac muscles and the supply of oxygen.13,14
Notably, in CKD patients, this treatment is found to be heterogeneous in some studies while many guidelines support its use in CKD and AKI patients.15 According to NICE guidelines, Bicarbonate therapy should be prescribed in patients with GFR less than 30 mL/min/1.73 (GFR category 4 or 5) and a serum bicarbonate concentration of 20 mmol/L.16 Similarly, KDIGO 2012 guidelines recommend the use of oral base therapy in CKD and AKI patients having serum Bicarb levels <22 mmol/L.17 However, lack of scientific data in this area of interest leaves physicians in the ICU in uncertainty whether to use this therapy in such patients or not.
This study was aimed to evaluate clinical outcomes in terms of safety and efficacy of the use of bicarbonate therapy in renally compromised critically ill patients with metabolic acidosis.
Methods
Ethical Approval
Ethical approval was obtained from the hospitals Ethics Review Board (ERB). Before taking part in the study all the participants were requested to sign a consent form, after the purpose and method of study was explained. In case the patient was unable to give informed consent, his guardians gave the permission on behalf of the patient. Confidentiality of patients’ data was in obedience with the Declaration of Helsinki as guaranteed by the institutional Review Boards of the hospitals.
Study Design
This prospective cohort longitudinal research has been conducted at three military hospitals in Rawalpindi Pakistan, ie, Combined Military Hospital (CMH), Pak Emirate Military Hospital (PEMH), and Armed Forces Institute of Urology (AFIU), Rawalpindi, Pakistan.
Study Sample and Sampling Technique
All patients fulfilling inclusion criteria which were admitted in CMH, PEMH, and AFIU from July 2019 to March 2020 were studied. A non-probability convenience sampling technique was used for sampling.
Inclusion and Exclusion Criteria
Adult patients of both genders, age ≥12 years admitted in the ICU either because of CKD or Acute Kidney Injury (AKI) having metabolic acidosis and already prescribed with IV Sodium Bicarbonate therapy along with normal saline and other IV fluids by the attending physician, were included. Both renal functions and acidemia were determined using KDIGO 2012 guidelines, where metabolic acidosis is defined as pH ≤7.20, and serum sodium bicarbonate concentration ≤22 mm (KDIGO) while renal failure is explained using the Glomerular filtration rate (GFR) <60 mL/min/1.73 and comorbidities like hypertension and diabetes has also been included in the factors. Exclusion criteria includes patients having respiratory and lactic acidosis because larger data may hinder the primary outcomes of the study and also the studied treatment has been found to be insignificant in the treatment of these diseases.8 Similarly, cancer patients having complex therapeutic intervention will also alter the calculations, therefore were excluded.
Data Collection
After explaining the study rationale and objectives to the subjects and getting the approved and signed consent form data from the patient’s medical record file is collected in the clinical checklist.
Clinical Evaluations and Study Tool
An evidence-based clinical checklist was developed from a similar preexisting checklist available in the published literature.18 This preexisting form was amended after the literature review, focus group discussion pursuant to the aims of this research and used as a data collection tool. The study covered the daily evaluation of clinical outcomes in each patient using standard management guidelines by an experienced clinical pharmacist in consultation with ICU consultants and physicians. The clinical assessment included evaluation of degree and severity of the disease, determination of rationality of the prescribed drugs and administered dose, interactions, risks, evaluation of various laboratory parameters by the severity of disease and in relation to administered drugs with specific doses. The clinical evaluation was based on the recommendations of KDIGO guidelines. The difference in values of all variables analyzed was assessed before and after the therapy commenced. The study followed clinical evaluation of these patients since the day they were admitted to their “discharge” or “death” after treatment. Drug–drug interactions were also observed by Stockley’s drug interaction. The clinical evaluation data was entered in SPSS version 23.0 for further statistical analysis.
Statistical Analyses
All data were analyzed by means of IBM SPSS (Statistical Package for Social Sciences) statistical software Version 23.0. The analyzed data was subjected to statistical analysis using both descriptive as well as inferential statistics. The descriptive statistics involved measure of frequencies, percentage, central tendencies like mean and cross-tabulation. Inferential analysis includes Pearson’s Chi-square test, one-way analysis of variance (ANOVA) tests, and Fischer’s exact test depending on the type of variable. Statistically significant association was considered with a P-value <0.05.
Results
Demographics
A total of 81 patients including 51 (63%) males and 30 (37%) females were recruited having a mean age of 55.6±19.5 and weight of 63.4±14.1. These demographic and clinical characteristics of all patients are summarized in Table 1.
 |
Table 1 Characteristics of Study Patients |
The serum creatinine levels were measured before the start and at the end of the study. During the study mean reduction of 1.65 mg/dL of the serum creatinine values were observed. The mean serum creatinine values before and at the end of study and the average difference in creatinine levels are summarized in Table 2.
 |
Table 2 Serum Creatinine Values for the Study Patients |
The end clinical outcomes of the patient in terms of mortality or recovery was also determined and an overall mortality rate of 45.7% was observed. The results of end clinical outcomes are provided in Table 3.
 |
Table 3 Clinical Outcomes of the Disease |
Factors Affecting the Clinical Outcomes
The association of progression of kidney disease and the clinical outcomes were determined and it was found that the mortality rate was not affected by the progression of kidney disease in acidemia patients. The results of the association of progression of kidney disease and clinical outcomes is provided in Table 4.
 |
Table 4 Association of Progression of Kidney Diseases and Clinical Outcomes |
The association of various laboratory parameters and the clinical outcomes was determined, and it was found that the mortality rate has a statistically significant association with hyperkalemia and hypertension. The results of association of laboratory parameters and clinical outcomes is provided in Table 5.
 |
Table 5 Association of Laboratory Parameters with Clinical Outcomes |
The association of various co-morbid conditions and the clinical outcomes was determined, and it was found that the co-morbid conditions has no statistically significant association with the clinical outcomes. The results of association of various co-morbid conditions and clinical outcomes is provided in Table 6.
 |
Table 6 Association of Various Co-Morbid Conditions with Clinical Outcomes |
Metabolic acidosis leads to certain complications including imbalance in serum concentrations and progression to secondary medical problems like hypertension, bone mineral disease, and multiple organ disorders.19,20 It was observed that disease complications including hypoxia, heart failure, and multiple organ failure has a statistically significant association with the overall mortality rate. The results of association of disease complications with clinical outcomes are provided in Table 7.
 |
Table 7 Association of Various Complications of CKD with Clinical Outcomes |
Effect of Sodium Bicarbonate Therapy
Sodium bicarbonate therapy was being administered in all the study population for treatment of metabolic acidosis. The majority of the study population were receiving sodium bicarbonate therapy with sodium chloride (normal saline) which has shown a greater recovery rate. However, a statistically significant difference in mortality rate was not observed. The results of mortality rate with sodium bicarbonate and sodium chloride therapy is provided in Table 8.
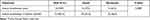 |
Table 8 Mortality Rate with Various Treatment Therapies |
Effect of Medications
The total number of medicines prescribed per patient is 18.75±6.244, of which 2.96±1.76 are antibiotics, 1.37±1.34 are antihypertensives, 0.7±0.68 are antidiabetics, 0.28±0.5 are immunosuppressants, 2.25±1.09 are IV fluids, and 13.69±5.42 are injectable medications. The brief description of medication prescribed among the study patient is provided in Table 9.
 |
Table 9 Number of Medicines of Various Categories Prescribed |
Several medications were prescribed and observed to determine their effect on mortality rate, of which some antihypertensive medications were found to play a role in mortality rate. The impact of various drug therapies on the end clinical outcomes are described in Table 10.
 |
Table 10 Effect of Medications on Mortality Rate |
Furthermore, drug interactions were also recorded in this study as 49 (60.5%) of the study population were receiving contraindicated medications having serious side-effects, 19 (23.5%) had significant interactions that needed monitoring, six (7.4%) had minor interactions, and l7 (8.6) received no such medications. The results of interactions among the prescribed medicine and their association with the mortality rate is descried in Table 11.
 |
Table 11 Impact of Drug–Drug Interactions on Clinical Outcomes |
Discussion
Metabolic acidosis is a serious medical condition characterized by low serum pH levels due to the reduction in HCO3 ions followed by compensatory decrease in paCO2.7 This condition is very common in ICU patients having complex medical conditions and receiving a multifaceted clinical interventions. This complexity interferes with the mortality rate and other end outcomes in critically ill patients. Renal diseases leading to metabolic acidosis are a common phenomenon as identified by Landmark studies that the reduction in the renal ammonia substantially leads to acidosis.19,20 Therefore, progression in stages of CKD should have been a complication in metabolic acidosis patients, but it has been observed conversely that CKD and AKI were both found to be non-significant with the mortality rate.
Impact of Co-Morbid Conditions on the Clinical Outcomes
The current study highlights medical complications caused by acidemia like multiple organ failure, hypoxia, risk of heart failure, and elevated heart rate affecting the end outcomes. Cardiovascular complications may be caused due to an increase in sodium levels of some patients. Several patients went into multiple organ failure during the study. One of the reasons of this complication is increased levels of lactate in the blood. Serum lactate is involved in tissue perfusion and oxygenation, therefore an increase in serum lactate indicates that the organ failure has started which corresponds with the results of hyperlactemia and multiple organ failure reported in this study. This phenomenon is supported by another study conducted in 2016, according to which hyperlactemia is associated with high mortality rate among acidemia patients.21 The relationship between kidney and cardiovascular diseases is well established, acidosis on top of it creates a definite cardiovascular risk.22 Acidosis causes sodium accumulation in blood serum, ultimately resulting in increased urinary excretion of sodium as well as an imbalance of other ions like sodium bicarbonate, hypercalcemia, etc., which eventually leads to cardiovascular problems.23
Impact of Biochemical Parameters on the Clinical Outcomes
One of the most significant outcomes reported in this study is the relationship between metabolic acidosis and biochemical parameters leading to an increased mortality rate. Although hypernatremia was not statistically significant in this study, a higher mortality rate was observed among hypernatremia patients. Other blood levels like urea, serum creatinine, magnesium, and calcium were also found to be high among those with higher mortality rate. These findings correspond with a similar study related to biochemical imbalance in critically ill patients with acidosis.25 However, factors like hypernatremia and hyperkalemia were found to influence mortality as both play a vital role in the regulation of heart rhythms.24
Impact of Drug Therapy on the Clinical Outcomes
Complexity in medical conditions of ICU patients allowed us to further expand these findings and determine which clinical interventions are being used to treat acidosis as well as the risk factor and complications. Sodium bicarbonate therapy was determined to be insignificant with mortality rate either with or without sodium chloride, although recovery has been shown in comparison to death, but this difference is not substantial. Furthermore, as hypothesized in the objective, the effect of sodium chloride when administered with sodium bicarbonate may increase the mortality rate, but it was observed that it promotes recovery, although these significant results are not quite pronounced. Similarly, various published studies have raised concerns over the concomitant administration of sodium chloride and sodium bicarbonate since they may cause hypernatremia and weight gain. On the contrary, this study has observed that a higher mortality rate was not observed, hence it is safe to administer sodium chloride and sodium bicarbonate together among the acidemic patients.
Several contraindicated drugs were prescribed to the study patients, especially antibiotics, which have been found to increase the mortality rate in patients with metabolic acidosis. Antibiotics not only worsen the acidosis they also reduce the ability of the kidneys to function properly.26 Some of the IV fluids were also interfering with the outcomes of the disease. Several drugs that were being prescribed were contraindicating, while fewer needed monitoring. The drug interactions were found to have a statistically significant relationship with the mortality rate. The incidence of such significant drug-interactions highlights the need for clinical pharmacists to review the drug therapy for critically ill patients in ICU settings, since none of the hospitals under study had a dedicated clinical pharmacist. It can be observed that drug-interactions were among the most significant factors which were affecting the mortality rate (P<0.05), hence the clinical pharmacy services should be introduced in the ICU settings to improve the clinical outcomes and reduce the higher mortality rate.
Study Limitations
The study limitations included small sample size and patient diversity. The study patients included patients with different stages of CKD as well as AKI which may create difficulties in developing and discussing the results.
Conclusion
Multiple factors including primary disease, complications, and biochemical parameters like serum lactate affect the mortality rate in renally compromised critically ill metabolic acidosis patients. Patients having cardiovascular complications due to acidosis and those having higher serum lactate levels were found to be at higher risk. Sodium bicarbonate therapy alone or in combination with sodium chloride was not found to affect the mortality rate in acidosis patients. Drug–drug interactions were found to be associated with higher mortality rate. The study has opened new ways to put forward research in this area.
Funding
No funding was received for this study.
Disclosure
The authors reported no conflicts of interest for this work.
References
1. Ellis MF. Use of bicarbonate in patients with metabolic acidosis. Crit Care Nurse. 2015;35:73–75. doi:10.4037/ccn2015502
2. Kraut JA, Madias NE. Lactic acidosis. N Engl J Med. 2014;371:2309–2319. doi:10.1056/NEJMra1309483
3. Kraut JA, Madias NE. Lactic acidosis: current treatments and future directions. Am J Kidney Dis. 2016;68(3):473–482. doi:10.1053/j.ajkd.2016.04.020
4. Jung B, Rimmele T, Le Goff C, et al. Severe metabolic or mixed acidemia on intensive care unit admission: incidence, prognosis and administration of buffer therapy. A prospective, multiple-center study. Crit Care. 2011;15(5):238. doi:10.1186/cc10487
5. Moranne O, Froissart M, Rossert J, et al.; NephroTest Study Group. Timing of onset of CKD-related metabolic complications. J Am Soc Nephrol. 2009;20(1):164–171. doi:10.1681/ASN.2008020159
6. Lo KB, Garvia V, Stempel JM, Ram P, Rangaswami J. Bicarbonate use and mortality outcome among critically ill patients with metabolic acidosis: a meta analysis. Heart Lung. 2020;49(2):167–174.
7. Cooper DJ, Walley KR, Wiggs BR, Russell JA. Bicarbonate does not improve hemodynamics in critically III patients who have lactic acidosis: a prospective, controlled clinical study. Ann Intern Med. 1990;112(7):492–498. doi:10.7326/0003-4819-112-7-492
8. Sabatini S, Kurtzman NA. Bicarbonate therapy in severe metabolic acidosis. J Am Soc Nephrol. 2009;20(4):692–695. doi:10.1681/ASN.2007121329
9. Kraut JA, Madias NE. Metabolic acidosis: pathophysiology, diagnosis and management. Nat Rev Nephrol. 2010;6(5):274. doi:10.1038/nrneph.2010.33
10. Adeva-Andany MM, Fern andez-Fern Andez C, Mourino-Bayolo D, Castro-Quintela E, Domínguez-Montero A. Sodium bicarbonate therapy in patients with metabolic acidosis. Sci World J. 2014;2014:1–10. doi:10.1155/2014/627673
11. Fujii T, Udy A, Licari E, Romero L, Bellomo R. Sodium bicarbonate therapy for critically ill patients with metabolic acidosis: a scoping and a systematic review. J Crit Care. 2019;51:184–191. doi:10.1016/j.jcrc.2019.02.027
12. Jaber S, Paugam C, Futier E, et al. Sodium bicarbonate therapy for patients with severe metabolic acidaemia in the intensive care unit (BICAR-ICU): a multicentre, open-label, randomised controlled, Phase 3 trial. Lancet. 2018;392(10141):31–40. doi:10.1016/S0140-6736(18)31080-8
13. Kraut JA, Madias NE. Treatment of acute metabolic acidosis: a pathophysiologic approach. Nat Rev Nephrol. 2012;8:589–601. doi:10.1038/nrneph.2012.186
14. Brochard L, Abroug F, Brenner M, et al. An official ATS/ERS/ ESICM/SCCM/SRLF statement: prevention and management of acute renal failure in the ICU patient: an international consensus conference in intensive care medicine. Am J Respir Crit Care Med. 2010;181:1128–1155. doi:10.1164/rccm.200711-1664ST
15. Kraut JA, Madias NE. Consequences and therapy of the metabolic acidosis of chronic kidney disease. Pediatr Nephrol. 2011;26(1):19–28. doi:10.1007/s00467-010-1564-4
16. National Institute for Health and Care Excellence (NICE). Chronic kidney diseasse and acute kidney injury: assessment and management [Internet]. [London] NICE. (Clinical guideline [CG182]); January 2014 [updated April, 2015; cited September 8, 2020]. Available form: https://www.nice.org.uk/guidance/cg182/chapter/1-Recommendations#pharmacotherapy.
17. Kidney Disease. Improving Global Outcomes (KDIGO). Clinical practice guidelines for the evaluation and management of chronic kidney disease. chapter 2 definition, identification, and prediction of CKD progression & chapter 3 management of progression and complications of CKD [Internet]. KDIGO. 2012 [cited September 8, 2020]. Available form: https://kdigo.org/wp-content/uploads/2017/02/KDIGO_2012_CKD_GL.pdf.
18. Ali M, Naureen H, Tariq MH, et al. Rational use of antibiotics in an intensive care unit: a retrospective study of the impact on clinical outcomes and mortality rate. Infect Drug Resist. 2019;12:493–499. doi:10.2147/IDR.S187836
19. Raphael KL. Metabolic acidosis and subclinical metabolic acidosis in CKD. J Am Soc Nephrol. 2018;29(2):376–382. doi:10.1681/ASN.2017040422
20. Lemann JR, Litzow JR, Lennon EJ. The effects of chronic acid loads in normal man: further evidence for the participation of bone mineral in the defense against chronic metabolic acidosis. J Clin Invest. 1966;45(10):1608–1614. doi:10.1172/JCI105467
21. Bolvardi E, Malmir J, Reihani H, et al. The role of lactate clearance as a predictor of organ dysfunction and mortality in patients with severe sepsis. Mater Sociomed. 2016;28(1):57–60. doi:10.5455/msm.2016.28.57-60
22. Abramowitz MK. Metabolic acidosis and cardiovascular disease risk in CKD. Clin J Am Soc Nephrol. 2018;13(10):1451–1452. doi:10.2215/CJN.10120818
23. Tuomilehto J, Jousilahti P, Rastenyte D, Moltchanov V, Tanskanen A, Pietinen P. Urinary sodium excretion and cardiovascular mortality in Finland: a prospective study. Lancet. 2001;357(9259):848–851. doi:10.1016/S0140-6736(00)04199-4
24. Urso C, Brucculeri S, Caimi G. Acid-base and electrolyte abnormalities in heart failure: pathophysiology and implications. Heart Fail Rev. 2015;20(4):493–503. doi:10.1007/s10741-015-9482-y
25. Zheng CM, Liu WC, Zheng JQ, et al. Metabolic acidosis and strong ion gap in critically ill patients with acute kidney injury. Biomed Res Int. 2014;2014. doi:10.1155/2014/819528
26. Kitterer D, Schwab M, Alscher MD, Braun N, Latus J. Drug-induced acid-base disorders. Pediatr Nephrol. 2015;30(9):1407–1423. doi:10.1007/s00467-014-2958-5
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
