Back to Journals » OncoTargets and Therapy » Volume 10
β-Catenin promotes cell proliferation, migration, and invasion but induces apoptosis in renal cell carcinoma
Authors Yang CM, Ji S, Li Y, Fu LY, Jiang T, Meng FD
Received 23 July 2016
Accepted for publication 2 November 2016
Published 20 February 2017 Volume 2017:10 Pages 711—724
DOI https://doi.org/10.2147/OTT.S117933
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Ingrid Espinoza
Chun-ming Yang,1 Shan Ji,2 Yan Li,3 Li-ye Fu,3 Tao Jiang,3 Fan-dong Meng3
1Department of Urology, The First Affiliated Hospital, China Medical University, 2Department of Endocrinology, The Fifth People’s Hospital of Shenyang, 3Department of Biotherapy, Cancer Research Institute, The First Affiliated Hospital, China Medical University, Shenyang, China
Abstract: β-Catenin (CTNNB1 gene coding protein) is a component of the Wnt signaling pathway that has been shown to play an important role in the formation of certain cancers. Abnormal accumulation of CTNNB1 contributes to most cancers. This research studied the involvement of β-catenin in renal cell carcinoma (RCC) cell proliferation, apoptosis, migration, and invasion. Proliferation, cell cycle, and apoptosis were analyzed by using Cell Counting Kit-8 and by flow cytometry. Migration and invasion assays were measured by transwell analysis. Real-time polymerase chain reaction and Western blot analysis were used to detect the expression of CTNNB1, ICAM-1, VCAM-1, CXCR4, and CCL18 in RCC cell lines. It was found that CTNNB1 knockdown inhibited cell proliferation, migration, and invasion and induced apoptosis of A-498 cells. CTNNB1 overexpression promoted cell proliferation, migration, and invasion and inhibited apoptosis of 786-O cells. Moreover, knockdown of CTNNB1 decreased the levels of ICAM-1, VCAM-1, CXCR4, and CCL18 expression, but CTNNB1 overexpression increased the expression of ICAM-1, VCAM-1, CXCR4, and CCL18. Further in vivo tumor formation study in nude mice indicated that inhibition of CTNNB1 delayed the progress of tumor formation through inhibiting PCNA and Ki67 expression. These results indicate that CTNNB1 could act as an oncogene and may serve as a promising therapeutic strategy for RCC.
Keywords: kidney cancer, oncogene, β-catenin, survival time, tumor migration-related protein
Introduction
Renal cell carcinoma (RCC), the most common type of adult kidney malignancy, accounts for 80% of all primary renal neoplasms and is composed of 2%–3% of all malignancies. The occurrence of RCC has increased in recent years; worldwide, ~100,000 patients die of disease every year.1 More than one-third of RCC patients present at advanced stages, and nearly half of the patients who experience curative therapies such as surgical resection suffer recurrence in the subsequent follow-up.2,3 Although the rate of detection of incidental RCC has increased with improved diagnostic techniques, metastatic lesions are still found at diagnosis in 30% of patients with RCC.4 Therefore, it is essential to find the molecular mechanism of tumor cell metastasis in primary lesions, which can improve the treatment and prognosis.
The canonical Wnt/β-catenin signaling pathway plays an important role in the embryonic development and cellular homeostasis and modulates cell growth and self-renewal in stem cells and progenitor cells.5,6 In the presence of Wnt signaling, β-catenin is accumulated in the cytosol, translocated into the nucleus, and forms a complex with transcription factors to activate target genes.7,8 Dysregulated β-catenin signaling is a common feature of many malignant tumors, including prostate, melanoma, myeloma, liver, colon, breast, and lung cancer.7,9–14 Activation of CTNNB1 promotes proliferation, migration, and invasion in glioma15 and oral squamous carcinoma.16
This study investigates the role of CTNNB1 in the pathobiology of RCC and shows that downregulation of this protein can effectively reduce proliferation, migration, and invasion of RCC cells. Taken together, these findings provide the evidence linking CTNNB1 signaling to tumor progression in human RCC.
Materials and methods
Cell lines
Normal kidney cell line (HK-2) and RCC cell lines (A-498, GRC-1, 786-O, and ACHN) were obtained from the Shanghai Cell Bank, Chinese Academy of Sciences (Shanghai, China). Cells were cultured in Dulbecco minimum essential medium (DMEM) (Invitrogen Life Technologies, Carlsbad, CA, USA) with the supplements of 10% heat-inactivated fetal bovine serum (Invitrogen Life Technologies), 100× penicillin-streptomycin solution (Invitrogen Life Technologies), and incubated in an incubator (Thermo Fisher Scientific Inc., Waltham, MA, USA) set to 37°C, 100% humidity, and 5% CO2.
Construction and infection
Oligonucleotide encoding shRNA-targeted human CTNNB1 mRNA and a scramble shRNA were both obtained from Sangon Biotech Inc. (Beijing, China). CTNNB1 cassette was amplified from the expression vector pMD18-T-CTNNB1 (Sangon Biotech Inc.). The recombinant pLKO.1-EGFP-CTNNB1-shRNA vector expressing CTNNB1-shRNA and pLKO.1-EGFP-CTNNB1 vector expressing CTNNB1 were digested with restriction enzymes, and the fragment containing CTNNB1-shRNA and CTNNB1 cassette was harvested from gel extraction by agarose gel electrophoresis. The infection of A498, 786-O, and ACHN cell lines by using of lentiviral expressing CTNNB1-shRNA and CTNNB1 were prepared as previously described.17 The recombinant pLKO.1-EGFP-scramble shRNA (NC-shRNA) and black pLKO.1-EGFP (NC-vector) were used as the negative control.
Cell proliferation assay
A498, 786-O, and ACHN cells were seeded in a 96-well plate (1×105 cells/well) and cultured at 37°C for indicated times. Cell proliferation was detected by Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Tokyo, Japan). After cells were infected 0, 12, 48, and 72 h, CCK-8 reagent was added to each group of wells at 1:10 (v/v) per 100 μL medium and incubated for 1 h at 37°C. Measurement was carried out in absorbance at 450 nm with a microplate reader according to the manufacturer’s instruction.
Flow cytometry assay
For cell cycle assay, A498, 786-O, and ACHN cells were collected and fixed with ice-cold ethanol overnight. Then the cells were washed three times with phosphate-buffered saline (PBS) and responded in PBS containing 500 ng/μL RNase A and incubated at 37°C for 30 min. All the samples were analyzed through flow cytometry (BD Biosciences, Franklin Lakes, NJ, USA) with the staining of propidium iodide (PI) and 0.1% TritonX-100. For cell apoptosis assay, A498 and 786-O cells were collected and washed twice with ice-cold PBS. And then 195 μL Annexin V-APC-binding buffer was used to resuspend the cells, followed by the addition of 5 μL Annexin V-FITC in each tube for 20 min, and 5 μL PI was added for another 10 min to incubate on ice. Finally, each sample was resuspended to a volume of 500 μL with 1× binding buffer and was analyzed through flow cytometry within 30 min.
In vitro transwell assay
For cell invasion, Transwell chamber (BD Biosciences) coated with Matrigel (BD Biosciences) was performed as described in the manufacturer’s protocol. After infection for 48 h, 1×105 cells in 500 μL serum-free DMEM were seeded into the upper well of the chamber. The lower chamber was filled with 750 μL DMEM containing 10% fetal bovine serum (FBS). Cells were incubated at 37°C for 48 h. Later, the cells that were able to pass through the filter were fixed with 4% paraformaldehyde and stained by crystal violet solution for 30 min. Cells on the top surface of the insert were removed with a cotton swab and counted under a microscope in five fields (×200). Similar procedure of cell invasion was also performed for cell migration transwell assay, except that the transwell membranes were not precoated with Matrigel.
Reverse transcription and real-time polymerase chain reaction (PCR)
Total RNA was extracted from normal kidney cells and four kidney cancer cell lines using Trizol reagent (Life Technologies) and detected by agarose gel electrophoresis according to the manufacturer’s instructions. As the template for PCRs, cDNA was synthesized from 5 μg of RNA using AMV reverse transcriptase (Fermentas, Waltham, MA, USA). Real-time quantitative PCR was performed using SYBR® Green 10× Supermix (Takara, Tokyo, Japan) in a total volume of 25 μL and on Roche Light Cycler® 480II System (Roche Diagnostics Ltd., Basel, Switzerland). Primer sequences were listed as follows: CTNNB1, forward: 5′-ATGACTCGAGCTCAGAGGGT-3′ and reverse: 5′-ATTGCACGTGTGGCAAGTTC-3′; GAPDH, forward: 5′-CTCCTCCTGGCCTCGCTGT-3′ and reverse: 5′-GCTGTCACCTTCACCGTTCC-3′; CXCR4, forward: 5′-GCTGTTGGCTGAAAAGGTGG-3′ and reverse: 5′-ATCTGCCTCACTGACGTTGG-3′; CCL18, forward: 5′-ATTCTGAAACCAGCCCCCAG-3′ and reverse: 5′-CCCTCAGGCATTCAGCTTCA-3′; ICAM-1, forward: 5′-CCCTGGAGCCCTTGAAGAAG-3′ and reverse: 5′-AGATGCACAACTTCTCGGCA-3′; VCAM-1, forward: 5′-CAGGACCACGAGAAGCTGTT-3′ and reverse: 5′-GATCTCCTTGGCATCCTCGG-3′. GAPDH was used as control of the input RNA level. The gene expression was calculated using the ΔΔCt method.
Western blot
Total protein was extracted from cells using ice-cold radio immunoprecipitation assay buffer (RIPA; Beyotime Institute of Biotechnology, Shanghai, China) containing 0.01% protease and phosphatase inhibitor (Sigma, Shanghai, China) and incubated on ice for 30 min. Cell lysis was centrifuged 12,000× g at 4°C for 10 min and the proteins in supernatant were separated by 10%–15% SDS gel electrophoresis and transferred to polyvinylidene fluoride membranes (Millipore, Shanghai, China), followed by blocking with 5% BSA in Tris-buffered saline with Tween 20 (TBST) (Amresco, Solon, OH, USA) and incubating with rabbit monoclonal antibodies anti-β-catenin (cat. no. ab6302; 1:4,000), anti-CXCR4 (cat. no. ab13854; 1:500), anti-CCL18 (cat. no. ab104867; 1:1,000), anti-GAPDH (cat. no. 5174; 1:1,500); mouse polyclonal antibody anti-ICAM-1 (cat. no. ab20; 1:500); rabbit polyclonal antibody anti-VCAM-1 (cat. no. ab174279; 1:5,000). All the primary antibodies were purchased from Abcam (Cambridge, MA, USA) expect for anti-GAPDH (Cell Signaling Technology, Inc., Danvers, MA, USA). Blots were then incubated for 1 h at 37°C with goat anti-mouse or anti-rabbit secondary antibody (cat. nos. A0208 and A0216; 1:1,000; Beyotime Institute of Biotechnology), and intensities were measured using enhanced chemiluminescence (Thermo Fisher Scientific). GAPDH antibody was used as an internal control. The blotting bands were quantified with ImageJ software.
In vivo experiments
For tumor growth assay, A-498 cells infected with NC-shRNA or CTNNB1-shRNA were trypsinized, washed, and resuspended in DMEM without FBS. Twelve male athymic nude mice (SLAC laboratory animal Center, Shanghai, China) were randomly divided into 2 groups (6 mice/group), and 2×106 A-498 cells were subcutaneously injected into the right armpit of the mice. The tumor size was determined every 3–4 days after the tumor is formed (around 1–2 weeks). At 45 days after injection, the mice were euthanized and the excised tumor tissues were formalin-fixed, paraffin-embedded, sectioned, and then analyzed with β-catenin, PCNA, or Ki67 immunohistochemistry, and the tumors were weighted on a digital balance. All animal-handling procedures were performed in strict accordance with the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (Ninth Edition, 2010). The animal use protocol was approved by the Institutional Animal Care and Use Committee of the First Affiliated Hospital, China Medical University (Shenyang, China).
Immunohistochemistry
Tissue sections were initial treatment for deparaffinization and hydration and then heated in EDTA (pH 8.0) and antigen-retrieved in 10 mm citrate buffer for 5 min at 100°C. The reaction of β-catenin, PCNA, or Ki67 antibody (Abcam) took place for 1 h at room temperature, followed by incubation by biotin-labeled secondary antibodies, and the slides were then developed using 3,3′-diaminobenzidine (DAB) solution and counterstained with hematoxylin staining (BASO, Shanghai, China). Immunohistochemical signals were calculated with the positive staining cells under a microscope (Olympus Corporation) with a magnification of ×200.
Statistical analysis
Results were shown as mean ± standard deviation. Data were analyzed by using the unpaired, two-tailed Student’s t-test with Graphpad Prism 6.0 software (GraphPad Software, La Jolla, CA, USA). All assays were performed in triplicate and repeated at least three times. All statistical analyses were two-sided and differences below P<0.05 were considered statistically significant.
Results
Upregulation of CTNNB1 associates with poor survival of RCC
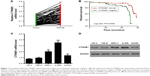
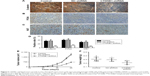
To study the biological role of CTNNB1 in RCC tumor tissues, real-time PCR was first used to detect the expression levels of CTNNB1 in 90 clear cell RCC patient tissues and their adjacent normal tissues. As shown in Figure 1A, CTNNB1 expression level was higher in clear cell RCC tissues compared with adjacent normal tissue control. Then, the correlation between CTNNB1 expression and clinical pathological features of the patients with clear cell RCC was investigated. Examination of the correlation between CTNNB1 expression and clinical pathological features showed that increased CTNNB1 expression was correlated with advanced stage, metastases, mutant von Hippel-Lindau (VHL) tumor suppressor, and poor survival (Table 1 and Figure 1B).
  | Table 1 Relationship between CTNNB1 and clinical characteristics of clear cell RCC patients |
CTNNB1 is overexpressed in RCC cell lines
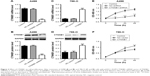
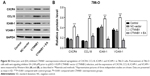
mRNA and protein expressions of CTNNB1 were examined in a normal kidney cell (HK-2) and four RCC cell lines (A-498, GRC-1, 786-O, and ACHN). Real-time PCR and Western blot assay observed that four RCC cell lines showed higher mRNA and protein levels of CTNNB1 compared with normal HK-2 cells (Figure 1C and D). Among that, A-498 cells are with the highest mRNA and protein levels of CTNNB1 and 786-O cells are with the lowest mRNA and protein levels of CTNNB1, compared with three other RCC cells. These two cell lines were therefore used for subsequent experiments.
Effect of CTNNB1 on A-498 and 786-O cell proliferation
To explore the biological significance of CTNNB1 in the tumorigenesis of RCC, CTNNB1 knockdown in A-498 cells and CTNNB1 overexpression in 786-O cells were introduced, respectively. Real-time PCR and Western blot assay at 48 h after infection showed that CTNNB1 knockdown in A-498 cells significantly decreased the mRNA and protein levels of CTNNB1 (Figure 2A and B), whereas overexpression of CTNNB1 in 786-O (Figure 2C and D) and ACHN cells (Figure S1A and B) significantly increased the mRNA and protein levels of CTNNB1. Moreover, immunohistochemistry staining was performed at 48 h after treatment with pLKO.1-EGFP-CTNNB1 in ACHN cells. We found that CTTNB1 translocated from the cytoplasm in the ACHN cells infected with the negative control vector to the nucleus in ACHN cells infected with pLKO.1-EGFP-CTNNB1 (Figure S2). The cell proliferation of A-498 and 786-O cells was assessed using CCK-8 assay. In A-498 cells with pLKO.1-EGFP-CTNNB1-shRNA infection, cell proliferation was significantly decreased by 24.8%, 29.7%, and 28.2% at 12, 24, and 48 h, respectively (Figure 2E). In 786-O and ACHN cells with pLKO.1-EGFP-CTNNB1 infection, cell proliferation was significantly increased by 22.7% and 11.6% at 24 h, and 22.0% and 13.3% at 48 h, respectively (Figures 2F and S1C).
Effect of CTNNB1 on A-498 and 786-O cell cycle and apoptosis
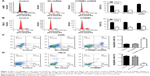
In order to further investigate the role of CTNNB1 in tumor cell growth, the cell cycle of A-498 and 786-O was measured using flow cytometry. Our data indicated that the cell number of G0/G1 phase was notably increased in CTNNB1-silenced A-498 cells, accompanied with decreased cell number of G2/M phase (Figure 3A). However, the cell numbers of G0/G1 and G2/M phases in CTNNB1-overexpressed 786-O and ACHN cells were against that in CTNNB1-silenced A-498 cells (Figures 3B and S1D). Negative control vector-infected A-498 (NC-shRNA), 786-O cells (NC-vector), and ACHN (NC-vector) had no cell cycle changes in G0/G1, S, and G2/M phases compared with corresponding control cells.
To figure out the effects of CTNNB1 on cell apoptosis of A-498 and 786-O cells, an Annexin V-FITC/PI staining and flow cytometry assay were performed. As shown in Figure 3C, it was revealed that CTNNB1 downregulation in A-498 cells significantly increased cell apoptosis by 2-fold compared with control cells. While, CTNNB1 upregulation in 786-O and ACHN cells showed a significant decrease in apoptotic rate by 78.2% and 82.8%, respectively (Figures 3D and S1E) compared with control cells. Taken together, these results demonstrate that CTNNB1 promotes cell proliferation and suppresses apoptosis of RCC cells in vitro.
Effect of CTNNB1 on A-498 and 786-O cell migration and invasion
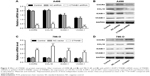
Evidences have shown that decrease in cell-cell and/or cell-matrix adhesion correlates with tumor invasion and metastasis. To investigate the effect of CTNNB1 in migration and invasion, A-498 and 786-O cells were cultured in a Boyden chamber. After 48 h of incubation, knockdown of CTNNB1 in A-498 cells significantly reduced the cell migration by 60.7% compared with control cells (Figure 4A). Meanwhile, CTNNB1 upregulation in 786-O and ACHN cells observed significant increase in cell migration by 45.3% and 68.7% compared with control cells (Figures 4B and S1F). As shown in Figure 4C, the invasion assay found that CTNNB1 downregulation in A-498 cells also markedly suppressed cell invasion by 63.9% compared with control cells. While, CTNNB1 upregulation in 786-O and ACHN cells observed significant increase in cell invasion by 49.3% and 92.6% compared with control cells (Figure 4D and S1G).
Effect of CTNNB1 on CXCR4, CCL18, ICAM-1, and VCAM-1 expression
To further explore the mechanism of CTNNB1 regulating cell migration and invasion of kidney cancer cell lines, two cell migration molecular markers, ICAM-1 and VCAM-1, and cytokines CXCR4 and CCL18, were predicated to be CTNNB1 targets. As shown in Figure 5A and B, the mRNA and protein levels of ICAM-1, VCAM-1, CXCR4, and CCL18 in A-498 cells were remarkably suppressed by CTNNB1 shRNA compared with control cells. However, the mRNA and protein levels of ICAM-1, VCAM-1, CXCR4, and CCL18 in 786-O cells were remarkably increased by CTNNB1 overexpression compared with control cells (Figure 5C and D).
Effect of CTNNB1 on tumor growth of A-498 cells in nude mice
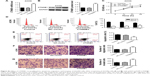
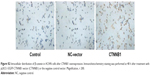
To further examine the effects of CTNNB1 on tumor growth in vivo, A-498 cells infected with NC-shRNA or CTNNB1-shNRA were subcutaneously injected into the right armpit of nude mice. High expression of β-catenin (Figure 6A and D), PCNA (Figure 6B and D), and Ki67 (Figure 6C and D) was found in the xenograft from the nude mice with NC-shRNA cell injection, but their expressions decreased by 78.3%, 68.6%, and 77.2% in that injected with CTNNB1-shRNA cells, respectively. Moreover, CTNNB1-shNRA tumors grew slower in mice, whereas NC-shRNA tumors grew fast in mice (Figure 6E). Tumor weights in CTNNB1-shRNA mice were notably decreased to 31.1% 45 days after injection compared with NC-shRNA group (Figure 6F). These data suggest that knockdown of CTNNB1 inhibits tumor growth in nude mice.
Discussion
In the last few decades, the incidence of RCC was increased and more than half of the patients whose survival time was poor had clinical limitations.2,18 Numerous candidate RCC biomarkers have been identified to date, while neither the expression pattern of these genes nor its localization and function in the tumor tissues has been investigated. At this point, identifying specific genetic markers associated with tumor metastasis may be useful to evaluate the disease progression. This study shows that CTNNB1 is overexpressed in RCC cell lines compared with renal tubular epithelial cell line HK-2 and is essential survival factor for RCC cells. In addition, these data also showed that CTNNB1 is a key factor associated with apoptosis, migration, and invasion of RCC cells. Lastly, it was indicated that CTNNB1 downregulation inhibits xenograft tumor growth in vivo.
In several types of cancers, including RCC, an enhanced activation of the Wnt signaling pathway is a major etiological factor.19 Although the number of papers focused on the investigation of Wnt/β-catenin pathway in renal carcinogenesis is small, the reported results are relevant.20,21 In the present study, CTNNB1 expression was high in clear cell RCC patients and in those associated with advanced stage, metastases, VHL expression and prognosis. In line with our findings that the mutation of VHL gene is observed in ~60% of sporadic clear cell RCCs22 and loss of pVHL leads to combined de-repression of HIF-β-catenin with repercussions on Wnt/β-catenin pathway,23 suggesting VHL status could have an influence on β-catenin. Although the role of CTNNB1 has been elucidated for many types of human cancer, its biological significance in RCC has not been examined in detail. CTNNB1 is widely expressed in the entire renal cancer cell lines being tested, demonstrating an oncogene feature of CTNNB1 in RCC. To test this hypothesis, the function of CTNNB1 in the cell proliferation, cell cycle, apoptosis, cell migration and migration was assessed, using RCC cell lines infected with pLKO.1-EGFP-CTNNB1-shRNA vector expressing CTNNB1-shRNA and pLKO.1-EGFP-CTNNB1 vector expressing CTNNB1, respectively. Our results showed that CTNNB1 knockdown significantly inhibited cell proliferation in vitro. However, overexpression of CTNNB1 promoted the proliferation of RCC cell lines significantly. The tumor growth in nude mice was also investigated using RCC cell lines stably infected with pLKO.1-EGFP-scramble shRNA or pLKO.1-EGFP-CTNNB1-shRNA. The inhibitory effect of CTNNB1 knockdown on renal tumor growth in mice was observed, which was evidenced by decreased expression of PCNA and Ki67. An increased nuclear translocation and activity of β-catenin was proposed to promote the proliferation of melanoma24 and deteriorate the survival of melanoma patients.25 In addition, our data showed G2/M phase blockage and increased apoptotic cells in CTNNB1-upregualted cells compared with empty vector cells. However, CTNNB1 overexpression observed increased cell number of G0/G1 phase and decreased cell number of G2/M phase. Pu et al26 reported that downregulation of CTNNB1 by RNA interference in U251 human glioma cells suppressed cell proliferation and invasion and induced cell apoptosis. Our results are consistent with this study. It has also been reported that CTNNB1 modulates early and late apoptotic progression in other cancer cells;27,28 hence, the present study did not conclusively prove the mechanisms of CTNNB1 function in RCC cell lines because some additional mechanisms are possible, including direct target genes and regulation of other signaling pathways.
Cell mobility is an important characteristic of many physiologic and pathologic phenomena, such as development, wound repair, angiogenesis, and metastasis.1 Downregulation of CTNNB1 significantly inhibited cell adhesion in invasive breast cancer, and metastatic disease is always accompanied with a higher activity of motility.29 It is arguable that activation of CTNNB1 is observed in migratory melanoma cells30 and associated with transendothelial migration of melanoma cells.31 Because the metastasis of cancer cells is dependent on the characteristics of migration and invasion, the relationship between the expression of CTNNB1 and the invasion or migration ability of RCC cell lines was studied. Indeed, it is found that CTNNB1 is essential for the migration and invasion of RCC cell lines. Knockdown of CTNNB1 expression in RCC cell lines reduced cell migration and invasion significantly, whereas overexpression of CTNNB1 increased cell migration and invasion. In order to determine the molecular mechanism of CTNNB1 action in the migration and invasion, the expression of various migration- and invasion-related genes was evaluated. CXCR4, CCL18, ICAM-1, and VCAM-1 expressions were significantly downregulated in the CTNNB1 knockdown cells. However, CTNNB1 overexpression increased the expression of these genes at both mRNA and protein levels. These cytokine factors, including CXCR4, CCL18, ICAM-1, and VCAM-1, have been reported to play critical role in RCC cell invasion and migration.32–34 It has also been reported that CXCR4 signaling activates CTNNB1 transcriptional activity in embryonic rat spinal cord neural progenitors, suggesting that CXCR4 signaling may interact with β-catenin to regulate the development of the central nervous system.35 A previous study found that loss of CTNNB1 affects VCAM-1 levels in multiple cell types.36 However, ICAM-1 was not downregulated by CTNNB1 expression in prostate cancer cells.37 Moreover, wnt-signaling inhibitor ethacrynic acid (100 μM) significantly decreased the expression of these proteins induced by CTNNB1 overexpression in 786-O (Figure S3). The precise molecular mechanisms of CTNNB1 regulating these gene expressions call for more research studies. However, our findings may be important in the studies of invasion and migration of RCC.
A limitation of the study is that CTNNB1 expression in papillary RCC and other histologic subtypes are not being addressed, which should be investigated in the future. Understanding the mechanisms involved in oncogenesis has wide-ranging implications for targeting the treatment of cancer. This seems to be the first report showing CTNNB1 involved in cell growth, apoptosis, migration, and invasion of RCC cell lines. Moreover, CTNNB1 involved in tumor cell motility, and invasion could represent a molecular target for RCC and could eventually provide new leads for improved therapeutic approaches against RCC.
Disclosure
The authors report no conflicts of interest in this work.
References
Garg M, Kanojia D, Khosla A, et al. Sperm-associated antigen 9 is associated with tumor growth, migration, and invasion in renal cell carcinoma. Cancer Res. 2008;68:8240–8248. | ||
Koul H, Huh JS, Rove KO, et al. Molecular aspects of renal cell carcinoma: a review. Am J Cancer Res. 2011;1:240–254. | ||
Yamasaki T, Seki N, Yamada Y, et al. Tumor suppressive microRNA138 contributes to cell migration and invasion through its targeting of vimentin in renal cell carcinoma. Int J Oncol. 2012;41:805–817. | ||
Hirata H, Hinoda Y, Nakajima K, et al. Wnt antagonist DKK1 acts as a tumor suppressor gene that induces apoptosis and inhibits proliferation in human renal cell carcinoma. Int J Cancer. 2011;128:1793–1803. | ||
Sinnberg T, Menzel M, Ewerth D, et al. β-Catenin signaling increases during melanoma progression and promotes tumor cell survival and chemoresistance. PLoS One. 2011;6:e23429. | ||
Gough NR. Focus issue: wnt and beta-catenin signaling in development and disease. Sci Signal. 2012;5:eg2. | ||
Ying Y, Tao Q. Epigenetic disruption of the WNT/beta-catenin signaling pathway in human cancers. Epigenetics. 2009;4:307–312. | ||
Katoh M, Katoh M. WNT signaling pathway and stem cell signaling network. Clin Cancer Res. 2007;13:4042–4045. | ||
Yang LH, Xu HT, Han Y, et al. Axin downregulates TCF-4 transcription via beta-catenin, but not p53, and inhibits the proliferation and invasion of lung cancer cells. Mol Cancer. 2010;9:25. | ||
Hsieh IS, Chang KC, Tsai YT, et al. MicroRNA-320 suppresses the stem cell-like characteristics of prostate cancer cells by downregulating the Wnt/beta-catenin signaling pathway. Carcinogenesis. 2013;34:530–538. | ||
Hallett RM, Kondratyev MK, Giacomelli AO, et al. Small molecule antagonists of the Wnt/beta-catenin signaling pathway target breast tumor-initiating cells in a Her2/Neu mouse model of breast cancer. PLoS One. 2012;7:e33976. | ||
Ji J, Yamashita T, Wang XW. Wnt/beta-catenin signaling activates microRNA-181 expression in hepatocellular carcinoma. Cell Biosci. 2011;1:4. | ||
Chien AJ, Moore EC, Lonsdorf AS, et al. Activated Wnt/beta-catenin signaling in melanoma is associated with decreased proliferation in patient tumors and a murine melanoma model. Proc Natl Acad Sci U S A. 2009;106:1193–1198. | ||
Kim Y, Schmidt M, Endo T, Lu D, Carson D, Schmidt-Wolf IG. Targeting the Wnt/beta-catenin pathway with the antifungal agent ciclopirox olamine in a murine myeloma model. In Vivo. 2011;25:887–893. | ||
Wang K, Wang X, Zou J, et al. miR-92b controls glioma proliferation and invasion through regulating Wnt/beta-catenin signaling via Nemo-like kinase. Neuro Oncol. 2013;15:578–588. | ||
Iwai S, Yonekawa A, Harada C, et al. Involvement of the Wnt-beta-catenin pathway in invasion and migration of oral squamous carcinoma cells. Int J Oncol. 2010;37:1095–1103. | ||
Zhang HJ, Tao J, Sheng L, et al. Twist2 promotes kidney cancer cell proliferation and invasion via regulating ITGA6 and CD44 expression in the ECM-receptor-interaction pathway. Biomed Pharmacother. 2016;81:453–459. | ||
Hadoux J, Vignot S, De La Motte Rouge T. Renal cell carcinoma: focus on safety and efficacy of temsirolimus. Clin Med Insights Oncol. 2010;4:143–154. | ||
Pulkkinen K, Murugan S, Vainio S. Wnt signaling in kidney development and disease. Organogenesis. 2008;4:55–59. | ||
Cojocaru E, Lozneanu L, Giusca SE, Caruntu ID, Danciu M. Renal carcinogenesis – insights into signaling pathways. Rom J Morphol Embryol. 2015;56:15–19. | ||
SA VONS-H, Schmeel LC, Schmeel FC, Schmidt-Wolf IG. Targeting the Wnt/beta-catenin pathway in renal cell carcinoma. Anticancer Res. 2014;34:4101–4108. | ||
Kim WY, Kaelin WG. Role of VHL gene mutation in human cancer. J Cin Oncol. 2004;22(24):4991–5004. | ||
Linehan WM, Rubin JS, Bottaro DP. VHL loss of function and its impact on oncogenic signaling networks in clear cell renal cell carcinoma. Int J Biochem Cell Biol. 2009;41:753–756. | ||
Moon RT, Kohn AD, De Ferrari GV, Kaykas A. WNT and beta-catenin signalling: diseases and therapies. Nat Rev Genet. 2004;5:691–701. | ||
Kielhorn E, Provost E, Olsen D, et al. Tissue microarray-based analysis shows phospho-beta-catenin expression in malignant melanoma is associated with poor outcome. Int J Cancer. 2003;103:652–656. | ||
Pu P, Zhang Z, Kang C, et al. Downregulation of Wnt2 and beta-catenin by siRNA suppresses malignant glioma cell growth. Cancer Gene Ther. 2009;16:351–361. | ||
Svedlund J, Auren M, Sundstrom M, et al. Aberrant WNT/beta-catenin signaling in parathyroid carcinoma. Mol Cancer. 2010;9:294. | ||
Li F, Chong ZZ, Maiese K. Winding through the WNT pathway during cellular development and demise. Histol Histopathol. 2006;21:103–124. | ||
Medrek C, Landberg G, Andersson T, Leandersson K. Wnt-5a-CKI{alpha} signaling promotes {beta}-catenin/E-cadherin complex formation and intercellular adhesion in human breast epithelial cells. J Biol Chem. 2009;284(16):10968–10979. | ||
Murakami T, Toda S, Fujimoto M, et al. Constitutive activation of Wnt/beta-catenin signaling pathway in migration-active melanoma cells: role of LEF-1 in melanoma with increased metastatic potential. Biochem Biophys Res Commun. 2001;288:8–15. | ||
Qi J, Chen N, Wang J, Siu CH. Transendothelial migration of melanoma cells involves N-cadherin-mediated adhesion and activation of the beta-catenin signaling pathway. Mol Biol Cell. 2005;16:4386–4397. | ||
Wang L, Wang Z, Yang B, Yang Q, Wang L, Sun Y. CXCR4 nuclear localization follows binding of its ligand SDF-1 and occurs in metastatic but not primary renal cell carcinoma. Oncol Rep. 2009;22:1333–1339. | ||
Jones J, Otu H, Spentzos D, et al. Gene signatures of progression and metastasis in renal cell cancer. Clin Cancer Res. 2005;11:5730–5739. | ||
Hemmerlein B, Scherbening J, Kugler A, Radzun HJ. Expression of VCAM-1, ICAM-1, E- and P-selectin and tumour-associated macrophages in renal cell carcinoma. Histopathology. 2000;37:78–83. | ||
Luo Y, Cai J, Xue H, Mattson MP, Rao MS. SDF1alpha/CXCR4 signaling stimulates beta-catenin transcriptional activity in rat neural progenitors. Neurosci Lett. 2006;398:291–295. | ||
Nemeth MJ, Mak KK, Yang Y, Bodine DM. β-Catenin expression in the bone marrow microenvironment is required for long-term maintenance of primitive hematopoietic cells. Stem Cells. 2009;27:1109–1119. | ||
Kim JH, Kim B, Cai L, et al. Transcriptional regulation of a metastasis suppressor gene by Tip60 and β-catenin complexes. Nature. 2005;434:921–926. |
Supplementary materials
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.