Back to Journals » Drug Design, Development and Therapy » Volume 10
Baicalein suppresses metastasis of breast cancer cells by inhibiting EMT via downregulation of SATB1 and Wnt/β-catenin pathway
Authors Ma X, Yan W, Dai Z , Gao X, Ma Y, Xu Q, Jiang J, Zhang S
Received 16 December 2015
Accepted for publication 20 January 2016
Published 18 April 2016 Volume 2016:10 Pages 1419—1441
DOI https://doi.org/10.2147/DDDT.S102541
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Wei Duan
Xingcong Ma,1 Wanjun Yan,1 Zhijun Dai,1 Xiaoyan Gao,1 Yinan Ma,1 Quntao Xu,2 Jiantao Jiang,3 Shuqun Zhang1
1Department of Oncology, the Second Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi, People's Republic of China, 2Department of Oncology, Institute of Health, China North Industries Group Corporation, Xi'an, Shaanxi, People's Republic of China, 3Department of Thoracic Surgery, the Second Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi, People's Republic of China
Background: The flavonoid baicalein, a historically used Chinese herbal medicine, shows a wide range of biological and pharmaceutical effects, among which its potent antitumor activity has raised great interest in recent years. However, the molecular mechanism involved in the antimetastatic effect of baicalein remains poorly understood. This study aimed to verify the inhibitory effects of baicalein on metastasis of MDA-MB-231 human breast cancer cells both in vitro and in vivo, as well as to investigate the related mechanisms.
Methods: MTT assay was used to examine the inhibition of baicalein on proliferation of MDA-MB-231 cells. Wound healing assay and the in vitro invasion assay was carried out to investigate the effects of baicalein on migration and invasion of MDA-MB-231 cells, respectively. In order to explore the effects of baicalein on tumor metastasis in vivo, xenograft nude mouse model of MDA-MB-231 cells was established. Animals were randomly divided into four groups (control, therapy group, and low-dose and high-dose prevention group, n=6), and treated with baicalein as designed. Following sacrifice, their lungs and livers were collected to examine the presence of metastases. qRT-PCR and Western blot were performed to study the effects of baicalein on expression of SATB1, EMT-related molecules, and Wnt/β-catenin signaling components of MDA-MB-231 cells as well as the metastatic tissue. Effects of baicalein on the expression of target proteins in vivo were also analyzed by immunohistochemistry.
Results: Our results indicated that baicalein suppressed proliferation, migration, and invasion of MDA-MB-231 cells in a time- and dose-dependent manner. Based on assays carried out in xenograft nude mouse model, we found that baicalein inhibited tumor metastasis in vivo. Furthermore, baicalein significantly decreased the expression of SATB1 in MDA-MB-231 cells. It suppressed the expression of vimentin and SNAIL while enhancing the expression of E-cadherin. Baicalein also downregulated the expression of Wnt1 and β-catenin proteins and transcription level of Wnt/β-catenin-targeted genes.
Conclusion: Our results demonstrate that baicalein has the potential to suppress breast cancer metastasis, possibly by inhibition of EMT, which may be attributed to downregulation of both SATB1 and the Wnt/β-catenin pathway. Taken together, baicalein may serve as a promising drug for metastasis treatment of breast cancer.
Keywords: breast cancer, baicalein, metastasis, EMT, SATB1, Wnt/β-catenin pathway
Background
Breast cancer is the most common malignancy and the leading cause of cancer-related death among women worldwide.1 Although reliable statistics suggests a downtrend of mortality in the past 20 years, the incidence of breast cancer is continuously climbing, especially in many developing countries where breast cancer incidence used to be very low.2 Women aged between 20 and 59 years are most likely to be inflicted by breast cancer, and the age of onset has reduced in recent years, posing a big threat to women’s health and life quality.3 Despite the improvement of surgical treatment, radiotherapy and chemotherapy, metastasis remains incurable and is the main cause of breast cancer-related death. Research in this field must concentrate on the investigation of metastatic mechanisms, reliable molecular markers, and therapeutic targets for breast cancer, thus promoting the exploration of effective drugs.
Metastasis is defined as the process by which malignant tumor cells break away from their primary site, invade the surrounding tissue, intravasate into microvasculature, then translocate to distant organs through lymphatic or blood vessels, exit from the circulatory system, survive in new microenvironment, and finally develop a homogeneous tumor at the secondary site.4 Invasiveness and metastatic potential are two important biological characteristics of cancers. These malignant behaviors are comprised of multiple steps and involve systemic damages, laying the pathologic basis of tumor relapse and disease deterioration. It is estimated that metastasis accounts for 90% of cancer-related mortality.5
The special AT-rich sequence-binding protein-1 (SATB1) is a specifically expressed matrix attachment regions (MARs)-binding protein. It is mainly expressed in the thymocytes, remains at low level in the testes, the fetal brain, and the osteoblasts, and is hardly detectable in other normal tissues. SATB1 specifically binds MARs with high affinity and provides an anchorage platform for hundreds of genes, thus regulating their expression and changing cell phenotype through excessive genetic recombination. The genes regulated by SATB1 count for 2%–10% of the genome.6,7 SATB1 was first identified in the T-cells. It regulates the spatial and temporal expression of genes during the differentiation and maturation of T-cells and guarantees their normal development.8 Recent studies have shown that SATB1 regulates the malignant progression of breast cancer. It is expressed during oncogenesis and changes the expression pattern of genes. Tumor cells then acquire aggressive phenotype and the ability to metastasize. Activation of SATB1 is indispensable for tumor metastasis, for it enhances the expression of various genes involved in the growth, proliferation, angiogenesis, invasion, and metastasis. High level of SATB1 predicts bad outcome for breast cancer patients.9 Further researches indicate that SATB1 is overexpressed in gastric,10,11 colorectal,12 liver,13 ovarian,14,15 prostate,16,17 and bladder18 cancers and promotes their metastases. All these findings suggest an important role of SATB1 in the initiation and development of cancers.
Wnt signaling pathway was first discovered in Drosophila. It is highly conserved and involved in multiple basic physiological functions of cells, such as the selection between survival or death during embryonic development, decision of cell polarity, regulation of cell proliferation, differentiation, cell cycle arrest, apoptosis, and intrinsic homeostasis.19,20 Because Wnt signaling plays a vital role in organ development and the maintenance of intrinsic homeostasis, abnormity of this pathway may cause developmental deficiency and is associated with various diseases. Dysfunction of the canonical Wnt/β-catenin pathway is one of the most common signaling abnormalities occurring in human malignancies.21 This pathway participates in the regulation of cancer stem cells phenotype and EMT (epithelial–mesenchymal transition). Plenty of studies have suggested that Wnt/β-catenin pathway is closely related to the initiation and progression of different cancers.22–26 Studies investigating the T-cell development and tumor progression indicate that some functions of the Wnt/β-catenin pathway overlap with that of SATB1.27 They share targets such as some key molecules in EMT program, suggesting that there may be an important functional link between SATB1 and Wnt/β-catenin pathway.
Baicalein is the effective component extracted from the dry root of Scutellaria baicalensis Georgi (SBG) and has a defined chemical structure (Figure 1), which is the basis of its pharmacological function. In recent years, both in vitro and in vivo experiments showed that baicalein exerts antitumor effects. It has a broad spectrum of action and multiple targets, and the related mechanisms are complicated and varied, including inducing tumor cell apoptosis and cell cycle arrest,28,29 inhibiting tumor proliferation and angiogenesis,29,30 and scavenging free radicals.31,32 The mechanisms involved in the antimetastatic effect of baicalein are not clear yet, making it a new hot spot for researchers.
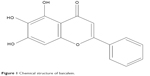  | Figure 1 Chemical structure of baicalein. |
In this study, we verified that baicalein significantly suppressed the proliferation, migration, and invasion of breast cancer cell line MDA-MB-231 in vitro. Results of assays carried out in xenograft nude mouse model also indicated an inhibitive effect of baicalein on tumor metastasis in vivo. Chung et al33 reported that baicalein suppresses the EMT of breast epithelial cells; the tumorigenic activity of breast cancer cells, which indicated that inhibition of EMT, may play an important part in antitumor effect of baicalein. Additionally, we reported a novel mechanism for the antimetastatic effect of baicalein – suppression of EMT – which may be attributed to the cooperative inhibition of SATB1 and Wnt/β-catenin pathway. Furthermore, appealing is that our findings suggest potential cross talk between SATB1 and Wnt/β-catenin signaling during the progression of breast cancer, providing a new perspective to study the regulation mechanisms of cancers.
Materials and methods
Cell culture and reagents
Immortalized mammary epithelial cells (MCF-10A) were obtained from Sagene Biological Technology Co., Ltd. (Guangzhou, People’s Republic of China). MCF7, SKBR3, and MDA-MB-231 human breast cancer cell lines were obtained from Shanghai Cell Biological Institute of the Chinese Academy of Science (Shanghai, People’s Republic of China). MCF-10A cells were cultured in mammary epithelial growth medium supplemented with 100 ng/mL cholera toxin (Sagene Biological Technology Co., Ltd). MCF7, SKBR3, and MDA-MB-231 cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS; Hyclone) and 1% penicillin–streptomycin solution (Thermo Fisher Scientific, Waltham, MA, USA) and maintained in a cell incubator with a humidified atmosphere of 95% air and 5% CO2 at 37°C.
MTT [3-(4,5-Dimethylthiazol-2-yl)-5-(3-carboxyme-thoxyphenyl)-2-(4-ulfophenyl)-2H-tetrazolium] and baicalein were purchased from Sigma-Aldrich (St Louis, MO, USA) and stored at −20°C in the dark. The stock solution of baicalein for incubation with cells was prepared in dimethyl sulfoxide (DMSO; MP Biomedicals, Santa Ana, CA, USA) and further diluted in culture medium. Final concentration of DMSO in the medium was 0.1% (to avoid its interference with cell viability). Carboxymethyl cellulose sodium (CMC-Na) was purchased from Sigma-Aldrich. 3,3′-Diaminobenzidine (DAB) was from Proteintech (Beijing, People’s Republic of China). β-Catenin rabbit mAb (D10A8), E-cadherin rabbit mAb (24E10), and vimentin rabbit mAb (D21H3) were purchased from Cell Signaling Technology (Danvers, MA, USA). SATB1 (ab92307), SNAIL (ab180714) monoclonal antibodies, and Wnt1 (ab85060) polyclonal antibody were purchased from Abcam (Cambridge, MA, USA). Anti-β-actin antibody was obtained from Santa Cruz Biotechnology Inc. (Dallas, TX). Horseradish peroxidase (HRP)-linked goat anti-mouse or anti-rabbit immunoglobulin G (IgG) antibodies were purchased from ComWin Biotech Co., Ltd. (Beijing, People’s Republic of China). Baicalein treatment and extraction of RNA and protein were all performed during the logarithmic growth phase of cells.
MTT assay
MTT assay was conducted to evaluate the effect of baicalein on proliferation of breast cancer cells. MDA-MB-231 cells were routinely digested, collected, and then seeded in 96-well plates at a density of 8×103 cells/well. After incubation for 12–24 hours, cells were treated with 0, 20, 40, 60, 80, 100, and 120 μmol/L baicalein according to their experimental grouping and then incubated at 37°C for 24, 48, and 72 hours. At the end of baicalein treatment, 20 μL MTT solution at a concentration of 5 mg/mL was added into each well and the incubation continued for another 4 hours. Then the medium was removed and 160 μL/well DMSO was added. The optical density (OD) value of each well was measured spectrophotometrically at 490 nm. The inhibition ratio (IR) was calculated according to the following formula: IR = (1–mean OD value of experimental group/mean OD value of control group) ×100%. Approximately 50% inhibition concentration (IC50) of baicalein at different time points was calculated using the Logit method.
Wound healing assay
Wound healing assay was used to evaluate the migration ability of breast cancer cells. MDA-MB-231 cells were seeded into the six-well plates at a density of 1×105 cells/well and incubated in medium containing 10% FBS. When the cell monolayer reached 80% confluency, a 200 μL pipette tip was used to scratch a straight wound tract through the middle of the cell monolayer. Each well was gently washed with phosphate-buffered saline (PBS) three times to remove the detached cells. Then, the MDA-MB-231 cells were incubated in medium containing 2% FBS and 0, 10, 20, and 40 μmol/L baicalein for up to 24 hours. Pictures were taken under 40× magnification at 0, 12, and 24 hours after baicalein was added. The cell migration distance was presented as an average value assessed by using Photoshop software (Adobe, San Jose, CA, USA). Each experiment was performed three times. The relative migration distance was determined as a percentage of the control.
In vitro invasion assay
The in vitro invasive potential of breast cancer cells was assessed by use of the Transwell chamber with 10 mm diameter and 8 μm pore size polycarbonate membrane (Millipore, Billerica, MA, USA) coated with Matrigel (BD, Franklin Lakes, NJ, USA), according to manufacturer’s instructions. MDA-MB-231 cells were pretreated with baicalein (0, 10, 20, and 40 μmol/L) for 24 hours. Then cells were collected and resuspended to a final density of 5×104 cells/mL and added into each upper chamber of Transwell system. After 24 hours of incubation at 37°C, cells that remained on the upper side of the polycarbonate membrane were removed with a cotton swab and the invaded cells were fixed and stained with crystal violet. Ten fields on each membrane were randomly selected and the number of invaded cells was counted. Pictures were taken under 100× magnification. The experiment was repeated three times.
Establishment of the xenograft nude mouse model of breast cancer metastasis
To explore the effects and mechanisms of baicalein on tumor metastasis in vivo, a xenograft nude mouse model of MDA-MB-231 cells was established. After 1 week acclimation, nude mice were injected (through the tail vein) with MDA-MB-231 human breast cancer cells (2×105/animal), which was resuspended in 0.1 mL serum- and antibiotic-free culture medium containing 50% Matrigel (BD) and 50% DMEM (Hyclone). After inoculation, the animals were randomly divided into four groups (n=6), and treated as indicated in Table 1. For the low-dose and high-dose prevention group, 50 or 100 mg/kg baicalein was given by intragastric gavage for 15 days starting from the day after tumor cells were injected. Control group was given NS (normal saline). Four weeks after tumor cell injection, the therapy group began to receive 100 mg/kg baicalein by intragastric gavage everyday for up to 15 days. To examine the presence of metastases, the mice were sacrificed 8 weeks after the injection of tumor cells. Their lungs and livers were removed, fixed, sectioned, and stained with hematoxylin and eosin (H&E). Representative fields for each group were photographed and the number of metastases was counted.
  | Table 1 Experimental grouping and treatment |
Quantitative real-time RT-PCR
Total cell RNA was extracted using the MiniBEST Universal RNA Extraction Kit (TaKaRa, Tokyo, Japan) according to the manufacturer’s instructions. cDNA was synthesized with PrimeScriptTM RT Master Mix (TaKaRa). Quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) reaction system was prepared following the instructions given in the SYBR®Premix Ex TaqII (Tli RNaseH Plus) RT-PCR Kit (TaKaRa) using 100 ng cDNA from each sample. Primers for specific genes investigated in this study are listed in Table 2. GAPDH was used as endogenous control. Each sample was analyzed in triplicate by using a StepOne™Real-Time PCR System (Thermo Fisher Scientific). Gene copy number data was analyzed in accordance with the 2−ΔΔCt method relative to expression of GAPDH and compared with the control.
  | Table 2 Primers used for qRT-PCR analysis |
Western blot analysis
MDA-MB-231 cells were separately treated with different concentrations of baicalein (0, 10, 20, and 40 μmol/L) for 24 and 48 hours. For comparing the protein level of SATB1, MCF-10A, MCF7, SKBR3, and MDA-MB-231 cells were directly lysed during their logarithmic growing phase. A total of 5×106 cells of each group were suspended in 320 μL of ice-cold lysis buffer, a mixture of 1% B, C, and D components in RIPA (A), and allowed to settle on ice for 1 hour. For the lysis of tissue protein, modified RIPA buffer (containing 50 mM Tris pH 8.0, 150 mM NaCl, 0.02% NaN3, 0.1% sodium dodecyl sulfate (SDS), 1% NP-40, 0.5% sodium deoxycholate, 1.56 mM protease inhibitor cocktail, 1 mM phenylmethanesulfonyl fluoride (PMSE), and 1 mM sodium orthovanadate) was used. Protein solutions from cell or tissue lysates were collected, and 15–20 μL of each sample (the quantity of total protein was made equal between all the groups) was separated by SDS–PAGE and transferred onto the polyvinylidene fluoride (PVDF) membranes (Millipore). The membranes were subsequently blocked in skim milk (5% in Tris-buffered saline with TWEEN 20 TBST buffer) at 25°C for 2 hours and then incubated at 4°C overnight with antibodies against SATB1, vimentin, SNAIL, E-cadherin, β-catenin, Wnt1, or β-actin in TBST containing 5% defatted milk separately. The membranes were then incubated with proper HRP-linked secondary antibody for 2 hours at 25°C. Finally, the bands were detected with an enhanced chemiluminescence kit (Thermo Fisher Scientific) and exposed by autoradiography using the chemiluminescence gel imaging system (G: BOX, Syngene, Cambridge, UK). The densitometric analysis was performed using Image-Pro Plus 6.0 (Media Cybernetics, Rockville, MD, USA), and the targeted protein level of each sample was evaluated relative to expression of the control. Comparison was also performed between the two prevention groups in the mouse model.
Immunohistochemistry
Effects of baicalein on the expression of target proteins in vivo were analyzed by immunohistochemistry. Metastatic lesions in the lung and liver of experimental mice were collected and paraffin-embedded. Then the paraffin-embedded tumor tissues were sliced into 5 μm sections. Antigen retrieval was performed with citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0) in a domestic stainless steel pressure cooker at 120°C for 15 minutes. The sections were incubated with SATB1 (1:100), Wnt1 (1:50), β-catenin (1:50), E-cadherin (1:100), vimentin (1:100), and SNAIL (1:80) primary antibody at 4°C overnight, washed twice with PBS, and incubated with HRP-conjugated secondary antibody at room temperature for 30 minutes. Then the slides were washed and incubated with an Avidin–Biotin solution (Biocare Medical, Concord, CA, USA). Finally, sections were incubated with DAB and counterstained with H&E. Sections that were not incubated with primary antibody served as negative control. Images of sections were taken using Zeiss microscope (Zeiss Microscopy, Jena, Germany) and analyzed by Image-Pro Plus 6.0 (Media Cybernetics).
Statistical methods
Experiments were all repeated three times. SPSS 20.0 (IBM Corporation, Armonk, NY, USA) statistics software was applied to assess the statistical significance of differences in numerical data using the Student’s t-test. All statistical tests and corresponding P-values were two sided. P<0.05 was considered to be statistically significant.
Results
Baicalein inhibits the proliferation of MDA-MB-231 cells
The antiproliferation effects of baicalein (0–120 μmol/L) on MDA-MB-231 cells at different time points are shown in Table 3 and Figure 2. The proliferation potential of MDA-MB-231 cells was suppressed by baicalein in a dose- and time-dependent manner. IC50 of baicalein at 24, 48, and 72 hours were 89.71, 59.50, and 33.61 μmol/L, respectively. To exclude the obvious antiproliferative effect of baicalein, concentration range lower than 50 μmol/L was chosen for all subsequent experiments.
  | Table 3 Effect of baicalein on proliferation of MDA-MB-231 cells ( |
Baicalein suppresses the motility of MDA-MB-231 cells in vitro
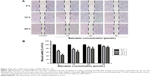
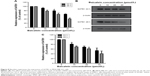
Cancer cell motility predicts its metastasis potential, so wound healing assay was applied to evaluate the effect of baicalein on migration of MDA-MB-231 cells. As shown in Figure 3A, cells of the control group migrated into the wound area continuously and significantly as time progressed, compared to the baicalein-treated groups. After treatment with 10, 20, and 40 μmol/L baicalein for 24 hours, breast cancer cell motility was inhibited by 31.17%, 61.09%, and 74.15%, respectively (Figure 3B).
Baicalein inhibits the invasion of MDA-MB-231 cells in vitro
Invasion is another important aspect that shows the metastatic capacity of cancer cells. Thus, the classic method – Transwell in vitro invasion assay – was used to assess the invasion ability of MDA-MB-231 cells. Figure 4A shows that baicalein reduced the invasiveness of MDA-MB-231 cells in a concentration-dependent pattern. Quantification analysis indicated that after 24 hours, the invasive potential of MDA-MB-231 cells were suppressed by 32.04%, 54.59%, and 81.76% when cells were treated with 10, 20, and 40 μmol/L baicalein, respectively (Figure 4B).
Baicalein suppresses breast cancer metastasis in vivo
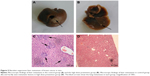
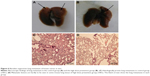
We evaluated the in vivo antitumor effects of baicalein by using the xenograft nude mouse model of breast cancer metastasis. After preventive or therapeutic treatment of baicalein, lung and liver tissues from each group were collected and analyzed. In the control group, obvious metastatic lesions in the liver (Figure 5A) and lung (Figure 6A) were visible to the naked eye. In all mice (6/6), liver and lung metastases were also detected when viewed under microscope (Figures 5C and 6C). In contrast, macroscopic nodules of the therapy or prevention group were rather smaller in size (Figures 5B and 6B), and some were hardly visible to the naked eye. At the microscopic level, baicalein treatment effectively reduced metastasis rate (liver: 4/6 in therapy group, 5/6 in low-dose prevention group, and 3/6 in high-dose prevention group; lung: 3/6 in therapy group, 4/6 in low-dose prevention group, and 3/6 in high-dose prevention group) and, most importantly, the lesion size (Figures 5D and 6D).
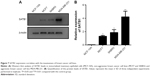
SATB1 expression correlates with the invasiveness of breast cancer cell lines
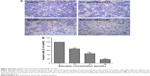
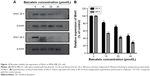
To investigate the correlation between SATB1 and the invasiveness of breast cancer, we used Western blot to examine the protein level of SATB1 in four breast epithelial cell lines, including the immortalized mammary epithelial cells (MCF-10A), two nonmetastatic cancer cell lines (MCF7 and SKBR3), and one highly metastatic cancer cell line (MDA-MB-231). As shown in Figure 7A, SATB1 protein expression level is highest in MDA-MB-231 cell line, (significantly higher than in MCF7 and SKBR3 cell lines), while MCF-10A cells barely expressed SATB1 protein. Quantification analysis (Figure 7B) showed that when using MCF-10A as the internal reference, SATB1 protein expression level are 1.31-, 1.87-, and 4.21-fold in MCF7, SKBR3, and MDA-MB-231 cells, respectively, indicating that SATB1 expression is correlated with the invasive phenotype of breast cancer cells.
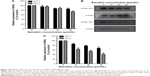
Baicalein suppresses the expression of SATB1 in MDA-MB-231 cells
A number of studies9–18 have indicated that SATB1 regulates the malignant progression of various cancers, including breast cancer, and its overexpression predicts poor outcome for cancer patients. To explore the possible antimetastatic mechanism of baicalein, the expression of SATB1 messenger RNA (mRNA) and protein were both examined in MDA-MB-231 cells treated with different concentrations of baicalein for 24 or 48 hours. As shown in Figure 8A, after treatment for 48 hours, 10, 20, and 40 μmol/L baicalein inhibited SATB1 mRNA expression by 21.58%, 37.29%, and 58.77%, respectively. Furthermore, SATB1 protein expression was suppressed by baicalein in a dose- and time-dependent manner compared with the control group (Figure 8B). Figure 8C showed that 10, 20, and 40 μmol/L baicalein inhibited the expression of SATB1 protein by 30.12%, 38.35%, and 59.18%, respectively, after 48 hours treatment.
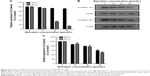
Baicalein regulates the expression of EMT-related molecules in MDA-MB-231 cells
EMT is one of the crucial mechanisms by which tumor cells detach from their primary site and invade into surrounding tissues as well as the vascular system. It promotes metastasis during the malignant progression of epithelium-originated cancers.34,35 To investigate whether baicalein inhibits breast cancer cell metastasis through regulating EMT, we chose four representative molecules and assessed their expression levels in MDA-MB-231 cells after baicalein treatment. Expression of E-cadherin represents the epithelial phenotype, while vimentin is the molecular marker of mesenchymal cells. Transcription factor SNAIL is regarded to be an important suppressor of several epithelial markers, mainly E-cadherin, thus inducing EMT. MDA-MB-231 cells were treated with 0, 10, 20, and 40 μmol/L baicalein for 24 or 48 hours and then subjected to qRT-PCR or Western blot. Figure 9A shows that the inhibition rate of vimentin transcription level was approximately 12.31%, 27.93%, and 35.18% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively; and 10, 20, and 40 μmol/L baicalein downregulated the transcription level of SANIL by 4.78%, 11.85%, and 26.08%, respectively, after 48 hours (Figure 10A). Figures 9B and 10B show that baicalein suppressed the protein expression of both vimentin and SNAIL in a dose- and time-dependent manner. The inhibition rate of vimentin protein was 16.47%, 28.92%, and 40.33% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively (Figure 9C). Figure 10C shows that 10, 20, and 40 μmol/L baicalein downregulated the protein level of SNAIL by 36.95%, 45.79%, and 58.83%, respectively, after 48 hours of treatment.
Figure 11A and B shows that baicalein upregulated the mRNA and protein levels of E-cadherin both in a dose- and time-dependent way. After treatment with 10, 20, and 40 μmol/L baicalein for 48 hours, the expression level of E-cadherin mRNA was increased to 1.49-, 2.45-, and 4.42-fold compared to the control. Further, Figure 11C shows that the expression level of E-cadherin protein was increased to 2.09-, 2.29-, and 2.56-fold, respectively.
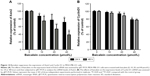
Inhibition effect of baicalein on Wnt/β-catenin pathway in vitro
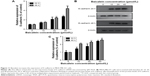
It has been verified that deregulation of Wnt/β-catenin pathway may contribute to the progression of various cancers.22,36 To further investigate the possible mechanisms by which baicalein exerts its anticancer effect, Western blot was applied to assess the expression level of Wnt1 protein – one of the main ligands in the Wnt/β-catenin pathway. Figure 12A shows that baicalein suppressed the protein expression of Wnt1 in a dose- and time-dependent manner. The inhibition rate of Wnt1 protein was 46.68%, 58.13%, and 72.02% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively (Figure 12B).
β-Catenin is the key effector molecule in the Wnt/β-catenin pathway. A study found that the translocation of β-catenin from the cytoplasm to the nucleus plays an important role in promoting the expression of related genes of EMT.37 Figure 13A shows that the inhibition rate of β-catenin transcription level was approximately 5.93%, 66.55%, and 86.74% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively. Figure 13B shows that baicalein suppressed the protein expression of β-catenin in a dose- and time-dependent manner. The inhibition rate of β-catenin protein was 11.09%, 23.30%, and 50.38% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively (Figure 13C).
Axin2 and Cyclin D1 are the downstream targets of the Wnt/β-catenin pathway. We used qRT-PCR to examine the effect of baicalein on the transcription level of Axin2 and Cyclin D1. Figure 14A shows that after treatment with 10, 20, and 40 μmol/L baicalein for 48 hours, the expression level of Axin2 mRNA was suppressed by 25.05%, 46.13%, and 61.24%, respectively. The inhibition rate of Cyclin D1 mRNA was also 4.62%, 11.38%, and 22.85% after 48 hours of treatment with 10, 20, and 40 μmol/L baicalein, respectively (Figure 14B).
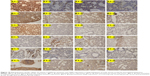
Baicalein downregulates the expression of SATB1, EMT, and Wnt/β-catenin pathway in vivo
To further investigate the mechanisms involved in the antimetastatic effects of baicalein on breast cancer in vivo and acquire more reliable evidences to support our in vitro experimental findings, we applied immunohistochemistry as well as Western blot to evaluate the influence baicalein exerts on expression of various proteins studied in the previous in vitro assays. According to product datasheets of the primary antibodies, location of SATB1 or SNAIL in cells was mainly limited to the nucleus, but cytoplasmic staining also appears in some tissue sections. For Wnt1, β-catenin, E-cadherin, and vimentin, cytoplasmic (and membranous) staining was considered positive.
As shown in Figures 15 and 16, treatment of baicalein significantly suppressed the protein expression of SATB1, Wnt1, and β-catenin in the metastases of the mouse model, further verifying our previous findings that baicalein effectively downregulates SATB1 and Wnt/β-catenin signaling. In addition, vimentin and SNAIL expression in the control groups was much higher than in the therapy group and the two prevention groups, while increased expression of E-cadherin was observed in the baicalein-treated groups. These results suggest that baicalein inhibits, and even possibly, reverses the EMT that occurs during liver or lung metastasis of breast cancer in vivo. Statistical analysis proved the significant difference between control and other three experimental groups that was caused by baicalein treatment. Comparison between low-dose and high-dose prevention groups was also performed. The results indicate that baicalein downregulates the expression of SATB1, EMT, and Wnt/β-catenin pathway in a dose-dependent manner. Moreover, high-dose prevention treatment (IG100 mg/kg) of baicalein exerts greater effects on expression of studied proteins (SATB1, Wnt1, β-catenin, E-cadherin, vimentin, and SNAIL) than therapy treatment (which began later). Expression differences of most of the analyzed proteins between the high-dose prevention group and therapy group are statistically significant (data not shown). These results indicate that early treatment with baicalein is more effective for the inhibition of tumor metastasis.
In addition to immunohistochemistry, Western blot was also performed to determine whether the in vivo antimetastatic effect of baicalein is associated with regulation of SATB1, EMT components, and Wnt/β-catenin signaling. Figure 17 shows that baicalein remarkably reduced the expression of SATB1, Wnt1, β-catenin, vimentin, and SNAIL, while increasing the expression of E-cadherin at the protein level, all in a dose-dependent manner. Results acquired by Western blot are in accordance with those of the immunohistochemistry assay, further verifying that treatment with baicalein can effectively inhibit the expression of SATB1 and downregulate Wnt/β-catenin pathway, which may cooperatively contribute to the prevention of EMT process and, eventually, suppress tumor metastasis in vivo.
Discussion
In recent years, the antitumor effect of the flavonoid baicalein has been confirmed in various cancers, including breast cancer.38–42 However, its antimetastatic effect and the specific mechanism(s) remain an unsolved problem. Our study revealed that baicalein significantly suppressed the in vitro metastatic potential of MDA-MB-231 cells possibly by inhibition of EMT via downregulation of SATB1 and Wnt/β-catenin pathway.
Normal human cells acquire uncontrolled proliferation ability through genetic variation, and resistance to apoptosis is the main mechanism of tumorigenesis. Immortalization of cancer cells also forms the biological basis for tumor growth and metastasis. We chose the highly metastatic breast cancer cell line MDA-MB-231 to investigate the antitumor effect of baicalein. According to the results of the MTT assay, we again confirmed that baicalein significantly inhibited the proliferation of MDA-MB-231 cells in a dose- and time-dependent manner. Wound healing assay has been widely used to assess the motility and migration ability of cells on the two-dimensional substratum. Tumor cells adhere to the basic membrane and extracellular matrix (ECM), secreting numerous proteases, such as matrix metalloproteinases (MMPs), to degrade the ECM and then migrate through the normal biological barriers; these constitute the crucial steps occurring during tumor metastasis.43 Transwell in vitro invasion assay has been effectively applied to evaluate the invasiveness of cancer cells. Our results suggested that baicalein significantly suppressed the migration and invasion capacity of MDA-MB-231 cells in vitro compared to the control, both in a dose- and time-dependent manner.
SATB1 is a nuclear protein required for T-cell development. It combines with the AT-rich sequence in the genome and constitutes a functional nuclear architecture that has a “cage-like” structure, which is also termed as “the SATB1 regulatory network”. As a genome organizer, SATB1 regulates the expression of thousands of genes through recruiting chromatin remodeling/modifying enzymes and transcription factors, thus determining the classification of cell types and functions.6,44 Han et al9 first revealed that SATB1 plays an important role during the progression of breast cancer. They pointed out that SATB1 is expressed during oncogenesis and reprograms the transcription profiles of breast tumors to promote tumor growth and metastasis. Silencing SATB1 in the highly metastatic breast cancer cell line MDA-MB-231 restored cell polarity; the invasive phenotype is reversed and cell metastatic capacity is significantly inhibited. Ectopic expression of SATB1 in the nonmetastatic breast cancer cell line SKBR3 led to gene expression patterns consistent with aggressive tumor-phenotype-induced cell invasiveness and enhanced cell metastatic activity in vivo. Although subsequent studies concerning various cancers have provided compelling evidences for the important function of SATB1 in cancer progression, there are dissenting views from some other research groups questioning this theory. Iorns et al45 found that the invasive ability of MDA-MB-231 cells was not suppressed after knocking down of SATB1. Upregulation of SATB1 in noninvasive cells did not promote cell invasiveness either. Furthermore, clinical statistical analysis showed no correlation between SATB1 and the overall survival of primary breast cancer patients. Similar opposition also came from a study concerning glioma.46
To investigate whether SATB1 is associated with metastatic potential of breast cancer, we performed Western blot to examine the expression level of SATB1 protein in immortalized mammary epithelial cells MCF-10A, nonmetastatic breast cancer cell lines MCF7 and SKBR3, and highly metastatic breast cancer cell line MDA-MB-231. Our results showed that the expression of SATB1 protein is positively related to the invasiveness of breast cancer cells, which is basically in accordance with the result of Han et al.9 To further study the antimetastatic mechanism of baicalein, we explored whether baicalein inhibits cell invasion by regulating the expression of SATB1. The results showed that baicalein significantly suppressed the expression of SATB1 mRNA and protein, both in a dose- and time-dependent pattern.
EMT happens when epithelial cells transform into mesenchymal phenotype during various physiological as well as pathological processes, such as embryotic development, fibrosis and wound healing, and tumor progression. Cells that have undergone EMT gain a higher invasive potential and acquire apoptosis resistance.47 EMT also induces the stemness of cells and participates in the regulation of immunosuppression.48 The biological activities that initiate and promote EMT process include activation of transcriptional factors, expression of certain cell-surface proteins, recognition and expression of cytoskeletal proteins, and production of ECM-degrading proteases.47
The β-catenin protein is a component of adheren junctions; it promotes cell adhesion by binding to the intracellular domain of the transmembrane protein cadherin.49 During EMT, β-catenin mediates the binding of cadherins and cytoskeleton, constitutes the transcription factor coactivation complex (β-catenin/TCF/LEF complex) with T-cell factor (TCF/LEF), which directly regulates EMT-associated genes, especially SNAIL.50,51 Transcriptional factor SNAIL has been proved to be the common downstream target shared by many signaling pathways that regulate EMT. SNAIL induces EMT through suppression of many epithelial markers, including E-cadherin and claudins (a family of proteins that are the most important components of tight junction). E-cadherin is one of the molecular markers of the epithelial phenotype; its expression decreases during EMT in embryotic development, fibrosis, and tumor progression.34,35 Downregulation or loss of E-cadherin results in the destruction of intercellular connection and contributes to metastasis of tumor cells, which is the most remarkable event during EMT.52 Vimentin remains a controversial marker of EMT. Many studies47,48,53 suggested that expression of vimentin positively correlates with the tumor metastasis; hence, it is regarded as a marker for cancer-related EMT.53 β-catenin also acts as the key effector molecule of the Wnt/β-catenin signaling pathway. In normal epithelial cells and nontumor cells, β-catenin is located on cell membrane; once EMT occurs, it translocates to the cytoplasm (dissociates from E-cadherin) or the nucleus (acts as transcription activator) and promotes the expression of related genes of EMT.36
Some studies suggested that the mechanism by which SATB1 regulates tumor metastasis is associated with its role in regulation of EMT. In breast cancer, Han et al9 found that SATB1 depletion blocks the upregulation of cell-structure genes typically seen in invasive breast cancers, among them vimentin and fibronectin. On the contrary, SATB1 depletion results in the upregulation of E-cadherin and β-catenin, and the E-cadherin suppressors – SNAIL and SIP1 – are also downregulated. Thus, EMT is reversed and breast cancer cells restore polarized cellular structures found in normal mammary epithelial cells. Similar results are mentioned in other studies regarding colon cancer54 and liver cancer.13
In this study, we examined the mRNA and protein level of E-cadherin, SNAIL, and vimentin in MDA-MB-231 cells that are pretreated with 0, 10, 20, and 40 μmol/L baicalein for 24 or 48 hours. Our results displayed that baicalein significantly downregulated SNAIL and vimentin while upregulating E-cadherin, all in a dose- and time-dependent manner. On the basis of the results obtained, two inferences can be drawn: 1) SATB1 contributes to breast cancer metastasis partly by regulating the EMT-associated molecules and promoting EMT and 2) the antimetastatic effect of baicalein is possibly associated with the downregulation of SATB1, and inhibition of EMT is probably the downstream effect.
The highly conserved canonical Wnt/β-catenin pathway is the most studied Wnt signal pathway. It plays a vital role in embryonic development and tissue homeostasis of adults.55 Deregulation of Wnt/β-catenin signaling is closely related to many diseases, including cancer.56 It has been suggested that Wnt/β-catenin is among the main pathways that regulate EMT, especially in tumor stem cells, which may be the key mechanism by which Wnt/β-catenin signal mediates breast cancer progression.36,57,58 Wnt1 is one of the canonical ligands for the Wnt/β-catenin pathway. Translocation into the nucleus as well as accumulation in cytoplasm of β-catenin indicates activation of Wnt/β-catenin pathway. Cyclin D1 and Axin2 are two downstream targets of Wnt/β-catenin pathway. Our study found that baicalein significantly inhibited the expression of Wnt1 and β-catenin in MDA-MB-231 cells and also suppressed the transcription level of both Cyclin D1 and Axin2, all in a dose- and time-dependent manner. These results indicate that baicalein inhibits breast cancer metastasis possibly by downregulating the activity of Wnt/β-catenin pathway.
In addition to the in vitro assays, the in vivo experiments have provided more evidence to support our findings in this study. We built the xenograft nude mouse model of breast cancer metastasis and performed a series of assays to explore the in vivo antimetastatic effects of baicalein. The results indicate that baicalein can effectively inhibit SATB1 protein expression, suppress EMT, and downregulate the Wnt/β-catenin pathway during lung and liver metastasis of breast cancer cells in vivo, which is in accordance with results from our in vitro assays.
It is known that SATB1 is mainly expressed in the T-cell linage (thymocytes and TH2 cells) and plays multiple roles in the normal development of thymus and differentiation of pluripotent embryonic stem cells.59 Wnt/β-catenin signaling is also indispensable for the differentiation of T-cells and maturation of thymus.55 Notani et al27 found that SATB1 recruits β-catenin and regulates TH2 cells differentiation in a Wnt-dependent manenr. In addition, Wnt/β-catenin and SATB1 share many target genes, such as c-Myc and Bcl-2.6,60,61 To sum up, the common involvement in regulation of thymus development, EMT, and stemness mentioned earlier indicate important associations between Wnt/β-catenin pathway and SATB1. We infer that they are also functionally connected during cancer progression. The question of whether the multifunctional “genome organizer” SATB1 exerts upstream regulation on Wnt/β-catenin pathway, and the mechanisms by which they interact with each other, warrants further studies.
Conclusion
In conclusion, we first report that baicalein has the potential to suppress MDA-MB-231 cells metastasis both in vitro and in vivo possibly by inhibition of EMT, which may be attributed to both downregulation of SATB1 and Wnt/β-catenin pathway. These findings provide a new perspective for the mechanisms involved in achieving the antimetastatic effect of baicalein and may serve as theoretical basics for therapeutic application of baicalein in antimetastatic therapy for breast cancer.
Ethics
Female Balb/c nude mice (4–8 weeks old, body weight 18–20 g) were purchased from Experimental Animal Center of Xi’an Jiao tong University (Xi’an, People’s Republic of China). Animal experiments performed in this study were conducted according to the recommended guidelines for the care and use of laboratory animals issued by the Chinese Council on Animal Research, and approved by the ethics committee of Xi’an Jiaotong University. Ethical approval for use of human cell lines was not needed as per Xi’an Jiaotong University ethical commitee guidelines. The breast cancer cell lines used in this study are all commonly used and subcultured cell lines obtained from public cell banks. They were not derived from our patients or healthy donors.
Author contributions
SQZ directed and supervised this study and provided funding. XCM designed the experiments, conducted the entire cell in vitro assays, performed statistical analysis, and drafted the manuscript. WJY conducted the in vivo assays. XCM and WJY performed qRT-PCR, Western blot, and IHC. ZJD, XYG, and YNM provided technological advice on the project. QTX and JTJ consulted on the project and the manuscript. All authors have approved the content of the final manuscript.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Number 81274136, Number 81471670); Program for New Century Excellent Talents in Universities of China (Number NCET-110439); China Postdoctoral Science Foundation (Number 2014M560791, Number 2015T81037); and the Fundamental Research Funds for the Central Universities, People’s Republic of China (Number 2014qngz-04).
Disclosure
The authors report no conflicts of interest in this work.
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: Globocan 2008. Int J Cancer. 2010;127(12):2893–2917. | ||
Benson JR, Jatoi I. The global breast cancer burden. Future Oncol. 2012;8(6):697–702. | ||
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. | ||
Steeg PS. Tumor metastasis: mechanistic insights and clinical challenges. Nat Med. 2006;12(8):895–904. | ||
Christofori G. New signals from the invasive front. Nature. 2006;441(7092):444–450. | ||
Cai ST, Han HJ, Kohwi-Shigematsu T. Tissue-specific nuclear architecture and gene expression regulated by SATB1. Nat Genet. 2003;34(1):42–51. | ||
Wen J, Huang SM, Rogers H, Dickinson LA, Kohwi-Shigematsu T, Noguchi CT. SATB1 family protein expressed during early erythroid differentiation modifies globin gene expression. Blood. 2005;105(8):3330–3339. | ||
Alvarez JD, Yasui DH, Niida H, Joh T, Loh DY, Kohwi-Shigematsu T. The MAR-binding protein SATB1 orchestrates temporal and spatial expression of multiple genes during T-cell development. Genes Dev. 2000;14(5):521–535. | ||
Han HJ, Russo J, Kohwi Y, Kohwi-Shigematsu T. SATB1 reprograms gene expression to promote breast tumour growth and metastasis. Nature. 2008;452(7184):187–193. | ||
Cheng C, Lu X, Wang G, et al. Expression of SATB1 and heparanase in gastric cancer and its relationship to clinicopathologic features. APMIS. 2010;118(11):855–863. | ||
Lu X, Cheng C, Zhu S, et al. SATB1 is an independent prognostic marker for gastric cancer in a Chinese population. Oncol Rep. 2010;24(4):981–987. | ||
Zhang J, Zhang B, Zhang X, et al. SATB1 expression is associated with biologic behavior in colorectal carcinoma in vitro and in vivo. PLoS One. 2013;8(1):e47902. | ||
Tu W, Luo M, Wang Z, et al. Upregulation of SATB1 promotes tumor growth and metastasis in liver cancer. Liver Int. 2012;32(7):1064–1078. | ||
Xiang J, Zhou L, Li S, et al. AT-rich sequence binding protein 1: contribution to tumor progression and metastasis of human ovarian carcinoma. Oncol Lett. 2012;3(4):865–870. | ||
Nodin B, Hedner C, Uhlen M, Jirström K. Expression of the global regulator SATB1 is an independent factor of poor prognosis in high grade epithelial ovarian cancer. J Ovarian Res. 2012;5(1):24. | ||
Shukla S, Sharma H, Abbas A, et al. Upregulation of SATB1 is associated with prostate cancer aggressiveness and disease progression. PLoS One. 2013;8(1):e53527. | ||
Mao L, Yang C, Wang J, et al. SATB1 is overexpressed in metastatic prostate cancer and promotes prostate cancer cell growth and invasion. J Transl Med. 2013;11:111. | ||
Han B, Luan L, Xu Z, Wu B. Expression and biological roles of SATB1 in human bladder cancer. Tumor Biol. 2013;34(5):2943–2949. | ||
Gough NR. Focus issue: Wnt and β-catenin signaling in development and disease. Sci Signal. 2012;5(206):eg2. | ||
Dickinson ME, McMahon AP. The role of Wnt genes in vertebrate development. Curr Opin Genet Dev. 1992;2(4):562–566. | ||
Brennan KR, Brown AM. Wnt proteins in mammary development and cancer. J Mammary Gland Biol Neoplasia. 2004;9(2):119–131. | ||
Howe LR, Brown AM. Wnt signaling and breast cancer. Cancer Biol Ther. 2004;3:36–41. | ||
Kurayoshi M, Oue N, Yamamoto H, et al. Expression of Wnt-5 is correlated with aggressiveness of gastric cancer by stimulating cell migration and invasion. Cancer Res. 2006;66(21):10439–10448. | ||
Gupta S, Iljin K, Sara H, et al. FZD4 as a mediator of ERG oncogene-induced Wnt signaling and epithelial-to-mesenchymal transition in human prostate cancer cells. Cancer Res. 2010;70(17):6735–6745. | ||
Kobune M, Chiba H, Kato J, et al. Wnt3/RhoA/ROCK signaling pathway is involved in adhesion-mediated drug resistance of multiple myeloma in an autocrine mechanism. Mol Cancer Ther. 2007;6(6):1774–1784. | ||
Lalli E, Wakil AE. The Wnt/β-catenin pathway in adrenocortical development and cancer. Mol Cell Endocrinol. 2010;332(1–2):32–37. | ||
Notani D, Gottimukkala KP, Jayani RS, et al. Global regulator SATB1 recruits β-catenin and regulates TH2 differentiation in Wnt-dependent manner. PLoS Biol. 2010;8(1):e1000296. | ||
Pidgeon GP, Kandouz M, Meram A, Honn KV. Mechanisms controlling cell cycle arrest and induction of apoptosis after 12-lipoxygenase inhibition in prostate cancer cells. Cancer Res. 2002;62(9):2721–2727. | ||
Lee HZ, Leung HW, Lai MY, Wu CH. Baicalein induced cell cycle arrest and apoptosis in human lung squamous carcinoma CH27 cells. Anticancer Res. 2005;25(2A):959–964. | ||
Hyde CA, Missailidis S. Inhibition of arachidonic acid metabolism and its implication on cell proliferation and tumour-angiogenesis. Int Immunopharmacol. 2009;9:701–715. | ||
Ling Y, Chen Y, Chen P, et al. Baicalein potently suppresses angiogenesis induced by vascular endothelial growth factor through the p53/Rb signaling pathway leading to G1/S cell cycle arrest. Exp Biol Med. 2011;236(7):851–858. | ||
Eliza LZ, Agnieszka W, Zofia BO. Anticancer properties of Scutellaria baicalensis root in aspect of innate immunity regulation. Adv Clin Exp Med. 2010;19(4):419–428. | ||
Chung H, Choi HS, Seo EK, Kang DH, Oh ES. Baicalin and baicalein inhibit transforming growth factor-β1-mediated epithelial-mesenchymal transition in human breast epithelial cells. Biochem Biophys Res Commun. 2015;458(3):707–713. | ||
Prasad CP, Rath G, Mathur S, Bhatnagar D, Parshad R, Ralhan R. Expression analysis of E-cadherin, slug and GSK3β in invasive ductal carcinoma of breast. BMC Cancer. 2009;9:325. | ||
Logullo AF, Nonogaki S, Pasini FS, Osório CA, Soares FA, Brentani MM. Concomitant expression of epithelial-mesenchymal transition biomarkers in breast ductal carcinoma: association with progression. Oncol Rep. 2010;23(2):313–320. | ||
Lamb R, Ablett MP, Spence K, Landberg G, Sims AH, Clarke RB. Wnt pathway activity in breast cancer sub-types and stem-like cells. PLoS One. 2013;8(7):e67811. | ||
Müller T, Bain G, Wang X, Papkoff J. Regulation of epithelial cell migration and tumor formation by β-catenin signaling. Exp Cell Res. 2002;208(1):119–133. | ||
Wang L, Ling Y, Chen Y, et al. Flavonoid baicalein suppresses adhesion, migration and invasion of MDA-MB-231 human breast cancer cells. Cancer Lett. 2010;297:42–48. | ||
Zheng YH, Yin LH, Grahn TH, Ye AF, Zhao YR, Zhang QY. Anticancer effects of baicalein on hepatocellular carcinoma cells. Phytother Res. 2014;28(9):1342–1348. | ||
Yan X, Rui X, Zhang K. Baicalein inhibits the invasion of gastric cancer cells by suppressing the activity of the p38 signaling pathway. Oncol Rep. 2015;33(2):737–743. | ||
Shang D, Li Z, Zhu Z, et al. Baicalein suppresses 17-β-estradiol-induced migration, adhesion and invasion of breast cancer cells via the G protein-coupled receptor 30 signaling pathway. Oncol Rep. 2015;33(4):2077–2085. | ||
Zhang Z, Lv J, Lei X, et al. Baicalein reduces the invasion of glioma cells via reducing the activity of p38 signaling pathway. PLoS One. 2014;9(2):e90318. | ||
Fingleton B. Matrix metalloproteinases: roles in cancer and metastasis. Front Biosci. 2006;11:479–491. | ||
Nakayama Y, Mian IS, Kohwi-Shigematsu T, Ogawa T. A nuclear targeting determinant for SATB1, a genome organizer in the T cell lineage. Cell Cycle. 2005;4(8):1099–1106. | ||
Iorns E, Hnatyszyn HJ, Seo P, Clarke J, Ward T, Lippman M. The role of SATB1 in breast cancer pathogenesis. J Natl Cancer Inst. 2010;102:1284–1296. | ||
Han S, Xia J, Qin X, Han S, Wu A. Phosphorylated SATB1 is associated with the progression and prognosis of glioma. Cell Death Dis. 2013;4:e901. | ||
Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119(6):1420–1428. | ||
Thiery JP, Acloque H, Huang RYJ. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139(5):871–890. | ||
Kemler R. From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet. 1993;9(9):317–321. | ||
Bienz M. β-Catenin: a pivot between cell adhesion and Wnt signaling. Curr Biol. 2005;15(2):R64–R67. | ||
Yook JI, Li XY, Ota I, et al. A Wnt-Axin2-GSK3β cascade regulates Snail1 activity in breast cancer cells. Nat Cell Biol. 2006;8(12):1398–1406. | ||
Hay ED, Zuk A. Transformations between epithelium and mesenchyme: normal, pathological, and experimentally induced. Am J Kidney Dis. 1995;26(4):678–690. | ||
Raymond WA, Leong AS. Vimentin – a new prognostic parameter in breast carcinoma? J Pathol. 1989;158(2):107–114. | ||
Frömberg A, Rabe M, Aigner A. Multiple effects of the special AT-rich binding protein 1 (SATB1) in colon carcinoma. Int J Cancer. 2014;135(11):2537–2546. | ||
Clevers H. Wnt/β-catenin signaling in development and disease. Cell. 2006;149(6):1192–1205. | ||
Moon RT, Kohn AD, De Ferrari GV, Kaykas A. WNT and β-catenin signaling: diseases and therapies. Nat Rev Genet. 2004;5(9):691–701. | ||
Teissedre B, Pinderhughes A, Incassati A, Hatsell SJ, Hiremath M, Cowin P. MMTV-Wnt1 and -ΔN89β-catenin induce canonical signaling in distinct progenitors and differentially activate Hedgehog signaling within mammary tumors. PLoS One. 2009;4:e4537. | ||
Ayyanan A, Civenni G, Ciarloni L, et al. Increased Wnt signaling triggers oncogenic conversion of human breast epithelial cells by a notch-dependent mechanism. Proc Natl Acad Sci U S A. 2006;103(10):3799–3804. | ||
Savarese F, Davila A, Nechanitzky R, et al. Satb1 and Satb2 regulate embryonic stem cell differentiation and Nanog expression. Genes Dev. 2009;23(22):2625–2638. | ||
Ma C, Zhang J, Durrin LK, et al. The BCL2 major breakpoint region (mbr) regulates gene expression. Oncogene. 2007;26(18):2649–2657. | ||
Li Q, Dashwood WM, Zhong X, Nakagama H, Dashwood RH. Bcl-2 overexpression in PhIP-induced colon tumors: cloning of the rat Bcl-2 promoter and characterization of a pathway involving β-catenin, c-Myc and E2F1. Oncogene. 2007;26(42):6194–6202. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

 ± s, n=6)
± s, n=6)