Back to Journals » Infection and Drug Resistance » Volume 13
Bacterial Profile and ESBL Screening of Urinary Tract Infection Among Asymptomatic and Symptomatic Pregnant Women Attending Antenatal Care of Northeastern Ethiopia Region
Authors Belete MA
Received 15 April 2020
Accepted for publication 5 July 2020
Published 28 July 2020 Volume 2020:13 Pages 2579—2592
DOI https://doi.org/10.2147/IDR.S258379
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Melaku Ashagrie Belete
Department of Medical Laboratory Science, College of Medicine and Health Science, Wollo University, Dessie, Ethiopia
Correspondence: Melaku Ashagrie Belete
Department of Medical Laboratory Science, College of Medicine and Health Sciences, Wollo University, Dessie 1145, Ethiopia
Tel +251 913867849
Fax +251 333115250
Email [email protected]
Background: Urinary tract infection is a common cause of morbidity in pregnant women. Emergence of antimicrobial resistance particularly ESBL production among bacterial uropathogens is increasing and becoming principal cause of treatment failure. The aim of this study was to determine the bacterial profile, their antimicrobial susceptibility patterns, risk factors and identify ESBL-producing bacterial uropathogens.
Patients and Methods: A hospital-based cross-sectional study was conducted in the Northeastern Ethiopia region. A total of 323 pregnant women were included and structured questionnaire was used to collect sociodemographic and risk factor-related data. About 10mL freshly voided midstream urine specimen was collected, transported and processed according to standard operating procedures. The data obtained were entered into SPSS version 22 and descriptive statistics, chi-square, bivariate and multivariate logistic regression analyses were performed. P-value ≤ 0.05 with corresponding 95% confidence interval were considered for statistical significance.
Results: The overall prevalence of UTI was 15.5% (50/323). The predominant bacterial isolates were Escherichia coli 17 (33.3%) followed by coagulase-negative staphylococci 15 (30.0%) and Staphylococcus aureus 14 (27.5%). Previous history of UTI (AOR=8.824, 95% CI: 3.769,20.654, P< 0.001) and history of catheterization (AOR=3.270, 95% CI: 1.316,8.122, P=0.011) were significantly associated with the occurrence of bacterial UTI. Gram-negative isolates showed high level of resistance to ampicillin 12 (60.0%) and relatively low level of resistance to nitrofurantoin 5(25.0%), norfloxacin 5 (25.0%) and ceftazidime 3 (15.0%). Gram-positive uropathogens showed higher resistance for penicillin 29 (93.5%) and trimethoprim-sulfamethoxazole 23 (79.3%) whereas all isolates were sensitive 29 (100.0%) to nitrofurantoin. Moreover, multidrug resistance was observed among 41 (80.4%) of the isolates, and 3 (15.8%) of isolated gram-negative bacteria were ESBL producers.
Conclusion: High prevalence of bacterial UTI and MDR for commonly prescribed drugs were observed with significant number of ESBL producers. Therefore, instant UTI culture assessment of pregnant women, especially those having possible risk factors such as previous histories of UTI and catheterization; moreover, appropriate prescription and use of antibiotics are necessary.
Keywords: urinary tract infection, pregnant women, bacterial profile, antimicrobial susceptibility pattern, risk factors, ESBL, multidrug resistance
Corrigendum for this paper has been published
Introduction
Urinary Tract Infection (UTI) is one of the most common infectious diseases, with approximately 150 million diagnosed cases each year worldwide.1 Pregnant women have 4 times higher rate of developing UTI compared to non-pregnant women.2,3 UTI is among the most commonly studied health problems in pregnancy ranging 3% to 35% prevalence worldwide in which increased prevalence is predominantly seen among developing countries especially Sub-Saharan Africa, Middle East, and Asia.4
The high incidence and risk of developing UTI in the course of pregnancy are related to abnormal anatomical and physiological changes that occur during this period.5–7 Furthermore, previous history of UTI, increased age, multiparity, sexual activity, history of catheterization, immunodeficiency and lower socioeconomic status are identified as factors likely to increase risk of UTI during pregnancy.1,8-10
Bacteria are the most common agents causing UTI including Escherichia coli, Proteus mirabilis, Klebsiella species, Pseudomonas aeruginosa, Enterobacter species, enterococci, Citrobacter, Staphylococcus aureus, coagulase-negative staphylococci, Streptococcus species and others.11 Gram-negative bacteria are the major isolates causing UTI in which the predominant isolates are Escherichia coli accounting majority (80–90%) of infection.8,9
UTI in pregnancy may be symptomatic or asymptomatic bacteriuria (ASB) which occurs in 2–11% of pregnancies worldwide and is a major predisposition to the development of acute pyelonephritis in 20–50% of untreated ASB cases.12 Furthermore, untreated UTI in pregnancy (symptomatic or asymptomatic) is associated with a 50% increase in the risk of maternal complications of pregnancy which raise extent of preterm labor, prematurity and low birth weight resulting in high perinatal morbidity and mortality.1,13 Early diagnosis and clinical management reduce the incidence of these complications.14 Nevertheless, in developing countries including Ethiopia, urine culture screening is not routinely done as part of antenatal care and treatment is empirical which may lead to emergence and spread of antimicrobial-resistant strains which is a leading cause of treatment failure in UTI.
One of the leading antimicrobial resistance mechanisms for many UTI causing Gram-negative bacteria is extended-spectrum β-lactamase enzyme production that hydrolyzes the β-lactam ring of antimicrobials, which confer bacterial resistance to commonly prescribed antibiotics including penicillins; first, second and third-generation cephalosporins, aztreonams.15,16
Few studies have been conducted showing the prevalence of UTI in different parts of Ethiopia including, Gondar (10.4%), Bahir dar (9.5%), Dire Dawa (14%), Addis Ababa (11.6%) and Hawassa (18.8%).9,17-20 However, all these studies did not determine the ESBL-producing uropathogenic bacteria, which are the current worldwide threat; and data on bacterial profile and detection of ESBL-producing bacteria from UTI among pregnant women was not assessed in the study area. Therefore, we sought to fill this information gap.
Patients and Methods
Study Design, Area and Period
A hospital-based cross-sectional study was conducted from February 2017 to May 2017 at Dessie Referral Hospital, Northeastern Ethiopia. A total of 323 pregnant women with and without symptoms of UTI were included using convenient sampling technique.
Data and Specimen Collection
A structured questionnaire was used to obtain information related to sociodemographic, clinical, and risk factor data. About 10mL freshly voided midstream urine specimen was collected using pre-labeled (date, time, identification code, age), leak-proof, wide mouth, sterile, screw-capped plastic container (FL Medical, Italy) by study participants after appropriate instructions were given.
Specimen Transportation
The collected specimens were then stored in a cold box and transported to Dessie Health Research Regional Laboratory and processed within two hours. In case of unavoidable delay, very few specimens were refrigerated at 4°C until being processed. Immediate inoculation had been performed for the rest of the specimens on arrival to the laboratory.
Cultivation and Identification of Isolates
Midstream urine samples were inoculated using calibrated wire loop (0.001mL) into Cystine Lactose Electrolyte Deficient medium (CLED) (Oxoid Ltd, UK). After cultures were incubated overnight under the aerobic condition at 37°C for 24 hours, colonies were counted to check the presence of significant growth. Colony counts yielding bacterial growth of ≥105 CFU/mL of urine were regarded as significant bacteriuria (SB), but specimens that produce <105CFU/mL were considered insignificant or due to contamination.9 Based on their gram staining reaction, the CLED agar colonies were subcultured onto MacConkey agar (Oxoid, Ltd), blood agar plates and Mannitol salt agar (HiMediaTM), then incubated at 37°C for 24 hours. Identification of bacterial species was done using colony characteristics, gram staining reaction, and biochemical tests following standard procedure. The gram-negative bacteria were identified by indole production, H2S production in Kligler Iron Agar (KIA), citrate utilization, urease test, motility test, and carbohydrate utilization tests. The gram-positive bacteria were identified using catalase and coagulase tests.21 Furthermore, all bacterial isolates were identified using standard clinical laboratory methods.21
Antimicrobial Susceptibility Testing
Antimicrobial susceptibility testing was performed using the Kirby–Bauer disk diffusion method recommended by the Clinical and Laboratory Standards Institute (CLSI).22 Pure culture colonies of 24-hour growth were suspended in a tube with 4mL of physiological saline to get bacterial inoculums equivalent to 0.5 McFarland turbidity standards. A sterile cotton swab was dipped, rotated across the wall of the tube to avoid excess fluid and was evenly inoculated on Muller-Hinton agar (Conda ltd, USA) and then the antibiotic discs were placed on MHA plates. The following antimicrobials were used based on the CLSI recommendations and local frequent prescriptions of these drugs for the treatment of UTI: clindamycin (CL,10μg), erythromycin (E,15μg), penicillin (PEN,10µg), chloramphenicol (CAF, 30μg), ciprofloxacin (CIP,5µg), tetracycline (TTC,30µg), trimethoprim-sulfamethoxazole (SXT,1.25/23.75µg), nitrofurantoin (F,300µg), norfloxacin (NOR,10µg) and vancomycin (VA,30µg) for Gram-positive organisms; and ciprofloxacin (CIP,5µg), tetracycline (TTC,30µg), trimethoprim-sulfamethoxazole (SXT,1.25/23.75µg), nitrofurantoin (F,300µg), norfloxacin (NOR,10µg), ceftriaxone (CRO,30μg), amoxicillin-clavulanic acid (AMC,20/10μg), cefotaxime (CTX,30µg), ceftazidime (CAZ,30µg), ampicillin (AMP,10μg), amikacin (AMK,30μg) and gentamicin (GN,10μg) for Gram-negative bacteria. Moreover, imipenem (10µg) was used to check the susceptibility pattern for ESBL-producing gram-negative bacterial uropathogens. All antibiotic discs were from Oxoid, Ltd, UK. The plates were then incubated at 37°C for 24 hours. Diameters of the zone of inhibition around the discs were measured using a digital caliper. The interpretation of the results of the antimicrobial susceptibility tests was based on the standardized table supplied by CLSI22 criteria as sensitive, intermediate, and resistant.
Extended-Spectrum Beta-Lactamase Detection
Initial screening for ESBL was done by the diameters of zones of inhibition produced by either of ceftriaxone (30µg), ceftazidime (30 µg) or cefotaxime (30µg) from the antimicrobial susceptibility test on Muller-Hilton media (Conda Ltd) according to the CLSI screening criteria. These breakpoints indicative of suspicion for ESBL production were: for ceftriaxone ≤ 25mm, for ceftazidime (30 µg) ≤22mm and cefotaxime ≤27mm. After this initial screening, phenotypic detection of ESBL production was confirmed by Combined Disk (Double Disk Potentiate) test according to CLSI guidelines.22 The organism to be tested was spread onto a Mueller–Hinton agar plate using similar procedures as for drug susceptibility testing. A ceftazidime (30 µg) disk and cefotaxime (30µg) disk were used alone and their combination with clavulanic acid (30 µg/10 µg) for phenotypic confirmation of the presence of ESBLs. After incubation at 37ºC for 24 hours, a ≥5mm increase in diameter of zone of inhibition for either of the cephalosporin-clavulanate disk combination versus the zone diameter of the respective Cephalosporin disk was considered positive, and the isolate was interpreted as ESBL producer.22
Quality Assurance
To generate quality and reliable data, all quality control checks were done before, during and after data collection. All the questions in structured questionnaire were prepared in a clear and precise way and translated into local language (Amharic). Data collectors were trained; the entire questionnaires were checked for completeness, during and after data collection by the data collectors. Moreover, all laboratory assays were done by maintaining quality control procedures. Standard Operating Procedures (SOPs) were strictly followed verifying that media meet expiration date and quality control parameters per CLSI guideline. Visual inspections of cracks in media or plates, unequal fill, hemolysis, evidence of freezing, bubbles, and contamination were performed. Quality control and sterility testing were performed to check the quality of medium. Reference strains of S. aureus (ATCC 25,923); E. coli (ATCC 25,922), E. faecalis (ATCC 29,212) and P. aeruginosa (ATCC 27,853) were used as quality control for culture and susceptibility testing throughout the study. Moreover, E. coli (ATCC 25,922) and K. pneumoniae (ATCC 700,603) reference strains were used as quality control for ESBL detection. All reference strains were obtained from Dessie Health Research Regional laboratory.
Statistical Analysis
The data were imported and analyzed by Statistical Package for Social Sciences (SPSS) version 22.0. (IBM, USA), and descriptive statistics, chi-square, binary and multivariate logistic regression were employed. Binary logistic regression was used to show the association of each variable with the dependent variable. Moreover, a multivariate analysis was computed to identify factors that independently influence the occurrence of dependent variable. P-value <0.05 with 95% confidence interval was considered statistically significant.
Ethics Approval and Consent to Participate
The study protocol was evaluated and approved by the Research Ethics Review Committee (RERC 0958/2017) of College of Health Sciences, Mekelle University, and ethical clearance was obtained. Official cooperation and permission were obtained from the Amhara Regional Health Bureau and Dessie Referral Hospital. Moreover, prior to commencing the study, as all of the study participants were above the age of 18, written informed consent was obtained from each study participant before data collection. Moreover, this study was conducted in accordance with the Declaration of Helsinki.
Results
Sociodemographic Characteristics
In this study, a total of 323 pregnant women 79 (24.5%) with and 244 (75.5%) without symptoms of UTI were investigated during the study period. The age of the study participants ranged from 19 to 43 years, with a mean age of 26.7±4.7 years. Slightly majority 183 (56.7%) of the study participants were in the age group of 25 to 34 years and 271 (83.9%) were urban dwellers (Table 1). Among the study participants, 301 (93.2%) were married, and majority were housewives 188 (58.2%).
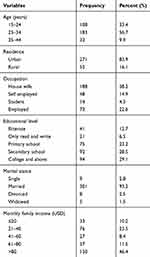 |
Table 1 Sociodemographic Characteristics of the Study Participants (n=323) in Dessie Referral Hospital (DRH), Northeastern Ethiopia |
Prevalence of Urinary Tract Infection
Out of 323 cultured urine specimens (244 asymptomatic and 79 symptomatic), significant bacteriuria was detected in 50 (29 from asymptomatic and 21 from symptomatic) pregnant women investigated for UTI (Figure 1). The prevalence of significant bacteriuria among asymptomatic and symptomatic pregnant women was 29 (11.8%) and 21 (26.6%), respectively. The overall prevalence of UTI was 15.5%.
 |
Figure 1 Frequency of UTI among asymptomatic and symptomatic pregnant women in DRH, Northeastern Ethiopia. |
Associated Risk Factors of UTI
In this study, 18 independent variables were considered during the bivariate analysis of risk factors for bacterial UTI. In multivariate analysis, history of previous UTI (AOR=8.824,95% CI: 3.769, 20.654, P<0.001) and history of the previous catheterization (AOR=3.270,95% CI: 1.316, 8.122, P=0.011) were found to have statistically significant association with UTI. Out of a total of 50 pregnant women who had significant bacteriuria, 38 (76.0%) had a history of UTI (P<0.001), and 19 (38.0%) had a history of catheterization (P=0.011) (Table 2).
 |
Table 2 Bivariate and Multivariate Logistic Regression Analyses of Factors Associated with UTI Among Pregnant Women (n=323) Attending ANC in DRH, Northeastern Ethiopia |
Bacterial Uropathogen Isolates
In this study, six different bacteria were isolated from midstream urine sample of study participants. Almost all 49 (98.0%) of the infected pregnant women had a single infection; while only one (2.0%) was dually infected (E. coli and CoNS), which makes the total number of bacterial isolates 51 (Table 3). Of the total 51 isolates, Gram-positive bacteria were more prevalent 31 (60.8%) than Gram-negative bacteria 20 (39.2%). Overall, the predominantly isolated bacteria were E. coli 17 (33.3%), followed by coagulase-negative staphylococci (CoNS) 15 (30.0%), S. aureus 14 (27.5%), K. pneumoniae 2(3.9%), S. agalactiae 2(3.9%), and Enterobacter species 1(2.0%).
 |
Table 3 Frequency of Bacterial Uropathogens of Symptomatic and Asymptomatic UTI Among Pregnant Women Attending ANC at DRH, Northeastern Ethiopia |
Antimicrobial Susceptibility Pattern of Bacterial Uropathogens
In this study, the majority of isolated Gram-negative uropathogens showed resistance rate of 13 (65.0%) for ampicillin. Rates of resistance of Gram-negatives against ciprofloxacin, tetracycline, trimethoprim-sulfamethoxazole, ceftriaxone, amoxicillin-clavulanic acid, cefotaxime, amikacin, gentamicin range from 6(30.0%) – 10(50.0%). However, all Gram-negative bacterial isolates showed a relatively low level of resistance against nitrofurantoin and norfloxacin, both accounting 5(25.0%) and ceftazidime 3(15.0%) (Table 4).
 |
Table 4 Antimicrobial Susceptibility Pattern of Gram-Negative Bacteria (n=20) Isolated from Urine Culture Among Pregnant Women (n=323) at DRH, Northeastern Ethiopia |
In general, Gram-positive uropathogens showed a high level of resistance for penicillin 29 (93.5%) and trimethoprim-sulfamethoxazole 23 (79.3%). On the other hand, all Gram-positive isolates showed full sensitivity 29 (100.0%) to nitrofurantoin. Moreover, 23 (74.2%), 20 (70.0%) and 22 (75.9%) of the gram-positive isolates were sensitive to clindamycin, ciprofloxacin, and norfloxacin, respectively (Table 5).
 |
Table 5 Antimicrobial Susceptibility Pattern of Gram-Positive Bacteria (n=31) Isolated from Urine Culture Among Pregnant Women (n=323) at DRH, Northeastern Ethiopia |
Multiple Drug Resistance Patterns of the Isolates
Overall, 48 (94.1%) bacterial isolates were resistant to at least one antimicrobial agent, whereas 42 (82.3%) isolates were resistant to ≥2 antimicrobials (Table 6). Multidrug resistance (defined as non-susceptible to  1 agent in
1 agent in  3 antimicrobial categories) was seen in 41 (80.4%) of all isolated bacterial uropathogens. Eighty percent of Gram-negative and 80.6% of Gram-positive bacteria showed multidrug resistance for the tested antimicrobial drugs. S. aureus, CoNS, and K. pneumoniae were found to be highly resistant to most of the antibiotics tested (Table 6).
3 antimicrobial categories) was seen in 41 (80.4%) of all isolated bacterial uropathogens. Eighty percent of Gram-negative and 80.6% of Gram-positive bacteria showed multidrug resistance for the tested antimicrobial drugs. S. aureus, CoNS, and K. pneumoniae were found to be highly resistant to most of the antibiotics tested (Table 6).
 |
Table 6 Multidrug Resistance Patterns of Bacterial Isolates (n=51) from Pregnant Women (n=323) at DRH, Northeastern Ethiopia |
ESBL-Producing Gram-Negative Uropathogens
Twenty (20) Enterobacteriaceae were isolated from the total study participants. The isolates were E. coli 17 (80.0%), K. pneumoniae 2 (10.0%) and Enterobacter aerogens 1 (5.0%). However, E. aerogens 1 (5.0%) was excluded from further screening for ESBL because these methods were not validated for these groups. As a result, out of the 19 Enterobacteriaceae clinical isolates that were screened for ESBL production, 3(15.8%) were positive for ESBL production. These positive ESBL isolates were Escherichia coli 2(66.7%) and Klebsiella pneumoniae 1(33.3%) (Table 7). The ESBL-producing isolates showed 100% resistance to all tested antibacterial drugs. However, all the ESBL-producing uropathogens were sensitive to Imipenem (100%).
 |
Table 7 Prevalence of ESBL Production Among Gram-Negative Uropathogens Isolated from Pregnant Women at DRH, Northeastern Ethiopia |
Associated Risk Factors of ESBL Production
In this study, history of hospitalization (p=0.010) and antibiotics usage in the last 3 months (p=0.036) were found to be significantly associated with ESBL production of the bacterial isolates. However, there was no statistically significant association between ESBL production of bacterial isolates and current symptom of UTI, history of UTI, history of catheterization, history of Diabetes Mellitus, history of HIV/AIDS and genitourinary abnormality (Table 8).
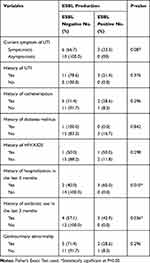 |
Table 8 Prevalence of ESBL-Producing Gram-Negative Bacterial Uropathogens vs Clinical Variables of Pregnant Women at DRH, Northeastern Ethiopia |
Discussion
In this study, the overall bacterial prevalence of UTI among pregnant women was found to be 15.5%. This is in line with study reports from Brazil 15.6%,6 Dire Dawa 14.4%,18 Tanzania 14.6%,23 and Iran 13.1%.24 Our finding is relatively higher than the findings from different parts of Ethiopia such as Gondar 10.4%,9 Addis Ababa 11.6%,19 and Bahir Dar 9.5%.17 This inconsistency in prevalence may be due to differences in the study participants; i.e., all of these studies took a small number of symptomatic pregnant women which might decrease the overall prevalence when compared with the present study. However, our finding was lower than the reports from Cameroon 23.5%,13 Nepal 30.5%,25 Nigeria 25.3%26 and studies from India 20.1%,27 24.0%,28 28.0%29 which might be due to geographical variations. Furthermore, the prevalence of UTI observed in our study is radically high compared to those reported in developed countries (2–10%)4 and this can be attributed to low socio-economic status and difference in the level of health-care development.30
The reported prevalence of symptomatic UTI among pregnant women in this study was 26.6%. This agrees with the previous studies conducted in India, 25.2%,31 25.0%28 and Southeastern Ethiopia 26%32 but it was relatively higher than the study performed in Addis Ababa 20%,19 Gondar 15.3%,9 Bahir Dar 8.5%,17 Dire Dawa 17.0%,18 Brazil 12.7%,6 Tanzania 17.9%,23 Sudan 12.1%,33 and Pakistan, 4.3%.8 Such variation might be attributed to the difference in risk factors with geographical areas. On the contrary, this finding was lower than reports of studies from Nigeria 46.5%34 and India 59.7%.27 The difference from the later studies might be attributable to the small number of symptomatic pregnant women involved in later studies. On the other hand, our finding of 11.9% prevalence of asymptomatic UTI among pregnant women is in line with study reports from Dire Dawa 11.0%,18 India 11.5%,35 Northeastern Ethiopia 11.5%36 and Tanzania 13%;23 but lower than study reports from our country including Bahir Dar 18.9%,17 Hawassa 18.8%,20 Adama 16.1%37 and Adigrat 21.2%;38 also, in other parts of the world such as Sudan 14.7%33 as well as India 16.9%14 and 40.3%.27 Conversely, this result was higher than previous studies reported locally from Gondar 9.0%9 and also from Brazil 5.2%,6 Ghana 5.5%,39 7.3%,40 Turkey 8.5%,12 Egypt 10.0%41 and Cameroon 7.8%.13 Such variations might be as a result of differences in the study population, varying geographical location, and deployment of diverse methodologies.
Unlike most of the study reports in our country and elsewhere in the world, our findings showed that majority of the etiological agents for UTI were gram-positive bacteria. This might be attributable to the fact that CoNS, which are gram-positive bacteria, are recently becoming a major cause of UTI especially among young women pertaining hand to urogenital transmission. In the present study, E. coli was the most predominant isolate with an isolation rate of 17 (33.3%) which is in line with most of the studies conducted locally such as Gondar,9 Bahir Dar,17 Addis Ababa19 and Dire Dawa,18 and also elsewhere in the world including Brazil,6 Iran,1 Nigeria,26 India,27 Nepal,25 Cameroon,13 Pakistan,42 and Kenya.43 The major contributing factor for isolating such higher rate of E. coli might be due to the significant abundance of E. coli as fecal flora which in turn via contamination ascend through genitalia to cause UTI and due to numerous virulence factors used for colonization and invasion of the urinary epithelium such as P-fimbriae or pili adherence factors which mediate the attachment of E. coli to uroepithelial cells. CoNS were the second dominant bacterial isolates in our study with an overall isolation rate of 30%, and comparable results were reported from other previous studies conducted in Gondar,9 Bahir Dar,17 Addis Ababa,19 Dire Dawa,18 Zimbabwe44 and India.28
Pregnancy is a critical period and boosts the occurrence of UTI due to the numerous risk factors. Physiological and hormonal changes that occur in the course of pregnancy, routes of infection as a result of reproductive physiology of females and urinary instrumentation are among the major factors predisposing pregnant women to acquire UTI. Among the investigated risk factors, previous history of UTI showed significant association (AOR=8.824,95% CI: 3.769, 20.654, p<0.001) with the prevalence of UTI indicating pregnant women with the previous history of UTI showed higher rate of significant bacteriuria, which agrees with studies from Gondar,9 Bahir Dar,17 Dire Dawa18 and Pakistan.8 This is possibly due to the presence of resistant bacterial strains from those who had previous history of UTI afterward poor diagnosis and ineffective treatment. The prevalence of UTI in pregnant women with previous history of catheterization was also significantly higher than those without a history of previous catheterization (AOR=3.270,95% CI: 1.316, 8.122, p=0.011). This finding agrees with similar reports from Gondar.9 This is possibly due to catheterization could bring urethral mucosa injury when staying for long duration and contamination during insertion of catheters. Also, it could be due to the ability of gram-negative bacteria such as E. coli to adhere to plastic materials to form a biofilm.
In this study, 15.8% of the gram-negative isolates were found to be ESBL producers while similar prevalence of 16 (20.0%) has been reported from Nigeria.45 This finding is higher than study reports from Norway 2.9%46 and Nepal 7.3%.25 The rise in the prevalence of ESBL-producing uropathogens might be attributed to the habit of empirical treatment practice without drug susceptibility testing, and poor compliance of patients with prescribed drugs. Whereas lower findings were obtained than India 44.4%47 which might be due to the difference in the study population and health-care trends. Despite there were no documented reports on the occurrence of ESBL-producing bacterial uropathogens causing UTI particularly among pregnant women in Ethiopia, ESBL production was assessed in different setups and study groups. For instance, 78.6% ESBL-producing Enterobacteriaceae among children with septicemia and UTIs at Tikur Anbessa Specialized Hospital,48 52.0% of ESBL-producing gastrointestinal colonizing Enterobacteriaceae among hospitalized patients at Tikur Anbessa Specialized Hospital,49 2.73% ESBL-producing Enterobacteriaceae among patients with UTI at University of Gondar Referral Hospital50 and 33.3% ESBL-producing Klebsiella species isolates in clinical specimens from Harar region, Eastern Ethiopia.51 In all these studies, similar to the present study, E. coli and Klebsiella species were the isolates that showed ESBL production. These findings collectively showed the significantly rising occurrence of ESBL-producing isolates showing resistance to commonly used antibiotic drugs in Ethiopia, which needs much attention. Moreover, our finding revealed that history of hospitalization (p=0.010) and antibiotics usage in the last 3 months (p=0.036) were found to be significantly associated with ESBL production of the bacterial isolates which might be an indicator for hospital-acquired resistance and improper prescription and use of antibiotic drugs.
Mainly due to the habit of empirical treatment, infrequent bacterial identification and absence of susceptibility testing, antimicrobial resistance among bacterial uropathogens to the commonly used antibiotics become increasing that make clinicians left with very limited choices of drugs for the treatment of urinary tract infection.19 In this study, the highest resistance was shown to ampicillin (60.0%) among gram-negative bacteria. This could be due to overuse of the drug for many years. On the other hand, lower resistance (higher rate of sensitivity) was observed against nitrofurantoin, ceftazidime, and norfloxacin which is an indication of the possible use of these drugs as empiric therapy, particularly in the study area. Similar findings have been reported in previous studies done in Ethiopia: Bahir Dar17 and Dire Dawa.18 The possible justification for such low-level resistance might be attributable to the infrequent prescription of these drugs. Hence, they could be considered as alternative options in the treatment of UTI.
Gram-positive bacteria were relatively resistant to penicillin (93.5%) and trimethoprim-sulfamethoxazole (79.3%). This might be due to the easy availability and indiscriminate use of these drugs, which could lead to an increase in resistance. On the contrary, all tested gram-positive isolates showed sensitivity to nitrofurantoin (100.0%), ceftriaxone (100.0%) and norfloxacin (75.9%) which was also in agreement with the findings of other studies from Gondar,9 Addis Ababa19 and Bahir Dar.17 According to the international standard for the definition of drug resistance,52 multidrug resistance (MDR = Non-susceptible to  1 agent in
1 agent in  3 antimicrobial categories) was observed in 80.4% of the total isolated bacterial uropathogens. This was higher than study from Addis Ababa (74.0%),19 and lower than reports from Bahir Dar 91.3%,53 Jigjiga 96.0%,54 Gondar 95.0%,9 Dire Dawa 100.0%18 and Kenya 96.0%.43 This indicates that MDR was found to be very high to the commonly used antibiotics in the study area. The possible reason for this rise in MDR might be repeated, inappropriate, and incorrect use of antimicrobial agents in empirical treatment and poor infection control strategies which in turn raise the prevalence of resistant microorganism in the community.
3 antimicrobial categories) was observed in 80.4% of the total isolated bacterial uropathogens. This was higher than study from Addis Ababa (74.0%),19 and lower than reports from Bahir Dar 91.3%,53 Jigjiga 96.0%,54 Gondar 95.0%,9 Dire Dawa 100.0%18 and Kenya 96.0%.43 This indicates that MDR was found to be very high to the commonly used antibiotics in the study area. The possible reason for this rise in MDR might be repeated, inappropriate, and incorrect use of antimicrobial agents in empirical treatment and poor infection control strategies which in turn raise the prevalence of resistant microorganism in the community.
In accordance to reported studies,42 ESBL-producing isolates showed 100.0% sensitivity to imipenem. All of the ESBL positive isolates showed a high level of resistance (100.0%) to amoxicillin, sulphamethoxazole-trimethoprim, cefotaxime, cefepime, ceftazidime, and the rest of tested antimicrobial drugs. These outcomes correspond with reports from Nigeria45 and Pakistan.42 Such findings imply that the use of these antibiotics for the treatment of urinary tract infections caused by ESBL-producing strains may not be effective and may result in a significant amount of treatment failure. On the other hand, ESBL positive strains can respond better for Carbapenem drugs such as imipenem which could be a better treatment option.
Limitations
The present study employed convenient sampling technique that could not be generalizable for the total population.
Conclusion
In this study, a relatively higher overall prevalence of UTI was observed compared with other similar studies in the country. Nitrofurantoin and norfloxacin were effective for most of the gram-positive and gram-negative isolates, whereas, penicillin, ampicillin, and trimethoprim-sulfamethoxazole were less effective for the management of UTI among our study participants. Notably, increasing rate of resistance to the commonly used antimicrobial agents has been observed for both gram-negative and gram-positive isolates. Moreover, a significant amount of ESBL producers (15.8%) and an alarmingly high multidrug resistance has been shown in most of (80.4%) the bacterial isolates. Therefore, instant UTI culture assessment of pregnant women, especially those having possible risk factors such as previous histories of UTI and catheterization; moreover, appropriate prescription and use of antibiotics is necessary. Furthermore, continuous screening and investigations, including genotypic characterization in ESBL-producing bacteria causing UTI among pregnant women in larger sample size, are needed to confirm these findings and assess other possible risk factors.
Abbreviations
AOR, adjusted odds ratio; ASB, asymptomatic bacteriuria; ANC, antenatal care; ATCC, American Type Culture Collection; CI, confidence interval; CLED, Cystine Lactose Electrolyte Deficient medium; CLSI, Clinical and Laboratory Standards Institute; CoNS, coagulase-negative staphylococci; COR, crude odds ratio; DRH, Dessie Referral Hospital; ESBL, extended-spectrum beta-lactamase; MDR, multidrug resistance; MHA, Muller-Hilton Agar; SOPs, standard operating procedures; SPSS, Statistical Package for Social Sciences; SB, significant bacteriuria; UTI, urinary tract infection; WHO, World Health Organization.
Acknowledgment
The author would like to acknowledge Mekelle University and Dessie Regional Health Research Laboratory for providing laboratory space and facilities to conduct the experiments. Dessie Referral Hospital and all study participants are acknowledged for their cooperation during sample collection.
Disclosure
The author reports no conflicts of interest in this work.
References
1. Amiri M, Lavasani Z, Norouzirad R, et al. Prevalence of urinary tract infection among pregnant women and its complications in their newborns during the birth in the hospitals of Dezful city, Iran, 2012–2013. Iran Red Crescent Med J. 2015;17(8). doi:10.5812/ircmj.26946
2. Rosana Y. Comparison of microbial pattern causing urinary tract infection in female out and hospitalized patients in Jakarta. Microbiol Indones. 2016;10(1):30–37. doi:10.5454/mi.10.1.5
3. Vasudevan R. Urinary tract infection: an overview of the infection and the associated risk factors. J Microbiol Exp. 2014;1(2):00008.
4. Gilbert NM, O’brien VP, Hultgren S, et al. Urinary tract infection as a preventable cause of pregnancy complications: opportunities, challenges, and a global call to action. Global Adv Health Med. 2013;2(5):59–69. doi:10.7453/gahmj.2013.061
5. Ferede G, Yismaw G, Wondimeneh Y, et al. The prevalence and antimicrobial susceptibility pattern of bacterial uropathogens isolated from pregnant women. Eur J Exp Biol. 2012;2(5):1497–1502.
6. Rahimkhani M, Khavari-Daneshvar H, Sharifian R. Asymptomatic bacteriuria and pyuria in pregnancy. Acta Med Iran. 2006;46(5):409–412.
7. Vasconcelos-Pereira EF, Figueiró-Filho EA, de Oliveira VM, et al. Urinary tract infection in high risk pregnant women. Rev Patol Trop. 2013;42(1):21–29. doi:10.5216/rpt.v42i1.23590
8. Haider G, Zehra N, Munir AA, et al. Risk factors of urinary tract infection in pregnancy. J Pak Med Assoc. 2010;60(3):213–216.
9. Alemu A, Moges F, Shiferaw Y, et al. Bacterial profile and drug susceptibility pattern of urinary tract infection in pregnant women at University of Gondar Teaching Hospital, Northwest Ethiopia. BMC Res Notes. 2012;5(1):197. doi:10.1186/1756-0500-5-197
10. Emiru T, Beyene G, Tsegaye W, et al. Associated risk factors of urinary tract infection among pregnant women at Felege Hiwot Referral Hospital, Bahir Dar, North West Ethiopia. BMC Res Notes. 2013;6(1):292. doi:10.1186/1756-0500-6-292
11. Guerra G, Costa BF, Nascimento F, et al. Urine test to diagnose urinary tract infection in high-risk pregnant women. Rev Bras Ginecol Obstet. 2012;34(11):488–493. doi:10.1590/S0100-72032012001100002
12. Çelen Ş, Oruç AS, Karayalçin R, et al. Asymptomatic bacteriuria and antibacterial susceptibility patterns in an obstetric population. ISRN Obstet Gynecol. 2011;2011:1–4. doi:10.5402/2011/721872
13. Mokube MN, Atashili J, Halle-Ekane GE, et al. Bacteriuria amongst pregnant women in the Buea Health District, Cameroon: prevalence, predictors, antibiotic susceptibility patterns and diagnosis. PLoS One. 2013;8(8):e71086. doi:10.1371/journal.pone.0071086
14. Jain V, Das V, Agarwal A, et al. Asymptomatic bacteriuria & obstetric outcome following treatment in early versus late pregnancy in north Indian women. Indian J Med Res. 2013;137(4):753–758.
15. Kateregga JN, Kantume R, Atuhaire C, et al. Phenotypic expression and prevalence of ESBL-producing Enterobacteriaceae in samples collected from patients in various wards of Mulago Hospital, Uganda. BMC Pharmacol Toxicol. 2015;16(1):14. doi:10.1186/s40360-015-0013-1
16. Hoban DJ, Lascols C, Nicolle LE, et al. Antimicrobial susceptibility of Enterobacteriaceae, including molecular characterization of extended-spectrum beta-lactamase–producing species, in urinary tract isolates from hospitalized patients in North America and Europe: results from the SMART study 2009–2010. Diagn Microbiol Infect Dis. 2012;74(1):62–67.
17. Demilie T, Beyene G, Melaku S, et al. Urinary bacterial profile and antibiotic susceptibility pattern among pregnant women in North West Ethiopia. Ethiop J Health Sci. 2012;22(2):121–128.
18. Derese B, Kedir H, Teklemariam Z, et al. Bacterial profile of urinary tract infection and antimicrobial susceptibility pattern among pregnant women attending at antenatal clinic in Dil Chora Referral Hospital, Dire Dawa, Eastern Ethiopia. Ther Clin Risk Manag. 2016;12:251–260. doi:10.2147/TCRM.S99831
19. Assefa A, Asrat D, Woldeamanuel Y, et al. Bacterial profile and drug susceptibility pattern of urinary tract infection in pregnant women at Tikur Anbessa Specialized Hospital Addis Ababa, Ethiopia. Ethiop Med J. 2008;46(3):227–235.
20. Tadesse E, Teshome M, Merid Y, et al. Asymptomatic urinary tract infection among pregnant women attending the antenatal clinic of Hawassa Referral Hospital, Southern Ethiopia. BMC Res Notes. 2014;7(1):155. doi:10.1186/1756-0500-7-155
21. Cheesbrough M. Medical Laboratory Practice in Tropical Countries. Part 2.
22. CLSI. Performance Standards for Antimicrobial Susceptibility Testing.
23. Masinde A, Gumodoka B, Kilonzo A, et al. Prevalence of urinary tract infection among pregnant women at Bugando Medical Centre, Mwanza, Tanzania. Tanzania J Health Res. 2009;11(3). doi:10.4314/thrb.v11i3.47704
24. Rejali M, Ahmadi SS. Prevalence and risk factors of urinary tract infection among pregnant women in Shahrekord, Iran. IJER. 2019;6(2):55–59.
25. Thapa R, Lamichhane P, Banjara MR, et al. Prevalence of extended spectrum beta lactamase producing uropathogens in pregnant women. Asian J Pharm Clin Res. 2015;8(1):207–210.
26. Ebidor L, Tolulope A, Deborah O. Urinary tract infection amongst pregnant women in Amassoma, Southern Nigeria. Afr J Microbiol Res. 2015;9(6):355–359. doi:10.5897/AJMR2014.7323
27. Vaijanathrao CY, Nalini YL, Reddy CM. Antibiotic sensitivity pattern of uropathogens: a comparative study between symptomatic and asymptomatic bacteriuria in pregnant women. Int J Curr Microbiol Appl Sci. 2015;4:689–695.
28. Sabharwal ER. Antibiotic susceptibility patterns of uropathogens in obstetric patients. North Am J of Med Sci. 2012;4(7):316. doi:10.4103/1947-2714.98591
29. Yasmin T, Sarwar MY, Sen A. Prevalence of urinary tract infection in pregnant women in Katihar district, Bihar. J Evol Med Dent Sci. 2018;7(3):372–375. doi:10.14260/jemds/2018/82
30. Ankur G, Namita S, Sapna G, et al. Prevalence of asymptomatic urinary tract infections in the three trimesters of pregnancy. Int J Curr Microbiol App Sci. 2015;1:110–117.
31. Rizvi M, Khan F, Shukla I, et al. Rising prevalence of antimicrobial resistance in urinary tract infections during pregnancy: necessity for exploring newer treatment options. J Lab Physicians. 2011;3(2):98. doi:10.4103/0974-2727.86842
32. Taye S, Getachew M, Desalegn Z, et al. Bacterial profile, antibiotic susceptibility pattern and associated factors among pregnant women with urinary tract infection in Goba and Sinana Woredas, Bale Zone, Southeast Ethiopia. BMC Res Notes. 2018;11(1):799. doi:10.1186/s13104-018-3910-8
33. Hamdan HZ, Ziad AHM, Ali SK, et al. Epidemiology of urinary tract infections and antibiotics sensitivity among pregnant women at Khartoum North Hospital. Ann Clin Microbiol Antimicrob. 2011;10(1):2. doi:10.1186/1476-0711-10-2
34. Onoh RC, Umeora O, Egwuatu V, et al. Antibiotic sensitivity pattern of uropathogens from pregnant women with urinary tract infection in Abakaliki, Nigeria. Infect Drug Resist. 2013;6:225–233. doi:10.2147/IDR.S46002
35. Dash M, Sahu S, Mohanty I, et al. Prevalence, risk factors and antimicrobial resistance of asymptomatic bacteriuria among antenatal women. J Basic Clin Reprod Sci. 2013;2(2):92–96. doi:10.4103/2278-960X.118647
36. Habteyohannes AD, Mekonnen D, Abate E, et al. Bacterial isolates and their current drug susceptibility profile from urine among asymptomatic pregnant women attending at a Referral Hospital, Northwest Ethiopia; cross-sectional study. EJRH. 2018;10(2):10.
37. Nisha AK, Etana AE, Tesso H. Prevalence of asymptomatic bacteriuria during pregnancy in Adama city, Ethiopia. Int J Microbiol Immunol Res. 2015;3(5):58–63.
38. Tadesse S, Kahsay T, Adhanom G, et al. Prevalence, antimicrobial susceptibility profile and predictors of asymptomatic bacteriuria among pregnant women in Adigrat General Hospital, Northern Ethiopia. BMC Res Notes. 2018;11(1):740. doi:10.1186/s13104-018-3844-1
39. Labi A, Yawson A, Ganyaglo G, et al. Prevalence and associated risk factors of asymptomatic bacteriuria in ante-natal clients in a large teaching hospital in Ghana. Ghana Med J. 2015;49(3):154–158. doi:10.4314/gmj.v49i3.5
40. Turpin C, Minkah B, Danso K, et al. Asymptomatic bacteriuria in pregnant women attending antenatal clinic at Komfo Anokye Teaching Hospital, Kumasi, Ghana. Ghana Med J. 2007;41(1):26.
41. Elzayat M-A-A, Barnett-Vanes A, Dabour MFE, et al. Prevalence of undiagnosed asymptomatic bacteriuria and associated risk factors during pregnancy: a cross-sectional study at two tertiary centres in Cairo, Egypt. BMJ. 2017;7(3):e013198.
42. Mohammed I, Abass E. Phenotypic detection of Extended Spectrum β-Lactamases (ESBL) among gram negative uropathogens reveals highly susceptibility to imipenem. Pak J Med Sci. 2019;35(4). doi:10.12669/pjms.35.4.207
43. Onyango HA, Ngugi C, Maina J, et al. Urinary tract infection among pregnant women at Pumwani Maternity Hospital, Nairobi, Kenya: bacterial etiologic agents, antimicrobial susceptibility profiles and associated risk factors. Adv Microbiol. 2018;8(3):175–187. doi:10.4236/aim.2018.83012
44. Rukweza J, Nziramasanga P, Gidiri M, et al. Antibiotic susceptibility of bacterial strains causing asymptomatic bacteriuria in pregnancy: a cross-sectional study in Harare, Zimbabwe. MOJ Immunol. 2018;6(1):00184.
45. Onwuezobe A, Orok F. Extended spectrum beta-lactamase producing uropathogens in asymptomatic pregnant women attending antenatal care in an urban community secondary health facility. Afr J Cln Exper Microbiol. 2015;16(2):49–53. doi:10.4314/ajcem.v16i2.1
46. Rettedal S, Löhr I, Bernhoff E, et al. Extended-spectrum β-lactamase-producing Enterobacteriaceae among pregnant women in Norway: prevalence and maternal–neonatal transmission. J Perinatol. 2015;35(11):907–912. doi:10.1038/jp.2015.82
47. Krishnakumar S, Rajan RA, Babu MM, et al. Antimicrobial susceptibility pattern of extended spectrum of beta lactamase (ESBL) producing uropathogens from pregnant women. IJMHC. 2012;1(8):189–192.
48. Legese MH, Weldearegay GM, Asrat D. Extended-spectrum beta-lactamase-and carbapenemase-producing Enterobacteriaceae among Ethiopian children. Infect Drug Resist. 2017;10:27–34. doi:10.2147/IDR.S127177
49. Desta K, Woldeamanuel Y, Azazh A, et al. High gastrointestinal colonization rate with extended-spectrum β-lactamase-producing Enterobacteriaceae in hospitalized patients: emergence of carbapenemase-producing K. pneumoniae in Ethiopia. PLoS One. 2016;11(8):e0161685. doi:10.1371/journal.pone.0161685
50. Eshetie S, Unakal C, Gelaw A, et al. Multidrug resistant and carbapenemase producing Enterobacteriaceae among patients with urinary tract infection at referral hospital, Northwest Ethiopia. Antimicrob Resis Infect Control. 2015;4(1):12. doi:10.1186/s13756-015-0054-7
51. Seid J, Asrat D. Occurrence of extended spectrum β-lactamase enzymes in clinical isolates of Klebsiella species from Harar region, eastern Ethiopia. Acta Trop. 2005;95(2):143–148. doi:10.1016/j.actatropica.2005.05.009
52. Magiorakos AP, Srinivasan A, Carey R, et al. Multidrug‐resistant, extensively drug‐resistant and pandrug‐resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
53. Biadglegne F, Abera B. Antimicrobial resistance of bacterial isolates from urinary tract infections at Felge Hiwot Referral Hospital, Ethiopia. Ethiop J Health Dev. 2009;23(3):236–238.
54. Negussie A, Worku G, Beyene E. Bacterial identification and drug susceptibility pattern of urinary tract infection in pregnant Women at Karamara Hospital Jigjiga, Eastern Ethiopia. Afr Res J Bacteriol Res. 2018;10(2):15–22.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
