Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Ataxia with Parkinsonism and dystonia after intentional inhalation of liquefied petroleum gas
Authors Godani M, Canavese F, Migliorini S, Del Sette M
Received 7 January 2015
Accepted for publication 19 February 2015
Published 6 May 2015 Volume 2015:11 Pages 1157—1159
DOI https://doi.org/10.2147/NDT.S80460
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Massimiliano Godani,1 Francesca Canavese,1 Sonia Migliorini,2 Massimo Del Sette1
1Neurology Unit, 2Department of Neuroradiology, Sant’Andrea Hospital, La Spezia, Italy
Abstract: The practice of inhaling liquefied petroleum gas (LPG) to commit suicide is uncommon and almost exclusively a prerogative of the prison population. Numerous cases of sudden deaths caused by intentional propane and/or butane inhalation have been described, but these cases survived and a description of the consequences is very rare. We describe a prisoner who survived after voluntary inhalation of LPG, and who developed ataxia, Parkinsonism, and dystonia. Brain MRI showed bilateral hyperintensity in the basal ganglia and in the cerebellar hemispheres. The clinical evolution and the MRI abnormalities are similar to those described in cases of poisoning by CO where the mechanism of brain injury is related to histotoxic hypoxia. We believe that LPG, considered until now a mixture of gas with low neurotoxic power, may have caused direct toxic damage to the brain, mediated by a mechanism of hypoxia, such as in CO intoxication.
Keywords: ataxia, Parkinsonism, dystonia, liquefied petroleum gas
Case report
Numerous cases of sudden deaths caused by intentional propane and/or butane inhalation have been described in the existing literature,1,2 but cases of survivors, with relative descriptions of the associated consequences, are very rare. We report a patient who survived after intentional inhalation of liquefied petroleum gas (LPG), and who developed progressive ataxia, Parkinsonism, and dystonia.
A 38-year-old prisoner with an unremarkable medical history was found unconscious inside his cell. He was lying on the bed with a large cellophane bag near his head. An empty gas cylinder containing LPG, often used by prisoners to cook in their cells, was found close to him.
The man was brought to the emergency room in a comatose state and with shortness of breath. He was promptly intubated and admitted to the intensive care unit. Analysis of arterial blood gases showed hypoxia, hypercapnia, and mixed acidosis. Intravenous administration of naloxone had no effect. The patient’s blood pressure and electrocardiography were normal. Serum basic chemistry was unremarkable and the alcohol level was zero. Urine toxicology was negative for amphetamines, cocaine, barbiturates, benzodiazepines, and opiates. He had no pulmonary edema.
Urgent computed tomography showed slight hypodensities in both cerebellar hemispheres. In the first 48 hours, the patient rapidly improved. He was able to open his eyes on command and began to move his arms and legs. Brain MRI showed isolated bilateral lesions involving the caudate nucleus and globus pallidus, as well as diffuse signal alterations in both cerebellar hemispheres (Figure 1). The imaging findings were consistent with a metabolic or toxic etiology.
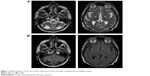  | Figure 1 Bilateral lesions on T2 and FLAIR sequences in both cerebellar hemispheres and pallidal regions. |
In the subsequent week, the clinical picture continued to improve gradually; he was awake and alert, and he was able to stand up, but unable to walk because of impaired balance. Despite significant motor impairment, his sensory and cognitive functions were normal (Mini-Mental State Examination score =30/30).
Two months after discharge, stiffness, bradykinesia, and hypomimia were more evident. He also developed a dystonic posture of his right foot and left hand.
123I-ioflupane single-photon emission computed tomography showed bilaterally decreased uptake in the caudate nucleus and putamen. He was treated with levodopa and dopamine agonists for the akinetic–rigid syndrome without any benefit.
At present, 1 year after the event, the patient is not able to walk and he is in a wheelchair. The main symptoms are severe ataxia, widespread plastic rigidity, bradykinesia, and dystonia. He is moderately dysarthric and his speech is very slow with frequent interruptions. He works as an art critic, and has written and published poetry. The current brain MRI scan has not shown significant changes when compared with the previous one.
The practice of inhaling hydrocarbons, despite being uncommon, has increased greatly in the last few years, especially among adolescents in search of getting high and among the prison population where drug addicts, who are unable to obtain their habitually abused substances, resort to this alternative practice.3 LPG is a flammable mixture of hydrocarbon gases and is used as a fuel, which contains propane as a major component and, in addition, isobutane. These substances are lipophilic, so that after being inhaled and after being taken up from the lungs into blood, they are distributed at high concentrations in lipid-rich tissues, especially in the brain and fat tissues.2
Pathologically, anoxic injuries can be classified as hypoxic hypoxia, from decreases in the partial pressure of blood oxygen (as in patients with hanging or drowning); histotoxic hypoxia, from the tissue’s inability to utilize oxygen (as in the case of exposure to mitochondrial toxins like carbon monoxide and cyanide); anemic hypoxia, from decreased hemoglobin content or function (as in cases of severe anemia); or hypoxic–ischemic encephalopathy, typically seen after cardiac arrest.4 The clinical evolution and the MRI abnormalities in our patient are similar to those described in cases of poisoning by CO, where the mechanism of anoxic brain injury is attributable to histotoxic hypoxia.5 The brain areas affected in CO toxicity are the ones with a high metabolic rate and oxygen demand, and include the basal ganglia, and particularly the globus pallidus, substantia nigra, and hippocampus. Laminar necrosis of the cerebellar cortex and Purkinje cell loss are often seen.5 Similar lesions have been reported following global cerebral hypoxia, but frequently with analogous alterations in the cerebral cortex, lacking in this case. Bilateral basal ganglia lesions can also be observed in patients with illegal drug abuse, such as those using heroin, cocaine, and ecstasy. In these cases, the pathomechanism is usually vascular related (vasospasm, ischemia, hemorrhage, and vasculitis), and the white matter is frequently involved.6 The progressive ataxia and the akinetic–rigid syndrome, which appeared a few days after the event, were the prominent clinical manifestation in our patient. Before brain insult, the patient had no neurological symptoms. He had a normal birth and development, and there was no relevant family history. Ataxia was caused by bilateral and symmetrical lesions of the cerebellar hemispheres, compatible with toxic or metabolic etiology. Akinetic–rigid syndrome and dystonia are instead clear signs of damage at the level of the basal ganglia, which are very sensible to hypoxic/anoxic damage, especially at the globi pallidi. Dystonia could be also explained by aberrant functional output from the cerebellum,7 which was extensively damaged in our patient.
Conclusion
In conclusion, we believe that LPG, considered until now a mix of gas with low neurotoxic power, may have caused direct toxic damage to the brain, mediated by a mechanism of histotoxic hypoxia, such as in CO intoxication. To our knowledge, this is the first reported case featuring a neurological syndrome that presented after LPG intoxication. It would be useful to know if LPG is really able to cause or amplify hypoxic/anoxic damage in humans. For this purpose, laboratory analysis of blood and urine samples up to 24 hours postexposure should be used to confirm the diagnosis of volatile substance intoxication and to demonstrate not only the concentration of inhaled gases, but also the duration of LPG exposure.8 Although blood butane levels can be measured by headspace gas chromatography–mass spectroscopy, the majority of clinical laboratories do not run these tests routinely, and their availability is extremely limited. Therefore, we recommend greater diffusion of this technique, especially in emergency departments.
Disclosure
The authors report no conflicts of interest in this work.
References
Pfeiffer H, Al Khaddam M, Brinkmann B, Köhler H, Beike J. Sudden death after isobutane sniffing: a report of two forensic cases. Int J Legal Med. 2006;120(3):168–173. | ||
Sugie H, Sasaki C, Hashimoto C, et al. Three cases of sudden death due to butane or propane gas inhalation: analysis of tissues for gas components. Forensic Sci Int. 2004;143(2–3):211–214. | ||
Howard MO, Balster RL, Cottler LB, Wu LT, Vaughn MG. Inhalant use among incarcerated adolescents in the United States: prevalence, characteristics, and correlates of use. Drug Alcohol Depend. 2008;93(3):197–209. | ||
Singhal AB, Topcuoglu MA, Koroshetz WJ. Diffusion MRI in three types of anoxic encephalopathy. J Neurol Sci. 2002;196(1–2):37–40. | ||
Busl KM, Greer DM. Hypoxic-ischemic brain injury: pathophysiology, neuropathology and mechanisms. Neuro Rehabilitation. 2010;26(1):5–13. | ||
Geibprasert S, Gallucci M, Krings T. Addictive illegal drugs: structural neuroimaging. AJNR Am J Neuroradiol. 2010;31(5):803–808. | ||
Jinnah HA, Hess EJ. A new twist on the anatomy of dystonia: the basal ganglia and the cerebellum? Neurology. 2006;67(10):1740–1741. | ||
Meredith TJ, Ruprah M, Liddle A, Flanagan RJ. Diagnosis and treatment of acute poisoning with volatile substances. Hum Toxicol. 1989; 8(4):277–286. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
