Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 14
Association of Metabolic Obesity Phenotypes and Total Testosterone in Chinese Male Population
Authors Liu L, Liu S, Song Q, Luo D, Su Y, Qi X, Wang Q, Ning J, Lv Y, Guan Q
Received 21 November 2020
Accepted for publication 22 December 2020
Published 29 January 2021 Volume 2021:14 Pages 399—408
DOI https://doi.org/10.2147/DMSO.S293259
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Juei-Tang Cheng
Luna Liu,1,2 Shuang Liu,1,2 Qianmei Song,1,2 Dandan Luo,1,2 Yu Su,1,2 Xiangyu Qi,1,2 Qian Wang,3 Jing Ning,1,2 Youyuan Lv,1,2 Qingbo Guan1,2,4
1Department of Endocrinology, Shandong Provincial Hospital, Cheeloo College of Medicine, Shandong University, Jinan, Shandong 250021, People’s Republic of China; 2Shandong Provincial Key Laboratory of Endocrinology and Lipid Metabolism, Institute of Endocrinology and Metabolism, Shandong Academy of Clinical Medicine, Jinan, Shandong 250021, People’s Republic of China; 3Department of Ultrasound, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Jinan, Shandong 250021, People’s Republic of China; 4Department of Endocrinology, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Jinan, Shandong 250021, People’s Republic of China
Correspondence: Qingbo Guan Tel +86-531-68776375
Fax +86-531-87068707
Email [email protected]
Objective: Obesity and metabolic syndrome have been reported to exert an impact on the male reproductive system with decreasing levels of serum total testosterone (TT); however, the effect of different metabolic obesity phenotypes on testosterone has been poorly studied. We aimed to evaluate the association of metabolic obesity phenotypes and total testosterone levels in a Chinese male population.
Methods: We performed a retrospective study based on an epidemiological investigation, a total of 4,081 male individuals aged from 40– 75 years old were recruited. The population was classified as metabolically healthy normal weight (MHNW), metabolically healthy overweight/obese (MHO), metabolically unhealthy normal weight (MUNW), and metabolically unhealthy overweight/obese (MUO) according to normal weight (BMI< 25.0) and overweight/obesity (BMI≥ 25.0) with or without metabolic syndrome.
Results: We collected 563 hypotestosteronemia among 4,081 male individuals. The odds ratios (ORs) (95% CIs) of hypotestosteronemia in obesity and metabolic syndrome were 3.072 (2.414– 3.911) and 3.294 (2.631– 4.125), respectively, after adjusting for age, luteinizing hormone, smoking status, and alcohol consumption. Compared to the MHNW group, male subjects in MHO, MUNW, and MUO groups had decreased serum TT levels. Additionally, the MUO group had a lowest concentration of serum TT and a highest proportion of hypotestosteronemia. There was no significant difference of TT levels between the MHO and MUNW groups.
Conclusion: Obesity and metabolic syndrome are independent risk factors of hypotestosteronemia in Chinese male populations. Our study also suggested that individuals with MHO, MUNW, and MUO have a higher risk of developing hypotestosteronemia.
Keywords: metabolic obesity phenotypes, testosterone, metabolically healthy overweight/obese, metabolically unhealthy normal weight, hypotestosteronemia
Introduction
Obesity is an important chronic disease all over the world because of the increasing prevalence and harms to public health. Obesity can result in multiple alterations in the endocrine systems, including altered circulating blood hormone concentrations, because of changes in the pattern of secretion and/or metabolism, abnormal hormonal transport, and/or actions at levels of target tissues.1 It has been demonstrated that obesity has an impact on male reproductive system. And obesity has been considered as a risk for male hypotestosteronemia with low testosterone levels. It has been reported that there are 20–64% of obese men with low total testosterone (TT) or free testosterone (FT) levels.2 Related studies have reported that the increasing BMI is associated with low testosterone and sex-hormone-binding-globulin (SHBG) levels in the male population.3–6 Weight reduction by lifestyle intervention or bariatric surgery can increase the levels of TT and SHBG.3
Metabolic syndrome (MS), gathering of abdominal obesity, hypertension, hyperglycemia, and dyslipidemia is an important challenge for health problem. The prevalence of metabolic syndrome ranges from 10–34% according to different definitions or populations.7 Several studies have pointed that metabolic syndrome can be regarded as an independent association with low levels of total testosterone in males.8–11 And low levels of TT and SHBG can be considered as independent risk factors for metabolic syndrome.12,13 However, the relationship of TT with metabolic syndrome remains inconsistent. A cross-sectional study reported that there was no significant difference of total testosterone among MS and non-metabolic syndrome group. It is supposed that the other parameters were abnormal, the lower levels of SHBG would be measured, without alternations of TT.14 And several studies have concluded that their data were not consistent with the hypothesis that low levels of TT were associated with incidence of metabolic syndrome.15,16
Recent studies have demonstrated that not all obese individuals have metabolic abnormal risk factors, and not all lean individuals are metabolically healthy.17 Therefore, different metabolic phenotypes of obesity have attracted researchers’ attention in recent years. Although obesity may induce decreased serum TT concentration by some related mechanisms, the fact is that only a small proportion of obese men are hypotestosteronemia, probably those genetically predisposed or morbidly obese.18 Metabolic healthy overweight/obesity (MHO) is considered to be overweight/obesity (BMI≧25 kg/m2) without metabolic diseases (hypertension, type 2 diabetes mellitus (T2DM), and dyslipidemia). In addition, metabolically unhealthy overweight/obesity (MUO) refers to a proportion of overweight/obese subjects with metabolic diseases. A recent study based on 4,945 men with sexual dysfunction and 231 male partners of infertile couples demonstrated that men in the MHO groups had lower total testosterone levels compared to men of normal weight.19 However, the effect of specific metabolic obesity phenotypes on total testosterone in the general male population remains unknown. Hypotestosteronemia is classified into primary hypotestosteronemia which results from testicular defects and is associated with low testosterone levels and elevated LH and FSH levels and secondary hypotestosteronemia which results from dysfunction of hypothalamus and/or pituitary and refers to patients with low testosterone levels and low or inappropriately normal LH and FSH levels. And whether the influence of different metabolic obesity phenotypes on TT levels is caused by affecting hypothalamus and/or pituitary levels or testicular levels remains unknown.
Thus, we performed the study to verify the separate influence of MS and overweight/obesity on male hypotestosteronemia, and investigate the relationship of different metabolic obesity phenotypes and male total testosterone levels in a large-scale male population.
Subjects and Methods
Subjects
We recruited nearly 11,000 persons who participated in the population-based cross-sectional study in Ningyang County (Taian, Shandong Province, China) from June to November 2011. All participants were local-registered residents aged 40 years and older who have lived there for at least 5 years. All participants were asked to complete a self-reported questionnaire and provided an overnight fasting blood sample. In our study, the exclusion criteria were as follows: i) female; ii) no information on vital statistics (such as age, sex, height, or weight) or missing data on serum total testosterone (TT), blood pressure, fasting serum glucose, or lipid levels; iii) taking medications that might affect TT level (such as androgens, steroid hormones); and iv) severe hepatic or renal disorders, lung diseases, hypothalamus and/or pituitary gland diseases, neurologic diseases, or tumors that might affect TT level (such as brain cancer or prostate cancer). At last, a total of 4,081 participants were recruited in this study. The study protocol conformed to the 1975 Declaration of Helsinki and was approved by the Committee on Human Research at Ruijin Hospital, Shanghai Jiao Tong University School of Medicine. All study participants provided written informed consent.
Data and Specimen Collection
Height and WC were measured adjusting by 0.1 cm. Weight was measured adjusting by 0.1 kg. BMI was obtained by dividing weight in kilograms by the square of height in meters. The average of the three blood pressure measurements in sitting position after a 5-minute rest was used in the analysis. Past medical history, smoking status, and alcohol consumption were obtained by a questionnaire. The diseases (hypertension, diabetes mellitus, and coronary heart disease) were based on previous diagnosis by a physician. Smoking status and alcohol consumption were defined as never, ever, and current.
Venous blood samples were collected from all patients after at least 10-hours overnight fast and samples were separated and preserved in −80°C. The concentrations of total cholesterol (TC), triglyceride (TG), high density lipoprotein cholesterol (HDL-C), fasting plasma glucose (FPG), and glycosylated hemoglobin (HbA1c) were measured directly with an ARCHITECT ci16200 Integrated System (Abbott). Serum TT, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) were measured using electrochemiluminescent procedures (Cobas E601; Roche) at the clinical laboratory of Shandong Provincial Hospital.
Study Outcome Definition
Total testosterone level <12 nmol/L is suggestive of hypotestosteronemia.20 Secondary hypotestosteronemia refers to patients with low testosterone levels and low or inappropriately normal LH and FSH levels. Primary hypotestosteronemia is associated with low testosterone levels and elevated LH (>8.6 mIU/L) and FSH (>12.4 mIU/L) levels. We used the Adult Treatment Panel III definition of metabolic syndrome in male population: having three or more following criteria: waist circumference >102 cm, triglycerides >1.7 mmol/L (or treatment for hyperlipidemia), HDL-C <1 mmol/L, plasma fasting glucose >100 mg/dL, and systolic/diastolic blood pressure >130/85 mmHg (or treatment for hypertension).21 And we classified the population as normal weight (BMI<25.0) and overweight/obesity (BMI≥25.0) with or without metabolic syndrome. Then, according to a previous study, the participants were classified as four groups: metabolically healthy normal weight (MHNW) (BMI<25.0, without metabolic syndrome); metabolically healthy overweight/obese (MHO) (BMI≥25.0, without metabolic syndrome); metabolically unhealthy normal weight (MUNW) (BMI<25.0, with metabolic syndrome), and metabolically unhealthy overweight/obese (MUO) (BMI≥25.0, with metabolic syndrome).22
Statistical Analysis
SPSS22.0 software was used for the statistical analysis. Continuous variables were presented as mean±standard deviations, and between-group differences were assessed by a single-factor analysis of variance (ANOVA). Categorical variables were presented as percentages, and between-group differences were assessed by Chi-squared test. Multiple logistic regression analysis was performed to assess the risk factors of hypotestosteronemia. All P-values were 2-sided, and less than 0.05 were considered statistically significant.
Results
Baseline Characteristics of Participants in Different Metabolic Obesity Phenotypes
A total of 4,081 male participants aged from 40–75 years were enrolled into the final analysis. There were 563 participants suffering from hypotestosteronemia (13.80%). Within metabolic categories, overweight/obese men tended to be younger with a high level of BMI and WC than men with normal weight. And overweight/obese men were more likely to have a high level of TC, TG, LDC-C, ALT, GGT, and DBP, and a low level of HDL-C, TT, LH, and FSH, without difference of AST, FBG, and HbA1C. As expected, compared to the metabolic healthy group, metabolic unhealthy patients were more likely to have a high level of TG, TC, ALT, AST, GGT, FPG, HbA1C, systolic blood pressure, and diastolic blood pressure, and a low level of HDL-C and TT. There was a high prevalence of diabetes mellitus, hypertension, and coronary heart disease in the metabolic unhealthy population. Compared to the MHNW group, the MHO, MUNW, and MUO groups tended to have a high level of BMI, WC, elevated liver function index, altered metabolic syndrome components, and high risk of developing hypertension and coronary heart disease, as well as decreased levels of serum TT, LH, and FSH (Table 1). There was no significant difference of serum testosterone level between the MHO and MUNW groups. The significant differences in the composition of the four groups with respect to smoking status and alcohol consumption are also shown in Table 1.
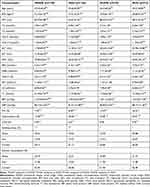 |
Table 1 Baseline Characteristics in Different Metabolic Obesity Phenotypes |
The Prevalence of Hypotestosteronemia in Different Metabolic Obesity Phenotypes
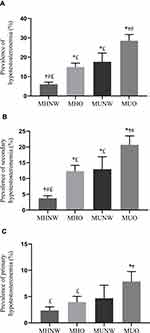
To compare the difference of prevalence of hypotestosteronemia among different metabolic obesity phenotypes, we performed a Chi-squared test between each two groups. We observed an obvious elevated trend of prevalence of hypotestosteronemia in these four metabolic obesity phenotypes in Figure 1. Compared to the MHNW group, the other three groups have been demonstrated to have a higher risk of hypotestosteronemia. At the same time, compared to the MUO group, the other three groups were more likely to have a lower risk of hypotestosteronemia. However, no difference existed between MUNW and MHO group (Figure 1A). The results of comparison of prevalence of secondary hypotestosteronemia in four groups were similar to hypotestosteronemia (Figure 1B). However, the prevalence of primary hypotestosteronemia in the MHNW and MHO groups was significantly lower than in the MUO group. No significant difference existed in the prevalence of primary hypotestosteronemia in other groups (Figure 1C). These differences demonstrated that different metabolic obesity phenotypes could have different impacts on serum testosterone levels.
The Separated and Synergistic Effect of Overweight/Obesity and Metabolic Status on Serum TT Concentration
To examine the separated and synergistic effect of overweight/obesity and metabolic status on serum TT concentration, we carried out multivariate logistic regression analysis of hypotestosteronemia with low serum TT levels (<12 nmol/L) adjusting for age, LH, smoking status (never, ever, current), and alcohol intake (never, ever, current). Both overweight/obesity and metabolic syndrome, considered separately, were risk factors of low serum TT levels. The multivariate-adjusted Odd Ratio (OR) of low serum TT levels in overweight/obesity men was 3.072 (95% CI= 2.414–3.911). The risk of low serum TT levels increased appropriately 3.294-fold in metabolic syndrome individuals compared to metabolic healthy individuals (Table 2). Although the risk of hypotestosteronemia in overweight/obesity and metabolic syndrome individuals was attenuated after mutual analysis, the OR of low serum TT levels remained statistically significant adjusting for age, LH, smoking status (never, ever, current), and alcohol intake (never, ever, current) (Table 2).
 |
Table 2 Adjusted Correlations of BMI and Presence of the MS with Total Testosterone |
 |
Table 3 Logistic Regression Analysis of Hypotestosteronemia According to Different Metabolic Phenotypes of Obesity |
The Independence of Different Metabolic Obesity Phenotypes for Hypotestosteronemia
To study the independence of different metabolic obesity phenotypes for male hypotestosteronemia, we performed logistic regression analysis to observe the OR of hypotestosteronemia in four groups. Compared to participants in the MHNW group, the risk of developing hypotestosteronemia in the MHO, MUNW, and MUO groups significantly increased. ORs of hypotestosteronemia in the MHO, MUNW, and MUO groups were 2.736 (95% CI=2.132–3.512), 3.332 (2.314–4.799), and 6.213 (4.842–7.972), respectively (Table 3). After adjusted for age, OR of hypotestosteronemia in MHO and MUO groups decreased, but it was still significant. Male subjects in the MHO, MUNW, and MUO groups also had a significantly increased risk of hypotestosteronemia in contrast to those in the MHO group after adjusted for age, LH, smoking status (never, ever, current), and alcohol intake (never, ever, current).
Discussion
Obesity and metabolic syndrome are both considered to be risk factors of low serum TT levels. We provided precise insight into the association between different metabolic obesity phenotypes and low serum TT levels in a large-scale population of Chinese men aged from 40–75 years old. It demonstrated that individuals with different metabolic obesity phenotypes including MHO, MUNW, and MUO, respectively, can all have a higher risk of developing low serum TT levels, especially significantly in the MUO group.
In our study, we draw a conclusion that both overweight/obese men and metabolic unhealthy men had a higher risk of developing low serum TT levels than normal weight and metabolic healthy individuals, which is consistent with the results in previous studies.8–11,13 It demonstrated that serum TT levels in the male population were associated with BMI and different components of metabolic syndrome. A Korean study has shown that serum TT levels were negatively associated with BMI, WC, FPG, TG, and blood pressure, and positively associated with HDL-L levels.11 A meta-analysis that included 9,525 men of different ethnicities proved that serum TT levels were negatively correlated with each component of metabolic syndrome.8 It has been reported that hyperinsulinemia which was associated with metabolic syndrome can inhibit the secretion of testosterone through insulin receptors which were expressed on Leydig cells.23 And excess adiposity tissue could lead to high levels of leptin, which can inhibit the steroidogenesis and the secretion of testosterone. Inflammatory cytokines, especially IL-1β, IL-6, and TNF-α, which were elevated in obesity or metabolic unhealthy individuals, can directly inhibit the steroidogenesis and subsequent testosterone production.24 Although there are several studies investigating the association of overweight/obesity and metabolic syndrome components with testosterone levels separately, there is still quite a gap in the literature considering the relationship between different metabolic obesity phenotypes and total testosterone levels.
As we have proved, different metabolic obesity phenotypes except for the MHNW group are at a high risk of developing low serum TT levels. Among these groups, MUO individuals are more likely to have a low level of serum TT than others. Studies have demonstrated that age, smoking status, and alcohol consumption were associated with different metabolic obesity phenotypes.25,26 However, after adjusting these above confounding factors, the relationship between TT levels and metabolic obesity phenotypes remained significant. To the best of our knowledge, the only study on this topic was performed to explore male sexual dysfunction with different metabolic obesity phenotypes.19 It showed that the men with MHO and metabolically complicated obesity (MCO) had lower testosterone levels and only MCO men had worse erectile function compared with normal weight men. However, the results were derived from male patients with sexual dysfunction or infertility, which could have different characteristics compared with the general male population. What’s more, the definition of MCO was the presence of at least one abnormality among hypertension, low HDL-C, and diabetes in obese men, and the contrast group was men with normal weight regardless of metabolic status.
In our study, there was a higher risk of developing hypotestosteronemia in male patients in MHO compared to the MHNW group, but a lower risk than the MUO group. MHO individuals referred to overweight/obese people without the risk factor of metabolic syndrome, and several studies have reported that these individuals were fat but fit with more favorable inflammatory and metabolic profiles.27 However, the recent systematic review and meta-analysis showed that MHO subjects had an increased risk for the development of cardiovascular diseases and all-cause mortality in contrast to MHNW subjects.28 And our results indicated that MHO individuals had a decreased total testosterone level compared to MHNW individuals. It has been reported that metabolic healthy obese individuals have certain differences in multiple aspects, such as fat distribution, inflammatory status, insulin resistance, and postprandial lipemia, compared with metabolic unhealthy obese individuals.29,30 Compared with MUO individuals, MHO was shown to have increased subcutaneous fat relative to visceral fat, lower liver fat, and insulin sensitivity. Excessive visceral fat or insulin resistance rather than general adiposity in obese individuals were at high risk of developing pre-diabetes and type 2 diabetes mellitus. Metabolic healthy subjects displayed lower postprandial response of plasma TG and higher postprandial response of hs-CRP, compared with those metabolic unhealthy, independent of whether or not they were obese. We also observed that testosterone levels were decreased in male subjects with MUO compared to MHO. Clinical features and metabolism difference caused by metabolic healthy obesity phenotype can partly explain the alternations of testosterone concentration in MHO group compared to MHNW and MUO group. Since MHO is a dynamic state (with a significant proportion of MHO subjects progressing to MUO over time), it is necessary to encourage weight control in MHO phenotypes to prevent the development of metabolic related disease including hypotestosteronemia.
We also observed that testosterone levels in male subjects with MUNW were decreased compared to the MHNW group, but these were increased in the MUNW group compared to the MUO group, and no significant difference to the MHO group. MUNW individuals displayed several metabolic abnormal risks including insulin resistance, atherogenic lipid profiles, visceral adiposity accumulation, and lower physical energy expenditures, despite having a normal BMI, and eventually had the increased risk of cardiovascular disease.31 It has been reported that MUNW phenotype exhibited increased arterial stiffness and carotid atherosclerosis compared to MHNW or MHO subjects.32 And another report has shown that MUNW individuals had a higher risk of cardiovascular disease than MHO, as well as reduced morbidity and mortality.33 The MHO phenotype was associated with a better overall metabolic profile and less oxidative stress than that observed in MUNW individuals.34 In our study, there is no significant difference of testosterone levels between MUNW and MHO group, which indicated that both MUNW and MHO can exert an influence on total testosterone levels. The underlying mechanism needs further study. It was shown that male subjects in the MUO group have decreased serum TT levels compared to MHO and MUNW groups, which can be explained by synergistic effect of obesity and metabolic unhealthy on TT levels in patients with MUO. It also indicated that male patients with MHO or MUNW should pay attention to losing weight or improving metabolic status in order to prevent the further drop of TT levels.
It is well known that testosterones are mainly synthesized in Leydig cells, which could be modulated by hypothalamus-pituitary levels. In our study, we also found that gonadotropins FSH and LH were significantly reduced in MHO and MUO groups compared to MHNW. However, there was no significant difference of FSH and LH levels between MHNW and MUNW, as well as MHO and MUO groups. There was an increasing conversion of androgen precursors into estrogens by aromatase system in adipose tissue among obese male population. Inappropriate effects of increasing estrogens might reduce LH secretory mass per secretory burst without any alternation in burst number.35 The exact mechanism for decreased gonadotrophins in male obesity could also include leptin, inflammatory mediators, and hypothalamic kisspeptin affecting gonadotropins- releasing hormone secretion.36 These results indicated that low levels of gonadotropins can partly explain the decreasing total testosterone levels in obesity participants, but not affected by metabolic status. To observe the effect of metabolic obesity phenotypes on pituitary or testicular levels, we further compared the difference of prevalence of secondary hypotestosteronemia and primary hypotestosteronemia in four metabolic obesity phenotypes. It showed that the difference of prevalence of secondary hypotestosteronemia and primary hypotestosteronemia in four groups was similar to hypotestosteronemia. The results were confusing, which was inconsistent with the results that LH and FSH levels were only affected by obesity. Although we did not get information about the pulse and frequency of LH and FSH secretory, which also played an important role in regulating the production of testosterone levels. Therefore, further studies about the pulse and frequency of LH and FSH secretory, the alternations of structure and function in testis need to be performed in different metabolic obesity phenotypes.
This is the first study to investigate the relationship of different metabolic obesity phenotypes with serum TT concentration in a large-scale male population. In our study, other potential risk factors of low TT levels such as age, LH levels, smoking status, and alcohol consumption were taken into account to make the result more convincing. However, several limitations also existed in our study. We did not obtain information about other sex hormones such as SHBG and estrogen, which can have an effect on TT levels. Meanwhile, we cannot draw a conclusion about the association of different metabolic obesity phenotypes and other sex hormones. What’s more, the cross-sectional study was unable to detect any causal relationship between different metabolic obesity phenotypes and testosterone levels. Large prospective studies are needed to validate this relationship.
In conclusion, both overweight/obesity and metabolic abnormalities are important risk factors for low serum TT levels. Different metabolic obesity phenotypes have influence on lower serum testosterone level in different degrees, especially in the MUO group with a lowest serum TT concentration. Male subjects in the MHO and MUNW groups had significantly decreased testosterone levels compared to the MHNW group, however, there was no significant difference of testosterone levels between the MHO and MUNW groups. Clinical physicians should pay attention to the weight combined with metabolic status of patients when we explore the reason of decreasing concentration of serum TT in male populations. Individual weight control and prevention of metabolic syndrome may be used for the primary prevention of male hypotestosteronemia. Patients with low level of serum TT can improve by losing weight or improving metabolic status.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author, Qingbo Guan, upon request.
Acknowledgments
The research used data collected as part of a longitudinal study (REACTION) which was a multi-center, prospective, observational cohort study in Chinese people. We gratefully acknowledge the REACTION study group for organizing the national epidemiological REACTION study in China.
Funding
This work was supported in part by grants from the National Natural Science Foundation of China (81770860, 81471078 and 81641030) and Key Research and Development Plan of Shandong Province (2016GSF201007).
Disclosure
The authors declare that there is no conflict of interest.
References
1. Pinkey JH, Kopelman PG. Endocrine Determinant of Obesity. In: Bray GA, Bouchard C, Handbook of Obesity. Etiology and Pathophysiology.
2. Kalyani RR, Dobs AS. Androgen deficiency, diabetes, and the metabolic syndrome in men. Curr Opin Endocrinol Diabetes Obes. 2007;14(3):226–234. doi:10.1097/MED.0b013e32814db856
3. Escobar-Morreale HF, Santacruz E, Luque-Ramírez M, Botella Carretero JI. Prevalence of ‘obesity-associated gonadal dysfunction’ in severely obese men and women and its resolution after bariatric surgery: a systematic review and meta-analysis. Hum Reprod Update. 2017;1–19.
4. Pasquali R, Vicennati V, Gambineri A. Adrenal and gonadal function in obesity. J Endocrinol Invest. 2002;25:893–898. doi:10.1007/BF03344053
5. Eriksson J, Haring R, Grarup N, et al. Causal relationship between obesity and serum testosterone status in men: a bi-directional mendelian randomization analysis. PLoS One. 2017;12(4):4. doi:10.1371/journal.pone.0176277
6. Renato Pasquali R. Obesity and androgens: facts and perspectives. Fertil Steril. 2006;85(5):1319–1340. doi:10.1016/j.fertnstert.2005.10.054
7. Munoz-Garach A, Cornejo-Pareja I, Tinahones FJ. Does metabolically healthy obesity exist? Nutrients. 2016;8(6):320. doi:10.3390/nu8060320
8. Brand JS, Rovers MM, Yeap BB, et al. Testosterone, sex hormone-binding globulin and the metabolic syndrome in men: an individual participant data meta-analysis of observational studies. PLoS One. 2014;9(7):e100409. doi:10.1371/journal.pone.0100409
9. Corona G, Monami M, Rastrelli G, et al. Testosterone and metabolic syndrome: a meta-analysis study. J Sex Med. 2011;8(1):272–283. doi:10.1111/j.1743-6109.2010.01991.x
10. Angelova P, Kamenov Z, Tsakova A, El-Darawish Y, Okamura H. Interleukin-18 and testosterone levels in men with metabolic syndrome.. Aging Male. 2018;21(2):130–137. doi:10.1080/13685538.2017.1401993
11. Hong D, Kim Y-S, Son ES, et al. Total testosterone and sex hormone-binding globulin are associated with metabolic syndrome independent of age and body mass index in Korean men. Maturitas. 2013;74:148–153. doi:10.1016/j.maturitas.2012.10.016
12. Sá EQ, Sá FC, Oliveira KC, Feres F, Verreschi IT. Association between sex hormone-binding globulin (SHBG) and metabolic syndrome among men. Sao Paulo Med J. 2014;132(2):111–115. doi:10.1590/1516-3180.2014.1322666
13. Zhang J, Huang X, Liao M, et al. Both total testosterone and sex hormone-binding globulin are independent risk factors for metabolic syndrome: results from fangchenggang area male health and examination survey in China. Diabetes Metab Res Rev. 2013;29:391–397. doi:10.1002/dmrr.2405
14. Siddiqui K, Al-Rubeaan K, Nawaz SS, Aburisheh KH, Alaabdin AMZ, Tolba IA. Serum Sex Hormone Binding Globulin (SHBG) relation with different components of metabolic syndrome in men with type 2 diabetes. Horm Metab Res. 2018;50(2):138–144. doi:10.1055/s-0043-123348
15. Callou de Sá EQ, Feijó de Sá FC, e Silva RDS, et al. Endogenous oestradiol but not testosterone is related to coronary artery disease in men. Clin Endocrinol (Oxf). 2011;75(2):177–183. doi:10.1111/j.1365-2265.2011.04017.x
16. Bhasin S, Jasjua GK, Pencina M, et al. Sex hormone-binding globulin, but not testosterone, is associated prospectively and independently with incident metabolic syndrome in men: the framingham heart study. Diabetes Care. 2011;34(11):2464–2470. doi:10.2337/dc11-0888
17. Lopez-Miranda J, Perez-Martinez P. It is time to define metabolically obese but normal-weight (MONW) individuals. Clin Endocrinol (Oxf). 2013;79(3):314–315. doi:10.1111/cen.12181
18. Martini AC, Molina RI, Ruiz RD, Fiol de Cuneo M. Obesity and male fertility. Rev Fac Cien Med Univ Nac Cordoba. 2012;69:2.
19. Lotti F, Rastrelli G, Maseroli E, et al. Impact of metabolically healthy obesity in patients with andrological problems. J Sex Med. 2019;16(6):821–832. doi:10.1016/j.jsxm.2019.03.006
20. Wang C, Nieschlag E, Swerdloff R, et al. Investigation, treatment, and monitoring of late-onset hypogonadism in males: ISA, ISSAM, EAU, EAA, and ASA recommendations. J Androl. 2008;30(1):1–9. doi:10.2164/jandrol.108.006486
21. Lauer MS, Fontanarosa PB. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel III). JAMA. 2001;285(19):2486–2497. doi:10.1001/jama.285.19.2486
22. Kim JM, Kim BH, Lee H, et al. The relationship between thyroid function and different obesity phenotypes in Korean euthyroid adults. Diabetes Metab J. 2019;43(6):867–878.
23. Pitteloud N, Hardin M, Dwyer AA, et al. Increasing insulin resistance is associated with a decrease in leydig cell testosterone secretion in men. J Clin Endocrinol Metab. 2005;90:2636–2641. doi:10.1210/jc.2004-2190
24. Lin T, Wang D, Stocco DM. Interleukin-1 inhibits leydig cell steroidogenesis without affecting steroidogenic acute regulatory protein messenger ribonucleic acid or protein levels. J Endocrinol. 1998;156:461–467. doi:10.1677/joe.0.1560461
25. Primeau V, Coderre L, Karelis AD, et al. Characterizing the profile of obese patients who are metabolic healthy. Int I Obes (Lond). 2011;35:971–981. doi:10.1038/ijo.2010.216
26. Wang B, Zhang M, Wang S, et al. Dynamic status of metabolically healthy overweight/obesity and metabolically unhealthy and normal weight and the risk of type 2 diabetes mellitus: a cohort study of a rural adult Chinese population. Obes Res Clin Pract. 2018;12(1):61–71. doi:10.1016/j.orcp.2017.10.005
27. Ortega FB, Cadenas-Sanchez C, Migueles JH, et al. Role of physical activity and fitness in the characterization and prognosis of the metabolically healthy obesity phenotype: a systematic review and meta-analysis. Prog Cardiovasc Dis. 2018;61(2):190–205. doi:10.1016/j.pcad.2018.07.008
28. Yeh T-L, Chen -H-H, Tsai S-Y, Lin C-Y, Liu S-J, Chien K-L. The relationship between metabolically healthy obesity and the risk of cardiovascular disease: a systematic review and meta-analysis. J Clin Med. 2019;8(8):1228. doi:10.3390/jcm8081228
29. Perez-Martinez P, Alcala-Diaz JF, Delgado-Lista J, et al. Metabolic phenotypes of obesity influence triglyceride and inflammation homoeostasis. Eur J Clin Invest. 2014;44(11):1053–1064. doi:10.1111/eci.12339
30. Wildman RP, Muntner P, Reynolds K, et al. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999–2004). Arch Intern Med. 2008;168:1617–1624. doi:10.1001/archinte.168.15.1617
31. Karelis AD, St-Pierre DH, Conus F, Rabasa-Lhoret R, Poehlman ET. Metabolic and body composition factors in subgroups of obesity: what do we know? J Clin Endocrinol Metab. 2004;89(6):2569e75. doi:10.1210/jc.2004-0165
32. Yoo HJ, Hwang SY, Hong HC, et al. Association of metabolically abnormal but normal weight (MANW) and metabolically healthy but obese (MHO) individuals with arterial stiffness and carotid atherosclerosis. Atherosclerosis. 2014;234(1):218–223. doi:10.1016/j.atherosclerosis.2014.02.033
33. Choi KM, Cho HJ, Choi HY, et al. Higher mortality in metabolically obese normal-weight people than in metabolically healthy obese subjects in elderly Koreans. Clin Endocrinol (Oxf). 2013;79(3):364–370. doi:10.1111/cen.12154
34. Kim M, Paik JK, Kim KR, et al. Increased oxidative stress in normal-weight postmenopausal women with metabolic syndrome compared with metabolically healthy overweight/obese individuals. Metabolism. 2012;62(4):554–560. doi:10.1016/j.metabol.2012.10.006
35. Vermeulen A, Kaufman JM, Deslypere JP, Thomas G. Attenuated luteinizing hormone (LH) pulse amplitude but normal LH pulse frequency, and its relation to plasma androgens in hypogonadism of obese men. J Clin Endocrinol Metab. 1993;76(5):1140–1146. doi:10.1210/jcem.76.5.8496304
36. George JT, Millar RP, Anderson RA. Hypothesis: kisspeptin mediates male hypogonadism in obesity and type 2 diabetes. Neuroendocrinology. 2010;91(4):302–307. doi:10.1159/000299767
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.