Back to Journals » Journal of Pain Research » Volume 13
Association Between Serum 25-Hydroxyvitamin D Concentrations and Chronic Pain: Effects of Drinking Habits
Authors Suzuki K , Tsujiguchi H, Miyagi S , Thi Thu Nguyen T, Hara A, Nakamura H, Shimizu Y, Hayashi K, Yamada Y, Minh Nguyen P , Tao Y, Kannon T , Tajima A , Nakamura H
Received 21 August 2020
Accepted for publication 9 October 2020
Published 19 November 2020 Volume 2020:13 Pages 2987—2996
DOI https://doi.org/10.2147/JPR.S277979
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Keita Suzuki,1 Hiromasa Tsujiguchi,1– 3 Sakae Miyagi,3,4 Thao Thi Thu Nguyen,2 Akinori Hara,1– 3 Haruki Nakamura,2 Yukari Shimizu,5 Koichiro Hayashi,1 Yohei Yamada,1 Phat Minh Nguyen,2 Yuichi Tao,2 Takayuki Kannon,3,6 Atsushi Tajima,3,6 Hiroyuki Nakamura1– 3
1Department of Public Health, Graduate School of Advanced Preventive Medical Sciences, Kanazawa University, Kanazawa, Ishikawa 920-8640, Japan; 2Department of Environmental and Preventive Medicine, Graduate School of Medical Science, Kanazawa University, Kanazawa, Ishikawa 920-8640, Japan; 3Kanazawa University Advanced Preventive Medical Sciences Research Center, Kanazawa, Ishikawa 920-8640, Japan; 4Innovative Clinical Research Center, Kanazawa University, Kanazawa, Ishikawa 920-8640, Japan; 5Department of Nursing, Faculty of Health Sciences, Komatsu University, Komatsu, Ishikawa 923-0961, Japan; 6Department of Bioinformatics and Genomics, Graduate School of Advanced Preventive Medical Sciences, Kanazawa University, Kanazawa, Ishikawa 920-8640, Japan
Correspondence: Keita Suzuki
Department of Public Health, Graduate School of Advanced Preventive Medical Sciences, Kanazawa University, 13-1, Takara-Machi, Kanazawa, Ishikawa 920-8640, Japan
Tel +81-76-265-2218
Fax +81-76-234-4233
Email [email protected]
Purpose: Although the explanation for inconsistencies in the reported association between serum 25-hydroxyvitamin D [25(OH)D] levels and chronic pain (CP) has not yet been determined, understanding this discrepancy is necessary for the development of vitamin D supplementation as an effective treatment for CP. The aim of this cross-sectional study was to examine the relationship between 25(OH)D concentrations and CP according to drinking habits in Japanese subjects.
Patients and Methods: We distributed invitation letters to 2314 individuals older than 40 years in Shika town, a rural area in Japan, and 724 subjects (386 females; mean age: 63.9 ± 10.4 years) were recruited. CP was defined as persistent pain lasting at least 3 months in any part of the body. Serum concentrations of 25(OH)D, a biomarker of the vitamin D status, were measured using a radioimmunoassay. A serum 25(OH)D level < 20 ng/mL was defined as serum 25(OH)D deficiency. Drinking habits were assessed using a self-administered questionnaire. There were three choices, “rarely drink”, “sometimes” and “everyday”. Respondents who answered “rarely drink” were labelled as non-drinkers and the others as drinkers.
Results: The prevalence of CP was 40.6%. A significant interaction between CP and drinking habits on 25(OH)D concentrations was observed (p = 0.098). A one-way analysis of covariance was performed to compare 25(OH)D concentrations between the subjects with and without CP in each drinking group, and the serum 25(OH)D levels of subjects with CP were significantly lower than those without CP among drinkers (p = 0.007). A logistic regression analysis revealed a correlation between serum 25(OH)D deficiency and CP in drinkers after adjustments for several confounding factors (odds ratio: 0.499; 95% confidence interval: 0.268 − 0.927; p = 0.028).
Conclusion: The present results suggest that low serum 25(OH)D concentrations are associated with the development of CP in drinkers.
Keywords: pain, vitamin D, alcohol, cross-sectional analysis, epidemiology
Introduction
The International Association for the Study of Pain (IASP) defines chronic pain (CP) as pain without an apparent biological cause that persists for longer than 3 months.1 A meta-analysis from the United Kingdom reported that the prevalence of CP ranged from 35.0% to 51.3%.2 Another review reported that 25%−76% of older people living in the community had CP.3 CP has a negative impact on health-related quality of life, work productivity and increases the risk of developing depression and anxiety.4 Furthermore, the national cost of pain in the United States in 2008 was estimated to be between 560 and 635 billion dollars.5 Therefore, the development of effective and reasonable treatments is urgently needed.
Vitamin D is a vital hormone for optimal neuromuscular, bone, and immune functions, and its sources are dietary intake and cutaneous synthesis. Vitamin D is metabolized into 25-hydroxyvitamin D [25(OH)D] in the liver and then into 1,25-dihydroxyvitamin D [1,25(OH)2D] in the kidneys.6 Serum 25(OH)D concentrations are regarded as the most useful marker of the vitamin D status stored from sunlight exposure, dietary intake, supplements, and other factors.7
Interventional studies previously reported the efficacy of vitamin D supplementation for the treatment of CP as well as good patient adherence.8–10 However, the 2015 Cochrane review concluded that vitamin D was not significantly beneficial for various CP conditions.11 Although observational studies examined the relationship between 25(OH)D concentrations and CP, the findings obtained have been inconsistent.12–14 The reasons for these disparities have not yet been identified, but need to be clarified for the development of vitamin D supplementation as an effective treatment for CP.
Several previous studies have reported that alcohol consumption is associated with a reduced risk of vitamin D deficiency,15,16 whereas other studies have reported a negative association17–19 or no association.20–22 Although alcohol has been shown to influence the expression of the parathyroid hormone23–25 or CYP24A1 induction26 in several studies, the mechanism underlying the association between alcohol and serum 25(OH)D levels remains unknown. However, one possibility is that alcohol consumption may affect CP by modulating the vitamin D status. Therefore, performing a subgroup analysis stratified by drinking habits is necessary to clearly evaluate the association between CP and serum 25(OH)D concentrations.
Based on these studies, we hypothesized that alcohol consumption may affect the association between CP and 25(OH)D concentrations. Therefore, we herein investigated the association between CP and serum 25(OH)D concentrations among individuals who do and do not consume alcohol.
Materials and Methods
Study Design and Subjects
We utilized cross-sectional data gathered between 2011 and 2015 in the Shika study. The Shika study is a longitudinal community-based observational study that has been conducted among the residents of Shika town, located on Noto peninsula in Ishikawa prefecture, since 2011. The town has more than 20,000 residents, and the climate is humid subtropical. We recruited subjects from two model districts in the town for the present study. We invited all adults older than 40 years (n = 2314) in the districts to participate. A self-administrated questionnaire was delivered to all subjects by trained interviewers, who explained the outline of the Shika study and how to fill in the questionnaire. In total, 2199 subjects (95%) responded to the questionnaire, and 972 (42%) who underwent a comprehensive health examination were included.
We omitted subjects with data missing on the variables used in the analysis. To avoid a treatment bias, we also excluded subjects who had been using vitamin D supplementation. Therefore, data collected from 724 subjects who completed the health examination and replied to the questionnaire were available for analysis in the present study. The details of recruitment are shown in Figure 1.
 |
Figure 1 Flow chart of subject recruitment. |
The present study was conducted with the approval of the Ethics Committee of Kanazawa University (No.1491). All subjects provided written informed consent for inclusion before they participated in the present study. The study was conducted in accordance with the Declaration of Helsinki.
Pain Questions
We asked subjects “Have you been having any pain persisting for 3 months or longer?” on a self-administered questionnaire. The definition of CP was according to that presented by IASP.1 Subjects with CP were assigned to the CP group.
Serum 25(OH)D Measurement
Fasting blood samples were collected from all subjects at the health examination. Serum 25(OH)D concentrations were measured using a radioimmunoassay (RIA) (25-hydroxyvitamin D 125I RIA Kit, DiaSorin Inc., Stillwater, Minnesota, USA). Coefficients of variation for inter-assay of high-performance liquid chromatography (HPLC), competitive protein binding, and RIA, have been reported to be 8.4%, 14%, and <12%, respectively.27 A previous study that measured serum 25(OH)D levels using HPLC-atmospheric pressure chemical ionization-mass spectrometry, RIA, and chemiluminescent immunoassays reported that the RIA value was between that of the other two techniques.28
According to several previous reports, the minimum desirable serum 25(OH)D level is 20 ng/mL (50 nmol/L).29,30 Therefore, a serum 25(OH)D level <20 ng/mL was defined as serum 25(OH)D deficiency in this study.
Questions on Drinking Habits
The drinking habits of the participant were collected by questionnaire. We used a structured-question to ask if the participants drink alcohol or not. The participant was asked, “How often do you drink alcohol?” There were three choices, “rarely drink”, “sometimes” and “everyday”. We assigned subjects who answered “rarely drink” as “non-drinkers”, and the others as “drinkers” for the analysis.
Other Variables
We measured age, height, weight, grip strength, calf circumference, C-reactive protein (CRP), and the osteo sono-assessment index in all subjects at the health examination. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters. The values of grip strength and calf circumference were divided by body weight to adjust for the influence of physique, and these values were used in the analysis.
To assess the smoking status and frequency of exercise, we used self-administered questionnaires. Subjects were classified into the following three groups according to their smoking status: the non-smoker group, the ex-smoker group, and the current smoker group. We also assigned subjects into three groups according to the frequency of walking for more than 30 min in 1 week as follows: more than 5 days, 1 to 4 days, and no exercise. The analgesics administration status was also assessed on the self-administered questionnaire.
We evaluated the nutritional status using a brief-type self-administered diet history questionnaire (BDHQ) that asked subjects about the consumption frequency of 58 food and beverage items during the previous month. These items are mainly from the food list used in the National Health and Nutrition Survey of Japan and commonly consumed in Japan. The validity of BDHQ has been demonstrated in other studies, and is considered to have a satisfactory ranking ability for many nutrients in Japanese subjects.31,32 We excluded all subjects who reported a total energy intake of less than 600 kcal/day (half of the energy intake required for the lowest physical activity category) or more than 4000 kcal/day (1.5-fold the energy intake required for the moderate physical activity category) to avoid under/overestimations leading to bias in the analysis of other nutrients. We obtained data on total energy, protein, lipid, carbohydrate, and vitamin D intake, and nutrient data were adjusted for daily energy intake using the density method.
Statistical Analysis
We summarized continuous variables as means and standard deviation (SD), and presented categorical variables as numbers (N) and percentages (%). To compare differences in the mean levels of continuous variables and categorical variables between subjects with and without CP in each drinking group, the Student’s t-test and chi-squared test were used. A two-way analysis of covariance (ANCOVA) was performed to assess the interaction on serum 25(OH)D concentrations between the CP status and drinking habits with adjustments for sex, age, BMI, the smoking status, exercise habit, the analgesics administration status, the osteo sono-assessment index, grip strength/body weight, and calf circumference/body weight. To confirm differences in the adjusted serum 25(OH)D concentrations between the subjects with and without CP in each drinking habits group, we used a one-way ANCOVA with adjustments for sex, age, BMI, the smoking status, exercise habit, the analgesics administration status, the osteo sono-assessment index, grip strength/body weight, and calf circumference/body weight. The Bonferroni post hoc test was then conducted for multiple comparisons. A multiple logistic regression analysis was performed to examine the association between CP and the categorized 25(OH)D levels in each drinking group after adjustments for the following independent factors: sex, age, BMI, the smoking status, exercise habit, the analgesics administration status, the osteo sono-assessment index, grip strength/body weight, and calf circumference/body weight.
We performed all statistical analyses using IBM SPSS Statistics version 21.0 (SPSS Inc., Armonk, NY, USA). A two-sided p-value < 0.05 was considered to be significant. A p-value < 0.10 was considered significant for the interaction test.
Results
Subject Characteristics
Table 1 shows the characteristics of subjects according to CP. The mean age of the 724 subjects analyzed in the present study was 63.9 ± 10.4 years. A total of 294 (40.6%) subjects had CP. Mean serum 25(OH)D concentrations in the groups with and without CP were 24.37 ± 8.25 and 23.58 ± 7.36 ng/mL, respectively. In total, 43.3% of non-CP subjects and 50.7% of CP subjects were drinkers.
 |
Table 1 Subject Characteristics |
Subject Characteristics According to CP and Drinking Habits
Table 2 shows the subject characteristics according to CP and drinking habits. In non-drinkers, no significant differences were observed in 25(OH)D concentrations between the groups with and without CP, whereas among drinkers, a slight difference was noted between these groups (p = 0.072). Among non-drinkers, significant differences were found in grip strength/body weight (p = 0.002) and the osteo sono-assessment index (p = 0.011) between the groups with and without CP. Drinkers with CP had significantly lower protein (p = 0.014) and vitamin D intakes (p = 0.009) than those without CP.
 |
Table 2 Comparison of Characteristics Between CP and Non-CP Subjects According to Drinking Habits |
Interaction Between CP and Drinking Habits on Serum 25(OH)D Concentrations and Comparison of Adjusted Serum 25(OH)D Values
A two-way ANCOVA was performed to examine the interaction between CP and drinking habits on serum 25(OH)D concentrations (Table 3). We observed a significant main effect of CP (p = 0.021) and drinking habits (p < 0.001). A significant interaction was noted between CP and drinking habits on serum 25(OH)D levels after adjustments for covariates (p = 0.098). The one-way ANCOVA revealed that the subjects without CP had higher 25(OH)D concentrations than those with CP (p = 0.007) in drinkers after adjustments for covariates (Table 3). In non-drinkers, no significant differences were found.
 |
Table 3 Interaction Between CP and Drinking Habits on Serum 25(OH)D Concentrations and Comparison of Adjusted Serum 25(OH)D Values |
Association Between CP and Drinking Habits
The association between CP and drinking habits is shown in Table 4. Drinking habits correlated with CP after adjustments for sex, age, BMI, the smoking status, exercise habit, the analgesics administration status, the osteo sono-assessment index, grip strength/body weight, and calf circumference/body weight (odds ratio: 1.467; 95% confidence interval: 1.018–2.114; p = 0.040).
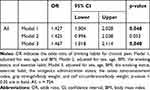 |
Table 4 Logistic Analysis of Chronic Pain as a Dependent Variable with Drinking Habits as an Independent Variable |
Association Between CP and Serum 25(OH)D Deficiency
The association between CP and serum 25(OH)D deficiency in each group stratified by drinking habits are shown in Table 5. The results obtained showed that serum 25(OH)D deficiency correlated with CP in drinkers after adjustments for sex, age, BMI, the smoking status, exercise habit, the analgesics administration status, the osteo sono-assessment index, grip strength/body weight, and calf circumference/body weight (odds ratio: 0.499; 95% confidence interval: 0.268−0.927; p = 0.028), but not in non-drinkers.
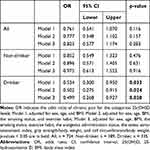 |
Table 5 Logistic Analysis of the Categorized Serum 25(OH)D Levels as a Dependent Variable with Chronic Pain as an Independent Variable |
Discussion
This cross-sectional study investigated the association between serum 25(OH)D concentrations and CP with stratification by drinking habits. The results obtained showed that drinkers with CP had lower 25(OH)D concentrations than those without CP. This result implies that lower 25(OH)D concentrations correlated with CP in drinkers only.
Previous studies reported discordant findings on the relationship between serum 25(OH)D concentrations and CP. Several large cross-sectional studies showed low 25(OH)D concentrations with widespread CP.33,34 Furthermore, a systematic review published in 2018 concluded that low 25(OH)D concentrations may be associated with pain conditions.35 On the other hand, large population-based studies found no relationships between 25(OH)D levels and self-reported CP after adjustments for several confounding factors.14,36 The reasons for inconsistencies in the relationship between serum 25(OH)D concentrations and CP currently remain unknown because these studies did not perform analyses with the stratification of subjects by factors considered to affect the association between CP and 25(OH)D. To the best of our knowledge, the present study is the first to investigate the association between serum 25(OH)D and CP according to drinking habits, which is assumed to influence this relationship.
The positive effects of moderate alcohol consumption on CP have been demonstrated.37–39 However, these findings may be attributed to “reverse causation”. “Reverse causation” means that individuals with CP reduce their alcohol intake because of the harmful effects of alcohol on their symptoms. A Mendelian randomization study using UK Biobank recently showed that alcohol consumption did not have a positive impact on the risk of widespread CP.40 In contrast, the present results suggested that drinking habits had negative effects on CP. Previous studies only included individuals with widespread CP.37–40 A review published in 2016 reported that the prevalence of widespread CP was 10–15%.41 However, 40.6% of subjects in the present study had CP. Therefore, we may have included not only subjects with widespread CP but also those with another CP status, such as osteoarthritis. Previous cross-sectional and longitudinal studies demonstrated a relationship between moderate alcohol consumption and the development of osteoarthritis.42,43 Therefore, the present results appear to support the relationship between alcohol consumption and CP.
Although previous studies investigated the effects of alcohol consumption on serum vitamin D concentrations, this relationship remains unclear.15–22 In studies that reported a positive relationship between frequent alcohol consumption and the vitamin D status, alcohol suppressed the secretion of parathyroid hormone, which controls metabolism from 25(OH)D to 1,25(OH)2D in the kidney.23–25 These findings suggested that an increase in unconverted 25(OH)D may have resulted in higher serum 25(OH)D concentrations in drinkers. Drinkers had higher 25(OH)D concentrations than non-drinkers in the present study. Therefore, high serum 25(OH)D concentrations induced by alcohol consumption seems to mean not a favorable status of vitamin D but rather a deficiency of 1,25(OH)2D, which is the active form of vitamin D.
Several studies have reported that vitamin D deficiency causes a decrease in bone density, muscle weakness, and bone sensitivity.44,45 Vitamin D deficiency can lead to muscle weakness, which can cause herniated discs, and the other study has shown that vitamin D levels are associated with pain severity in patients with low back pain.46 In addition, several studies have demonstrated an association between vitamin D status and serum creatine kinase levels or inflammatory cytokines.47,48 Furthermore, the link between vitamin D deficiency and alteration of nociception via changes in gut microbiota has previously been detected in mice.49 Therefore, the analgesic effects of vitamin D may be compromised in subjects with drinking habits, owing to alcohol-induced disturbances in the metabolism of 25(OH)D to 1.25(OH)2D. Furthermore, insufficient vitamin D intake may adversely affect CP in drinkers. Drinkers with CP had a significantly lower intake of vitamin D than those without CP in the present study. Therefore, the association found between low 25(OH)D concentrations and the development of CP in drinkers only appears to support our hypothesis that drinking habits affect the inhibitory effects of vitamin D on pain.
These results indicate the importance of maintaining sufficient serum 25(OH)D concentrations, particularly in drinkers. Furthermore, vitamin D supplementation appears to be more effective for the treatment of CP in drinkers. However, there are several limitations in the present study that need to be addressed. The causality between serum 25(OH)D concentrations, CP, and drinking habits remains unclear because of the nature of this cross-sectional study. Furthermore, we need to consider the selection bias. In the present study, the percentage of health-conscious individuals may have been high because the health examination was only conducted on voluntary subjects. In addition, because the participation rate in this study was low, this study’s findings are limited in their generalizability. Moreover, we did not include information on the mental conditions of subjects, which may have affected CP, because the number of subjects with complete data of these variables was small.
Conclusion
We herein performed a cross-sectional study on Japanese subjects to assess the association between serum 25(OH)D concentrations and CP in drinkers and non-drinkers. An association between 25(OH)D concentrations and CP was only observed in drinkers. These results suggest that drinkers need to maintain sufficient serum 25(OH)D concentrations by an adequate vitamin D intake in order to inhibit CP.
Acknowledgments
The present study was funded by a Grant-in-Aid for Scientific Research (B) by the Japan Society for the Promotion of Science, number 19H03882. The authors wish to thank all the field workers and laboratory personnel of the Shika Study for their efforts.
Disclosure
The authors declare no conflicts of interest.
References
1. Treede RD, Rief W, Barke A, et al. Chronic pain as a symptom or a disease: the IASP classification of chronic pain for the international classification of diseases (ICD-11). Pain. 2019;160(1):19–27. doi:10.1097/j.pain.0000000000001384
2. Fayaz A, Croft P, Langford RM, Donaldson LJ, Jones GT. Prevalence of chronic pain in the UK: a systematic review and meta-analysis of population studies. BMJ Open. 2016;6(6):e010364. doi:10.1136/bmjopen-2015-010364
3. Abdulla A, Adams N, Bone M, et al. Guidance on the management of pain in older people. Age Ageing. 2013;42(Suppl 1):i1–57. doi:10.1093/ageing/afs199
4. Kawai K, Kawai AT, Wollan P, Yawn BP. Adverse impacts of chronic pain on health-related quality of life, work productivity, depression and anxiety in a community-based study. Fam Pract. 2017;34(6):656–661. doi:10.1093/fampra/cmx034
5. Gaskin DJ, Richard P. The economic costs of pain in the United States. J Pain. 2012;13(8):715–724. doi:10.1016/j.jpain.2012.03.009
6. Wimalawansa SJ. Non-musculoskeletal benefits of vitamin D. J Steroid Biochem Mol Biol. 2018;175:60–81. doi:10.1016/j.jsbmb.2016.09.016
7. Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the institute of medicine: what clinicians need to know. J Clin Endocrinol Metab. 2011;96(1):53–58. doi:10.1210/jc.2010-2704
8. Ghai B, Bansal D, Kanukula R, et al. Vitamin D supplementation in patients with chronic low back pain: an open label, single arm clinical trial. Pain Physician. 2017;20(1):E99–e105. doi:10.36076/ppj.2017.1.E99
9. Lakkireddy M, Karra ML, Patnala C, et al. Efficiency of vitamin D supplementation in patients with mechanical low back ache. J Clin Orthop Trauma. 2019;10(6):1101–1110. doi:10.1016/j.jcot.2019.06.018
10. Schlogl M, Chocano-Bedoya P, Dawson-Hughes B, et al. Effect of monthly vitamin D on chronic pain among community-dwelling seniors: a randomized, double-blind controlled trial. J Am Med Dir Assoc. 2019;20(3):356–361. doi:10.1016/j.jamda.2018.09.004
11. Straube S, Derry S, Straube C, Moore RA. Vitamin D for the treatment of chronic painful conditions in adults. Cochrane Database Syst Rev. 2015;5:Cd007771.
12. McCabe PS, Pye SR, Beth JM, et al. Low vitamin D and the risk of developing chronic widespread pain: results from the European male ageing study. BMC Musculoskelet Disord. 2016;17:32. doi:10.1186/s12891-016-0881-6
13. Thorneby A, Nordeman LM, Johanson EH. No association between level of vitamin D and chronic low back pain in Swedish primary care: a cross-sectional case-control study. Scand J Prim Health Care. 2016;34(2):196–204. doi:10.1080/02813432.2016.1183557
14. Wu Z, Camargo CA, Sluyter JD, et al. Association between serum 25-hydroxyvitamin D levels and self-reported chronic pain in older adults: a cross-sectional analysis from the ViDA study. J Steroid Biochem Mol Biol. 2019;188:17–22. doi:10.1016/j.jsbmb.2018.11.018
15. Larose TL, Chen Y, Camargo CA, Langhammer A, Romundstad P, Mai XM. Factors associated with vitamin D deficiency in a Norwegian population: the HUNT Study. J Epidemiol Community Health. 2014;68(2):165–170. doi:10.1136/jech-2013-202587
16. Gorter EA, Krijnen P, Schipper IB. Vitamin D deficiency in adult fracture patients: prevalence and risk factors. Eur J Trauma Emerg Surg. 2016;42(3):369–378. doi:10.1007/s00068-015-0550-8
17. Sobral-Oliveira MB, Faintuch J, Guarita DR, Oliveira CP, Carrilho FJ. Nutritional profile of asymptomatic alcoholic patients. Arq Gastroenterol. 2011;48(2):112–118.
18. Naude CE, Carey PD, Laubscher R, Fein G, Senekal M. Vitamin D and calcium status in South African adolescents with alcohol use disorders. Nutrients. 2012;4(8):1076–1094. doi:10.3390/nu4081076
19. Santori C, Ceccanti M, Diacinti D, et al. Skeletal turnover, bone mineral density, and fractures in male chronic abusers of alcohol. J Endocrinol Invest. 2008;31(4):321–326. doi:10.1007/BF03346365
20. Janssen HC, Emmelot-Vonk MH, Verhaar HJ, van der Schouw YT. Determinants of vitamin D status in healthy men and women aged 40–80 years. Maturitas. 2013;74(1):79–83. doi:10.1016/j.maturitas.2012.10.008
21. Kühn T, Kaaks R, Teucher B, et al. Dietary, lifestyle, and genetic determinants of vitamin D status: a cross-sectional analysis from the European prospective investigation into cancer and nutrition (EPIC)-Germany study. Eur J Nutr. 2014;53(3):731–741. doi:10.1007/s00394-013-0577-8
22. Hirani V, Cumming RG, Blyth FM, et al. Vitamin D status among older community dwelling men living in a sunny country and associations with lifestyle factors: the concord health and ageing in men project, Sydney, Australia. J Nutr Health Aging. 2013;17(7):587–593. doi:10.1007/s12603-013-0013-z
23. McCarty MF, Thomas CA. PTH excess may promote weight gain by impeding catecholamine-induced lipolysis-implications for the impact of calcium, vitamin D, and alcohol on body weight. Med Hypotheses. 2003;61(5–6):535–542. doi:10.1016/S0306-9877(03)00227-5
24. Rapuri PB, Gallagher JC, Balhorn KE, Ryschon KL. Alcohol intake and bone metabolism in elderly women. Am J Clin Nutr. 2000;72(5):1206–1213. doi:10.1093/ajcn/72.5.1206
25. Sampson HW. Alcohol, osteoporosis, and bone regulating hormones. Alcohol Clin Exp Res. 1997;21(3):400–403. doi:10.1111/j.1530-0277.1997.tb03782.x
26. Shankar K, Liu X, Singhal R, et al. Chronic ethanol consumption leads to disruption of vitamin D3 homeostasis associated with induction of renal 1,25 dihydroxyvitamin D3-24-hydroxylase (CYP24A1). Endocrinology. 2008;149(4):1748–1756. doi:10.1210/en.2007-0903
27. Zerwekh JE. The measurement of vitamin D: analytical aspects. Ann Clin Biochem. 2004;41(Pt4):272–281. doi:10.1258/0004563041201464
28. Snellman G, Melhus H, Gedeborg R, et al. Determining vitamin D status: a comparison between commercially available assays. PLoS One. 2010;5(7):e11555. doi:10.1371/journal.pone.0011555
29. Rosen CJ. Vitamin D insufficiency. N Engl J Med. 2011;364(3):248–254. doi:10.1056/NEJMcp1009570
30. Manson JE, Brannon PM, Rosen CJ, Taylor CL. Vitamin D deficiency - is there really a pandemic? N Engl J Med. 2016;375(19):1817–1820. doi:10.1056/NEJMp1608005
31. Kobayashi S, Murakami K, Sasaki S, et al. Comparison of relative validity of food group intakes estimated by comprehensive and brief-type self-administered diet history questionnaires against 16 d dietary records in Japanese adults. Public Health Nutr. 2011;14(7):1200–1211. doi:10.1017/S1368980011000504
32. Sasaki S, Yanagibori R, Amano K. Self-administered diet history questionnaire developed for health education: a relative validation of the test-version by comparison with 3-day diet record in women. J Epidemiol. 1998;8(4):203–215. doi:10.2188/jea.8.203
33. Atherton K, Berry DJ, Parsons T, Macfarlane GJ, Power C, Hypponen E. Vitamin D and chronic widespread pain in a white middle-aged British population: evidence from a cross-sectional population survey. Ann Rheum Dis. 2009;68(6):817–822. doi:10.1136/ard.2008.090456
34. McBeth J, Pye SR, O’Neill TW, et al. Musculoskeletal pain is associated with very low levels of vitamin D in men: results from the European male ageing study. Ann Rheum Dis. 2010;69(8):1448–1452. doi:10.1136/ard.2009.116053
35. Wu Z, Malihi Z, Stewart AW, Lawes CM, Scragg R. The association between vitamin D concentration and pain: a systematic review and meta-analysis. Public Health Nutr. 2018;21(11):2022–2037. doi:10.1017/S1368980018000551
36. Hirani V, Blyth FM, Naganathan V, et al. Active vitamin D (1,25 dihydroxyvitamin D) is associated with chronic pain in older Australian men: the concord health and ageing in men project. J Gerontol a Biol Sci Med Sci. 2015;70(3):387–395. doi:10.1093/gerona/glu126
37. Beasley MJ, Macfarlane TV, Macfarlane GJ. Is alcohol consumption related to likelihood of reporting chronic widespread pain in people with stable consumption? Results from UK biobank. Pain. 2016;157(11):2552–2560. doi:10.1097/j.pain.0000000000000675
38. Kim CH, Vincent A, Clauw DJ, et al. Association between alcohol consumption and symptom severity and quality of life in patients with fibromyalgia. Arthritis Res Ther. 2013;15(2):R42. doi:10.1186/ar4200
39. Macfarlane GJ, Beasley M. Alcohol consumption in relation to risk and severity of chronic widespread pain: results from a UK population-based study. Arthritis Care Res (Hoboken). 2015;67(9):1297–1303. doi:10.1002/acr.22604
40. Beasley M, Freidin MB, Basu N, Williams FMK, Macfarlane GJ. What is the effect of alcohol consumption on the risk of chronic widespread pain? A Mendelian randomisation study using UK Biobank. Pain. 2019;160(2):501–507. doi:10.1097/j.pain.0000000000001426
41. Mansfield KE, Sim J, Jordan JL, Jordan KP. A systematic review and meta-analysis of the prevalence of chronic widespread pain in the general population. Pain. 2016;157(1):55–64. doi:10.1097/j.pain.0000000000000314
42. Haugen IK, Magnusson K, Turkiewicz A, Englund M. The prevalence, incidence, and progression of hand osteoarthritis in relation to body mass index, smoking, and alcohol consumption. J Rheumatol. 2017;44(9):1402–1409. doi:10.3899/jrheum.170026
43. Magnusson K, Mathiessen A, Hammer HB, et al. Smoking and alcohol use are associated with structural and inflammatory hand osteoarthritis features. Scand J Rheumatol. 2017;46(5):388–395. doi:10.1080/03009742.2016.1257736
44. Garg MK, Tandon N, Marwaha RK, Menon AS, Mahalle N. The relationship between serum 25-hydroxy vitamin D, parathormone and bone mineral density in Indian population. Clin Endocrinol (Oxf). 2014;80(1):41–46. doi:10.1111/cen.12248
45. Cauley JA, Parimi N, Ensrud KE, et al. Serum 25-hydroxyvitamin D and the risk of hip and nonspine fractures in older men. J Bone Miner Res. 2010;25(3):545–553. doi:10.1359/jbmr.090826
46. Gokcek E, Kaydu A. Assessment of relationship between vitamin D deficiency and pain severity in patients with low back pain: a retrospective, observational study. Anesth Essays Res. 2018;12(3):680–684. doi:10.4103/aer.AER_96_18
47. Pennisi M, Di Bartolo G, Malaguarnera G, Bella R, Lanza G, Malaguarnera M. Vitamin D serum levels in patients with statin-induced musculoskeletal pain. Dis Markers. 2019;2019:3549402. doi:10.1155/2019/3549402
48. Choi M, Park H, Cho S, Lee M. Vitamin D3 supplementation modulates inflammatory responses from the muscle damage induced by high-intensity exercise in SD rats. Cytokine. 2013;63(1):27–35. doi:10.1016/j.cyto.2013.03.018
49. Guida F, Boccella S, Belardo C, et al. Altered gut microbiota and endocannabinoid system tone in vitamin D deficiency-mediated chronic pain. Brain Behav Immun. 2020;85:128–141. doi:10.1016/j.bbi.2019.04.006
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
