Back to Journals » Journal of Pain Research » Volume 17
Association Between Epidural Labor Analgesia and Autism Spectrum Disorder in Offspring: A Systematic Review and Meta-Analysis
Authors Hu X, Wang B, Chen J , Han D, Wu J
Received 28 September 2023
Accepted for publication 21 December 2023
Published 12 January 2024 Volume 2024:17 Pages 227—240
DOI https://doi.org/10.2147/JPR.S442298
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jinlei Li
Xinyue Hu,1– 3 Beibei Wang,1– 3 Jing Chen,1– 3 Dong Han,1– 3 Jing Wu1– 3
1Department of Anesthesiology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, People’s Republic of China; 2Institute of Anesthesia and Critical Care Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, People’s Republic of China; 3Key Laboratory of Anesthesiology and Resuscitation (Huazhong University of Science and Technology), Ministry of Education, Wuhan, People’s Republic of China
Correspondence: Jing Wu, Department of Anesthesiology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, People’s Republic of China, Tel +86-27-85351618, Email [email protected]
Purpose: Many studies have focused on the association between Autism spectrum disorder (ASD) and epidural labor analgesia (ELA), which is the most effective way to manage labor pain. The purpose of this meta-analysis was to summarize the current state of the association between ELA and ASD.
Methods: A search of the literature yielded 201 relevant studies, of which 7 cohort studies met our inclusion criteria. Two independent reviewers screened the inclusion results, extracted data, and assessed the risk of bias and quality of evidence.
Results: Compared to parturient who did not receive ELA, parturient who received ELA had a slightly increased risk of ASD (adjusted hazard ratio [aHR], 1.12; 95% confidence interval [CI], 1.06– 1.17; I2, 69%; P < 0.001; seven studies). After excluding one literature (aHR, 1.09; 95% CI, 1.06– 1.12; I2, 4%; P < 0.001; six studies). The sensitivity analyses had consistent outcomes with the main analyses involving siblings (aHR 1.11; 95% CI 1.03– 1.19), cesarean section and instrumental deliveries (aHR 1.07; 95% CI 1.03– 1.10), non-overlapping populations (aHR 1.09; 95% CI 1.05– 1.12), full-term birth populations (aHR 1.10; 95% CI 1.06– 1.14), and studies assessed to have moderate risk of bias (aHR 1.09; 95% CI 1.02– 1.16).
Conclusion: This meta-analysis revealed a modest positive association between ELA and ASD, acknowledging a slight potential risk. However, it is important to note that this risk cannot be completely dismissed due to the possibility of bias and this association is based on low-quality evidence. Future studies are required to assess and mitigate different confounding biases and investigate the time-dose-response relationship.
Keywords: Autism spectrum disorder, epidural analgesia, epidural labor analgesia, ELA, neuraxial labor analgesia, pregnancy, meta-analysis
Introduction
Autism spectrum disorder (ASD), a neurodevelopmental disorder characterized by social impairment, limitations, and repetitive behaviors, is associated with a variety of genetic and environmental factors.1–3 Among the genetic factors, the overall recurrence rate is approximately 25% between siblings, there is a 50%–80% probability of autism in identical twins, and up to a 30% probability of autism in dizygotic twins.2 The environmental factors involve parental age, maternal status, and toxin exposure. Many studies have focused on the relationship between perinatal events in which both factors coexist and childhood autism. Perinatal events include birth asphyxia, breech or transverse lie, and pre-eclampsia; these perinatal events are potential risks for ASD.4 It has been shown that cesarean delivery might increase the risk of ASD in the offspring.5
Epidural labor analgesia (ELA) is the most effective way to relieve labor pain. To date, several studies have been conducted in the United States, Canada, and Germany to determine the association between ELA and ASD, and confirm whether ELA is a safe option. It has been shown that ELA has no direct effect on neonatal outcomes;6 however, a population-based study including 147,895 live births showed that ELA was associated with a 37% increased risk of ASD in the offspring, which led to widespread debate and concern.7
The purpose of this systematic review and meta-analysis was to determine the relationship between ELA and ASD in offspring by including all available evidence (seven studies through August 2022) and making a primary analysis, and then further conducting a comprehensive meta-analysis with different sensitivity analyses.
Material and Methods
This systematic review and meta-analysis was initiated in August 2022 and strictly followed the principles set out in the Cochrane Handbook. The primary design and protocol of this synthesis were previously described and registered in PROSPERO (CRD42022359029). We reported the evidence selection, quality assessment, evidence synthesis, and research results according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses.8,9
Eligibility Criteria, Databases, Search Strategy, and Study Selection
The eligibility criteria for inclusion were as follows: (1) cohort study; (2) all studies included parturient who had and had not received ELA; and (3) the diagnosis of ASD was reasonably determined.
According to these criteria, a literature search was performed in PubMed, Embase, Scopus and Web of Science databases by the free text and medical subject headings (MeSH in PubMed and Emtree in Embase) for all articles published before August 2022 using the relevant terms “epidural anesthesia, neuraxial labor anesthesia, labor, autism”. In addition, citations from relevant trials and reviews were screened. A total of 201 studies were identified, and ultimately seven cohort studies formed this review.7,10–15
Two authors (Wu and Hu) independently screened titles, abstracts and full-text articles, then reviewed the studies to confirm that the studies fully conformed with the specified inclusion criteria. Discrepancies were resolved by consensus. The remaining search results are shown in a flowchart in Figure 1.
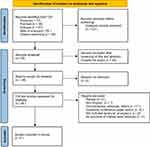 |
Figure 1 PRISMA flowchart. |
Data Extraction, Risk of Bias Assessment, and Quality Assessment
Two authors (Wu and Hu) independently extracted data from the included studies, which were recorded on a data collection form for each study. Extracted study characteristics included study population, study period, data source/classification for ELA, data source/classification for ASD, and outcomes (shown in Table 1).
 |
Table 1 Data Collection Characteristics of Included Studies |
Considering the critical role of genetic and environmental factors in ASD, it was not possible to fully control for confounding bias in the studies. Therefore, studies with the mother’s psychiatric family history, and history of psychotropic drug use as confounding factors were rated as moderate risk and studies without those factors were rated as high risk.
The quality of the selected studies was assessed for risk of bias using the Risk Of Bias In Non-randomized Studies-of Interventions (ROBINS-I) tool, as recommended by the Cochrane Handbook16 (shown in Figure 2).
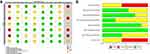 |
Figure 2 (A) Risk of bias assessment of each individual study. (B) Risk of bias assessment of included studies summary. |
In addition, the quality of available evidence was assessed using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach. Discrepancies were resolved by consensus17 (shown in Figure 3).
 |
Figure 3 The GRADE quality of evidence for the primary outcomes. |
Statistical Synthesis and Analysis
Data were statistically synthesized and analyzed using Review Manager (RevMan) [Computer program]. Version 5.4.1, The Cochrane Collaboration, 2020. The dichotomous outcomes of selected studies were analyzed using the hazard ratio (HR) for survival prognosis, quantitative synthesis, and analysis, and presented as forest plots. The results were expressed as the HR and 95% confidence interval (CI), and a p-value < 0.05 was considered a statistically significant difference. The inverse variance method was used to aggregate the data, and a random-effects model was used to calculate the overall effect. The I-squared and p-values of the Cochran’s Q statistic were calculated as a measure of statistical heterogeneity to assess the quality of the pooled results. If an I-squared value was > 50% or a Cochran Q p-value was < 0.10, high heterogeneity was assumed to exist, and further sensitivity analysis was performed to investigate the source of heterogeneity.
Results
Search Results
A total of 201 studies were identified from PubMed, Embase, Scopus, Web of Science, and citations; 123 studies were excluded due to duplication.
Of the remaining 78 studies, 40 were excluded after screening the title and abstract because the scope of the studies was not considered relevant. Two studies were not retrieved. The remaining 36 studies were reviewed for full text, 29 of which were excluded for the following reasons: reviews (n = 3); non-English (n = 1); commentaries, editorials, and letters (n =17); guidelines, conferences, papers, and patents (n =5); ELA was not included (n =2); and no outcomes of interest were obtained (n = 1). Ultimately, seven cohort studies formed the basis of this review and were included in our meta-analysis. The remaining results are shown in a flowchart in Figure 1. The search form is in Supplementary Material 1.
Characteristics of the Included Studies
Seven cohort studies were ultimately included in our meta-analysis. The characteristics of these studies are summarized in Table 1, with sample sizes ranging from 123,175–1,607,579, of whom 1,894,266 and 2,127,140 did and did not receive ELA. Of the seven studies, three were conducted in Canada, two in the United States with overlap populations,7,14 and two studies in Denmark based on cohort studies in the same population.12,13
Risk of Bias Assessment and Quality Assessment
The risk of bias for individual studies is presented in Figure 2A, and the risk of bias summary is presented in Figure 2B. The overall risk of bias was assessed according to the principle of overall bias according ROBIN-I, as follows: if the risk of bias was low in all seven evaluation areas, the overall risk of bias was low; if all seven evaluation areas were low or moderate risk, the overall risk of bias was moderate; and if at least one evaluation area was high risk, but none were extremely high risk, the overall risk of bias was high. Therefore, the overall bias of the seven studies was moderate- or high-risk bias.16
A summary Figure was constructed with the support of GRADEpro software,17 and the GRADE quality of evidence for the primary outcomes is presented in Figure 3. The association between ELA and ASD was graded as low-quality evidence due to the inevitably high risk of bias and inconsistency in each outcome.
Primary Outcomes
Results
The primary analysis aimed to assess the association between ELA and the risk of ASD in offspring, and ultimately included 7 observational studies. Estimates were aggregated using a random-effects model. The results of the meta-analysis suggested that children of mothers who received ELA had a slightly increased risk of ASD. There was a high degree of heterogeneity between studies (aHR, 1.12; 95% CI, 1.06–1.17; I2, 69%; P < 0.0001). The study by Qiu et al7 was subsequently excluded. The results were thus slightly attenuated (aHR, 1.09; 95% CI, 1.06–1.12; I2 = 4%; P < 0.00001) with significantly lower heterogeneity, which confirmed that the study by Qiu et al7 was indeed substantially different from other studies. This might be associated with residual confounding specific to the study population (maternal and family histories of ASD were not considered adequately). The forest plot for this analysis is shown in Figure 4.
Figure 4 Continued. Figure 4 (A) All forest plots for this analysis. (B) All forest plots for this analysis.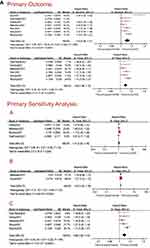

Sensitivity Analysis
After adjusting for potential confounders, the pooled results of seven observational studies showed a high degree of heterogeneity. Thus, it was necessary to perform sensitivity analysis to explore the sources of heterogeneity.
- Studies with sibling analysis (siblings with different exposure status) were selected and the pooled results were consistent (aHR,1.11; 95% CI, 1.03–1.19; I2 = 3%; P = 0.006) with no change in conclusions.
- Studies including cesarean section and instrumental vaginal deliveries were selected. The risk of ASD in children of mothers who received ELA was 8% higher (aHR, 1.07; 95% CI, 1.03–1.10; I2=0%; P = 0.0003), result without major differences.
- The study populations of Qiu et al and Straub et al7,14 overlapped, as did the populations studied by Mikkelsen et al and Ren et al.12,13 Therefore, only studies with a low risk of bias assessed by the ROBINS-I tool were included in the primary outcomes. This sensitivity analysis was finalized for 5 studies, and the pooled study results did not differ significantly (aHR, 1.09; 95% CI, 1.05–1.12; I2 = 19%; P < 0.00001).
- Three studies with full-term birth study populations were selected and assessed as moderate-risk bias (aHR, 1.10; 95% CI, 1.06–1.14; I2 = 47%; P < 0.00001).
- Three studies with a moderate risk of bias assessed by the ROBINS-I tool were selected (aHR, 1.09; 95% CI, 1.02–1.16; I2 = 57%; P = 0.007), and the results were consistent with the primary analysis.
ASD is associated with multiple genetic and environmental factors, therefore we selected other factors from the original study, rather than ELA, as another two groups for sensitivity analysis.
F) Only the study population that received ELA at first birth was not positively associated with the risk of ASD(aHR, 1.06; 95% CI, 0.99–1.12; I2 = 0%; P = 0.09).
G) There were pooled results of studies reporting socioeconomic and demographic factors (aHR, 1.29; 95% CI, 1.23–1.36; I2 = 0%; P < 0.00001). The maternal risk of ASD in the ELA group was 29% compared with that of the non-ELA group, suggesting that autism is a multifactorial disorder, especially with prenatal exposure and genetic factors.
All forest plots for this analysis are shown in Figure 4.
The funnel plots for this analysis are shown in Figure 5 and Supplementary Material 2.
 |
Figure 5 The funnel plot. |
Discussion
A total of 7 cohort studies including 4,021,406 participants were included in which the risk of ASD in offspring of mothers who did and did not receive were compared. Our meta-analysis showed a weak association between exposure to ELA and the risk of ASD in offspring; the association was based on low-quality evidence, though a small increase risk could not be excluded considering the possibility of bias. We should be cautious about the association between LEA and autism, but it does not imply causation.
Analysis Results
With the current systematic review with meta-analysis, there was a modest positive association between ELA and ASD in the offspring, acknowledging a slight potential risk. However, it is important to note that this risk cannot be completely dismissed due to the presence of residual confounding and selection bias.
First, offspring head injuries are common control outcomes in vaccine studies, therefore it may provide some assurance of the validity of the methods. The observational study by Murphy et al15 was the first to use head injuries in offspring as a negative control outcome. Murphy et al15 showed that ELA was not associated with all-cause head injuries in offspring (aHR, 1.01; 95% CI, 1.00–1.02), further confirming the weak link between ELA and ASD should be treated with caution.
Second, whether intrapartum oxytocin has potentially deleterious effects on neurodevelopment in children has been debated for many years because of the possible physiologic effects of oxytocin entering the fetal circulation on brain development.18 Several studies have reported that ELA prolongs the second stage and increases instrumental vaginal delivery and that the combined factors of prolonged labor and assisted labor increase the risk of oxytocin exposure during labor.19,20 Interestingly, we recently found a slight increase in risk between intrapartum oxytocin exposure and ASD with a small gender-specific association, with a modest 13% increase in risk in boys, but no association in girls.21 The original studies included in this meta-analysis also showed that mothers who chose ELA were more likely to have increased use of oxytocin. Due to the lack of original data and studies on the relationships between ELA, increased oxytocin use, and ASD, it was not possible to complete a correlation analysis, but this may become a direction for future research.
Third, maternal health and pregnancy status with ELA are also associated with ASD risk. Previous studies have shown that selective serotonin reuptake inhibitors /serotonin norepinephrine reuptake inhibitors during pregnancy had a weak association with the risk of ASD.22
Fourth, Hanley et al raised questions about the diagnostic accuracy of using International Classification of Diseases, Ninth Revision (ICD-9) codes to identify children with ASD, and argued that there was a misclassification that would bias the results towards no association.23 Therefore, International Statistical Classification of Diseases and Related Health Problems,10th Revision (ICD-10) code was used to identify ASD in an observational study in Denmark, while the authors stated that ICD-10 has a high-positive predictive value for neurodevelopmental disorders in the Danish population.13 In addition, some studies used a comprehensive diagnosis of ASD by multiple physicians and diagnosis codes recorded by health insurance plans, while other studies used an alternative definition of ASD to maximize high-positive outcomes, both of which estimated reduced HR bias.11 It is also important to emphasize that 7 cohort studies included have neglected that patients with borderline and ambiguous ASD symptoms are often overlooked when common symptom diagnoses are used to assess ASD. A recent study showed that the use of health administrative data (including prospectively collected clinical data on both ASD-positive and ASD-negative status and validity measures (reporting negative predictive value and positive predictive value) were more effective in identifying ASD than diagnostic codes.24
Fifth, Lee et al and Kern-Goldberg et al25,26 commented that lacking a discussion of delivery complications. If delivery complications were included, confounding bias might be increased because it is impossible to judge whether the outcome was caused before or after exposure.
Based on the above findings, it is difficult to collect all related factors as covariates so that ascertainment bias cannot be avoided.
To further explore this association, we performed sensitivity analysis and found that five studies showed a slightly increased risk of ASD in offspring of mothers who had received ELA, but we should be cautious about the association between LEA and autism, it does not imply causation. Sensitivity analysis applying sibling-matched designs also balanced possible confounding by unmeasured shared environmental and genetic factors within families, which has an important role in explaining any positive association between ELA and ASD risk. Recently, several studies have reported non-shared environmental factors that have a role in ASD during the perioperative period, such as parental age at birth, maternal prenatal medications, gestational diabetes, eclampsia, uterine bleeding, birth order, cord complications, fetal distress, birth injury, birth trauma, multiple births, maternal hemorrhage, low birth weight, less than gestational age, congenital malformations, and low 5-minute Apgar score.27 The original studies, including the above factors, did not completely circumvent the bias caused by these residual confounders. Additionally, two groups of sensitivity analyses showed that other factors, but not ELA, were associated with the risk of ASD, and both confirmed that parturient who chose ELA have greater access to healthcare, and the evaluation for and diagnosis of ASD. Thus, it is critical to limit the effect of bias when assessing the presence and extent of the ELA-outcome association.
Qiu et al7 overlooked the selection of appropriate covariates in statistical analysis models, which may have contributed to a significant association with a risk up to 37%, as follows: balancing patient-, home-, and hospital-level residual confounding bias; and selection bias.
Outlook
A recent study by Morton et al28 confirmed the association between ELA and intrapartum hyperthermia, and any cause of intrapartum hyperthermia was associated with neonatal brain injury. Interestingly, the most popular mechanistic hypothesis of epidural-related maternal fever (ERMF) is non-infectious inflammation through immunomodulation and cellular injury, which typically occurs during labor.29 Several studies have suggested that oxidative stress, inflammation, immune system dysfunction, and mitochondrial dysfunction may be associated with the pathogenesis and/or severity of ASD;30,31 however, the current research power and evidence are insufficient to prove that ERMF is associated with the risk of ASD in offspring. Nevertheless, this finding provides many ideas for improving ELA during labor because the risk of ASD may be related to the type of epidural catheter, medication regimen, and mode of administration. ELA is still an important bias factor in the perinatal period and a huge gap in the current original studies.
In future corollary studies, all of the evidence should be evaluated and balanced, such as the strength of the ELA-outcome association, the specificity and consistency of sensitivity analysis, the biological rationality, and the time and dose-response relationship.
Limitations
First, the seven original studies initially included in this meta-analysis had high heterogeneity in the combined statistical outcome results, and we applied sensitivity analysis to reduce heterogeneity. By eliminating the article by Qiu et al7 with low-quality bias evaluation, the heterogeneity was greatly reduced (I2 = 69%→4%), thus accounting for the main source of heterogeneity.7 In addition, high heterogeneity may be due to unmeasured confounding bias, ascertainment bias, and unspecified environmental, genetic, socioeconomic, and neurobiological factors, which resulted in study populations that could not be prospectively controlled and evaluated, and outcomes that could not be reliably measured.
Second, the study is currently being conducted only in the United States, Canada, and Germany. The lack of diversity of study sources (lack of Asian populations) may affect the generalizability of results to different populations and clinical settings.
Third, due to the small number of included studies, we did not perform a complex publication bias analysis. Simultaneously, we implemented the exclusion of non-English articles to mitigate potential bias introduced by linguistic variations. However, this measure may have further increased publication bias.
The above reasons may lead to the low quality of the included studies, suggesting that clinical trial design should be more rigorous and cautious to improve the quality of research.
Conclusion
This meta-analysis demonstrated a modest positive association between ELA and ASD, acknowledging a slight potential risk. However, it is important to note that this risk cannot be completely dismissed due to the presence of residual confounding and selection bias, and this association is based on low-quality evidence. It is imperative to reassure the public that no conclusive evidence of ELA causing ASD has been found identified, affirming that ELA remains a safe option during labor. For the majority of women, making a truly informed choice about ELA during labor is not only for themselves, but also for the health of their newborns.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This work was supported by National Natural Science Foundation of China (grant no. 81772131).
Disclosure
The authors report no conflicts of interest in this work.
References
1. First MB. Diagnostic and statistical manual of mental disorders, 5th edition, and clinical utility. J Nerv Ment Dis. 2013;201(9):727–729. doi:10.1097/NMD.0b013e3182a2168a
2. Muhle RA, Reed HE, Stratigos KA, Veenstra-VanderWeele J. The emerging clinical neuroscience of autism spectrum disorder: a review. JAMA Psychiatry. 2018;75(5):514–523. doi:10.1001/jamapsychiatry.2017.4685
3. Ronald A, Hoekstra RA. Autism spectrum disorders and autistic traits: a decade of new twin studies. Am J Med Genet B Neuropsychiatr Genet. 2011;156B(3):255–274. doi:10.1002/ajmg.b.31159
4. Cheng J, Eskenazi B, Widjaja F, Cordero JF, Hendren RL. Improving autism perinatal risk factors: a systematic review. Med Hypotheses. 2019;127:26–33. doi:10.1016/j.mehy.2019.03.012
5. Chien LN, Lin HC, Shao YHJ, Chiou ST, Chiou HY. Risk of autism associated with general anesthesia during cesarean delivery: a population-based birth-cohort analysis. J Autism Develop Disorder. 2015;45(4):932–942. doi:10.1007/s10803-014-2247-y
6. Anim-Somuah M, Smyth RMD, Cyna AM, Cuthbert A. Epidural versus non-epidural or no analgesia for pain management in labour (Review). Cochrane Database Syst Rev. 2018;5. doi:10.1002/14651858.CD000331.pub4
7. Qiu C, Lin JC, Shi JM. Association between epidural analgesia during labor and risk of autism spectrum disorders in offspring. JAMA Pediatr. 2020;174(12):1168. doi:10.1001/jamapediatrics.2020.3231
8. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021:
9. Cumpston M, Li T, Page MJ, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10(10):ED000142. doi:10.1002/14651858.ED000142
10. Wall-Wieler E, Bateman BT, Hanlon-Dearman A, Roos LL, Butwick AJ. Association of epidural labor analgesia with offspring risk of autism spectrum disorders. JAMA Pediatr. 2021;175(7):698. doi:10.1001/jamapediatrics.2021.0376
11. Hanley GE, Bickford C, Ip A. Association of epidural analgesia during labor and delivery with autism spectrum disorder in offspring. JAMA. 2021;326(12):1178. doi:10.1001/jama.2021.14986
12. Mikkelsen AP, Greiber IK, Scheller NM, Lidegaard Ø. Association of labor epidural analgesia with autism spectrum disorder in children. JAMA. 2021;326(12):1170. doi:10.1001/jama.2021.12655
13. Ren T, Zhang J, Yu Y, et al. Association of labour epidural analgesia with neurodevelopmental disorders in offspring: a Danish population-based cohort study. Br J Anaesth. 2022;128(3):513–521. doi:10.1016/j.bja.2021.10.042
14. Straub L, Huybrechts KF, Mogun H, Bateman BT. Association of neuraxial labor analgesia for vaginal childbirth with risk of autism spectrum disorder. JAMA Network Open. 2021;4(12):e2140458. doi:10.1001/jamanetworkopen.2021.40458
15. Murphy MSQ, Ducharme R, Hawken S, et al. Exposure to intrapartum epidural analgesia and risk of autism spectrum disorder in offspring. JAMA Network Open. 2022;5(5):e2214273. doi:10.1001/jamanetworkopen.2022.14273
16. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016:
17. Guyatt G, Oxman AD, Akl EA, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64(4):383–394. doi:10.1016/j.jclinepi.2010.04.026
18. Wahl RUR. Could oxytocin administration during labor contribute to autism and related behavioral disorders?--A look at the literature. Med Hypotheses. 2004;63(3):456–460. doi:10.1016/j.mehy.2004.03.008
19. Cambic CR, Wong CA. Labour analgesia and obstetric outcomes. Br J Anaesth. 2010;105(Suppl 1):i50–i60. doi:10.1093/bja/aeq311
20. Costley PL, East CE. Oxytocin augmentation of labour in women with epidural analgesia for reducing operative deliveries. Cochrane Database Syst Rev. 2013;2013(7):CD009241. doi:10.1002/14651858.CD009241.pub3
21. Weisman O, Agerbo E, Carter CS, et al. Oxytocin-augmented labor and risk for autism in males. Behav Brain Res. 2015;284:207–212. doi:10.1016/j.bbr.2015.02.028
22. Leshem R, Bar-Oz B, Diav-Citrin O, et al. Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin Norepinephrine Reuptake Inhibitors (SNRIs) During Pregnancy and the Risk for Autism spectrum disorder (ASD) and Attention deficit hyperactivity disorder (ADHD) in the Offspring: a True Effect or a Bias? A Systematic Review & Meta-Analysis. Curr Neuropharmacol. 2021;19(6):896–906. doi:10.2174/1570159X19666210303121059
23. Hanley GE, Oberlander TF, Oberlander TF. Epidural analgesia and autism spectrum disorder risk - the challenges inherent in complex observational research. JAMA Pediatr. 2021;175(7):675–677. doi:10.1001/jamapediatrics.2021.0382
24. Bickford CD, Oberlander TF, Lanphear NE, et al. Identification of pediatric autism spectrum disorder cases using health administrative data. Autism Res. 2020;13(3):456–463. doi:10.1002/aur.2252
25. Lee A, Guglielminotti J, Landau R. Methodologic concerns with concluding a link between epidural and autism spectrum disorder. Comment in QIU. JAMA Pediatr. 2021;175(5):535–536. doi:10.1001/jamapediatrics.2020.6686
26. Kern-Goldberger AR, Burris HH, Levine LD. Methodologic concerns with concluding a link between epidural and autism spectrum disorder. JAMA Pediatr. 2021;175(5):536–537. doi:10.1001/jamapediatrics.2020.6692
27. Gardener H, Spiegelman D, Buka SL. Perinatal and neonatal risk factors for autism: a comprehensive meta-analysis. Pediatrics. 2011;128(2):344–355. doi:10.1542/peds.2010-1036
28. Morton S, Kua J, Mullington CJ. Epidural analgesia, intrapartum hyperthermia, and neonatal brain injury: a systematic review and meta-analysis. Br J Anaesth. 2021;126(2):500–515. doi:10.1016/j.bja.2020.09.046
29. Li CJ, Xia F, Xu SQ, Shen XF. Concerned topics of epidural labor analgesia: labor elongation and maternal pyrexia: a systematic review. Chin Med J. 2020;133(5):597–605. doi:10.1097/CM9.0000000000000646
30. Pangrazzi L, Balasco L, Bozzi Y. Oxidative stress and immune system dysfunction in autism spectrum disorders. Int J Mol Sci. 2020;21(9):E3293. doi:10.3390/ijms21093293
31. Rose S, Niyazov DM, Rossignol DA, Goldenthal M, Kahler SG, Frye RE. Clinical and molecular characteristics of mitochondrial dysfunction in autism spectrum disorder. Mol Diagn Ther. 2018;22(5):571–593. doi:10.1007/s40291-018-0352-x
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
