Back to Journals » Neuropsychiatric Disease and Treatment » Volume 15
Association between CD14 rs2569190 C>T polymorphism and ischemic stroke susceptibility: a meta-analysis based on 5,277 subjects
Authors Wu YQ, Cheng SY, Xu XC, Li WC
Received 25 August 2018
Accepted for publication 17 November 2018
Published 21 December 2018 Volume 2019:15 Pages 47—55
DOI https://doi.org/10.2147/NDT.S185313
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Yuping Ning
Yan-Qiong Wu,1,* Shi-Yan Cheng,2,* Xian-Cheng Xu,1 Wen-Cui Li3
1Department of Anesthesiology, Taihe Hospital, Hubei University of Medicine, Shiyan 442000, China; 2Department of Respiratory Medicine, Suizhou Central Hospital, Hubei University of Medicine, Suizhou 441300, China; 3Department of Laboratory Medicine, Taihe Hospital, Hubei University of Medicine, Shiyan 442000, China
*These authors contributed equally to this work
Introduction: Previous epidemiological studies have suggested that CD14 rs2569190 C>T polymorphism plays an important role in ischemic stroke (IS) risk, but the results were inconsistent. Therefore, we conducted a meta-analysis to determine the association between CD14 rs2569190 C>T polymorphism and IS susceptibility.
Methods: Online databases were searched from inception up to July 1, 2018, for studies concerning CD14 rs2569190 C>T polymorphism and its association with IS susceptibility. ORs and corresponding 95% CIs were calculated in the genetic models of each polymorphism locus with Stata Version 14.0. Furthermore, heterogeneity, meta-regression, accumulative analyses, sensitivity analyses, and publication bias were examined.
Results: Overall, 10 observed studies involving 5,277 subjects were included in this meta-analysis on CD14 rs2569190 C>T polymorphism. Generally, no significant associations were found between CD14 rs2569190 C>T polymorphism and IS risk (allele contrast of T vs C: OR =1.03, 95% CI =0.96–1.12, P=0.41, I2=27.8%; co-dominant models of CT vs CC: OR =1.01, 95% CI =0.81–1.25, P=0.95, I2=51.9%; co-dominant models of TT vs CC: OR =1.04, 95% CI =0.89–1.22, P=0.62, I2=25.1%; dominant model of CT + TT vs CC: OR =1.02, 95% CI =0.84–1.25, P=0.82, I2=51.4%; recessive model of TT vs CC + CT: OR =1.07, 95% CI =0.95–1.22, P=0.28, I2=0%), similar to the results in the subgroup analysis.
Conclusion: The current evidence indicated that CD14 rs2569190 C>T polymorphism was not a critical risk factor for IS development.
Keywords: CD14, ischemic stroke, polymorphism
Introduction
Stroke is the second most common cause of mortality, accounting for more than 11.8% of all deaths globally.1 The World Health Organization estimated that stroke affects 9.0 million people and causes 6.15 million deaths worldwide, and the number of deaths is expected to increase to 7.8 million in 2030.2,3 Stroke and its complications, such as hemiplegia, depression, and death, have been considered the sixth common cause of reduced disability-adjusted life years and impose intolerable economic and mental burden on individuals and the society in general.4 Ischemic stroke (IS) is a major type of stroke, accounting for more than 80% of all stroke cases, and its most common pathological presentation is arterial atherosclerosis.5 Multiple factors, such as high blood pressure, intracranial atherosclerosis, dyslipidemia, and cigarette smoking, contribute to the development of IS. However, these factors do not account for all IS cases and the pathogenesis of IS is still unclear.
Recent evidence indicated that multiple pro- and anti-inflammatory factors are involved in the pathogenesis of IS. CD14 is the receptor of bacterial lipopolysaccharide (LPS), which is an important glycoprotein expressed as membrane CD14 on the surface of monocytes, neutrophils, and macrophages and soluble CD14 (sCD14) in the serum.6 CD14 transfers the LPS and other bacterial signals through the LPS-binding protein/CD14/myeloid differentiation factor 2 (MD-2)/Toll-like receptor (TLR) 4 complex.7 This multimolecule complex triggers cascade signal amplification and activates innate host defense mechanisms, thereby promoting the release of cytokines and increase in antigen presentation, which stimulate an immune response.8
Animal research had suggested that low-dose LPS intraperitoneal injection results in IS and systemic inflammatory conditions, aggravates cerebrovascular balance, enhances blood–brain barrier injury and leads to brain edema formation.9,10 Moreover, considerably high levels of plasma sCD14 are reported in patients with IS and closely associated with the risk of death.11,12 CD14 is a 3.9-kb gene located on chromosome 5q23-31, which encodes a 55 kDa glycoprotein with 375 amino acids.13 rs2569190 (C-260T, sometimes referred to as C-159T) is the most common polymorphism locus in the promoter region of the CD14 gene, and this single-nucleotide polymorphism is related to the Sp proteins.14 The T allele of rs2569190 decreases the strength of the bond between the CD14 promoter GC box and Sp consensus sequence, changes the transcriptional capacity, and increases the protein expression level of CD14.15,16 In 2000, Ito et al17 conducted the first case–control study and reported that rs2569190 C>T polymorphism is not associated with IS susceptibility. Since then, other case–control studies on the association between rs2569190 C>T polymorphism and IS susceptibility have been conducted, but the results were inconsistent. Thus, we conducted this meta-analysis on the basis of all published studies to explore a precise assessment of the association between the rs2569190 C>T polymorphism and IS susceptibility.
Methods
This current meta-analysis was conducted according to the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement.18 All collected data were extracted from published studies, and there is no ethical issue.
Search strategy
Three online databases (PubMed, Embase, and Web of Science) were searched for all published case–control studies that focused on the association between rs2569190 C>T polymorphism and IS susceptibility from inception up to July 1, 2018. Only English studies were included. The bibliographies of the included studies were reviewed for the inquiry of some potential studies. The search terms were “((cluster of differentiation 14) OR CD14 OR rs2569190) AND (polymorphism OR variant OR mutation) AND (stroke OR (ischemic stroke) OR (cerebral infarction))”, and the strategy was listed (eg, in PubMed) as follows:
#1 cluster of differentiation 14 | |
#2 CD14 | |
#3 rs2569190 | |
#4 #1 OR #2 OR #3 | |
#5 polymorphism | |
#6 variant | |
#7 mutation | |
#8 #5 OR #6 OR #7 | |
#9 stroke | |
#10 ischemic stroke | |
#11 cerebral infarction | |
#12 #9 OR #10 OR #11 | |
#13 #4 AND #8 AND #12. |
Inclusion criteria
In this meta-analysis, all included studies met the following criteria: 1) case–control studies focused on the association between rs2569190 C>T polymorphism and IS susceptibility, 2) patients with IS were diagnosed with magnetic resonance imaging or computed tomography, 3) there were sufficient data of the genotypes in the case–control groups to evaluate the ORs and 95% CIs, 4) studies were published only in the English language, and 5) duplicate publications or overlapping data were deleted and only the largest or most recently updated data were retained.
Data extraction and quality evaluation
Two investigators (Yan-Qiong Wu and Shi-Yan Cheng) independently reviewed and collected the eligible information from the selected studies, such as surname of the first author, date of publication, country or region, ethnicity, control design, genotyping method, sample sizes of the cases and controls, and frequency data of the genotype distribution. Moreover, the Hardy–Weinberg equilibrium (HWE) and minor allele frequency assessment in the controls were calculated and presented in Table 1. The qualities of all the selected studies were evaluated by the first two authors according to the modified Newcastle–Ottawa scale (NOS).19 The scores ranged from 0 (worst) to 11 points (best) (Table 1), and the studies with the score of 8 or higher were considered as high quality. Potential divergences were solved by discussion with all the authors.
  | Table 1 Scale for quality evaluation |
Statistical analysis
The association between rs2569190 C>T polymorphism and IS susceptibility was evaluated by calculating the pooled ORs and 95% CIs. Heterogeneity between the included studies was first determined with Cochran’s Q test and I2 test. The presence of I2>40% and P<0.1 was considered as an indicator of significant heterogeneity. When P>0.1 or I2≤40%, the fixed-effect model was adopted (the Mantel–Haenszel method), otherwise a random-effects model was applied (I–V heterogeneity method).20,21 Four genetic models were examined, including allele contrast (T vs C), co-dominant models (heterozygote comparison: CT vs CC and homozygote comparison: TT vs CC), dominant model (CT + TT vs CC), and recessive model (TT vs CC + CT). Subgroup analyses were conducted according to HWE status, ethnicity, control design, subject number, and NOS evaluation. Meta-regression was conducted to identify potential factors that contribute to existing heterogeneity. Cumulative meta-analyses and sensitivity analyses were performed to assess the statistical tendency and stability of the results of each study. Egger’s linear regression test and Begg’s funnel plots were used for the identification of publication biases.22 All the statistical outcomes were processed with Stata Version 14.0 (StataCorp LP, College Station, TX, USA). A two-sided P-value <0.05 was considered statistically significant.
Results
Study characteristics
In total, we identified 74 potential case–control studies through a systematic literature search. Figure 1 presents the inclusion procedures of the related studies. A total of 62 studies were excluded. Ten eligible studies involving 2,535 patients and 2,742 controls were included in the meta-analysis.17,23–31 Five studies focus on Asian population,17,26,29–31 and five studies focus on Caucasian population.23–25,27,28 Three studies deviated from the HWE in terms of the genotype distributions in the control groups.25,29,31 Nine studies used PCR-restriction fragment length polymorphism (PCR-RFLP) method, and one study used PCR-fluorescent-labeled oligonucleotide hybridization (PCR-FLOH) method.28 All included characteristics are shown in Table 2.
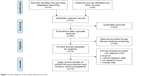  | Figure 1 Flow diagram of the study selection process. |
Quantitative and subgroup analyses
The pooled results on the association between CD14 rs2569190 C>T polymorphism and IS susceptibility are presented in Table 3. Overall, no significant association between the CD14 rs2569190 C>T polymorphism and IS susceptibility was observed in all genetic models (T vs C: OR =1.03, 95% CI =0.96–1.12, P=0.41, I2=27.8%, Figure 2; CT vs CC: OR =1.01, 95% CI =0.81–1.25, P=0.95, I2=51.9%; TT vs CC: OR =1.04, 95% CI =0.89–1.22, P=0.62, I2=25.1%; CT + TT vs CC: OR =1.02, 95% CI =0.84–1.25, P=0.82, I2=51.4%; TT vs CC + CT: OR =1.07, 95% CI =0.95–1.22, P=0.28, I2=0%) (Table 3). In the subsequent analysis, some similar negative associations were demonstrated between CD14 rs2569190 C>T polymorphism and IS susceptibility, such as in the subgroup of the Asian (T vs C: OR =1.01, 95% CI =0.91–1.11, P=0.92, I2=0%; CT vs CC: OR =0.94, 95% CI =0.71–1.23, P=0.65, I2=50.4%; TT vs CC: OR =0.97, 95% CI =0.79–1.19, P=0.80, I2=0%; CT + TT vs CC: OR =0.96, 95% CI =0.82–1.14, P=0.66, I2=37.8%; TT vs CC + CT: OR =1.05, 95% CI =0.90–1.23, P=0.56, I2=0%) and Caucasian population groups (T vs C: OR =1.07, 95% CI =0.85–1.35, P=0.56, I2=52.8%; CT vs CC: OR =1.02, 95% CI =0.75–1.67, P=0.58, I2=59.9%; TT vs CC: OR =1.13, 95% CI =0.75–1.70, P=0.57, I2=50.2%; CT + TT vs CC: OR =1.12, 95% CI =0.75–1.66, P=0.58, I2=63.9%; TT vs CC + CT: OR =1.12, 95% CI =0.90–1.38, P=0.30, I2=0%) (Table 3).
  | Figure 2 OR and 95% CIs of the associations between CD14 rs2569190 C>T polymorphism and ischemic stroke susceptibility in T vs C model. |
Furthermore, heterogeneity was observed in the heterozygote comparison and dominant model. Meta-regression analysis was performed according to the abovementioned stratified factors, but no apparent factor was found to contribute to the existing heterogeneity (for CT vs CC: PHWE=0.63, Pethnicity=0.52, Pcontrol design=0.19, Psubjects=0.23, PNOS=0.86; for CT + TT vs CC: PHWE=0.50, Pethnicity=0.53, Pcontrol design=0.16, Psubjects=0.28, PNOS=0.85). Accumulative (Figure 3 for T vs C model) and sensitive (Figure 4 for T vs C) analyses were conducted based on the published date. No significant fluctuations were found, which indicated that the results of all genetic models were stable and credible.
  | Figure 3 Cumulative meta-analyses according to publication year in T vs C model of CD14 rs2569190 C>T polymorphism. |
  | Figure 4 Sensitivity analysis through deleting each study to reflect the influence of the individual dataset to the pooled ORs in T vs C model of CD14 rs2569190 C>T polymorphism. |
Publication bias was measured and did not present any significant asymmetry in the five funnel plots. These results were confirmed using Egger’s test (T vs C, P=0.91; Figure 5; CT vs CC: P=0.65; TT vs CC, P=0.88; CT + TT vs CC, P=0.81; TT vs CC + CT, P=0.24).
  | Figure 5 Funnel plot analysis to detect publication bias for T vs C model of CD14 rs2569190 C>T polymorphism. |
Discussion
According to the new published report by Wang et al, IS is one of the most important leading causes of morbidity and mortality in China. More than 47 billion China Yuan (CNY) was spent in the treatment of IS, and the average annual growth rate has been 24.96% since 2004.32 IS can lead to serious injury to the nervous system and results in a series of complications. IS has become one of the major public health problems and imposed heavy economic and spiritual burden to families and society.
CD14 has been considered as a major component of LPS receptor complex and known to trigger immune cell recognition along with TLR-4 and MD-2.33 Increased serum CD14 levels have been suggested with the development of IS and contributes to a series of neuroinflammation in cerebral ischemia.34,35 Furthermore, high CD14 levels may be associated with vascular endothelial cell damage, which facilitates atherosclerosis formation and increases the risk of IS subsequently.36,37 rs2569190 C>T polymorphism located at the 5′ untranslated region (UTR) of CD14 gene. The T allele had been proven to be a risk factor for myocardial infarction in European ethnicity,38 suggesting that the T allele can increase the CD14 level by evaluating CD14 gene transcription and expression.39
In 2000, Ito et al17 investigated whether rs2569190 C>T polymorphism contributes to a disposition to IS and did not find any significant association between rs2569190 C>T polymorphism and IS susceptibility. Since then, a number of studies were conducted to evaluate the role of rs2569190 C>T polymorphism in IS events in different ethnicities. Grau et al,23 Banerjee et al,30 and Das et al31 reported a negative association between rs2569190 C>T polymorphism and IS susceptibility. However, Lichy et al25 found that the TT genotype is associated with a risk of the stratified micro- or macroangiopathic IS. In contrast, Cole et al40 suggested that C allele rs2569190 C>T polymorphism increases the risk of IS among smokers (OR =2.05, 95% CI =1.09–3.86).
In epidemiological studies, small sample sizes may contribute to a low statistical power of the results and lead to an inaccurate conclusion. To our knowledge, a meta-analysis is an effective method of combining the quantitative results of previous studies in order to derive a pooled summary conclusion through statistical measures, which can reduce the risk of drawing incorrect conclusions based on small sample sizes. Therefore, we conducted this meta-analysis using eligible case–control studies to explore a more reliable association between rs2569190 C>T polymorphism and IS risk by increasing the sample number of subjects. In this meta-analysis, the overall pooled results revealed that rs2569190 C>T polymorphism had no significant influence on IS risk in all genetic models. Moreover, no significant association between rs2569190 C>T polymorphism and IS risk was found in Asian or Caucasian populations, indicating no significant correlation with ethnicity. In addition, the stratified analyses were conducted on the basis of HWE status, control design, subject number, and NOS evaluation, but no significant results could be found. The possible explanation may be that CD14 rs2569190 C>T polymorphism is not involved in IS susceptibility directly but plays a pathogenic role in synergy with other abnormally expressed proteins or gene polymorphisms.
In 2009, Banerjee41 conducted a meta-analysis on the association between CD14 rs2569190 C>T polymorphism and cerebrovascular diseases but did not conduct a subgroup analysis on IS susceptibility. Thereafter, Misra et al conducted a meta-analysis with only six published case–control studies and failed to find any significant association between CD14 rs2569190 C>T polymorphism and IS susceptibility.42 We conducted this meta-analysis by integrating 10 published case–control studies with a comprehensive research strategy and large sample size. In addition, more rigorous methodology, such as stratified analysis, cumulative analyses, sensitivity analyses, meta-regression, and quality evaluation, was adopted to guarantee the accuracy of all results. However, some limitations in this meta-analysis should be addressed. First, heterogeneity was observed in the heterozygote model and dominant models and meta-regression was conducted but no apparent factor that contributes to the current heterogeneity was found. The heterogeneity can be partly alleviated in the subgroup analysis. Second, only English language studies were included and all included studies were from Asian and Caucasian populations; thus, a population bias may be present and may restrict the application of our conclusion to other races. Finally, these results were conducted on the basis of a single factor (genotype distribution in the case and control groups) and the potential interaction mechanisms with age, sex and other risk factors cannot be interpreted because of insufficient original data.
Conclusion
The evidence obtained suggested that rs2569190 C>T polymorphism may not be an independent risk factor for IS susceptibility. Considering the importance of CD14 in IS development, further larger studies with gene–environment interactions in diverse populations should be conducted to clarify the association between CD14 rs2569190 C>T polymorphism and IS susceptibility.
Disclosure
The authors report no conflicts of interest in this work.
References
Feigin VL, Norrving B, Mensah GA. Global Burden of Stroke. Circ Res. 2017;120(3):439–448. | ||
Norrving B, Kissela B. The global burden of stroke and need for a continuum of care. Neurology. 2013;80(3 Suppl 2):S5–S12. | ||
Writing Group Members, Mozaffarian D, Benjamin EJ, et al. Executive Summary: Heart Disease and Stroke Statistics – 2016 Update: A Report From the American Heart Association. Circulation. 2016;133(4):447–454. | ||
Vijayan M, Reddy PH. Stroke, vascular dementia, and Alzheimer’s disease: Molecular links. J Alzheimers Dis. 2016;54(2):427–443. | ||
Maasz A, Melegh B. Three periods of one and a half decade of ischemic stroke susceptibility gene research: lessons we have learned. Genome Med. 2010;2(9):64. | ||
Zhang DE, Hetherington CJ, Tan S, et al. Sp1 is a critical factor for the monocytic specific expression of human CD14. J Biol Chem. 1994;269(15):11425–11434. | ||
Haziak K, Herman AP, Wojtulewicz K, et al. Effect of CD14/TLR4 antagonist on GnRH/LH secretion in ewe during central inflammation induced by intracerebroventricular administration of LPS. J Anim Sci Biotechnol. 2018;9:52. | ||
Leon CG, Tory R, Jia J, Sivak O, Wasan KM. Discovery and development of toll-like receptor 4 (TLR4) antagonists: a new paradigm for treating sepsis and other diseases. Pharm Res. 2008;25(8):1751–1761. | ||
Doll DN, Hu H, Sun J, et al. Mitochondrial crisis in cerebrovascular endothelial cells opens the blood-brain barrier. Stroke. 2015;46(6):1681–1689. | ||
Dénes A, Ferenczi S, Kovács KJ. Systemic inflammatory challenges compromise survival after experimental stroke via augmenting brain inflammation, blood-brain barrier damage and brain oedema independently of infarct size. J Neuroinflammation. 2011;8:164. | ||
Klimiec E, Pera J, Chrzanowska-Wasko J, et al. Plasma endotoxin activity rises during ischemic stroke and is associated with worse short-term outcome. J Neuroimmunol. 2016;297:76–80. | ||
Klimiec E, Pasinska P, Kowalska K, et al. The association between plasma endotoxin, endotoxin pathway proteins and outcome after ischemic stroke. Atherosclerosis. 2018;269:138–143. | ||
Ulevitch RJ, Tobias PS. Receptor-dependent mechanisms of cell stimulation by bacterial endotoxin. Annu Rev Immunol. 1995;13:437–457. | ||
Jiménez-Sousa MÁ, Liu P, Medrano LM, et al. Association of CD14 rs2569190 polymorphism with mortality in shock septic patients who underwent major cardiac or abdominal surgery: A retrospective study. Sci Rep. 2018;8(1):2698. | ||
Levan TD, Bloom JW, Bailey TJ, et al. A common single nucleotide polymorphism in the CD14 promoter decreases the affinity of Sp protein binding and enhances transcriptional activity. J Immunol. 2001;167(10):5838–5844. | ||
Baldini M, Lohman IC, Halonen M, et al. A Polymorphism* in the 5′ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am J Respir Cell Mol Biol. 1999;20(5):976–983. | ||
Ito D, Murata M, Tanahashi N, et al. Polymorphism in the promoter of lipopolysaccharide receptor CD14 and ischemic cerebrovascular disease. Stroke. 2000;31(11):2661–2664. | ||
Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535. | ||
Niu YM, Weng H, Zhang C, et al. Systematic Review by Multivariate Meta-analyses on the Possible Role of Tumor Necrosis Factor-alpha Gene Polymorphisms in Association with Ischemic Stroke. Neuromolecular Med. 2015;17(4):373–384. | ||
Dersimonian R. Meta-analysis in the design and monitoring of clinical trials. Stat Med. 1996;15(12):1237–1248. | ||
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22(4):719–748. | ||
Hayashino Y, Noguchi Y, Fukui T. Systematic evaluation and comparison of statistical tests for publication bias. J Epidemiol. 2005;15(6):235–243. | ||
Grau AJ, Aulmann M, Lichy C, et al. Increased cytokine release by leucocytes in survivors of stroke at young age. Eur J Clin Invest. 2001;31(11):999–1006. | ||
Zee RY, Bates D, Ridker PM. A prospective evaluation of the CD14 and CD18 gene polymorphisms and risk of stroke. Stroke. 2002;33(4):892–895. | ||
Lichy C, Meiser H, Grond-Ginsbach C, et al. Lipopolysaccharide receptor CD14 polymorphism and risk of stroke in a South-German population. J Neurol. 2002;249(7):821–823. | ||
Park MH, Min JY, Koh SB, et al. Helicobacter pylori infection and the CD14 C(-260)T gene polymorphism in ischemic stroke. Thromb Res. 2006;118(6):671–677. | ||
Lalouschek W, Schillinger M, Hsieh K, et al. Polymorphisms of the inflammatory system and risk of ischemic cerebrovascular events. Clin Chem Lab Med. 2006;44(8):918–923. | ||
Kis Z, Sas K, Gyulai Z, et al. Chronic infections and genetic factors in the development of ischemic stroke. New Microbiol. 2007;30(3):213–220. | ||
Lin TM, Chen CH, Wu HL, Wang CH, Chen YL, Eng HL. The association of C (-260) – >T polymorphism in CD14 promoter and Chlamydia pneumoniae infection in ischemic stroke patients. Am J Clin Pathol. 2008;130(4):595–601. | ||
Banerjee I, Gupta V, Ahmed T, et al. Inflammatory system gene polymorphism and the risk of stroke: a case-control study in an Indian population. Brain Res Bull. 2008;75(1):158–165. | ||
Das S, Kaul S, Jyothy A, Munshi A. Role of TLR4 (C1196T) and CD14 (C-260T) Polymorphisms in Development of Ischemic Stroke, Its Subtypes and Hemorrhagic Stroke. J Mol Neurosci. 2017;63(3–4):300–307. | ||
Wang L, Wang J, Peng B, Xu M. The summary of Chinese Stroke Prevention report 2016. Chin J Cerebrovasc Dis. 2017;14(4):217–224. | ||
Triantafilou K, Triantafilou M, Dedrick RL. A CD14-independent LPS receptor cluster. Nat Immunol. 2001;2(4):338–345. | ||
Zhou M, Wang CM, Yang WL, Wang P. Microglial CD14 activated by iNOS contributes to neuroinflammation in cerebral ischemia. Brain Res. 2013;1506:105–114. | ||
Kaito M, Araya S, Gondo Y, et al. Relevance of distinct monocyte subsets to clinical course of ischemic stroke patients. PLoS One. 2013;8(8):e69409. | ||
Kulkarni M, Bowman E, Gabriel J, et al. Altered Monocyte and Endothelial Cell Adhesion Molecule Expression Is Linked to Vascular Inflammation in Human Immunodeficiency Virus Infection. Open Forum Infect Dis. 2016;3(4):ofw224. | ||
Mendel I, Feige E, Yacov N, et al. VB-201, an oxidized phospholipid small molecule, inhibits CD14- and Toll-like receptor-2-dependent innate cell activation and constrains atherosclerosis. Clin Exp Immunol. 2014;175(1):126–137. | ||
Kane JP, Havel RJ. Polymorphism of the lipopolysaccharide receptor (CD14) and myocardial infarction. New evidence for a role of gram-negative bacterial infection? Circulation. 1999;99(25):3210–3212. | ||
Hubacek JA, Rothe G, Pit’ha J, et al. C(-260) – >T polymorphism in the promoter of the CD14 monocyte receptor gene as a risk factor for myocardial infarction. Circulation. 1999;99(25):3218–3220. | ||
Cole JW, Brown DW, Giles WH, et al. Ischemic stroke risk, smoking, and the genetics of inflammation in a biracial population: the stroke prevention in young women study. Thromb J. 2008;6:11. | ||
Banerjee I. CD14 C260T promoter polymorphism and the risk of cerebrovascular diseases: a meta-analysis. J Appl Genet. 2009;50(2):153–157. | ||
Misra S, Kumar P, Kumar A, et al. Genetic association between inflammatory genes (IL-1α, CD14, LGALS2, PSMA6) and risk of ischemic stroke: A meta-analysis. Meta Gene. 2016;8:21–29. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


