Back to Journals » Vascular Health and Risk Management » Volume 15
Association Between Adherence To The French Dietary Guidelines And Lower Resting Heart Rate, Longer Diastole Duration, And Lower Myocardial Oxygen Consumption. The NUTRIVASC Study
Authors Fysekidis M , Kesse-Guyot E, Valensi P, Arnault N, Galan P, Hercberg S, Cosson E
Received 15 May 2019
Accepted for publication 28 September 2019
Published 30 October 2019 Volume 2019:15 Pages 463—475
DOI https://doi.org/10.2147/VHRM.S215795
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Harry Struijker-Boudier
Marinos Fysekidis,1,2 Emmanuelle Kesse-Guyot,2 Paul Valensi,1 Nathalie Arnault,2 Pilar Galan,2 Serge Hercberg,2 Emmanuel Cosson1,2
1AP-HP, Department of Endocrinology-Diabetology-Nutrition, CRNH-Idf, CINFO, Paris 13 University, Hôpital Jean Verdier, Bondy, France; 2Equipe de Recherche en Epidémiologie Nutritionnelle (EREN), Université Paris 13, Inserm (U1153), Inra (U1125), Centre d’Epidémiologie et Statistiques Paris Cité, Cnam, COMUE Sorbonne-Paris-Cité, Bobigny F-93017, France
Correspondence: Emmanuel Cosson
Department of Endocrinology-Diabetology-Nutrition, Jean Verdier Hospital, avenue du 14 Juillet, Bondy cedex 93143, France
Tel +33 148026580
Fax +33 148026579
Email [email protected]
Background: To investigate whether chronic adherence to the French Nutrition and Health Program (PNNS) guidelines was associated with better cardiovascular health.
Methods: A study nested within the SU.VI.MAX2 cohort was conducted on participants without cardiovascular risk factors. Long-term adherence to the PNNS guidelines was estimated using validated dietary scores from 2007 and 2012. Individuals who did (PNNS+) and did not (PNNS−) continuously adhere to the PNNS guidelines were included. Applanation tonometry, impedance cardiography, laser doppler flowmetry, heart rate, heart rate variability, endothelial function was used for the assessment of cardiovascular health.
Results: A total of 49 subjects (mean age 65.4 ± 5.6 years, 75.5% women) had been included. Those in the PNNS+ group (n=26) were older, had a higher BMI and fat mass than those in the PNNS− group, both groups had similar metabolic parameters. After adjusting for sex, age, and BMI, PNNS+ subjects were found to have a lower heart rate (60.2 ± 8.0 vs 64.3 ± 8.4 beats/min, p=0.042), a lower heart rate × systolic blood pressure product (7166 ± 1323 vs 7788 ± 1680 beats× mmHg/min, p = 0.009), a longer diastole duration (66.7 ± 3.1% vs 64.6 ± 4.1% of the cardiac cycle duration, p=0.049), and a shorter tension–time index (2145 ± 489 vs 2307 ± 428 ms * mmHg, p=0.018) compared to the PNNS− group.
Conclusion: Long-term adherence to the PNNS guidelines had a favorable impact on heart rate, diastole duration, and myocardial oxygen consumption.
Clinical Trial Registration number: NCT01579409.
Keywords: French Health and Nutrition Program, PNNS, heart rate, arterial stiffness, cutaneous blood flow, endothelial function, heart rate variability
Introduction
Cardiac vago-sympathetic activity, arterial stiffness, and endothelial function have been considered as integrators of cardiovascular risk factors given that they reflect the duration and intensity of exposure to such factors. Cardiac vago-sympathetic activity is altered in diabetes, obesity, and hypertension.1 Nutritional factors can acutely activate the autonomous nervous system during food intake through afferent vagal nerve fibers via the release of neuro-endocrine hormones.2 Accordingly, peripheral sympathetic or parasympathetic nervous system activity regulates the function of organs (such as the liver), white and brown fat, muscles, and alpha and beta cells of pancreatic islets, thereby controlling metabolism and energy expenditure and storage.3 Daily dietary habits, such as drinking coffee, have been known to increase vagal nerve activity,4 whereas mixing energy drinks with alcohol can provoke reduced parasympathetic tone.5 Energy expenditure is closely associated with both heart rate (HR)6 and blood pressure (BP).7 Notably, a higher HR has been shown to predict worse cardiovascular outcomes8–10 whereas lower HR has been associated with lower mortality.11 Arterial stiffness is influenced by several modifiable determinants that represent current therapeutic targets: obesity, glucose intolerance, type 1 and 2 diabetes, dyslipidemia, metabolic syndrome,12 and smoking.13 The endothelium plays a major role in vascular tone regulation and atherosclerosis development.14,15 Apart from gender and age,16 components of the metabolic syndrome, such as diabetes,17 hypertension, dyslipidemia, and obesity, have also been associated with endothelial dysfunction.12
Intervention studies involving both primary and secondary prevention have shown the beneficial effect of a Mediterranean diet in reducing the occurrence of major cardiovascular events.18 Nutrition can influence arterial stiffness by as early as age 10,19 while the introduction of healthy nutritional habits early in life can reduce arterial stiffness later during adult life.20 Although impaired endothelial function has been observed with a high-fat diet, improvements therein have been found with the Mediterranean diet.21
The protective effect of antioxidant vitamins against endothelial dysfunction suggests oxidative stress as a potential mechanism.22 Currently, a discrepancy exists between results from short- and long-term intervention studies involving antioxidant treatment for arterial stiffness and oxidative stress markers.23 A study conducted on a sample of over 1000 subjects included in the SU.VI.MAX (Supplémentation en Vitamines et Minéraux Antioxydants) cohort revealed that long-term supplementation with antioxidant vitamins and minerals had no beneficial effect on carotid atherosclerosis and arterial stiffness.24 However, only a few studies compared cardiovascular parameters according to dietary patterns and attempted to determine the mechanisms involved. Most studies presented relatively short-term results in populations that had confounding factors, like cardiovascular risk factors.
The French Health and Nutrition Program [Programme National Nutrition Santé (PNNS)], launched in 2001, has developed nutritional guidelines with the objective of preventing chronic diseases (cardiovascular diseases, cancer, and obesity) through diet and lifestyle counseling.
The main objective of the NUTRIVASC study was to investigate the protective mechanisms of nutrition provided by the long-term adherence to the PNNS guidelines in subjects who have stable dietary habits and no cardiovascular risk factors. We hypothesized that subjects who did not continuously adhere to the PNNS recommendations would have worse cardiovascular health, higher heart rate, greater sympathetic nervous system activity, higher arterial stiffness, and worse endothelial dysfunction compared to those who did.
Subjects And Methods
Study Design
The longitudinal, randomized, double-blind study SU.VI.MAX was conducted between 1994 and 2002.25 Moreover, a total of 6850 agreed to participate in SU.VI.MAX2, a prospective cohort study. Subjects aging 45 to 70 years were evaluated from 2007 to 2009 according to their anthropometric and metabolic criteria and responses to validated food frequency questionnaires.26 The NUTRIVASC study was a registered study nested within the SU.VI.MAX2 cohort (NCT 01579409, French Agency for the Security of Health Products, registration number Afssaps: B111351-70, 08/11/2011 and CNIL: 1552838, 15/12/2011). The study was conducted according to the Declaration of Helsinki of 1975 as revised in 1983, and written informed consent was obtained from all subjects prior to inclusion. Data collection had been completed by August 2016.
Pre-Screening
Among the participants included in the SU.VI.MAX2 cohort, those who had filled out the validated food frequency questionnaire from 2007 to 2009 were 50 to 75 years old and were living within the Paris area were screened. The exclusion criteria included a history of cancer, a history of cardiovascular disease (stroke, myocardial infarction, or amputation), hypertension [systolic blood pressure (SBP) ≥ 140 mmHg, diastolic blood pressure (DBP) ≥ 90 mmHg, or anti-hypertensive treatment]. dyslipidemia (LDL cholesterol > 4.9 mmol/L or lipid-lowering treatment), active smoking or smoking cessation within the last 3 years, known diabetes before inclusion (fasting glycemia ≥ 126 mg/dL or anti-diabetic therapy), fasting hyperglycemia (fasting glycemia ≥ 110 mg/dL), and the presence of a pacemaker.
Screening
The objective of the present study was to compare subjects who adhered to the PNNS recommendations with those who did not. Screening was performed using the 2007–2009 database. Accordingly, the PNNS guideline scores were used after removing the physical activity component in 2007 and 2012 (mPNNS-GS), a method that has been previously validated.27 Nutritional quality scores have been often shown to positively correlate with energy intake.28 Therefore, the probability of adhering to the recommendations increases with high-energy intake, i.e., by eating large quantities of food. To avoid this potential bias, a penalty system had been developed. Energy requirements were calculated using the Schofield equations, which include age, sex, size, and bodyweight of the subject, adjusted for a coefficient of physical activity.29 If the estimated energy intake determined from the food questionnaire was at least 5% higher than that determined from theoretical calculations using the Schofield equation, then the score was penalized as previously detailed.27 The penalized score was an important predictor of weight change within the SU.VI.MAX cohort.30 Accordingly, 28% of the SU.VI.MAX2 cohort had a penalized score.
We preselected subjects who had a 2007–2009 mPNNS-GS that fell between the 1st and 40th (lower adherence to PNNS recommendations) and between the 60th and 100th (higher adherence to PNNS recommendations) percentile.
Selection
In 2012, an information leaflet detailing the protocol, a questionnaire aiming to confirm eligibility (screening criteria), a participation request form, and a new food frequency questionnaire were mailed to the preselected subjects. The 2012 mPNNS-GS was then calculated for patients who satisfied the inclusion and non-inclusion criteria (Figure 1).
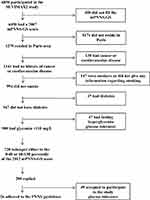 |
Figure 1 The NUTRIVASC study flow chart. |
Finally, individuals who did (exposed) and did not (non-exposed) continuously adhere to the PNNS guidelines were identified. Those who did not adhere to the guidelines (PNNS−) had a 2007 mPNNS-GS between the 1st and 40th percentile and a 2012 mPNNS-GS below the first quartile. Meanwhile, those who adhered to the guidelines (PNNS+) had a 2007 mPNNS-GS between the 60th and 100th percentile and a 2012 mPNNS-GS at the 4th quartile. Once selected subjects were prospectively included in the study.
Physical Activity Questionnaire
Physical activity in 2007 was also assessed using a validated questionnaire. The questionnaire included questions regarding physical activity and duration during leisure and working hours or at home according to intensity (weak, medium, or high), as well as sedentary behavior and duration.31
Cardiovascular Investigations
Tests were performed between September 2012 and December 2013 at the Department of Endocrinology-Diabetology-Nutrition, Jean Verdier Hospital, Bondy, France. The investigator (M.F) was blinded for the group of participants. Participants arrived at 07:30 in the morning after an overnight fast and were instructed to abstain from drinking alcoholic beverages, coffee, or tea on the morning prior to the study and to empty their bladder. Approximately 20 min after catheter insertion on the right arm for drawing blood, measurements were conducted at an ambient temperature of 22 °C to 24 °C in a room where noise and light were kept to a minimum. Participants remained at rest in the supine position throughout the tests. All cardiovascular tests were non-invasive and lasted for approximately 2 hrs (Figure S1). The following tests had been performed.
Cardiovascular Autonomic Nervous System Activity
Finger arterial BP and HR were recorded continuously in the supine position for 6 min at a paced breathing rate of 12 breaths/min using photoplethysmographic sensors and synchronous 6-lead ECG recordings (Task force monitor®, CNSystems Medizintechnik, Austria).
Cardiac autonomic nervous system activity was assessed using spectral analysis of HR and SBP variations.1 The opposite brachial artery was used to calibrate digital arterial BP measurements. Cardiac autonomic activity was expressed as normalized spectral HR variability: i) normalized spectral index [i.e., the percentage of the high-frequency band (HF: 0.15–0.40 Hz/total spectrum) of HR variations (HF-HR: vagal function); ii) the ratio between low frequency (LF: 0.03–0.15 Hz/total spectrum) and HF bands of HR variations (LF/HF-HR: sympathetic–parasympathetic balance), and iii) LF band of the SBP spectrum (LF-SBP: 0.075–0.15 Hz; vascular sympathetic activity).
Hemodynamic Parameters
Transthoracic bioimpedance cardiography (Task Force Monitor®, CNSystems Medizintechnik, Austria) was used for the measurement of the following hemodynamic parameters: stroke volume, cardiac output, total peripheral resistance, ventricular ejection time, left ventricular work index, and thoracic fluid content.
Arterial Stiffness And Central BP
The pulse wave velocity (PWV) was measured as previously described, in the supine position.32 BP was measured just before PWV measurements with an Omron 750 IT BP device. Briefly, PWV was measured as the arterial distance divided by the transit time from the carotid to the femoral artery. The arterial distance was assessed from surface measurements subtracting the sternal notch femoral distance to the carotid-sternal notch distance. Transit time was automatically calculated from the foot of carotid and femoral pulses measured successively by applanation tonometry using R-wave of an ECG as a timing reference. Only measurements with a beat to beat transit time standard deviation below 5% were considered of good quality and used in the study.
Using applanation tonometry on the radial artery (Sphygmocor®, Atcor Medical, Australia), the following measurements had been obtained: (i) radial and central BPs, pulse pressures (PP = SBP − DBP), and PP augmentation ratio; (ii) two arterial stiffness indices [carotid-to-femoral PWV and augmentation index adjusted for a HR of 75 beats/min (AIx@75)]; and (iii) cardiac cycle, systole, and diastole durations based on the BP curve. Myocardial oxygen demand was determined by calculating the double product (HR × central SBP) and the tension–time index (systolic pressure–time integral × HR), whereas myocardial perfusion were determined by calculating the subendocardial viability ratio (diastolic time index/tension–time index).33
Arterial Endothelial Function
Arterial endothelial function and HR were assessed in one of the digital arteries by recording finger arterial pulsatile volume changes for 15 min using the Endopat2000® system (Itamar Medical, Israel), which makes use of plethysmographic biosensors. Reactive hyperhemia was measured after occluding brachial artery blood flow for 5 min. The reactive hyperhemia index represents the post-to-pre occlusion signal ratio on the occluded side normalized to the control side.
Cutaneous Blood Flow Measurement
Cutaneous blood flow (CBF) at rest and during 1 min of paced breathing (6 breaths/min) was measured for 6 min in the forearm using laser Doppler flowmetry (Periflux System 5000® PERIMED, Sweden). CBF fluctuations during the 1 min of paced breathing reflect microvascular autonomic activity. Moreover, CBF decreases during inhalation due to sympathetic nervous system activation. The percentage decrease in the signal wavelet from the zenith to nadir was calculated, and the mean decrease during the three last breaths was used as the index of microvascular sympathetic activity.
Changes in CBF after transcutaneous administration of acetylcholine (Ach) (Miochol-E®, 20 mg of Ach chloride/vial; CHAUVIN®, topical application of 4 mg) through iontophoresis were also measured to explore endothelial function. Ach chloride was diluted in 1 mL of sterile water, after which 200 µL of this solution was placed into a special chamber of a sponge that was used for skin application. After Ach administration, we measured i) maximal CBF, ii) CBF incremental area under the curve 3 min after Ach administration, which is considered an integrator of microvascular endothelial function, iii) delay from Ach administration to maximal CBF (peak delay), iv) peak tangent from basal to maximal CBF (tangent increase represents acceleration in tissue perfusion), and v) vasodilation duration from Ach administration until return to levels at least 20% higher than those for basal CBF.34
Body Composition Analysis
Body composition (lean and fat mass) was determined using bioelectrical impedance (Bodystat® Quadscan, EuroMedix, Germany).
Biochemistry
Blood samples were collected from fasted subjects upon arrival and were stored at −80 °C at the CRB (liver disease biobank), Groupe Hospitalier Paris Seine-Saint-Denis BB-0033-00027. Glucose levels in venous plasma were measured through colorimetry using the glucose oxidase method (Kone Optima, Thermolab System, France). HbA1c measurements were based on the turbidimetric inhibition immunoassay principle (Dimension® technology, Siemens Healthcare Diagnosis Inc., Newark, NJ, USA), while HbA1c levels were standardized according to the Diabetes Control and Complications Trial. Serum fructosamine was measured using the nitroblue tetrazolium colorimetric procedure (COBAS; Roche Diagnostics GmBH, Penzberg, Germany).
Total cholesterol, HDL cholesterol, and triglycerides were measured using enzymatic colorimetry (Hitachi 912, Roche Diagnostic, France), whereas LDL cholesterol was calculated using the Friedwald formula. None of the participants had triglyceride concentrations greater than 400 mg/dL. Laser immunonephelometry (BN100; Dade-Behring) was used to measure the urinary albumin excretion rate from urine specimens.
Sample Size Calculation
To obtain 80% power to detect a 1 m/s difference in PWV and a 1.7 m/s standard deviation between the two groups using Student’s t-test, the number of participants per group was set at 47. Alpha (α) was set at 5%, while, for all other parameters, the sample size yielded 80% power to detect a difference between groups at an effect size (Δ/σ) of about 0.6. Differences in PWV were interpreted according to results from a Finnish study on the consumption of fruits and vegetables during childhood and adolescence in young healthy subjects (age range: 40–45 years). PWV was 0.47 m/s lower among those who consumed fruits and vegetables.20 The non-adherence to the PNNS guidelines had been considered to cause a 1 m/s difference in PWV between both groups. The clinical relevance of this difference in PWV was demonstrated by Blacher et al35 in a population with a very high cardiovascular risk who found that a 1 m/s increase in PWV was consistent with a 14% increase in cardiovascular and total mortality.
Statistical Analyses
All quantitative parameters were summarized according to group and time collected. Descriptive statistics, which included means and standard deviations, was used to describe all quantitative parameters at each time. Qualitative parameters have been presented as frequency distributions. The level of significance was set at 5% for all statistical analyses conducted between both groups.
Relationships between parameters were measured during a fasted state. Analysis of covariance (ANCOVA) was used to compare quantitative parameters between the PNNS+ and PNNS− groups according to their status. Age, BMI, and waist circumference were considered as covariates, with the first model including age and BMI (model 1), the second one including age and waist circumference (model 2), and the third one gender, age, and BMI (model 3).
Results
Study Flow Chart, Demographics, And Biological Parameters Of The Patients
Figure 1 shows the flow chart of the study. A total of 49 subjects were ultimately included, 37 of whom were women. Subject characteristics and a comparison between those who did (PNNS+: n = 26) and did not (PNNS−: n = 23) adhere to the PNNS guidelines are shown in Table 1. Compared to those in the PNNS− group, those in the PNNS+ group were older, had a higher BMI, had greater waist and hip circumferences, and had more fat mass. Other characteristics were similar, including physical activity (evaluated in 2007) and biological parameters (Table 1). Age correlated to heart rate (p=0.014), BMI was not correlated neither with age nor with heart rate (data not shown).
 |
Table 1 Participant Characteristics |
Weight change between 2007 and 2012 was similar in the PNNS− and PNNS+ groups (0.4 ± 3.3 vs −0.2 ± 3.3 kg, p=0.529). Energy and nutrient intake in 2007 and 2012 are shown in Tables S1 (for all participants) and S2 (for each group). Individuals in the PNNS+ group had declared lesser intake of calories, added sugars, and saturated fat and higher intake of vegetable proteins than those in the PNNS− group.
Heart Rate Measurements
HR was lower after adjustment in the PNNS+ group when measured with applanation tonometry on the radial, carotid, and femoral arteries. HR after adjustment was also lower in the PNNS+ group during an average 15 min recording with finger plethysmography (Table 2).
 |
Table 2 Heart Rate Measured In The Radial, Carotid, Femoral Arteries (applanation Tonometry) And Digital Artery (plethysmography) |
Cardiovascular Autonomic Nervous System Activity And Hemodynamic Parameters
No significant difference in any of the cardiovascular autonomic activity indices and parameters provided by cardiac impedancemetry were found between the PNNS− and PNNS+ groups (Table 3).
 |
Table 3 Cardiac Autonomic Nervous System Activity And Cardiac Impedancemetry Measurements |
Central And Peripheral Blood Pressures, Arterial Stiffness, And Cardiac Cycle Parameters
Table 4 shows a comparison of parameters obtained using applanation tonometry. After adjusting for age, BMI and gender (model 3), subjects in the PNNS− group had greater double product, higher tension–time index, lesser percent diastole duration (diastole duration as a percentage of the cardiac cycle), and a trend toward lower subendocardial viability ratio compared to those in PNNS+ group.
 |
Table 4 Peripheral And Central Blood Pressures Measured Through Applanation Tonometry, Indices Of Arterial Stiffness, And Cardiac Cycle Duration |
Measured PWV values were similar in both groups (p=0.522). Age-predicted PWV values in all participants were also analyzed. Given the higher age of our subjects (60–65 years) and the increase in PWV with age, the full quadratic equation would provide better R2 values than a linear fit (0.000 × age + 0.83 × 10–3 × age2 + 5.55).36 Measured values were significantly lower than age-predicted values (p<0.001), with mean predicted values being 9.3 ± 0.6 and 8.9 ± 0.6 m/s for the PNNS+ and PNNS− groups, respectively (PNNS− vs PNNS+, p=0.306) (Figure S2). We also compared predicted PWV values with the direct path length measurements according to the equation from the paper that established the reference values36 and the results remained the same (Table S3).
After adjusting for HR, cardiac cycle duration (p=0.352), diastole duration (p = 0.370), percent diastole duration (p=0.966) and SEVR (p=0.945) were similar in both groups.
Endothelial Function
Endothelial function in small arteries (reactive hyperhemia index) and cutaneous microcirculation (CBF after Ach administration), CBF, and sympathetic microvascular activity were all similar between both groups (Table 5).
 |
Table 5 Endothelial Function In Small Arteries And Laser Doppler Results |
Discussion
The NUTRIVASC study has been the first to extensively examine the effect of long-term adherence to the French nutritional guidelines on cardiovascular risk integrators in patients without cardiovascular risk factors. Compared to participants who did not adhere to the nutritional guidelines, those who did adhere were found to have higher intake of vegetable protein and lesser intake of calories, saturated fat, and added sugar. Despite sharing similar physical activity and metabolic profiles, those who adhered to the guidelines were older and presented worse anthropometric characteristics (i.e., higher BMI, higher waist circumference and higher fat mass) compared to those who did not. After adjustment for these differences, subjects in the PNNS+ group had a lower HR, longer diastole duration, lower HR × BP product, and lower tension–time index. Both groups had similar SBP, arterial stiffness, endothelial function, and vago-sympathetic cardiac activity. These results suggest that aside from the favorable effect on myocardial oxygen consumption as suggested by lower HR x BP product, nutritional adequacy in terms of adherence to the PNNS guidelines could prevent deterioration in cardiovascular risk integrators with age.
Arterial stiffness indices, like the carotid to femoral PWV and digital, radial and aortic AIx, were not affected. AIx measurements remained similar despite differences in age, even after adjusting for a HR of 75 beats/min (AIx@75). A diet favoring meat, alcohol and low micronutrient intake has been associated with increased arterial stiffness.37 On the contrary, a meal rich in ω-3 polyunsaturated fatty acids has been found to acutely reduce arterial stiffness in healthy subjects.38 Nutrient quality, especially in low-glycemic-index diets, have been shown to lower insulin secretion in healthy subjects, while the preventive properties of this type of diet in large cohort studies have shown to decrease the risk of developing diabetes and cardiovascular diseases.39 Notably, the mean measured PWV values in both PNNS+ and PNNS− groups were less than the 10th percentile of reference values (7.9 m/s) obtained from a group of subjects from the general population who had the same age category, optimal or normal BP, and no additional CV risk factors.36 Although the study was designed based on the assumption that adherence to the PNNS guidelines could lead to a 1 m/s difference in PWV between both groups, the actual difference was only 0.36 m/s. Measured PWV values were lower than age predicted ones for both groups (mean PWV difference of 1.8 m/s), probably because we selected patients without any cardiovascular risk factor. This may have attenuated differences in arterial stiffness.
One of the major findings of the present study was that participants who adhered to the PNNS guidelines had lower HR independent of age, with vago-sympathetic cardiac activity and sympathetic vascular activity being similar. Lower HR was confirmed using two different methods, the first of which was applanation tonometry of the radial, carotid and femoral arteries during AIx and PWV measurements. The second method utilized finger plethysmography (average HR recording of 15 min), which evaluated arterial endothelial function. It is important to note that Mediterranean Diet Scores and PNNS-GS scores predicted obesity risk equally well among French adults and that strong adherence to the PNNS guidelines was protective against weight gain and obesity.30 The current results are in line with those of previous studies that reported lower HRs with adherence to the Mediterranean diet.40 However, although the aforementioned trial had used a semi-automatic oscillometer during HR assessment, such a finding has yet to be confirmed in a prospective 5-year diet intervention.41 A resting HR between 50 and 70 beats/min has been associated with lower rates of mortality among the general population.11 However, obtaining the same HR using a β-blocker doubled the mortality,42 suggesting that a naturally occurring slow resting HR was indicative of good overall health. The present study selected subjects who had no confounding factors, like hypertension and anti-hypertensive therapy.
Higher resting HR has been independently associated with arterial stiffness. Accordingly, HR reduction could also slow accelerated age-related endothelial dysfunction through two potential mechanisms: reduction in mechanical stress and prolongation of the period of steady laminar flow.43 In the current study, subjects who adhered to the nutritional guidelines had a longer diastole, which was associated with better myocardial perfusion as suggested by SEVR measurements. This positive association was HR-dependent given that differences between the two groups were no longer significant after adjusting for HR.
Another important finding was that the HR × SBP double product44 and the tension–time index, both hemodynamic determinants of myocardial oxygen demand, were also lower in the PNNS+ group than in the PNNS− group. The presence of similar stroke volume, preload, afterload, and contractility with a lower HR suggested a more efficient myocardial metabolism for achieving similar cardiac workload. These results could not have been attributed to physical activity considering that no difference in physical activity existed between those who did and did not adhere to the PNNS recommendations. Nutritional factors, which could account for the lower HR, remain to be determined. A study on cultured myocardial cells suggested that enrichment with n−3 fatty acids, like docosahexaenoic acid, had a positive influence on β-adrenergic transduction.45 Intrinsic dietary effects on the myocardium have been described to be independent from dietary effects on circulating hormone or substrate levels in murine models.46 In the current study, although no difference had been found between the PNNS+ and PNNS− groups in terms of estimated docosahexaenoic acid intake, the PNNS+ group had a lower saturated fat intake (Table S2). Dietary patterns, such as the Mediterranean diet or the Dietary Approach to Stop Hypertension (DASH) diet have shown greater benefits. Potential health benefits of fruits and vegetables, olive oil, nuts, wine and fiber have been shown by many epidemiological studies. A recent meta-analysis47 showed that a higher intake of fruits or vegetables was inversely associated with the levels of inflammatory biomarkers. Recently, icosapent ethyl, an omega-3 fatty acid found in fish oil, decreased ischemic events and cardiovascular death compared to placebo despite the use of statins.48 Other substances like lycopene, phytosterols and polyphenols have also antioxidant actions.49 Nonetheless, the effects of docosahexaenoic acid intake on HR merit further investigation.
The hemodynamic profile of both groups, which included stroke volume, peripheral resistance, myocardial contractility, left ventricular ejection time and venous return, was also similar. Previous studies have shown that the consumption of high-fat meals with high levels of saturated fat over the course of several weeks may lead to higher total peripheral resistance compared to the consumption low-fat meals.50 BP was measured on the radial artery, while central BP was derived from the generalized transfer function. DBP has been known to decrease during the sixth decade, whereas SBP increases with age, usually leading to increased PP. Moreover, a decrease in PP amplification with age has been due to large artery stiffening.51
The absence of a difference in endothelial function indices, measured either at the digital artery level or using CBF at rest and after Ach iontophoresis, is also worth considering. The role of nutrients in endothelial function has been thoroughly studied: intra-arterial glucose infusion impairs endothelium-dependent vasodilation in healthy humans in vivo,52 transient hypertriglyceridemia decreases brachial artery reactivity, presumably through both endothelium-dependent and endothelium-independent mechanisms,53 while intravenous infusion of
Our study has limitations and strengths. We included a smaller sample size than initially designed which resulted in a loss of statistical power. One of the limitations of questionnaires is that unanswered answers were not assessed and the inevitable of forgetfulness by interviewed subjects. The strengths of the present study were that participants were tested for their adherence to nutritional guidelines at two time points over a 5-year period and were selected because their evaluations were concordant throughout the study period. Moreover, to avoid high-energy intake bias, a penalty system was used when both groups were selected. Both groups had no cardiovascular confounders given that they were free of cardiovascular risk factors and did not take any drug that could modify cardiovascular measurements. After considering anthropometric measurements and metabolic profiles, both groups were found to have probably shared a healthy way of living prior to study participation, thus increasing selection accuracy based on the mPNNS-GS. Finally, a follow-up evaluation of physical activity level in 2012 would have been useful. Pronounced changes in physical activity would have been unlikely considering that practically no weight change had been observed over a 5-year period. However, it is known that prolonged modest changes in physical fitness in individuals can have important effects on cardiovascular health not mediated by changes in BMI. We also compared the indices for physical activity from 2001 to 2007 (data not shown) during leisure (p=0.510), working hours (p=0.059, tendency to decrease) and at home (p=0.250). Our subjects had a mean age of 65.4 ± 5.6 years, and at this age were less likely to start a physical activity and more likely to decrease their physical activity.56 We also observed that the weight change (data not shown) from 2001 to 2012 was not significant (0.655 ± 3.9 kg, p=0.256).
In conclusion the NUTRIVASC study suggests that adherence to the French nutritional guidelines has a positive impact on resting HR and myocardial perfusion indices. Beneficial effects of adhering to nutritional guidelines also encompass delayed vascular aging and decreased myocardial oxygen consumption. Molecular pathways of nutritional intervention need to be studied, since information about possible mechanisms implicated in the cardioprotective effect of diet patterns, nutrients or bioactive compounds is lacking.
These results not only describe a mechanism whereby nutrition protects against cardiovascular diseases but also underline the importance of programs geared toward preventing cardiovascular diseases as recommended in the PNNS. Future randomized controlled studies in order to confirm the results of the current study are necessary.
Abbreviations
Ach, acetylcholine; AIx@75, augmentation index adjusted for a heart rate of 75 beats/min; BMI, body mass index; BP, blood pressure; CBF, cutaneous blood flow; DBP, diastolic blood pressure; HF nu-RRI, high-frequency band of the heart rate spectrum in normalized units expressed as a percentage of the total spectrum; HF-HR, High-frequency band of heart rate variations; HR, heart rate; LF/HF-HR, ratio between the low- and high-frequency bands of heart rate variations; LF-SBP, low-frequency band of systolic blood pressure spectrum; PNNS, Programme National Nutrition Santé (French Nutrition and Health Program); PNNS+ and PNNS−, individuals who did (PNNS+) and did not (PNNS−) continuously adhere to the “Programme National Nutrition Santé” guidelines; mPNNS-GS, validated dietary score reflecting the adherence to the PNNS guidelines after removing the physical activity component in 2007 and 2012; PP, pulse pressure; PWV, carotid-to-femoral pulse wave velocity; SBP, systolic blood pressure; SU.VI.MAX, Supplémentation en Vitamines et Minéraux Antioxydants.
Data Sharing Statement
Data in the current study are property of the French institute for medical research (Inserm) any requests for data sharing or other study-related documents should be addressed to Professor Serge Hercberg ([email protected]).
Acknowledgments
We would like to thank INSERM (Institut national de la santé et de la recherché médicale) for promoting this work. We would also like to thank for the storage of our samples CRB (CRB (liver disease biobank), Groupe Hospitalier Paris Seine-Saint-Denis BB-0033-00027). We would like to thank the University of Paris 13 for a research grant (Bonus Qualité Recherche, faculty decision - 26/01/2010).
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bernardi L, Spallone V, Stevens M, et al. Investigation methods for cardiac autonomic function in human research studies. Diabetes Metab Res Rev. 2011. doi:10.1002/dmrr.1224
2. Williams EK, Chang RB, Strochlic DE, Umans BD, Lowell BB, Liberles SD. Sensory neurons that detect stretch and nutrients in the digestive system. Cell. 2016;166:209–221. Epub May 26. doi:10.1016/j.cell.2016.05.011
3. Thorens B. Sensing of glucose in the brain. Handb Exp Pharmacol. 2012;277–294. doi:10.1007/978-3-642-24716-3_12
4. Hibino G, Moritani T, Kawada T, Fushiki T. Caffeine enhances modulation of parasympathetic nerve activity in humans: quantification using power spectral analysis. J Nutr. 1997;127:1422–1427. doi:10.1093/jn/127.7.1422
5. Wiklund U, Karlsson M, Ostrom M, Messner T. Influence of energy drinks and alcohol on post-exercise heart rate recovery and heart rate variability. Clin Physiol Funct Imaging. 2009;29:74–80. doi:10.1111/j.1475-097X.2008.00837.x
6. Spurr GB, Prentice AM, Murgatroyd PR, Goldberg GR, Reina JC, Christman NT. Energy expenditure from minute-by-minute heart-rate recording: comparison with indirect calorimetry. Am J Clin Nutr. 1988;48:552–559. doi:10.1093/ajcn/48.3.552
7. Wareham NJ, Wong M-Y, Hennings S, et al. Quantifying the association between habitual energy expenditure and blood pressure. Int J Epidemiol. 2000;29:655–660. doi:10.1093/ije/29.4.655
8. Jouven X, Empana JP, Schwartz PJ, Desnos M, Courbon D, Ducimetiere P. Heart-rate profile during exercise as a predictor of sudden death. N Engl J Med. 2005;352:1951–1958. doi:10.1056/NEJMoa043012
9. Palatini P. Heart rate and the cardiometabolic risk. Curr Hypertens Rep. 2013;15:253–259. doi:10.1007/s11906-013-0342-7
10. Vazir A, Claggett B, Cheng S, et al. Association of resting heart rate and temporal changes in heart rate with outcomes in participants of the atherosclerosis risk in communities study. JAMA Cardiol. 2018;3:200–206. doi:10.1001/jamacardio.2017.4974
11. Kannel WB, Kannel C, Paffenbarger RS
12. Czernichow S, Greenfield JR, Galan P, et al. Macrovascular and microvascular dysfunction in the metabolic syndrome. Hypertens Res. 2010. doi:10.1038/hr.2009.228
13. Kool MJ, Hoeks AP, Struijker Boudier HA, Reneman RS, Van Bortel LM. Short- and long-term effects of smoking on arterial wall properties in habitual smokers. J Am Coll Cardiol. 1993;22:1881–1886. doi:10.1016/0735-1097(93)90773-t
14. Verma S, Buchanan MR, Anderson TJ. Endothelial function testing as a biomarker of vascular disease. Circulation. 2003;108:2054–2059. doi:10.1161/01.CIR.0000089191.72957.ED
15. Lerman A, Zeiher AM. Endothelial function: cardiac events. Circulation. 2005;111:363–368. doi:10.1161/01.CIR.0000153339.27064.14
16. Celermajer DS, Sorensen KE, Spiegelhalter DJ, Georgakopoulos D, Robinson J, Deanfield JE. Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol. 1994;24:471–476. doi:10.1016/0735-1097(94)90305-0
17. Antony I, Nitenberg A, Foult JM, Aptecar E. Coronary vasodilator reserve in untreated and treated hypertensive patients with and without left ventricular hypertrophy. J Am Coll Cardiol. 1993;22:514–520. doi:10.1016/0735-1097(93)90058-9
18. Liyanage T, Ninomiya T, Wang A, et al. Effects of the mediterranean diet on cardiovascular outcomes—a systematic review and meta-analysis. PLoS One. 2016;11:e0159252. doi:10.1371/journal.pone.0159252
19. Schack-Nielsen L, Molgaard C, Larsen D, Martyn C, Michaelsen KF. Arterial stiffness in 10-year-old children: current and early determinants. Br J Nutr. 2005;94:1004–1011. doi:10.1079/bjn20051518
20. Aatola H, Koivistoinen T, Hutri-Kähönen N, et al. Lifetime fruit and vegetable consumption and arterial pulse wave velocity in adulthood: the cardiovascular risk in young finns study. Circulation. 2010;122:2521–2528. doi:10.1161/CIRCULATIONAHA.110.969279
21. Davis N, Katz S, Wylie-Rosett J. The effect of diet on endothelial function. Cardiol Rev. 2007;15:62–66. doi:10.1097/01.crd.0000218824.79018.cd
22. Title LM, Cummings PM, Giddens K, Nassar BA. Oral glucose loading acutely attenuates endothelium-dependent vasodilation in healthy adults without diabetes: an effect prevented by vitamins C and E. J Am Coll Cardiol. 2000;36:2185–2191. doi:10.1016/s0735-1097(00)00980-3
23. Plantinga Y, Ghiadoni L, Magagna A, et al. Supplementation with vitamins C and E improves arterial stiffness and endothelial function in essential hypertensive patients. Am J Hypertens. 2007;20:392–397. doi:10.1016/j.amjhyper.2006.09.021
24. Zureik M, Galan P, Bertrais S, et al. Effects of long-term daily low-dose supplementation with antioxidant vitamins and minerals on structure and function of large arteries. Arterioscler Thromb Vasc Biol. 2004;24:1485–1491. doi:10.1161/01.ATV.0000136648.62973.c8
25. Hercberg S, Galan P, Preziosi P, et al. The SU.VI.MAX study: a randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch Intern Med. 2004;164:2335–2342. doi:10.1001/archinte.164.21.2335
26. Kesse-Guyot E, Amieva H, Castetbon K, et al. Adherence to nutritional recommendations and subsequent cognitive performance: findings from the prospective supplementation with antioxidant vitamins and minerals 2 (SU.VI.MAX 2) Study. Am J Clin Nutr. 2011;93:200–210. doi:10.3945/ajcn.2010.29761
27. Estaquio C, Kesse-Guyot E, Deschamps V, et al. Adherence to the French Programme National Nutrition Sante Guideline Score is associated with better nutrient intake and nutritional status. J Am Diet Assoc. 2009;109:1031–1041. doi:10.1016/j.jada.2009.03.012
28. Waijers PMCM, Feskens EJM, Ocké MC. A critical review of predefined diet quality scores. BJN. 2007;97:219. doi:10.1017/S0007114507250421
29. Schofield WN. Predicting basal metabolic rate, new standards and review of previous work. Hum Nutr Clin Nutr. 1985;39(Suppl 1):5–41.
30. Lassale C, Fezeu L, Andreeva VA, et al. Association between dietary scores and 13-year weight change and obesity risk in a French prospective cohort. Int J Obes (Lond). 2012;36:1455–1462. doi:10.1038/ijo.2011.264
31. Charreire H, Kesse-Guyot E, Bertrais S, et al. Associations between dietary patterns, physical activity (leisure-time and occupational) and television viewing in middle-aged French adults. Br J Nutr. 2011;105:902–910. Epub 2011 Jan 21. doi:10.1017/S000711451000440X
32. Chen Q, Chiheb S, Fysekidis M, et al. Arterial stiffness is elevated in normotensive type 2 diabetic patients with peripheral neuropathy. Nutr Metab Cardiovasc Dis. 2015;25:1041–1049. doi:10.1016/j.numecd.2015.08.001
33. Crilly M, Coch C, Bruce M, Clark H, Williams D. Indices of cardiovascular function derived from peripheral pulse wave analysis using radial applanation tonometry: a measurement repeatability study. Vasc Med. 2007;12:189–197. doi:10.1177/1358863X07081134
34. Fysekidis M, Cosson E, Takbou K, et al. Effects of insulin analogs as an add-on to metformin on cutaneous microcirculation in type 2 diabetic patients. Microvasc Res. 2017;116:6–14. doi:10.1016/j.mvr.2017.09.005
35. Blacher J, Safar ME, Guerin AP, Pannier B, Marchais SJ, London GM. Aortic pulse wave velocity index and mortality in end-stage renal disease. Kidney Int. 2003;63:1852–1860. doi:10.1046/j.1523-1755.2003.00932.x
36. Boutouyrie P, Vermeersch SJ. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: establishing normal and reference values. Eur Heart J. 2010;31:2338–2350. doi:10.1093/eurheartj/ehq165
37. Kesse-Guyot E, Vergnaud A-C, Fezeu L, et al. Associations between dietary patterns and arterial stiffness, carotid artery intima-media thickness and atherosclerosis. Eur J Cardiovasc Prev Rehabil. 2010;17:718–724. doi:10.1097/HJR.0b013e32833a197f
38. Chong MF, Lockyer S, Saunders CJ, Lovegrove JA. Long chain n-3 PUFA-rich meal reduced postprandial measures of arterial stiffness. Clin Nutr. 2010. doi:10.1016/j.clnu.2010.02.001
39. Jenkins DJ, Kendall CW, Augustin LS, et al. Glycemic index: overview of implications in health and disease. Am J Clin Nutr. 2002;76:266S–73S. doi:10.1093/ajcn/76/1.266S
40. Garcia-Lopez M, Martinez-Gonzalez MA, Basterra-Gortari FJ, Barrio-Lopez MT, Gea A, Beunza JJ. Adherence to the Mediterranean dietary pattern and heart rate in the SUN project. Eur J Prev Cardiol. 2014;21:521–527. doi:10.1177/2047487312467871
41. Garcia-Lopez M, Toledo E, Beunza JJ, et al. Mediterranean diet and heart rate: the PREDIMED randomised trial. Int J Cardiol. 2014;171:299–301. doi:10.1016/j.ijcard.2013.11.074
42. Bangalore S, Messerli FH, Ou FS, et al. The association of admission heart rate and in-hospital cardiovascular events in patients with non-ST-segment elevation acute coronary syndromes: results from 135 164 patients in the CRUSADE quality improvement initiative. Eur Heart J. 2010;31:552–560. doi:10.1093/eurheartj/ehp397
43. Thorin E, Thorin-Trescases N. Vascular endothelial ageing, heartbeat after heartbeat. Cardiovasc Res. 2009;84:24–32. doi:10.1093/cvr/cvp236
44. Nelson RR, Gobel FL, Jorgensen CR, Wang K, Wang Y, Taylor HL. Hemodynamic predictors of myocardial oxygen consumption during static and dynamic exercise. Circulation. 1974;50:1179–1189. doi:10.1161/01.cir.50.6.1179
45. Grynberg A, Fournier A, Sergiel JP, Athias P. Effect of docosahexaenoic acid and eicosapentaenoic acid in the phospholipids of rat heart muscle cells on adrenoceptor responsiveness and mechanism. J Mol Cell Cardiol. 1995;27:2507–2520. doi:10.1006/jmcc.1995.0238
46. Liu J, Wang P, Douglas SL, Tate JM, Sham S, Lloyd SG. Impact of high-fat, low-carbohydrate diet on myocardial substrate oxidation, insulin sensitivity, and cardiac function after ischemia-reperfusion. Am J Physiol Heart Circ Physiol. 2016;311:H1–H10. doi:10.1152/ajpheart.00809.2015
47. Hosseini B, Berthon BS, Saedisomeolia A, et al. Effects of fruit and vegetable consumption on inflammatory biomarkers and immune cell populations: a systematic literature review and meta-analysis. Am J Clin Nutr. 2018;108:136–155. doi:10.1093/ajcn/nqy082
48. Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380:11–22. doi:10.1056/NEJMoa1812792
49. Casas R, Castro-Barquero S, Estruch R, Sacanella E. Nutrition and cardiovascular health. Int J Mol Sci. 2018;19(12). doi:10.3390/ijms19123988
50. Rontoyanni VG, Hall WL, Pombo-Rodrigues S, Appleton A, Chung R, Sanders TA. A comparison of the changes in cardiac output and systemic vascular resistance during exercise following high-fat meals containing DHA or EPA. Br J Nutr. 2012;108:492–499. doi:10.1017/S0007114511005721
51. Franklin SS, Gustin W, Wong ND, et al. Hemodynamic patterns of age-related changes in blood pressure. The Framingham Heart Study. Circulation. 1997;96:308–315. doi:10.1161/01.cir.96.1.308
52. Williams SB, Goldfine AB, Timimi FK, et al. Acute hyperglycemia attenuates endothelium-dependent vasodilation in humans in vivo. Circulation. 1998;97:1695–1701. doi:10.1161/01.cir.97.17.1695
53. Lundman P, Eriksson M, Schenck-Gustafsson K, Karpe F, Tornvall P. Transient triglyceridemia decreases vascular reactivity in young, healthy men without risk factors for coronary heart disease. Circulation. 1997;96:3266–3268. doi:10.1161/01.cir.96.10.3266
54. Piatti P, Fragasso G, Monti LD, et al. Acute intravenous L-arginine infusion decreases endothelin-1 levels and improves endothelial function in patients with angina pectoris and normal coronary arteriograms: correlation with asymmetric dimethylarginine levels. Circulation. 2003;107:429–436. doi:10.1161/01.cir.0000046489.24563.79
55. Bjelakovic G, Nikolova D, Gluud C. Antioxidant supplements and mortality. Curr Opin Clin Nutr Metab Care. 2014;17:40–44. doi:10.1097/MCO.0000000000000009
56. Sun F, Norman IJ, While AE. Physical activity in older people: a systematic review. BMC Public Health. 2013;13:449. doi:10.1186/1471-2458-13-449
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
