Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Asn563Ser polymorphism of CD31/PECAM-1 is associated with atherosclerotic cerebral infarction in a southern Han population
Authors Song Y, Li Q, Long L, Zhang N, Liu Y
Received 28 September 2014
Accepted for publication 11 November 2014
Published 18 December 2014 Volume 2015:11 Pages 15—20
DOI https://doi.org/10.2147/NDT.S75065
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Wai Kwong Tang
Yanmin Song, Qunfang Li, Lili Long, Ning Zhang, Yunhai Liu
Department of Neurology, Xiangya Hospital, Central South University, Changsha, Hunan Province, People’s Republic of China
Background: CD31, also called platelet endothelial cell adhesion molecule-1 (PECAM-1), is thought to play a role in the pathological mechanisms of atherosclerosis. Leu125Val polymorphism and elevated plasma levels of soluble PECAM-1 (sPECAM-1) were found to be associated with cerebral infarction. Our aim was to investigate the association between the Asn563Ser polymorphism of CD31/PECAM-1, plasma level of sPECAM-1, and the risk of atherosclerotic cerebral infarction (ACI) in the southern Han population of the People’s Republic of China.
Subjects and methods: A total of 147 subjects with ACI and 114 controls were enrolled in the study. The Asn563Ser CD31/PECAM-1 polymorphism was detected using the polymerase chain reaction–restriction fragment length polymorphism method. The plasma spECAM-1 level was measured using the enzyme-linked immunosorbent assay method.
Results: In this study, statistically significant differences in Asn563Ser genotype and allele distribution were found between the cases and controls (P<0.05). Furthermore, logistic regression analysis showed that the GG genotype is associated with increase in ACI risk (odds ratio =4.862, P<0.001). The plasma level of sPECAM-1 was associated with ACI (odds ratio =1.431, P=0.038). In both the ACI and the control groups, the plasma sPECAM-1 level in subjects with the GG genotype was higher than that in subjects carrying the AA or GA genotype (P<0.05).
Conclusion: Our study showed that the Asn563Ser polymorphism of CD31/PECAM-1 gene and elevated plasma sPECAM-1 level are related to ACI risk in the southern Han population of People’s Republic of China.
Keywords: genetic polymorphism, CD31, platelet endothelial cell adhesion molecule-1 (PECAM-1)
Introduction
Atherosclerosis is the principal pathological basis of cerebral infarction. The inflammatory response may play a vital role in the pathological mechanisms of atherosclerosis and cerebral vascular disease.1,2 Cell adhesion molecules mediate inflammatory cell aggregation and adhesion to endothelial cells, which are important initial steps in the development of atherosclerosis.3,4 CD31, also called platelet endothelial cell adhesion molecule-1 (PECAM-1), as a cell adhesion molecule, is mainly expressed on the surface of endothelial cells, circulating platelets, monocytes, neutrophils, and certain T-cell subsets.5,6 CD31/PECAM-1 has been shown to play dual roles (proinflammatory or anti-inflammatory) in the inflammatory response. It can mediate cell adhesion through the upregulation of integrin and plays important roles in the transendothelial migration of leukocytes, the inhibition of T-cell-mediated signal transduction, and the regulation of platelet function.7–10 On the other hand, it can exert anti-inflammatory effects by increasing the threshold for leukocyte activation, suppressing the generation of proinflammatory cytokines, and maintaining the integrity of the vascular barrier.10
Three common polymorphisms (Leu125Val, Asn563Ser, and Gly670Arg) are located on the CD31/PECAM-1 gene.11 These CD31/PECAM-1 polymorphisms have been deemed to be associated with myocardial infarction or coronary artery disease (CAD).12–15 However, only one study16 to date has focused on the relationship between CD31/PECAM-1 polymorphisms and cerebral infarction, and it found that Leu125Val polymorphism and plasma sPECAM-1 levels are associated with cerebral infarction.
We conducted a case–control study by genotyping the Asn563Ser polymorphism and testing the plasma sPECAM-1 levels in southern Han patients with ACI from the People’s Republic of China to determine whether the Asn563Ser genotype is associated with elevated plasma sPECAM-1 levels and ACI risk.
Materials and methods
Study populations
We enrolled acute ACI patients who were hospitalized in the Department of Neurology, Xiangya Hospital, Central South University, People’s Republic of China, continuously between September 2007 and February 2008. A total of 147 cases (87 males and 60 females; mean age: 62.61±8.51 years) were enrolled in this study. All subjects were examined by computed tomography (CT) and/or magnetic resonance imaging (MRI). The ACI diagnostic criteria were based on the criteria of the Fourth National Cerebrovascular Disease Conference of China.17 The exclusion criteria were as follows: cerebral infarction caused by cardiogenic factors; arthritis, blood diseases, tumor, or cerebrovascular malformation; hemorrhage after cerebral infarction; the use of contraceptives; severe liver and kidney disease; disseminated intravascular coagulation; autoimmune diseases; and pregnancy.
A total of 114 healthy controls (66 males and 48 females; average age: 61.04±7.75 years) were recruited from the Health Examination Center of the Xiangya Hospital during the same period. CT and/or MRI was conducted to exclude cerebral infarction. The exclusion criteria were as follows: stroke history or family history of stroke; thrombosis history; the use of oral contraceptives or anticoagulants; severe liver and kidney disease; disseminated intravascular coagulation; autoimmune diseases; and pregnancy.
All subjects were nonconsanguineous. This study was approved by the Ethics Committee of the Xiangya Hospital, and all subjects signed a written informed consent.
DNA extractions and blood biochemistry tests
A total of 10 mL of peripheral venous blood was collected into ethylenediamine tetraacetic acid (EDTA)-containing tubes for each subject; 5 mL of peripheral blood was used to extract genomic DNA using the traditional phenol–chloroform method, and another 5 mL was used for biochemistry tests (fasting blood sugar and blood lipids) and the measurement of the plasma levels of sPECAM-1. The Human sPECAM-1 Instant ELISA Kit (Bender MedSystems GmbH, Vienna, Austria) was used for determination of the plasma level of sPECAM-1 by enzyme-linked immunosorbent assay according to the manufacturer’s instructions.
Primer designs and polymerase chain reaction–restriction fragment length polymorphism
The primers for Asn563Ser were designed according to Wenzel et al18 and were synthesized by Invitrogen (Shanghai, People’s Republic of China). The primer sequences were as follows: 5′–TGTGAATCCTTTTCCTGCTTTTC–3′ (upstream primer); and 5′–GCTTGCTATGGAGACCCTGAC–3′ (downstream primer). The polymerase chain reaction (PCR) was performed as follows: predenaturation at 94°C for 5 minutes; followed by 35 cycles of amplification comprising denaturation at 94°C for 20 seconds, annealing at 55°C for 35 seconds, and then extension at 72°C for 35 seconds; with a final extension at 72°C for 10 minutes.
A variant of G→A (Asn→Ser) can lead to the appearance of the Nhe I restriction site. PCR amplification products were digested by restriction endonuclease Nhe I (Thermo Fisher Scientific, Waltham, MA, USA) and detected by electrophoresis on 2% agarose gel with 0.5× Tris–borate–EDTA buffer (115 V, 30–40 minutes). After electrophoresis, an ultraviolet light–based gel-imaging system (Tanon Science & Technology Co, Shanghai, People’s Republic of China) was used to observe and photograph the bands. The detection of Asn563Ser genotype was as follows: wild-type homozygotes had only one 81-bp band; heterozygotes had three fragments with lengths of 338 bp, 217 bp, and 121 bp; and the mutant homozygotes had only one fragment with a length of 180 bp.
Statistical analysis
Statistical analyses were performed with SPSS version 18.0 (SPSS Inc, Chicago, IL, USA). The direct counting method was used to calculate the allele and genotype frequencies. The chi-square test was used to examine Hardy–Weinberg equilibrium and the differences in genotype and allele frequencies between the two groups. Comparison of the measurement data between the two groups was performed using the independent samples t-test. The odds ratios (ORs) and 95% confidence intervals (95% CIs) were calculated, and a P-value <0.05 was considered statistically significant.
Results
Clinical data of subjects
As shown in Table 1, there were no significant differences in sex, age, body mass index, smoking history, drinking history, and plasma level of total cholesterol between the ACI and the control groups (P>0.05). Common risk factors for stroke were higher in the ACI group than in the control group (P<0.05); these include a history of hypertension, type 2 diabetes mellitus (T2DM), CAD, and hyperlipidemia, as well as the plasma levels of triglycerides and low-density-lipoprotein cholesterol. The plasma levels of high-density-lipoprotein cholesterol in the ACI group were lower, than in the control group (P<0.05).
Genotyping of Asn563Ser
The PCR-amplified product was 338 bp in length, which was presented by the three genotypes deduced by agarose gel electrophoresis after being digested by restriction endonuclease Nhe I. The genotype and allele frequencies of Asn563Ser in the ACI and the control groups are shown in Table 2. These data were consistent with Hardy–Weinberg equilibrium. The genotypes and allele frequencies of Asn563Ser were significantly different between the ACI and the control groups (P=0.018 and 0.05, respectively).
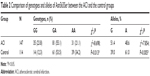  | Table 2 Comparison of genotypes and alleles of Asn563Ser between the ACI and the control groups |
Logistic regression analysis
We built a logistic regression model for ACI risk and the eight traditional risk factors of cerebrovascular disease. We selected the “Backward: LR” method and set 0.10 and 0.15, respectively, as the significance levels of the entered variables and eliminated variables. As shown in Table 3, the results indicated that the GG genotype, a history of hypertension, CAD, T2DM, and hyperlipidemia were major risk factors for ACI. After adjustment to exclude the interference of confounding factors, the GG genotype still showed a significant correlation with ACI (OR=4.862, 95% confidence interval [CI] =2.059–11.479, P<0.001).
Relation between sPECAM-1 levels and the Asn563Ser genotype
As shown in Table 4, plasma sPECAM-1 levels in the ACI group were higher than those of the control group (P=0.004). After adjustment by logistic regression analysis to eliminate the effects of confounding factors, the plasma level of sPECAM-1 was still significantly associated with ACI (OR=1.431, 95% CI=1.020–2.006, P=0.038). Plasma sPECAM-1 levels of GG, GA, and AA genotypes in the ACI and the control groups are shown in Table 4. Single-factor variance analysis showed that in both the ACI and the control groups, the plasma sPECAM-1 levels among the various Asn563Ser genotypes were significantly different (P=0.003 and 0.049, respectively). Multiple comparison results showed that the plasma sPECAM-1 level in subjects carrying the GG genotype was significantly higher than the level in those carrying the GA or AA genotypes (P<0.05).
Discussion
CD31/PECAM-1, a 130-kDa member of the immunoglobulin (Ig) superfamily, is thought to play a role in the pathogenesis of atherosclerosis and the underlying mechanism may involve inflammation. CD31/PECAM-1 mediates cell migration by regulating integrins and stimulates integrin activation.19,20 Some studies have found that polymorphisms of CD31/PECAM-1 are associated with several diseases such as myocardial infarction or acute coronary syndrome, bronchial asthma, and severe malaria;12–15,21–23 however, studies to date seldom focus on cerebral infarction risk. Asn563Ser is a polymorphism that is located on exon 8 at codon 563 of the CD31/PECAM-1 gene and it leads to a change from an asparagine to a serine. Our study showed a significant difference in the genotypes and alleles of CD31/PECAM-1 Asn563Ser between the ACI and the control groups. These findings suggest a possible correlation between the Asn563Ser polymorphism and ACI risk. The logistic regression analysis showed that the GG genotype is an independent risk factor for ACI. This result was consistent with the report by Wei et al.16 Zaremba et al24 and Marquardt et al25 also reported that significantly increased sPECAM-1 levels were found in the early stages of acute cerebral infarction. Our study also indicated that plasma sPECAM-1 levels in the ACI group were significantly higher than in controls; the sPECAM level was also associated with the risk of ACI after adjustment. The plasma sPECAM-1 level of subjects carrying the GG genotype was significantly higher than the level in those carrying the GA or AA genotype.
The underlying mechanism of how Asn563Ser polymorphism affects ACI risk and leads to the upregulated levels of plasma sPECAM-1 remains unclear. Asn563Ser is located on the sixth Ig-like homology domain of CD31 and plays an important role in cation binding. Substituting an amino acid may influence the binding affinity with integrins by altering the structure of CD31 or by affecting Ca2+-dependent cell interactions; this may influence signaling pathways that involve cell migration, differentiation, or cell apoptosis.26–28 More research is needed to elucidate the underlying mechanism.
The Leu125Val, Asn563Ser, and Gly670Arg polymorphisms in the CD31/PECAM-1 gene are found in close genetic linkages, although these single-nucleotide polymorphisms (SNPs) show population diversity.29,30 A limitation of our study is the lack of genotyping and haplotype analysis for these three SNPs together. It is necessary to expand the sample sizes in the southern Han population and verify this correlation in other populations.
In summary, the CD31/PECAM-1 Asn563Ser polymorphism is associated with genetic susceptibility to ACI in the southern Han Chinese population, independent of traditional risk factors. The Asn563Ser polymorphism may influence the plasma level of sPECAM-1 and play an important role in the pathogenesis of ACI.
Conclusion
Our study showed that the Asn563Ser polymorphism of the CD31/PECAM-1 gene and elevated soluble PECAM-1 levels are related to ACI risk in the southern Han population of the People’s Republic of China.
Acknowledgments
The study was supported by the Science and Technology Plan of Hunan Province (#2013SK5017) and Natural Science Foundation of Hunan Province, People’s Republic of China (#13JJ5005).
Disclosure
The authors report no conflicts of interest in this work.
References
Legein B, Temmerman L, Biessen EA, Lutgens E. Inflammation and immune system interactions in atherosclerosis. Cell Mol Life Sci. 2013;70(20):3847–3869. | ||
Chávez-Sánchez L, Espinosa-Luna JE, Chávez-Rueda K, Legorreta-Haquet MV, Montoya-Díaz E, Blanco-Favela F. Innate immune system cells in atherosclerosis. Arch Med Res. 2014;45(1):1–14. | ||
Ling S, Nheu L, Komesaroff PA. Cell adhesion molecules as pharmaceutical target in atherosclerosis. Mini Rev Med Chem. 2012;12(2):175–183. | ||
Galkina E, Ley K. Vascular adhesion molecules in atherosclerosis. Arterioscler Thromb Vasc Biol. 2007;27(11):2292–2301. | ||
Woodfin A, Voisin MB, Nourshargh S. PECAM-1: a multi-functional molecule in inflammation and vascular biology. Arterioscler Thromb Vasc Biol. 2007;27(12):2514–2523. | ||
Privratsky JR, Tilkens SB, Newman DK, Newman PJ. PECAM-1 dampens cytokine levels during LPS-induced endotoxemia by regulating leukocyte trafficking. Life Sci. 2012;90(5–6):177–184. | ||
Jonas NK, Saleh M. The expression of vascular endothelial platelet endothelial cell adhesion molecule-1 is not regulated by IFN-gamma treatment of C6 tumors in vivo. Int J Oncol. 2000;17(5):939–945. | ||
Newman PJ, Newman DK. Signal transduction pathways mediated by PECAM-1: new roles for an old molecule in platelet and vascular cell biology. Arterioscler Thromb Vasc Biol. 2003;23(6):953–964. | ||
Muller WA, Randolph GJ. Migration of leukocytes across endothelium and beyond: molecules involved in the transmigration and fate of monocytes. J Leukoc Biol. 1999;66(5):698–704. | ||
Privratsky JR, Newman DK, Newman PJ. PECAM-1: conflicts of interest in inflammation. Life Sci. 2010;87(3–4):69–82. | ||
Novinska MS, Pietz BC, Ellis TM, Newman DK, Newman PJ. The alleles of PECAM-1. Gene. 2006;376(1):95–101. | ||
Shalia KK, Mashru MR, Soneji SL, et al. Leucine125Valine (Leu125Val) gene polymorphism of platelet endothelial cell adhesion molecule-1 (PECAM-1) and myocardial infarction in Indian population. Indian J Clin Biochem. 2010;25(3):273–279. | ||
Listì F, Caruso C, Di Carlo D, et al. Association between platelet endothelial cellular adhesion molecule-1 polymorphisms and atherosclerosis: results of a study on patients from northern Italy. Rejuvenation Res. 2010;13(2–3):237–241. | ||
Elrayess MA, Webb KE, Bellingan GJ, et al. R643G polymorphism in PECAM-1 influences transendothelial migration of monocytes and is associated with progression of CHD and CHD events. Atherosclerosis. 2004;177(1):127–135. | ||
Listì F, Candore G, Lio D, et al. Association between platelet endothelial cellular adhesion molecule 1 (PECAM-1/CD31) polymorphisms and acute myocardial infarction: a study in patients from Sicily. Eur J Immunogenet. 2004;31(4):175–178. | ||
Wei YS, Lan Y, Liu YG, Meng LQ, Xu QQ, Xie HY. Platelet-endothelial cell adhesion molecule-1 gene polymorphism and its soluble level are associated with ischemic stroke. DNA Cell Biol. 2009;28(3):151–158. | ||
The Fourth National Cerebrovascular Diseases Conference. Diagnostic criteria and disability scale for cerebrovascular diseases. Zhonghua Shenjingke Zazhi. 1996;29:379. Chinese. | ||
Wenzel K, Baumann G, Felix SB. The homozygous combination of Leu125Val and Ser563Asn polymorphisms in the PECAM1 (CD31) gene is associated with early severe coronary heart disease. Hum Mutat. 1999;14(6):545. | ||
Saidi H, Vakilian M, Noori GH, Ghafouri HB, Abazarian N. Alterations in circulating adhesion molecules in acute myocardial infarction before and after thrombolysis with streptokinase. J Cardiovasc Thorac Res. 2013;5(4):139–141. | ||
Mestas J, Ley K. Monocyte-endothelial cell interactions in the development of atherosclerosis. Trends Cardiovasc Med. 2008;18(6):228–232. | ||
Nadi E, Hajilooi M, Babakhani D, Rafiei A. Platelet endothelial cell adhesion molecule-1 polymorphism in patients with bronchial asthma. Iran J Allergy Asthma Immunol. 2012;11(4):276–281. | ||
Casals-Pascual C, Allen S, Allen A, et al. Short report: codon 125 polymorphism of CD31 and susceptibility to malaria. Am J Trop Med Hyg. 2001;65(6):736–737. | ||
Kikuchi M, Looareesuwan S, Ubalee R, et al. Association of adhesion molecule PECAM-1/CD31 polymorphism with susceptibility to cerebral malaria in Thais. Parasitol Int. 2001;50(4):235–239. | ||
Zaremba J, Losy J. sPECAM-1 in serum and CSF of acute ischaemic stroke patients. Acta Neurol Scand. 2002;106(5):292–298. | ||
Marquardt L, Ruf A, Mansmann U, et al. Course of platelet activation markers after ischemic stroke. Stroke. 2002;33(11):2570–2574. | ||
Wang Y, Sheibani N. Expression pattern of alternatively spliced PECAM-1 isoforms in hematopoietic cells and platelets. J Cell Biochem. 2002;87(4):424–438. | ||
Wang Y, Su X, Sorenson CM, Sheibani N. Tissue-specific distributions of alternatively spliced human PECAM-1 isoforms. Am J Physiol Heart Circ Physiol. 2003;284(3):H1008–H1017. | ||
Goodman RS, Kirton CM, Oostingh GJ, et al. PECAM-1 polymorphism affects monocyte adhesion to endothelial cells. Transplantation. 2008;85(3):471–477. | ||
Bayat B, Werth S, Sachs UJ, Newman DK, Newman PJ, Santoso S. Neutrophil transmigration mediated by the neutrophil-specific antigen CD177 is influenced by the endothelial S536N dimorphism of platelet endothelial cell adhesion molecule-1. J Immunol. 2010;184(7):3889–3896. | ||
Robbins FM, Hartzman RJ. CD31/PECAM-1 genotyping and haplotype analyses show population diversity. Tissue Antigens. 2007;69(1):28–37. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



