Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 15
Long-Term Use of Antiperspirant is Associated with a Low Risk of Postoperative Complications in Patients with Axillary Osmidrosis
Authors Ho WT ![]() , Yang CY, Tsai MF
, Yang CY, Tsai MF
Received 6 July 2022
Accepted for publication 13 October 2022
Published 30 October 2022 Volume 2022:15 Pages 2335—2343
DOI https://doi.org/10.2147/CCID.S381380
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
Wen-Tsao Ho,1,* Chin-Yi Yang,2– 4,* Ming-Feng Tsai5– 7
1Department of Dermatology, Ho Wen Tsao Skin Clinic, New Taipei City, 244, Taiwan; 2Department of Dermatology, New Taipei Municipal TuCheng Hospital, New Taipei City, 236, Taiwan; 3Department of Dermatology, Chang Gung Memorial Hospital, Linkou Branch, Taoyuan, 333, Taiwan; 4Department of Cosmetic Science, Chang Gung University of Science and Technology, Linkou, Taoyuan, 333, Taiwan; 5Division of Plastic Surgery, Department of Surgery, Mackay Memorial Hospital, Taipei City, 104, Taiwan; 6Department of Medicine, MacKay Medical College, New Taipei, 25245, Taiwan; 7Graduate Institute of Biomedical Informatics, College of Medical Science and Technology, Taipei Medical University, Taipei City, 110, Taiwan
*These authors contributed equally to this work
Correspondence: Ming-Feng Tsai, Division of Plastic Surgery, Department of Surgery, Mackay Memorial Hospital, No. 92, Sec. 2, Zhongshan N. Road, Zhongshan Dist, Taipei City, 104, Taiwan, Tel +886-2-2543-3535, Fax +886-2-2543-3642, Email [email protected]
Purpose: Many patients with axillary osmidrosis (AO) cannot tolerate the local irritation of strong antiperspirants and discontinue AO use within a short time. This study evaluates the effect of long-term antiperspirant use on postoperative complications after osmidrosis surgery.
Patients and Methods: A total of 116 females (66 antiperspirant and 50 non-antiperspirant cases) who underwent osmidrosis surgery were retrospectively reviewed. Postoperative complications were compared between the 2 groups.
Results: Patients with long-term antiperspirant use had a lower risk of full-thickness skin necrosis compared with those who did not use antiperspirants (odds ratio [OR] = 0.048, 95% confidence Interval [CI]: 0.006– 0.392, p = 0.005). Patients with antiperspirants use also had a lower risk of moderate-to-severe erythema compared to those without antiperspirants use (moderate vs mild erythema: OR = 0.351, 95% CI: 0.129– 0.959, p = 0.041; severe vs mild erythema: OR = 0.161, 95% CI: 0.047– 0.550, p = 0.004). Patients who used antiperspirants also had a lower risk of severe skin erosion compared to those who did not use antiperspirants (severe vs mild skin erosion: OR = 0.164, 95% CI: 0.037– 0.725, p = 0.017). There was a trend of lower risk in moderate skin erosion in patients with antiperspirant use compared to those without antiperspirant use, but it was not statistically significant (moderate vs mild epidermal damage and peeling: OR = 0.406, 95% CI: 0.158– 1.043, p = 0.061).
Conclusion: Postoperative complications in patients with AO who undergo osmidrosis surgery are lower in those with a long-term antiperspirant use compared to patients who did not use antiperspirants.
Keywords: axillary osmidrosis, antiperspirant, complications, surgery
Introduction
The prevalence of axillary osmidrosis (AO) in China is about 4% to 8%, and there is no report on the prevalence of AO in foreign countries.1 Although the odor does not affect the life of the survivors, the odor seriously affects the daily life and normal social interaction of the patients, and brings great psychological pressure to the patients.2 AO is a distressing condition that results in considerable psychological stress and low quality of life due to an offensive odor and yellowish staining of clothing.3 AO is characterized by axillary malodor secondary to ammonia and short-chain fatty acid production resulting from bacterial decomposition of apocrine gland secretions.4,5 Apocrine glands in the armpit extend from the lower part of the dermis to the subcutaneous fat, and it is more difficult to remove firmly attached apocrine glands from the lower layer of the dermis than those located under the skin.6 Incomplete removal of apocrine glands attached to the lower dermis can lead to a recurrent rash.5
Current treatments for AO include antiperspirants, deodorants, botulinum toxin injections, subcutaneous laser treatments, percutaneous ethanol injections, and various types of surgery.7–12 To permanently resolve AO, apocrine glands must be eradicated through surgery.4,13,14 However, various surgical complications have been reported such as hematoma, seroma, skin necrosis, scarring, and a prolonged recovery period.6,15 Moreover, aggressive removal of apocrine glands may be associated with severe complications such as hematoma formation and skin necrosis.13 Thus, reducing the incidence of complications and successfully removing the apocrine glands is important for the treatment of AO.
Antiperspirants reduce sweat release from the eccrine sweat glands, thereby limiting the amount of secretion in the armpit area.16 Aluminum salts and other metal salts, such as zirconium, are known to reduce sweating by causing mechanical obstruction of glandular ducts.16 Therefore, by releasing less sweat, less bacterial production and hence alter the environment in which bacterial overgrowth takes place and odor reduces to occur.16 A previous study recommended antiperspirant use (based on a 90% success rate for the desired effects) as first-line treatment of AO.17 However, topical antiperspirants are only a transient solution for AO,18 while the surgery can permanently resolve AO.13 Nevertheless, additional evidences indicate that antiperspirants increase the vascular proliferation and ectasia, decrease the sweat and sebaceous glands, and decrease the infiltration of inflammatory cells.19–21 Surgical treatment reduces microcirculation of axillary skin, resulting in the occurrence of postoperative complication (eg, skin necrosis).22,23 Thus, we hypothesize that AO patients applied antiperspirants may reduce the postoperative complication due to the above effects. The purpose of this study is to investigate if the long-term use of antiperspirants has an effect on postoperative complications after osmidrosis surgery.
Patients and Methods
Patients and Study Design
The records of females who underwent osmidrosis surgery for severe AO (defined as Level 3 [moderate odor] to Level 5 [severe odor] by a previously described gauze test3 by the single surgeon from January 2019 to December 2021 were retrospectively reviewed. Before osmidrosis surgery, AO patients tracked for the status of antiperspirant use in the clinic for over 6 months. The patients were grouped into antiperspirant and non-antiperspirant groups. For antiperspirant group, patients continuously applied antiperspirant (1–2 times daily) for a minimum of 6 months prior to surgery. For non-antiperspirant group, patients never used antiperspirants during 6 months prior to surgery. Patients with antiperspirant-related rare side effects, including history of smoking or drinking, keloids, abnormal blood coagulation, contact eczema, ulceration or infection in axilla, previous laser hair removal or microwave thermolysis treatment, a history of a preoperative hematoma, and hormones, psycho-pharmaceuticals or any other drugs use, were excluded. Of the 116 patients, there were 66 in the antiperspirant group and 50 in the non-antiperspirant group. The Chang Gung Medical Foundation Institutional Review Board approved this study (approval no. 202200098B0), which adhered to the tenets of the 1975 Declaration of Helsinki and was obtained informed consent from each patient.
Surgical Procedure
After the sterilization of the axilla, local anesthesia with tumescent water were administered. Tumescent water (a mixture of 500 mL normal saline, 20 mL sodium bicarbonate and 1 mL epinephrine) evenly injected between skin flap and hypodermis with 150 mL on one side of axilla. One 5 mm incision was made in the axilla using a 15# surgical blade, and an dissecting scissors are used to separate subcutaneous tissue from the skin. An shaver (E9000 System, ConMed Linvatec Corporation, Largo, Fla.) was used for removal of the apocrine glands under the separated skin flap, which did not influence the thickness of skin flap. After complete removal of the apocrine glands, the axillary wound was covered with a bulky ball-shaped gaze, about the size of a fist, and was fixed in place with sutures such that the skin flap and subcutaneous skin were firmly pressed together (te-over dressing). The procedure was performed on an outpatient basis, and patients returned after 3 days for tie-over dressing and suture removal.
Data Collection and Evaluation
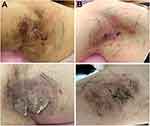
Patients were asked to report the effect of the operation 14 days after surgery, and pictures of the skin flaps were also taken 14 days after the operation. Postoperative complications, including erythema (percentage of the total area), skin erosion (percentage of the total area), and full-thickness skin necrosis, were evaluated for all patients. With respect to erythema, the severity of mild, moderate, and severe were respectively defined as “<50%”, ‘50–75%’, and ‘>75%’. For skin erosion, the severity of mild, moderate, and severe were respectively defined as “<50%”, “50–75%”, and “>75%”. For full-thickness skin necrosis, it was defined as any full-thickness skin necrosis. To increase the reliability of the measures, the same camera was used to record images under the same settings for all patients. To rule out the possibility of potential bias, the images were mixed and were then given to the same dermatologist who was unaware of the patient to assess the condition of the skin flaps.
At 8 weeks of post-surgery, postoperative malodor elimination was assessed by surgeons and patients. The degree of axillary malodor elimination was evaluated as excellent (no odor occurs after intense exercise), good (mild odor occurs after intense exercise), fair (mild odor occurs after daily activity or moderate odor occurs after intense exercise), and poor (moderate-to-severe odor occurs after daily activity).
Statistical Analysis
Continuous variables were represented as median with the corresponding range, and comparisons between groups was performed with the Mann–Whitney U-test. Categorical variables were expressed as number (percentage) and assessed by Fisher’s exact test following Bonferroni correction for comparisons between groups. Multivariate logistic regression analysis was performed to examine associations between patient characteristics and postoperative complications. SPSS version 25.0 software (IBM Corp., New York, USA) was used for all statistical analysis. A value of p < 0.05 was considered to indicate a statistically significant difference.
Results
Patient Characteristics
The median age of the 116 female patients was 28 years (range: 20–42 years) (Table 1). Sixty-six (52.6%) patients had a history of antiperspirant use, and 50 (47.4%) had not used antiperspirants. At 14 days of post-surgery, 80 (69.0%) patients had mild erythema, 19 (16.4%) patients had moderate erythema and 17 (14.7%) patients had severe erythema, and 78 patients (67.2%) had mild skin erosion, 28 (24.1%) patients had moderate skin erosion and 10 (8.6%) had severe skin erosion. Fourteen (12.1%) patients had full-thickness skin necrosis.
 |
Table 1 Patient Demographics and Clinical Characteristics |
Association of Antiperspirant Use and Postoperative Complications
Compared with the non-antiperspirant use group, patients with antiperspirant use significantly had a low proportion of postoperative full-thickness skin necrosis (antiperspirant vs non-antiperspirant group: 1.5% vs 26.0%, p < 0.001, Table 2 and Figure 1). Antiperspirant use was negatively associated with the severity of postoperative erythema (p = 0.001, Table 2 and Figure 1). Antiperspirant use was negatively associated with the severity of postoperative skin erosion, although the association was not statistically significant (p = 0.056, Table 2 and Figure 1). In addition, age was not significantly different between the 2 groups (p = 0.076, Table 2).
 |
Table 2 Comparison of Postoperative Complications Between the Non-Antiperspirant and Antiperspirant Groups |
Multivariate logistic regression analysis showed that patients who used antiperspirants had a lower risk of postoperative full-thickness skin necrosis compared with those who did not use antiperspirants (odds ratio [OR] = 0.048, 95% confidence Interval [CI]: 0.006–0.392, p = 0.005, Table 3). Patients who used antiperspirants also had a lower risk of moderate-to-severe erythema compared to those who did not use antiperspirants (moderate vs mild erythema: OR = 0.351, 95% CI: 0.129–0.959, p = 0.041; severe vs mild erythema: OR = 0.161, 95% CI: 0.047–0.550, p = 0.004, Table 3). Patients who used antiperspirants also had a lower risk of severe skin erosion compared to those who did not use antiperspirants (severe vs mild skin erosion: OR = 0.164, 95% CI: 0.037–0.725, p = 0.017, Table 3). Moreover, there was a trend of lower risk of moderate skin erosion in patients who used antiperspirants compared to those who did not use antiperspirants; however, statistical significance was not reached (moderate vs mild skin erosion: OR = 0.406, 95% CI: 0.158–1.043, p = 0.061, Table 3).
 |
Table 3 Multivariate Logistic Regression Analysis of Antiperspirant Use and Postoperative Complications |
Discussion
Antiperspirants can effectively reduce local perspiration in the axilla. The principle behind antiperspirants is to use aluminum molecules to infiltrate the sweat ducts. The entrance of aluminum into sweat glands causes changes in the cells and the formation of a complex that temporarily blocks sweat ducts, thus, inhibiting perspiration. However, topical antiperspirants are only a transient solution for AO. The current analysis of prior antiperspirant use and risk of postoperative complications yielded the following new key findings. First, prior antiperspirant use was a independent predictive factor for full-thickness skin necrosis or severe erythema. Second, prior antiperspirant use was associated with a lower risk of moderate and severe skin erosion, although it was not significantly different.
Generally, apocrine gland secretions and the activity of bacteria create the characteristic stench of AO. Previous studies have been shown that the apocrine glands of AO patients are hypertrophic and hyperplastic.24 In order to permanently treat AO, it is necessary to remove or destroy the axillary apocrine glands.24 Surgical methods provide a definitive treatment for AO, and the ideal surgical treatment should be a simple, effective, and be able to be performed under local anesthesia in an outpatient setting.25 However, aggressive removal of apocrine glands can be associated with relatively severe complications such as hematoma formation and necrosis.13,24 A early complication, superficial epidermal necrosis, was reported to occur in 37% of the AO patients after performing a subcutaneous gland excision technique.26 Although many methods have been used to reduce the frequency of postoperative complications, the incidence of complications with traditional surgical approaches is relatively high. If a hematoma occurs it should be removed immediately to avoid skin flap necrosis, and then a compression dressing should be applied. Debridement and daily dressing changes should be performed when skin flap necrosis occurs. However, larger skin defects may need to be repaired by skin grafts. Nevertheless, in contrast to traditional surgical approach, small incision surgery with shaver is now widely used because postoperative scarring is minimized and limb function is preserved.27 However, due to the small incision and the limitations of blind excision of secretory glands, the risk of subcutaneous hematoma formation and incomplete cleaning of apocrine glands is high, which directly affects the postoperative survival of the skin flap.25 Particularly, although a smaller incision with a length of about 5 mm were used in the present study, expert surgeon could avoid the occurrence of this state and traditional approach-related postoperative scarring and impaired limb function. By this procedure, the excision of subcutaneous apocrine glands was performed through a single incision that allowed the apocrine glands to be completely removed, which did not influence the postoperative thickness of skin flap. Overall, only 9.5%, 10.7%, and 7.1% of AO patients, respectively, developed full-thickness skin necrosis, severe erythema, and severe skin erosion during a follow-up period of 14 days. Moreover, at 8 weeks of post-surgery, all patients had an elimination of malodour. This is similar to our previous study, indicating that more than 90% patients have an improvement of malodour after surgery removed apocrine glands.28 The previous study demonstrates that removing a majority of the deeply located apocrine contributes to the elimination of malodour, while removing eccrine glands may be slightly less effective for treating hyperhidrosis than osmidrosis.14 Thus, the low complications and effective elimination of malodour suggest that the procedure described in this report is a good choice for the treatment of AO.
Antiperspirants are classified as drugs, and are therefore subject to rules and regulations set forth by the Food and Drug Administration. The active ingredient in antiperspirants is usually aluminum-based, which reduces sweat by causing obstruction of the apocrine glands.29 There are several possible explanations for the finding that long-term use of antiperspirants is associated with a lower rate of postoperative complications. It is speculated that there are 2 possible reasons for the finding: 1) antiperspirant use improves axillary microbiota and 2) microcirculation.
Human axillary odour is commonly attributed to the bacterial degradation of precursors in sweat secretions.30 The axillary microbiota consists mainly of gram-positive bacteria of the genera Staphylococcus, Micrococcus, Propionibacterium, and Corynebacterium, which is the primary cause of the foul odor.30 Recently, Troccaz et al reported that the underarms of non-antiperspirant users have significantly higher sweat odour intensities and harboured on average about 50 times more bacteria than those of antiperspirant users.31 These potentially pathogenic species can cause postoperative complications when forced out of their natural habitat by surgical utensils or needles and possess the ability to develop chronic infections in opportunistic locations,32 suggesting that strategies for preparing and managing patients undergoing surgery should be adjusted to reduce the chances of postoperative complications. Taken together, this may be an explanation of why the skin flaps of patients who have used antiperspirants over a long period of time exhibit improved healing. Certainly, randomized control trials are needed to determine the association of antiperspirant use and all aspects of postoperative outcomes and complications.
In rabbit skin treated with antiperspirants, the dermis showed features of edema, increased vascularity, a decrease in sweat and sebaceous glands, and less infiltration of inflammatory cells.21 The remarkable vascular proliferation and ectasia may be caused by a contact reaction to an antiperspirant.19,20 Surgical treatment reduces microcirculation of axillary skin, resulting in the occurrence of postoperative complication (eg, skin necrosis).22,23 In addition, a previous study analyzed AO patients 2 years after treatment with a mini-incision and subdermal vascular preservation, and found a low incidence of postoperative complications including hematomas, epidermal erosions, infections, necrosis, incision dehiscence, and skin ripples.33 Based on these findings, we can assume that the antiperspirants when applied for a long period of time will cause a subclinical contact reaction resulting in local microvascular proliferation, which then may improve the skin microcirculation. Although biopsies were not performed to support this claim, our clinical observations may confirm this hypothesis.
Antiperspirants help block the production of bothersome sweat, but nevertheless are associated with some side effects. They may cause mild and transient skin irritation,34 and 26% of the patients have reported that the irritation is treatment-limiting.35 One of the most common adverse reactions of antiperspirants is sensitive skin and itching, tingling, and rash.36,37 In addition, axillary hyperpigmentation, a type of post-inflammatory hyperpigmentation, can occur due to the continuous irritation related to antiperspirant use.38,39 Consequently, the present study did not include AO patients with antiperspirant-related side effects. Further, we only included female patients with AO, and demonstrated that long-term antiperspirant use resulted in a lower rate of postoperative complications than that without antiperspirant use. However, the previous literature revealed that the proportion of postoperative complications are not significantly different between male and female patients with AO.40 Thus, we suggest that antiperspirants use may be recommended preoperatively to lower the risk of postoperative complications in all patients with AO, providing a benefit strategy for surgical treatment of AO.
Several limitations of this study must be acknowledged. First, since this is a retrospective study in a single-center with a relatively minor sample size, selection bias may not be completely excluded, which may be affected by confounding factors. Therefore, it still remains necessary to carry out large-scale, multicenter and prospective studies to confirm the results of the present study. Second, this retrospective study can only establish associations, not cause-and-effect associations. Therefore, prospective large-sample, multicenter studies are still needed to confirm the results of the present study.
Conclusions
AO patients with prior antiperspirant use have a low risk of postoperative complications after osmidrosis surgery, particularly in postoperative full-thickness skin necrosis and erythema. Nevertheless, all wounds in our patients were completely healed and all patients had a good or excellent elimination of malodour at 8 weeks of post-surgery.
Abbreviation
AO, axillary osmidrosis.
Data Sharing Statement
The data used to support the findings of this study are included within the article.
Ethics Approval and Informed Consent
The Chang Gung Medical Foundation Institutional Review Board approved this study (approval no. 202200098B0), which adhered to the tenets of the 1975 Declaration of Helsinki and informed consent was obtained from each patient.
Funding
There is no funding to report.
Disclosure
The authors have no conflicts of interest to declare.
References
1. Dong Z, Tan Z, Chen Z. Association of BMI and lipid profiles with axillary osmidrosis: a retrospective case-control study. J Dermatolog Treat. 2021;32(6):654–657. doi:10.1080/09546634.2019.1688232
2. Zhang L, Chen F, Kong J, Li Z, Wen X, Liu J. The curative effect of liposuction curettage in the treatment of bromhidrosis: a meta-analysis. Medicine. 2017;96(33):e7844. doi:10.1097/MD.0000000000007844
3. Morioka D, Ohkubo F, Amikura Y. Clinical features of axillary osmidrosis: a retrospective chart review of 723 Japanese patients. J Dermatol. 2013;40(5):384–388. doi:10.1111/1346-8138.12115
4. Shin JY, Roh SG, Lee NH, Yang KM. Osmidrosis treatment approaches: a systematic review and meta-analysis. Ann Plast Surg. 2017;78(3):354–359. doi:10.1097/SAP.0000000000000911
5. Wang R, Yang J, Sun J, Minimally Invasive A. Procedure for axillary osmidrosis: subcutaneous curettage combined with trimming through a small incision. Aesthetic Plast Surg. 2015;39(1):106–113. doi:10.1007/s00266-014-0431-2
6. Park YJ, Shin MS. What is the best method for treating osmidrosis? Ann Plast Surg. 2001;47(3):303–309. doi:10.1097/00000637-200109000-00014
7. He J, Wang T, Dong J. A close positive correlation between malodor and sweating as a marker for the treatment of axillary bromhidrosis with Botulinum toxin A. J Dermatolog Treat. 2012;23(6):461–464. doi:10.3109/09546634.2011.594869
8. He J, Wang T, Dong J. Excision of apocrine glands and axillary superficial fascia as a single entity for the treatment of axillary bromhidrosis. J Eur Acad Dermatol Venereol. 2012;26(6):704–709. doi:10.1111/j.1468-3083.2011.04149.x
9. Ichikawa K, Miyasaka M, Aikawa Y. Subcutaneous laser treatment of axillary osmidrosis: a new technique. Plast Reconstr Surg. 2006;118(1):170–174. doi:10.1097/01.prs.0000221005.86108.0d
10. Ou LF, Yan RS, Chen IC, Tang YW. Treatment of axillary bromhidrosis with superficial liposuction. Plast Reconstr Surg. 1998;102(5):1479–1485. doi:10.1097/00006534-199810000-00021
11. Shim HS, Min SK, Lim JS, Han KT, Kim MC. Minimal subdermal shaving by means of sclerotherapy using absolute ethanol: a new method for the treatment of axillary osmidrosis. Arch Plast Surg. 2013;40(4):440–444. doi:10.5999/aps.2013.40.4.440
12. Traupe B, Folster H, Max H, Schulz J. Effective axillary malodour reduction by polyquaternium-16-containing deodorants. Int J Cosmet Sci. 2017;39(2):141–148. doi:10.1111/ics.12358
13. Jung SK, Jang HW, Kim HJ, et al. A prospective, long-term follow-up study of 1444 nm Nd: yAGLaser: a new modality for treating axillary bromhidrosis. Ann Dermatol. 2014;26(2):184–188. doi:10.5021/ad.2014.26.2.184
14. Wu WH, Ma S, Lin JT, Tang YW, Fang RH, Yeh FL. Surgical treatment of axillary osmidrosis: an analysis of 343 cases. Plast Reconstr Surg. 1994;94(2):288–294. doi:10.1097/00006534-199408000-00011
15. Perng CK, Yeh FL, Ma H, et al. Is the treatment of axillary osmidrosis with liposuction better than open surgery? Plast Reconstr Surg. 2004;114(1):93–97. doi:10.1097/01.prs.0000127801.15386.99
16. Oliveira ECV, Salvador DS, Holsback V, Shultz JD, Michniak-Kohn BB, Leonardi GR. Deodorants and antiperspirants: identification of new strategies and perspectives to prevent and control malodor and sweat of the body. Int J Dermatol. 2021;60(5):613–619. doi:10.1111/ijd.15418
17. Togel B, Greve B, Raulin C. Current therapeutic strategies for hyperhidrosis: a review. Eur J Dermatol. 2002;12(3):219–223.
18. Inaba M, Inaba Y. Human Body Odor: Etiology, Treatment, and Related Factors. Springer-Verlag; 1992:xii, 285 p.
19. Northcutt AD, Nelson DM, Tschen JA. Axillary granular parakeratosis. J Am Acad Dermatol. 1991;24(4):541–544. doi:10.1016/0190-9622(91)70078-g
20. English JC, Derdeyn AS, Wilson WM, Patterson JW. Axillary granular parakeratosis. J Cutan Med Surg. 2003;7(4):330–332. doi:10.1007/s10227-002-0131-4
21. Rashid M, Gupta S. A study to assess the effects of antiperspirant aluminium nitrate on the rabbit skin. JK Sci. 2019;21(2):88–93.
22. Bechara FG, Sand M, Stucker M, Georgas D, Hoffmann K, Altmeyer P. Laser Doppler scanning study of axillary skin before and after liposuction curettage in patients with focal hyperhidrosis. Dermatology. 2008;216(2):173–179. doi:10.1159/000111518
23. Ho WT. Preconditioning and postoperative hyperbaric oxygen therapy to reduce skin flap erosion after osmidrosis surgery. J Plast Reconstr Aesthet Surg. 2019;72(4):685–710. doi:10.1016/j.bjps.2019.01.020
24. Kim WO, Song Y, Kil HK, Yoon KB, Yoon DM. Suction-curettage with combination of two different cannulae in the treatment of axillary osmidrosis and hyperhidrosis. J Eur Acad Dermatol Venereol. 2008;22(9):1083–1088. doi:10.1111/j.1468-3083.2008.02717.x
25. Tung TC, Wei FC. Excision of subcutaneous tissue for the treatment of axillary osmidrosis. Br J Plast Surg. 1997;50(1):61–66. doi:10.1016/s0007-1226(97)91285-6
26. Qian JG, Wang XJ. Effectiveness and complications of subdermal excision of apocrine glands in 206 cases with axillary osmidrosis. J Plast Reconstr Aesthet Surg. 2010;63(6):1003–1007. doi:10.1016/j.bjps.2009.05.004
27. Yang H, Zhang MY, Ding SL, Li CY, Tan WQ. Modified tumescent liposuction-curettage through mini incisions for the treatment of secondary axillary bromhidrosis with subcutaneous scarring. Plast Reconstr Surg. 2012;130(6):916e–918e. doi:10.1097/PRS.0b013e31826da1db
28. Ho WT, Lee LJ, Pan JY. Following changes in the axillary secretions of two patients before and after bromhidrosis surgery using liquid chromatography-mass spectrometry. Dermatol Surg. 2017;43(3):459–462. doi:10.1097/DSS.0000000000000934
29. Benohanian A. Antiperspirants and deodorants. Clin Dermatol. 2001;19(4):398–405. doi:10.1016/s0738-081x(01)00192-4
30. James AG, Austin CJ, Cox DS, Taylor D, Calvert R. Microbiological and biochemical origins of human axillary odour. FEMS Microbiol Ecol. 2013;83(3):527–540. doi:10.1111/1574-6941.12054
31. Troccaz M, Gaia N, Beccucci S, et al. Mapping axillary microbiota responsible for body odours using a culture-independent approach. Microbiome. 2015;3(1):3. doi:10.1186/s40168-014-0064-3
32. Bay L, Barnes CJ, Fritz BG, et al. Universal dermal microbiome in human skin. mBio. 2020;11(1). doi:10.1128/mBio.02945-19
33. Zhao H, Li S, Nabi O, Hu L, Gao X, Luo F. Treatment of axillary bromhidrosis through a mini-incision with subdermal vascular preservation: a retrospective study in 396 patients. Int J Dermatol. 2016;55(8):919–925. doi:10.1111/ijd.13313
34. Sammons JE, Khachemoune A. Axillary hyperhidrosis: a focused review. J Dermatolog Treat. 2017;28(7):582–590. doi:10.1080/09546634.2017.1309347
35. Glent-Madsen L, Dahl JC. Axillary hyperhidrosis. Local treatment with aluminium-chloride hexahydrate 25% in absolute ethanol with and without supplementary treatment with triethanolamine. Acta Derm Venereol. 1988;68(1):87–89.
36. Holze E. Therapie der Hyperhidrosis [Therapy of hyperhidrosis]. Hautarzt. 1984;35(1):7–15. German.
37. Nyamekye IK. Current therapeutic options for treating primary hyperhidrosis. Eur J Vasc Endovasc Surg. 2004;27(6):571–576. doi:10.1016/j.ejvs.2004.01.023
38. Kwong HL, Lim SPR. Pigmented contact dermatitis in the axillae caused by hydroperoxides of limonene. JAAD Case Rep. 2020;6(6):476–478. doi:10.1016/j.jdcr.2020.04.007
39. Pincelli C, Magni R, Motolese A. Pigmented contact dermatitis from deodorant. Contact Dermatitis. 1993;28(5):305–306. doi:10.1111/j.1600-0536.1993.tb03447.x
40. Fang CL, Tsai CB, Chen MS, Yang HY, Fang KJ. Factors affecting the efficacy of suction curettage using an arthroscopic shaver for bromhidrosis. Dermatol Surg. 2021;47(2):245–249. doi:10.1097/DSS.0000000000002768
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
Recommended articles
Complications and Mortality After Surgery in Patients with Chronic Kidney Disease: A Retrospective Cohort Study Based on a Multicenter Clinical Database
Liao CC, Liu CC, Lee YW, Chang CC, Yeh CC, Chang TH, Chen TL, Lin CS
Journal of Multidisciplinary Healthcare 2024, 17:3535-3544
Published Date: 23 July 2024
Comparison of Ultrasound Energy Delivered to the Anterior Segment Across Different Phacoemulsification Surgical Platforms
Nelson TK, Ricks RG, Cardenas IA, Whitaker T, Jensen JL, Olson RJ, Pettey JH
Medical Devices: Evidence and Research 2025, 18:29-35
Published Date: 14 January 2025
Postoperative Adverse Outcomes in Patients With Frailty Undergoing Urologic Surgery Among American Patients: A Propensity-Score Matched Retrospective Cohort Study
Hsu CW, Chang CC, Lam F, Liu MC, Yeh CC, Chen TL, Lin CS, Liao CC
Clinical Epidemiology 2025, 17:241-250
Published Date: 12 March 2025