Back to Journals » Journal of Inflammation Research » Volume 15
Gold Nanoparticle-Based Therapy for Muscle Inflammation and Oxidative Stress
Authors Pinho RA , Haupenthal DP , Fauser PE, Thirupathi A, Silveira PCL
Received 29 January 2022
Accepted for publication 27 May 2022
Published 31 May 2022 Volume 2022:15 Pages 3219—3234
DOI https://doi.org/10.2147/JIR.S327292
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ning Quan
Ricardo A Pinho,1,2 Daniela PS Haupenthal,3 Paulo Emílio Fauser,3 Anand Thirupathi,2 Paulo CL Silveira3
1Laboratory of Exercise Biochemistry in Health, Graduate Program in Health Sciences, School of Life Sciences and Medicine, Pontifícia Universidade Católica Do Paraná, Curitiba, Paraná, Brazil; 2Faculty of Sports Science, Ningbo University, Ningbo, People’s Republic of China; 3Laboratory of Experimental Pathophysiology, Graduate Program in Health Sciences, Universidade Do Extremo Sul Catarinense, Criciúma, Santa Catarina, Brazil
Correspondence: Ricardo A Pinho, Laboratory of Exercise Biochemistry in Health, Graduate Program in Health Sciences, School of Life Sciences and Medicine, Pontifícia Universidade Católica do Paraná, Imaculada Conceição Street, 1155, Prado Velho, Curitiba, Paraná, Zip Code 80215-901, Brazil, Email [email protected]
Abstract: Proinflammatory cytokines and reactive oxygen species are released after muscle damage, and although they are necessary for the muscle regeneration process, an excess of these substances leads to the destruction of biomolecules and impairment of the repair system. Several drugs have emerged in recent years to control the muscle inflammatory response, and studies have shown that gold nanoparticles (AuNPs) have anti-inflammatory and antioxidant properties. This review reveals the effects of AuNPs on the inflammatory and redox mechanisms of muscles. We assessed the results of several studies published in different journals over the last 20 years, with a focus on the effects of AuNPs on possible aspects of muscle regeneration or recovery, namely, inflammatory processes and redox system mechanisms. A systematic database search was conducted using PubMed, Medline, Bireme, Web of Science, and Google Scholar to identify peer-reviewed studies from the 2000s. Combinations of keywords related to muscle damage, regeneration or repair, AuNPs, oxidative stress, and antioxidants were used in the search. This review did not address other variables, such as specific diseases or other biological effects; however, these variables should be considered for a complete understanding of the effects of AuNPs on skeletal muscles.
Keywords: gold nanoparticles, redox homeostasis, antioxidant, muscle regeneration, inflammation, nanomaterials, oxidative stress
Introduction
In the last decade, gold nanoparticles (AuNPs) have been widely explored as tools with applications in the medical field. Several studies performed by our group and other researchers have revealed the potential application of AuNPs in the medical field based on their optical, anti-inflammatory, antioxidant, and anti-cancer properties. Paula et al1 revealed that N-acetylcysteine (NAC) plus AuNPs promotes pronounced anti-inflammatory and antioxidant effects under conditions associated with lung inflammation. Haupenthal et al2 showed that chronic administration of AuNPs in Mdx mice increased the antioxidant potential and reduced inflammation in the gastrocnemius muscle. Tartuce et al3 also demonstrated the potential anti-inflammatory and antioxidant activities of AuNPs by conjugating AuNPs with 2-methoxy-isobutyl-isonitrile, which improved the redox and inflammatory profiles in infarcted rats. Other studies have indicated the efficacy of AuNPs as anticancer drug nanocarriers4–6 and in disease diagnosis.7
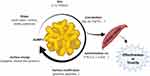
Based on their demonstrated properties and effectiveness, AuNPs have become the focus of numerous studies in the research and development of new tools for applications in numerous diseases. However, for applications in the medical field, the physical and chemical properties of AuNPs need to be precisely controlled8,9 because these properties have a direct influence on the interaction of AuNPs with biological cells/tissues,10,11 as represented in Figure 1. The intracellular release rate of AuNPs can affect their associated toxicity depending on the dose, size, shape, and/or surface charge,6,12–15 and different routes of administration can result in various effects on the biodistribution of drug carriers.16 Different sizes and shapes of AuNPs tend to induce different responses when applied in biological media.11,17 In vitro studies have revealed that AuNPs present concentration-dependent cytotoxic effects, including increased reactive oxygen species (ROS) production and consequent oxidative damage.18,19 However the complexity of a whole organism is much greater than that of a single cell, thus suggesting that a broad analysis is required that includes general health indicators and possible tissue toxicities.20 For example, Chen et al21 studied the in vivo effects of different concentrations of AuNPs and observed that concentrations of 3, 5, 50, and 100 nm did not show harmful effects; however, the intermediate size range of 8–37 nm had lethal effects on mice and induced severe sickness, clinical changes, and shorter average lifespans.
In relation to their shape, spherical AuNPs are the most commonly used because they easily synthesize and produce nanoparticles with different physicochemical properties.22 Several studies have shown that these properties of AuNPs have direct implications in biological media.10,11,23,24 The study of Della Vechia et al11 demonstrated that particle size directly influences biological behavior. In this study, the authors showed that small variations in size (10–30 nm) could directly influence therapeutic efficacy as well as toxicity. Other studies have demonstrated that the shape of AuNPs is essential for their effectiveness when applied in biological media.6,25 Xie et al25 demonstrated that different AuNP shapes induced different cellular uptake mechanisms; and Xia et al26 showed that the cellular uptake of AuNPs in human hepatoma cells (HepG2 cells) was dependent on their size and shape. The results showed that spherical AuNPs presented a higher uptake and star-shaped AuNPs presented lower uptake by HepG2 cells.
Another important property of AuNPs for application in the medical field is the surface charge. In conjunction with the size and shape, the surface charge can also directly affect the efficiency and cellular uptake of AuNPs. AuNPs can be positively and negatively charged or have a neutral charge. Gupta and Rai27 evaluated the effect of the size and surface charge of AuNPs on skin permeability and showed that neutral AuNPs (2 nm) presented maximum permeability while cationic AuNPs (3 nm) exhibited the lowest permeability. In general, negatively charged AuNPs exhibit lower toxicity than positively charged AuNPs. AuNPs tend to disrupt cell membrane integrity, leading to increased toxicity.28,29
The surface of AuNPs can be easily linked with different types of ligands/biomolecules depending on the application, thereby ensuring the higher effectiveness of these nanoparticles.30 The surface chemistry of AuNPs and surface modifications with biomolecules can significantly improve the permeability of these particles in cells/tissues, thereby increasing the effectiveness of AuNPs for the treatment and diagnosis of diseases.31–34 The surface chemistry of AuNPs is considered a key factor in the toxicity of the particles. Özçiçek et al35 showed that the surface chemistry of the AuNPs was a more important parameter than the size in terms of in vivo histological toxicity. Surface modifications of AuNPs can induce a synergistic effect, thereby potentiating the performance of these particles for biomedical applications.33
All of these properties mentioned above are critical parameters for the effective application of nanomaterials in the medical field. However, the effectiveness and toxicity of AuNPs are also related to cell or tissue type, the concentration used, and the administration route.11,26,36 In summary, spherical AuNPs with sized ≥50 nm tend to present an improved interaction with the biological medium and thus present higher effectiveness and lower toxicity.6,29 Table 1 summarizes a number of the influences of physicochemical properties on the effectiveness of nanoparticles from in vitro and in vivo studies.
 |
Table 1 Influence of the Physicochemical Properties of AuNPs on Their Effectiveness from in vitro and in vivo Studies |
Gold Nanomaterials Alter ROS Function and Support Redox Homeostasis in the Muscle
From a historical perspective, the effect of oxidizing free radicals in various biological samples and the mechanism that induces toxicity and injury were first verified in the 1950s.37,38 The effect of reactive oxygen species (ROS) on aging was discovered by Harman.39 At that time, it was believed that ROS induced aging. Another milestone study confirmed the presence of ROS in living organisms by isolating superoxide dismutase (SOD) from bovine erythrocytes.40 However, subsequent studies by Murad et al revealed the importance of ROS in biological functions; in particular, the role of nitric oxide (NO) in the vascular endothelial system revealed the importance of ROS as a secondary messenger in cell signaling to regulate various cellular functions.41 As a result, several extraordinary studies on free radical biology have been conducted. Studies have well established that ROS and reactive nitrogen species (RNS) are crucial in regulating various cellular functions by acting as messengers in biological systems during cellular metabolism. Almost every cellular event, including survival, has been linked with the removal and acceptance of electrons (called redox), which sometimes impacts other molecules and causes oxidative stress during physiological and pathological events. High levels of ROS formation create an “oxidative distress” environment during the pathological event instead of oxidative stress, and various cellular and molecular functions are organized.42 In addition, the level and steady-state of endogenous antioxidants determine the toxicity of these molecules. Indeed, exogenous antioxidant research has demonstrated for several decades the need to scavenge ROS. As a result, several molecules from natural products or synthesized products have been approved for treating various diseases, including cardiovascular and inflammatory diseases. However, addressing the limitations of these small molecules, such as their nonspecific distribution and low delivery efficiency, is difficult. The development of the nanotechnology field has significantly reduced the aforementioned burdens and could ultimately advance the field of antioxidant therapy.
Most importantly, several nanomaterials have been shown to efficiently increase the stability of endogenous antioxidants and improve their functions by protecting them from decay under harsh pH conditions in the cellular environment.43,44 Furthermore, nanomaterials can act as artificial redox systems by mimicking various antioxidants or delivering antioxidants to targeted tissues, which can significantly combat ROS-induced damage.45 However, the delivery of single antioxidants with nanomaterial doping can induce additional oxidative injury. For example, SOD-doped nanoparticles may be able to stop superoxide-induced oxidative damage but not concurrently induced hydrogen peroxide (H2O2)-mediated oxidative damage,46 suggesting that nanomaterials must not only break the free radical chain but also prevent ROS-induced damage. Although studies have demonstrated that nanomaterials with doping molecules can affect ROS scavenging activity in vitro, transferring these findings to clinical trials has been limited by the low biocompatibility, biodegradability, hydrophobicity, and hydrophilicity of loading molecules.46 Therefore, these factors should be considered when selecting nanomaterials with loading molecules to facilitate the treatment of ROS-mediated diseases. In this context, the use of gold materials, such as gold nanosheets, gold nanoconjugates, and AuNPs, has been reported to be effective in the treatment of various diseases, either with doping materials or AuNPs alone; however, the ability of such materials to affect ROS responses is the ultimate indicator of their benefits.47 Based on their size, shape, and oxidation state, AuNPs induce ROS production or prevent ROS formation. In addition, AuNPs alter various redox signaling pathways, including threonine-protein kinase B (AKT), by supporting the controlled release of ROS. For example, bioconjugated AuNPs regulate ROS formation and corresponding signaling pathways to induce angiogenesis without altering inflammatory cascades in human umbilical vein endothelial cells.48 The use of gold-coated nanoparticles upregulates cluster of differentiation 163 (CD163) in macrophages in atherosclerosis.49 Moreover, CD163 plays a crucial role in detoxifying ROS and preventing oxidative stress.50
Physical and Chemical Properties of AuNPs in the Formation of ROS and Oxidative Stress
The size of the AuNPs alters their impact on oxidative stress. For example, smaller-sized AuNPs promoted greater ROS formation, with approximately 2 nm AuNPs significantly increasing ROS by decreasing intracellular glutathione.51 In contrast, 5 nm AuNPs increased cell survival under normoxic conditions compared to hypoxic conditions,52 which may be due to the higher uptake efficiency and increased endocytosis facilitating less exposure to the cellular environment and extending the half-life.53,54 At the molecular level, 1 and 3 h of AuNP treatment activated extracellular signal-regulated kinase (ERK) signaling in response to oxidative stress.55 In addition to size, the shape of the AuNPs affects the formation of ROS and associated damage. For example, hexagonal AuNPs increase ROS formation compared to triangular and spherical AuNPs.56 Other shapes, such as gold nanorods, significantly increased ROS formation compared to gold nanospheres, indicating that hexagonal and spherical AuNPs are more crucial for forming ROS. The surface chemistry of AuNPs affects the formation of ROS. Studies have shown that positively charged AuNPs with an approximate size of 1.5 nm increase apoptosis,57 whereas other studies have shown that negatively charged AuNPs induce apoptosis in neutrophils.58
Neutral-charged AuNPs have also been shown to cause necrosis,57 and AuNP-induced ROS could be a significant factor underlying apoptosis. However, AuNP-induced ROS formation may be due to the different AuNP sizes, cell types, and ligand structures. For example, AuNPs with suitable ligands can modify the redox properties of particles, which may enhance the redox-mediated cellular actions either by decreasing or increasing ROS. Previous studies have reported that PNP ligands and carbanionic ligands alter the redox properties of gold.59,60 In addition, ligands could facilitate the internalization of AuNPs at the cellular level, which may occur through the endocytosis process.61 Although ligands help stabilize nanoparticles during their synthesis and facilitate their internalization, their presence in cells mainly affects the catalytic activity of their substrate. Furthermore, removing the ligands influences the AuNP size, which in turn modifies the specific function of the AuNPs.62 Such changes could eventually limit the redox-mediated benefits in the cells and cause ROS formation and oxidative stress. Developing a procedure to remove ligands that does not affect the size and morphology of the AuNPs would possibly enhance the redox properties of AuNPs.
In addition, the chiral structure of gold influences the formation of ROS. For example, a study showed that D-glutathione-decorated AuNPs increased ROS levels and depolarization of mitochondria compared to L-glutathione-decorated AuNPs in the human gastric cancer cell line 803 (MGC-803) cells.63 Other metallic and non-metallic coated AuNPs also significantly influence ROS formation, such as platinum-coated gold nanorods and mesoporous silica nanoparticles with gold nanorods. Furthermore, AuNPs affect ROS-mediated autophagy according to their physicochemical properties. A possible mechanism is that ROS-activated adenosine monophosphate-activated protein kinase (AMPK) may inhibit the mammalian target of rapamycin (mTOR) and promote autophagy activation.64 Another possible mechanism is that increased ROS formation inactivates autophagy-related proteins (Atg), particularly Atg4, consequently inducing autophagy,65 suggesting that AuNPs induce autophagy in response to oxidative damage. On the other hand, this mechanism causes lysosome alkalinization, which results in the inhibition of autophagy and further cell death. In summary, all of these processes are associated with AuNP-induced ROS production and oxidative stress.
AuNPs with Endogenous Antioxidant Properties
As previously mentioned, nanoparticles have been exploited as artificial redox systems in nanomedicine in recent years. For example, cerium oxide nanoparticles mimic SOD and catalase (CAT) and display higher catalytic rates than inherent antioxidants.66,67 Similarly, AuNPs exhibit CAT mimetic activity, which may be due to the mixed valence state of AuNPs that permits them to react with superoxide and hydrogen peroxide to detoxify ROS. However, coating the materials with AuNPs prevents an increase in the stability of the antioxidants. For example, Pt nanoparticles showed excellent glutathione peroxidase (GPx) and SOD-activating abilities along with CAT activity, thus promoting the maintenance of ROS at a physiological concentration,68 and Pt-coated AuNPs would efficiently stabilize this activity. A previous study showed that Pt-coated gold nanorods effectively detoxify ROS to prevent oxidative damage during plasmonic photothermal therapy.68 In addition, AuNPs with antioxidants could alter the deoxyribonucleic acid (DNA) epigenetic pattern, thus affecting various cellular functions.69 Furthermore, AuNPs (3.3 nm in diameter) doped with chiral L- and D-glutathione (D-GSH) inhibit tissue damage caused by ROS and are crucial for developing a potential therapeutic agent for various diseases, including neurodegenerative diseases, such as Alzheimer’s disease (AD).70,71 For example, AuNPs with capped mesoporous silica decreased cell membrane disruption and inhibited ROS-mediated apoptosis in AD.72 However, all of these effects caused by AuNPs depend on the specific pH environment; otherwise, the Haber-Weiss and Fenton reactions could be induced.73 Interestingly, a study showed that the pro-oxidant effect in biological microenvironments is inhibited at different hydrogen potentials (pH) when gold nanoclusters are entrapped into amine-terminated dendrimers; however, CAT activity is maintained.66 In contrast, at acidic pH, the pro-oxidant effect is observed to induce ROS and inhibit enzyme intrinsic activity,66 suggesting that the appropriate application of AuNPs could allow them to act as pH-responsive nanoantioxidants to modulate oxidative stress-mediated damage in cells.
Role of AuNPs in Muscle Oxidative Stress and Inflammation
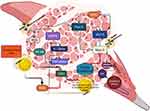
Although ROS-mediated molecular signaling contributes to structural and functional alterations in muscle, a small increase in local or systemic ROS alters this scenario and causes redox disturbances in the muscle. Indeed, skeletal muscle-like structures increase ROS levels owing to their contractile activities. Therefore, additional sources are required to balance this scenario, particularly when endogenous antioxidants have a lower buffering capacity for ROS. As previously mentioned, within specific physicochemical conditions, AuNPs can effectively neutralize ROS and contribute to muscle homeostasis. To date, various mechanisms have been proposed for AuNPs and oxidative stress. Notably, AuNPs with specific decorating molecules have been reported to influence inflammatory molecules to regulate ROS and vice versa. However, less attention has been directed toward using AuNPs in treating muscle-related disorders. Studies have shown that AuNPs improve muscle morphology during muscle injury. Here, we discuss some possible mechanisms. The regeneration phase after muscle injury requires ROS to activate specific redox signaling. For example, AuNPs activate ROS-mediated redox signaling, such as mitogen-activated protein kinase (MAPK) and factor nuclear kappa B (NF-κB), to induce a protective muscle response. AuNPs interact with cysteine (Cys-179) of kinase-beta (IKK-beta) to influence NF-κB, which could decrease the levels of proinflammatory cytokines, including tumor necrosis factor-alpha (TNF-alpha) and Interleukin-1beta (IL-1beta), thereby regulating ROS formation at physiological concentrations (Figure 2). Moreover, excess ROS induces secondary damage in nearby and uninjured muscle fibers.
The interaction between AuNPs and Cys may depend on neutral hydrogen bonding or zwitterionic interactions, which facilitate the aggregation of AuNPs in the muscle to activate redox-sensitive signaling.74 We reported that AuNPs with suitable decorating molecules, such as diclofenac and taurine, improved tissue repair in the later phase of the repair process by controlling ROS formation, as evidenced by an increased level of interleukin-4 (IL-4) and interleukin-10 (IL-10). A possible reason is that AuNPs with taurine or diclofenac increase myogenic differentiation and support tissue remodeling by regulating ROS in macrophage-1 (M1) and macrophage-2 (M2) cells. For example, our group showed that spherical AuNPs with taurine upregulate myogenic regulatory factor 5 (Myf-5), thereby reducing oxidative damage during muscle recovery.47 Another study also showed that AuNPs and gold-silver nanoparticles (Au-AgNPs) increased the myogenic differentiation of C2C12 myoblasts by activating myogenic genes, such as MyoD protein (MYOD) and MyoG protein (MyoGenin), and promoting myotube formation by activating p38 MAPK.75 However, these effects may be size-dependent. For example, AuNPs with a higher surface area can effectively support the intoxication of ROS when decorated with other doping molecules.
In contrast, electron donor or acceptor molecules of proteins such as GSH could decrease according to the gold surface area, thus allowing for higher ROS formation. These effects may be size-dependent in the absence of steric hindrance and lead to increased electron transfer-induced chemical reactions, which reduce the glutathione content in the cytosol.51 In particular, spherical AuNPs (approximately 1.4 nm in diameter) could induce mitochondrial damage and cell death in various cell lines, suggesting that ROS-induced damage is linked to these approximate sizes.76 Therefore, these sizes would produce significant damage, even in muscle. However, the doping of molecules with AuNPs can alter these effects. For example, AuNPs significantly upregulate myogenic differentiation and muscle degeneration. A recent study showed that AuNPs (5 nm in diameter) could selectively bind with the heparin-binding domain (HBD) of glycoproteins and are crucial for muscle repair because the HBD plays a major role in the retention of several growth factors, such as platelet-derived growth factors and vascular endothelial cell growth factors,77,78 which may support muscle repair during muscle injury. Taken together, the selective size and shape of AuNPs with a larger surface area would effectively prevent ROS-induced damage in the muscle, which further supports muscle homeostasis in muscle injury. Table 2 summarizes some of the potentially toxic effects of AuNPs on living systems from in vitro and in vivo studies.
 |
Table 2 Potential Toxicity of AuNPs on Living Systems from in vitro and in vivo Studies |
AuNPs in Muscle Regeneration and Inflammation
The injured muscle tissue regeneration process is dependent on an organized and sequential response mediated by cytokines and growth factors that are released by muscle fibers, and it involves macrophages, inflammatory cells, and connective tissue, including three distinct phases: degeneration, repair, and remodeling.91,92 In this scenario, the regulation of the inflammatory response is crucial for the effectiveness of the repair mechanisms. However, when the release of inflammatory mediators is stimulated, adjacent tissues suffer secondary damage from excess leukocyte infiltration and consequent oxidative stress, which requires pharmacological intervention to restore or maintain an ideal cellular environment for muscle regeneration. In this sense, several recent studies have suggested the use of AuNPs for inflammatory and redox control in muscle injury models.75,82,85
Previous in vitro studies evaluated the effects of AuNPs on tissue regeneration mediators.1–6 For example, Patel et al81 showed that the application of 10 nm AuNPs to mesenchymal stem cells from human adipose tissue regulates muscle cell differentiation within 7 days via the expression of myogenic regulatory factors. In another study, Gê et al75 showed that AuNP hydrogels with a mean diameter of 30 to 40 nm and concentration between 20 and 80 µg/mL caused increases in the myogenic differentiation of C2C12 myoblasts through the formation of myotubes and upregulation of the expression of myogenesis-inducing genes. Recently, Ko et al93 applied 3 to 5 nm AuNPs to rat aortic vascular smooth muscle cells and revealed that the treatment increased the expression and phosphorylation of NFR2 mediated by a redox-related reaction and p38 MAPK activation.
A variety of in vivo studies on the use of AuNPs for muscle regeneration have been conducted in rats5,8 and9 and mice.10,11,15 Most in vivo studies have used AuNPs at 10 to 30 nm, although variable sizes between 0.6 and 400 nm have been observed. The dosages used are also varied, with a predominance of 20 mg/kg.85,86,94,95 Raimondo and Mooney15 analyzed the effects of the injectable application of 30, 60 and 100 nm AuNPs conjugated to IL4 on the regeneration of the tibialis anterior muscle of C57BL/6J female mice after an ischemic injury model and revealed that the use of these nanoparticles favors the polarization of macrophages M1 to M2 and increases muscle regeneration, strength and contraction speed. AuNP rods at 30 nm (diameter) and 4500 nm (length) were added to hydrogel and applied to the temporal muscle of Sprague-Dawley rats, and the results showed better actin filament restructuring, greater myosin formation, and less fibrosis.84 Effective results on gastrocnemius regeneration in Wistar Rats were also observed by Da Rocha et al85 with the use of AuNPs with a size of 20 nm and concentration of 20 mg/kg, and these effects were associated with the microcurrent after a muscle injury. Moreover, the use of AuNPs restored the tissue histoarchitecture and promoted a reduction in proinflammatory and oxidative stress markers.
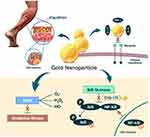
The effects of using AuNPs as an anti-inflammatory agent in regenerating muscle are due to the ability of AuNPs to permeate muscle tissue and interact with different molecules, which synergistically reduces the production of cellular oxidants (Figure 3). For example, Zortea et al82 investigated the effect of ultrasound with AuNPs on inflammatory and oxidative stress parameters in an experimental model of muscle overuse. The anti-inflammatory action observed in this study was related to the ability of AuNPs to inhibit the expression of NF-kB, reduce the binding of inflammatory cytokines to their membrane receptors, and downregulate vascular endothelial growth factor (VEGF) expression, thereby reducing cellular infiltration. The authors also suggested that the antioxidant effects may be secondary to the anti-inflammatory action. A lower proinflammatory response results in lower production of oxidants, primarily from the nicotinamide adenine dinucleotide phosphate-oxidase (NADPH-oxidase) complex. In addition, AuNPs cause an increase in the endogenous production of antioxidants. These effects may be related to the ability of AuNPs to interact with the thiol group of Kelch-like ECH-associated protein 1 (KEAP1), thus allowing for the translocation of nuclear factor erythroid 2-related factor 2 (NRF2) for subsequent transcription of antioxidant genes.46,96
Silveira et al97 analyzed the effect of phonophoresis using ultrasound associated with AuNPs and dimethyl sulfoxide (DMSO) on oxidative stress parameters after trauma using a contusion model. The authors suggested that the benefits arising from the use of AuNPs may be related to the inhibition of nitric oxide synthase inducible (iNOS) production, suppression of ROS, and potential reduction in the production of CD68, which favored muscle tissue repair. In addition, AuNPs can increase myogenic differentiation by activating the p38 MAPK signaling pathway through the expression of myosin/MHC heavy chain proteins75 or by targeting and activating regulatory genes of myogenesis, such as MyoD, myogenin, Myf5, and myogenic regulatory factor (Mrf).81 When associated with anti-inflammatory cytokines, such as IL4, AuNPs can induce the polarization of M2 macrophages and promote the infiltration of immunosuppressive/proregenerative regulatory T cells (Tregs), thus favoring the muscle regeneration process.15,81
Conclusion and Perspectives
Although several in vitro studies and investigations in healthy and sick animals have reported the therapeutic efficacy of AuNPs based on different mechanisms underlying muscle regeneration, the extrapolation of these results to humans has not been performed. Future investigations are needed to better understand the safety and efficacy of AuNPs for different aspects of muscle physiology. Despite the research-based evidence presented here on the anti-inflammatory and antioxidant effects of AuNPs on muscle injury, treatment methods that apply these results and new applications have not yet been developed. Moreover, current data from different studies are fragmented from the experimental methods to the final conclusions, which has introduced confusion and uncertainty to the applicability of AuNPs. Although AuNPs may represent a therapeutic target of clinical relevance for living beings, their potential toxicity must be carefully and precisely studied. Several studies have reported that AuNPs are nontoxic, while others have shown dose- and route-dependent toxic effects. Moreover, the toxicity of AuNPs associated with ligands must be considered since certain cationic ligands seem to induce toxic effects in living organisms independent of AuNPs. Therefore, investigating the therapeutic use of AuNPs in muscle injury may be a promising approach, although it requires further preclinical and applied research, especially in terms of the formulation of new molecules and the association with ligands that transport these molecules to the target tissue and help tissue regeneration. Future challenges include evaluating the effects of AuNPs on the treatment of muscle injury in humans and determining the best shape, size, dose, and administration method based on the type, severity, and chronicity of the muscle injury.
Methodological Considerations About This Revision
This manuscript was written using data from several databases, including PubMed, Medline, Bireme, Web of Science, and Google Scholar, and a broad range of synonyms and related terms were applied, namely, AuNPs, inflammation, oxidative stress, muscle-skeletal, and muscle damage. We included prospective cohort studies, cross-sectional studies, randomized clinical trials, experimental models, systematic revisions, and meta-analyses. For studies related to the effects of AuNPs on muscle, we considered the following eligibility criteria: (1) studies reporting human participants or animals (rats and mice); (2) search outputs that included only articles that were peer-reviewed and published in English language journals; and (3) papers that included parameters related to the effects of AuNPs on skeletal muscle. For each study, the study characteristics (authors, published year, and journal), specimens (human, rat, or mouse), study design (experimental design and groups), general aims, AuNP protocol (synthesis, type, size, dose, concentration), samples (tissue), biomarkers, and main outcomes were only extracted from studies related to the effects of AuNPs on skeletal muscle. The results of this search were organized in an electronic spreadsheet for further analysis.
Acknowledgments
The authors wish to thank the Pontifical Catholic University of Paraná (Brazil), the Universidade do Extremo Sul Catarinense (Brazil), and Coordination for the Improvement of Higher Education Personnel (Brazil) for their support in the studies cited in this manuscript.
Author Contributions
All authors made a significant contribution to the work reported, such as in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; participated in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; agreed on the journal to which the article has been submitted; and agreed to be accountable for all aspects of the work.
Funding
This research was supported by the Brazilian National Council for Scientific and Technological Development (grant number 307669/2019-0).
Disclosure
The authors declare no conflicts of interest.
References
1. Paula MM, Petronilho F, Vuolo F, et al. Gold nanoparticles and/or N-acetylcysteine N -acetylcysteine mediate carrageenan-induced inflammation and oxidative stress in a concentration-dependent manner. J Biomed Mater Res A. 2015;103(10):3323–3330. doi:10.1002/jbm.a.35469
2. Haupenthal D, Dias FM, Zaccaron RP, et al. Effects of phonophoresis with Ibuprofen associated with gold nanoparticles in animal model of traumatic muscle injury. Eur J Pharm Sci. 2019;143:105120. doi:10.1016/j.ejps.2019.105120
3. Tartuce LP, Brandt FP, Dos Santos Pedroso G, et al. 2-methoxy-isobutyl-isonitrile-conjugated gold nanoparticles improves redox and inflammatory profile in infarcted rats. Colloids Surf B. 2020;192:111012. doi:10.1016/j.colsurfb.2020.111012
4. Chugh H, Sood D, Chandra I, Tomar V, Dhawan G, Chandra RJA. Role of gold and silver nanoparticles in cancer nano-medicine. Artif Cells Nanomed Biotechnol. 2018;46(sup1):1210–1220. doi:10.1080/21691401.2018.1449118
5. Lombardo D, Kiselev MA, Caccamo MTJ. Smart nanoparticles for drug delivery application: development of versatile nanocarrier platforms in biotechnology and nanomedicine. J Nanomater. 2019;2019:1–26. doi:10.1155/2019/3702518
6. Bromma K, Chithrani DBJ. Advances in gold nanoparticle-based combined cancer therapy. Nanomaterials. 2020;10(9):1671.
7. Singh P, Pandit S, Mokkapati V, Garg A, Ravikumar V, Mijakovic IJI. Gold nanoparticles in diagnostics and therapeutics for human cancer. Int J Mol Sci. 2018;19:1979.
8. Ojea-Jiménez I, Romero FM, Bastús NG, Puntes VJT. Small gold nanoparticles synthesized with sodium citrate and heavy water: insights into the reaction mechanism. J Phys Chem. 2010;114(4):1800–1804.
9. Elbialy NS, Fathy MM, Khalil WMJ. Doxorubicin loaded magnetic gold nanoparticles for in vivo targeted drug delivery. Int J Pharm. 2015;490(1–2):190–199. doi:10.1016/j.ijpharm.2015.05.032
10. Gatoo MA, Naseem S, Arfat MY, Mahmood Dar A, Qasim K, Zubair SJB. Physicochemical properties of nanomaterials: implication in associated toxic manifestations. BioMed Res Int. 2014;2014. doi:10.1155/2014/498420
11. Della Vechia IC, Steiner BT, Freitas ML, et al. Comparative cytotoxic effect of citrate-capped gold nanoparticles with different sizes on noncancerous and cancerous cell lines. J Nanoparticle Res. 2020;22:133.
12. Sani A, Cao C, Cui DJB. Toxicity of gold nanoparticles (AuNPs): a review. Biochem Biophys Rep. 2021;26:100991. doi:10.1016/j.bbrep.2021.100991
13. Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662–668. doi:10.1021/nl052396o
14. Labouta HI, el-Khordagui LK, Kraus T, Schneider M. Mechanism and determinants of nanoparticle penetration through human skin. Nanoscale. 2011;3(12):4989–4999. doi:10.1039/c1nr11109d
15. Raimondo TM, Mooney DJ. Functional muscle recovery with nanoparticle-directed M2 macrophage polarization in mice. Proc Natl Acad Sci U S A. 2018;115(42):10648–10653. doi:10.1073/pnas.1806908115
16. Boisselier E, Astruc D. Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem Soc Rev. 2009;38(6):1759–1782. doi:10.1039/b806051g
17. Menon S, Rajeshkumar S, Kumar VJR. A review on biogenic synthesis of gold nanoparticles, characterization, and its applications. Resor Effic Technol. 2017;3:516–527.
18. Sacks D, Baxter B, Campbell BCV, et al. Multisociety consensus quality improvement revised consensus statement for endovascular therapy of acute ischemic stroke. Int J Stroke. 2018;13(6):612–632. doi:10.1177/1747493018778713
19. Oberdörster G, Oberdörster E, Oberdörster J. Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ Health Perspect. 2005;113(7):823–839. doi:10.1289/ehp.7339
20. Aillon KL, Xie Y, El-Gendy N, Berkland CJ, Forrest ML. Effects of nanomaterial physicochemical properties on in vivo toxicity. Adv Drug Deliv Rev. 2009;61(6):457–466. doi:10.1016/j.addr.2009.03.010
21. Chen Y-S, Hung Y-C, Liau I, Huang GS. Assessment of the in vivo toxicity of gold nanoparticles. Nanoscale Res Lett. 2009;4(8):858. doi:10.1007/s11671-009-9334-6
22. Stiufiuc R, Iacovita C, Nicoara R, et al. One-step synthesis of PEGylated gold nanoparticles with tunable surface charge. J Nanomater. 2013;2013.
23. Albanese A, Tang PS, Chan WC. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16. doi:10.1146/annurev-bioeng-071811-150124
24. Adewale OB, Davids H, Cairncross L, Roux SJI. Toxicological behavior of gold nanoparticles on various models: influence of physicochemical properties and other factors. Int J Toxicol. 2019;38(5):357–384. doi:10.1177/1091581819863130
25. Xie X, Liao J, Shao X, Li Q, Lin YJS. The effect of shape on cellular uptake of gold nanoparticles in the forms of stars, rods, and triangles. Sci Rep. 2017;7:1–9.
26. Xia Q, Huang J, Feng Q, et al. Size-and cell type-dependent cellular uptake, cytotoxicity and in vivo distribution of gold nanoparticles. Int J Nanomedicine. 2019;14(6957).
27. Gupta R, Rai BJS. Effect of size and surface charge of gold nanoparticles on their skin permeability: a molecular dynamics study. Sci Rep. 2017;7:1–13.
28. Fornaguera C, Feiner-Gracia N, Dols-Perez A, García-Celma MJ, Solans CJP. Versatile methodology to encapsulate gold nanoparticles in plga nanoparticles obtained by nano-emulsion templating. Pharm Res. 2017;34(5):1093–1103. doi:10.1007/s11095-017-2119-1
29. Foroozandeh P, Aziz AAJ. Insight into cellular uptake and intracellular trafficking of nanoparticles. Nanoscale Res Lett. 2018;13:1–12.
30. Spampinato V, Parracino MA, La Spina R, Rossi F, Ceccone GJF. Surface analysis of gold nanoparticles functionalized with thiol-modified glucose SAMs for biosensor applications. Front Chem. 2016;4:8. doi:10.3389/fchem.2016.00008
31. Yeh YC, Creran B, Rotello VM. Gold nanoparticles: preparation, properties, and applications in bionanotechnology. Nanoscale. 2012;4(6):1871–1880. doi:10.1039/C1NR11188D
32. Cardoso Avila PE, Rangel Mendoza A, Pichardo Molina JL, et al. Biological response of HeLa cells to gold nanoparticles coated with organic molecules. Toxicol In Vitro. 2017;42:114–122. doi:10.1016/j.tiv.2017.04.013
33. Zhang J, Mou L, Jiang X. Surface chemistry of gold nanoparticles for health-related applications. Chem Sci. 2020;11(4):923–936. doi:10.1039/C9SC06497D
34. Li T, Wang Y, Wang M, et al. Impact of albumin pre-coating on gold nanoparticles uptake at single-cell level. Nanomaterials. 2022;12:749.
35. Özçiçek İ, Ulaş Aytürk N, Aysit N. Histological changes in mice tissues induced by gold nanoparticles with different surface coatings and sizes. Exp Biol Med. 2022;5:73–87.
36. Bednarski M, Dudek M, Knutelska J, et al. The influence of the route of administration of gold nanoparticles on their tissue distribution and basic biochemical parameters: in vivo studies. Pharmacol Rep. 2015;67(3):405–409. doi:10.1016/j.pharep.2014.10.019
37. Commoner B, Townsend J, Pake GEJ. Free radicals in biological materials. Nature. 1954;174(4432):689–691. doi:10.1038/174689a0
38. Gerschman R, Gilbert DL, Nye SW, Dwyer P, Fenn WOJ. Oxygen poisoning and x-irradiation: a mechanism in common. Science. 1954;119(3097):623–626. doi:10.1126/science.119.3097.623
39. Harman DJF. Free radical theory of aging: history. Free Radic Aging. 1992;275:1–10.
40. McCord JM, Fridovich IJJ. Superoxide dismutase: an enzymic function for erythrocuprein (hemocuprein). J Biol Chem. 1969;244(22):6049–6055.
41. Murad FJA. Discovery of some of the biological effects of nitric oxide and its role in cell signaling (Nobel lecture). Biosci Rep. 1856–1868;1999(38).
42. Sies HJR. Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: oxidative eustress. Redox Biol. 2017;11:613–619.
43. Lin J, Oh SH, Jones R, et al. The peptide-binding cavity is essential for Als3-mediated adhesion of Candida albicans to human cells. J Biol Chem. 2014;289(26):18401–18412. doi:10.1074/jbc.M114.547877
44. Wu J, Wang X, Wang Q, et al. Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymes (II). Chem Soc Rev. 2019;48(4):1004–1076. doi:10.1039/c8cs00457a
45. Sims CM, Hanna SK, Heller DA, et al. Redox-active nanomaterials for nanomedicine applications. Nanoscale. 2017;9(15226–15251).
46. Liu Y, Shi JJN. Antioxidative nanomaterials and biomedical applications. Nano Today. 2019;27:146–177.
47. Thirupathi A, Pinho RA, Ugbolue UC, He Y, Meng Y, Gu YJ. Effect of running exercise on oxidative stress biomarkers: a systematic review. Front Physiol. 2021;1789.
48. Nethi SK, Barui AK, Mukherjee S, Patra CRJA, signaling R. Engineered nanoparticles for effective redox signaling during angiogenic and antiangiogenic therapy. Antioxid Redox Signal. 2019;30(5):786–809. doi:10.1089/ars.2017.7383
49. Tarin C, Carril M, Martin-Ventura JL, et al. Targeted gold-coated iron oxide nanoparticles for CD163 detection in atherosclerosis by MRI. Sci Rep. 2015;5:1–9.
50. Subramaniam SR, Chesselet M-F. Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog Neurobiol. 2013;106:17–32. doi:10.1016/j.pneurobio.2013.04.004
51. Jawaid P, Rehman MU, Zhao Q-L, et al. Small size gold nanoparticles enhance apoptosis-induced by cold atmospheric plasma via depletion of intracellular GSH and modification of oxidative stress. Cell Death Discov. 2020;6:1–12.
52. Ding F, Li Y, Liu J, et al. Overendocytosis of gold nanoparticles increases autophagy and apoptosis in hypoxic human renal proximal tubular cells. Int J Nanomedicine. 2014;9:4317. doi:10.2147/IJN.S68685
53. Alexis F, Pridgen E, Molnar LK, Farokhzad OCJ. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol Pharm. 2008;5(4):505–515. doi:10.1021/mp800051m
54. Coulter JA, Jain S, Butterworth KT, et al. Cell type-dependent uptake, localization, and cytotoxicity of 1.9 nm gold nanoparticles. Int J Nanomedicine. 2012;7:2673. doi:10.2147/IJN.S31751
55. Rezatabar S, Karimian A, Rameshknia V, et al. RAS/MAPK signaling functions in oxidative stress, DNA damage response and cancer progression. J Cell Physiol. 2019;234(14951–14965).
56. Tian F, Clift MJ, Casey A, et al. Investigating the role of shape on the biological impact of gold nanoparticles in vitro. Nanomedicine. 2015;10(17):2643–2657. doi:10.2217/nnm.15.103
57. Schaeublin NM, Braydich-Stolle LK, Schrand AM, et al. Surface charge of gold nanoparticles mediates mechanism of toxicity. Nanoscale. 2011;3(2):410–420. doi:10.1039/c0nr00478b
58. Durocher I, Noël C, Lavastre V, Girard DJI. Evaluation of the in vitro and in vivo proinflammatory activities of gold (+) and gold (−) nanoparticles. Inflamm Res. 2017;66(11):981–992. doi:10.1007/s00011-017-1078-7
59. Vreeken V, Siegler MA, van der Vlugt JI. Controlled interconversion of a dinuclear au species supported by a redox-active bridging PNP ligand facilitates ligand-to-gold electron transfer. Chemistry. 2017;23(23):5585–5594. doi:10.1002/chem.201700360
60. Sun C, Mirzadeh N, Guo SX, et al. Electrochemical interconversion of Au (I)–Au (I), Au (II)–Au (II), and Au (I)–Au (III) in binuclear complexes containing the carbanionic ligand C6F4PPh2. Inorg Chem. 2019;58(20):13999–14004. doi:10.1021/acs.inorgchem.9b01983
61. Li H, Ye X, Guo X, Geng Z, Wang G. Effects of surface ligands on the uptake and transport of gold nanoparticles in rice and tomato. J Hazard Mater. 2016;314:188–196. doi:10.1016/j.jhazmat.2016.04.043
62. Lopez-Sanchez JA, Dimitratos N, Hammond C, et al. Facile removal of stabilizer-ligands from supported gold nanoparticles. Nat Chem. 2011;3(7):551–556. doi:10.1038/nchem.1066
63. Zhang XX, Liao C. Perspectives in medicinal chemistry: current progress in the development of metalloprotein inhibitors. Curr Top Med Chem. 2015;16(5):467–469. doi:10.2174/156802661605151022150043
64. Wen X, Wu J, Wang F, et al. Deconvoluting the role of reactive oxygen species and autophagy in human diseases. Free Radic Biol Med. 2013;65:402–410. doi:10.1016/j.freeradbiomed.2013.07.013
65. Scherz‐Shouval R, Shvets E, Fass E, Shorer H, Gil L, Elazar ZJTE. Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. EMBO J. 2007;26(7):1749–1760. doi:10.1038/sj.emboj.7601623
66. Eleftheriadou D, Kesidou D, Moura F, Felli E, Song WJS. Redox‐responsive nanobiomaterials‐based therapeutics for neurodegenerative diseases. Small. 2020;16(1907308). doi:10.1002/smll.201907308
67. Heckert EG, Karakoti AS, Seal S, Self WTJ. The role of cerium redox state in the SOD mimetic activity of nanoceria. Biomaterials. 2008;29(18):2705–2709. doi:10.1016/j.biomaterials.2008.03.014
68. Aioub M, Panikkanvalappil SR, El-Sayed MAJ. Platinum-coated gold nanorods: efficient reactive oxygen scavengers that prevent oxidative damage toward healthy, untreated cells during plasmonic photothermal therapy. ACS Nano. 2017;11(579–586):579–586. doi:10.1021/acsnano.6b06651
69. Ma Y, Fu H, Zhang C, et al. Chiral antioxidant-based gold nanoclusters reprogram DNA epigenetic patterns. Sci Rep. 2016;6:1–12.
70. Mueller A, Bullich S, Barret O, et al. Tau PET imaging with 18F-PI-2620 in patients with Alzheimer disease and healthy controls: a first-in-humans study. J Nucl Med. 2020;61(6):911–919. doi:10.2967/jnumed.119.236224
71. Hou K, Zhao J, Wang H, et al. Chiral gold nanoparticles enantioselectively rescue memory deficits in a mouse model of Alzheimer’s disease. Nat Commun. 2020;11:1–11.
72. Yang L, Yin T, Liu Y, Sun J, Zhou Y, Liu J. Gold nanoparticle-capped mesoporous silica-based H(2)O(2)-responsive controlled release system for Alzheimer’s disease treatment. Acta Biomater. 2016;46:177–190. doi:10.1016/j.actbio.2016.09.010
73. Limbach LK, Wick P, Manser P, et al. Exposure of engineered nanoparticles to human lung epithelial cells: influence of chemical composition and catalytic activity on oxidative stress. Environ Sci Technol. 2007;41(11):4158–4163. doi:10.1021/es062629t
74. Acres RG, Feyer V, Tsud N, Carlino E, Prince KCJ. Mechanisms of aggregation of cysteine functionalized gold nanoparticles. J Phys Chem C. 2014;118:10481–10487.
75. Ge J, Liu K, Niu W, et al. Gold and gold-silver alloy nanoparticles enhance the myogenic differentiation of myoblasts through p38 MAPK signaling pathway and promote in vivo skeletal muscle regeneration. J Biomaterials. 2018;175:19–29.
76. Pan Y, Leifert A, Ruau D, et al. Gold nanoparticles of diameter 1.4 nm trigger necrosis by oxidative stress and mitochondrial damage. Small. 2009;5(18):2067–2076. doi:10.1002/smll.200900466
77. Mukherjee P, Bhattacharya R, Wang P, et al. Antiangiogenic properties of gold nanoparticles. Clin Cancer Res. 2005;11(9):3530–3534. doi:10.1158/1078-0432.CCR-04-2482
78. Ishihara J, Ishihara A, Fukunaga K, et al. Laminin heparin-binding peptides bind to several growth factors and enhance diabetic wound healing. Nat Commun. 2018;9:1–14.
79. Victor EG, Silveira PC, Possato JC, et al. Pulsed ultrasound associated with gold nanoparticle gel reduces oxidative stress parameters and expression of pro-inflammatory molecules in an animal model of muscle injury. J Nanobiotechnology. 2012;10(1):11. doi:10.1186/1477-3155-10-11
80. Leite PEC, Pereira MR, Santos CAD, Campos APC, Esteves TM, Granjeiro JM. Gold nanoparticles do not induce myotube cytotoxicity but increase the susceptibility to cell death. Toxicol Vitro. 2015;29(5):819–827. doi:10.1016/j.tiv.2015.02.010
81. Patel S, Yin PT, Sugiyama H, Lee KB. Inducing stem cell myogenesis using nanoscript. ACS Nano. 2015;9(7):6909–6917.
82. Zortéa D, Silveira PCL, Souza PS, et al. Effects of phonophoresis and gold nanoparticles in experimental model of muscle overuse: role of oxidative stress. Ultrasound Med Biol. 2015;41(1):151–162.
83. Silveira PCL, Victor EG, Notoya FD, et al. Effects of phonophoresis with gold nanoparticles on oxidative stress parameters in a traumatic muscle injury model. Drug Deliv. 2016;23(3):926–932. doi:10.3109/10717544.2014.923063
84. Kim W, Jang CH, Kim GH. A myoblast-laden collagen bioink with fully aligned au nanowires for muscle-tissue regeneration. Nano Lett. 2019;19(12):8612–8620. doi:10.1021/acs.nanolett.9b03182
85. Da Rocha FR, Haupenthal DPD, Zaccaron RP, et al. Therapeutic effects of iontophoresis with gold nanoparticles in the repair of traumatic muscle injury. J Drug Target. 2020;28(3):307–319. doi:10.1080/1061186X.2019.1652617
86. Haupenthal D, Possato JC, Zaccaron RP, et al. Effects of chronic treatment with gold nanoparticles on inflammatory responses and oxidative stress in Mdx mice. J Drug Target. 2020;28(1):46–54. doi:10.1080/1061186X.2019.1613408
87. Haupenthal DPD, Zortea D, Zaccaron RP, et al. Effects of phonophoresis with diclofenac linked gold nanoparticles in model of traumatic muscle injury. Mater Sci Eng C Mater Biol Appl. 2020;110:13.
88. Raimondo TM, Mooney DJ. Anti-inflammatory nanoparticles significantly improve muscle function in a murine model of advanced muscular dystrophy. Sci Adv. 2021;7(26):10. doi:10.1126/sciadv.abh3693
89. Thirupathi A, Sorato HR, Silva PRL, et al. Effect of taurine associated gold nanoparticles on oxidative stress in muscle of mice exposed to overuse model. An Acad Bras Cienc. 2021;93(2):12. doi:10.1590/0001-3765202120191450
90. Zhang YP, Le Friec A, Chen ML. 3D anisotropic conductive fibers electrically stimulated myogenesis. Int J Pharm. 2021;606:10. doi:10.1016/j.ijpharm.2021.120841
91. Järvinen TA, Järvinen TL, Kääriäinen M, et al. Muscle injuries: optimising recovery. Best Pract Res Clin Rheumatol. 2007:21(317–331):317–331.
92. Tidball JG, Villalta SA. Regulatory interactions between muscle and the immune system during muscle regeneration. Am J Physiol Regul Integr Comp Physiol. 2010;298(5):R1173–R1187. doi:10.1152/ajpregu.00735.2009
93. Ko WC, Shieh JM, Wu WB. P38 MAPK and Nrf2 activation mediated naked gold nanoparticle induced heme oxygenase-1 expression in rat aortic vascular smooth muscle cells. Arch Med Res. 2020;51(5):388–396. doi:10.1016/j.arcmed.2020.04.015
94. Islam NU, Khan I, Rauf A, Muhammad N, Shahid M, Shah MR. Antinociceptive, muscle relaxant and sedative activities of gold nanoparticles generated by methanolic extract of Euphorbia milii. BMC Complement Altern Med. 2015;15(1):160. doi:10.1186/s12906-015-0691-7
95. Sun PP, Lai CS, Hung CJ, et al. Subchronic oral toxicity evaluation of gold nanoparticles in male and female mice. Heliyon. 2021;7(3):e06577. doi:10.1016/j.heliyon.2021.e06577
96. Yu Z-M, Wan X-M, Xiao M, Zheng C, Zhou X-L. Puerarin induces Nrf2 as a cytoprotective mechanism to prevent cadmium-induced autophagy inhibition and NLRP3 inflammasome activation in AML12 hepatic cells. J Inorg Biochem. 2021;217:111389. doi:10.1016/j.jinorgbio.2021.111389
97. Silveira PC, Venâncio M, Souza PS, et al. Iontophoresis with gold nanoparticles improves mitochondrial activity and oxidative stress markers of burn wounds. Mater Sci Eng C Mater Biol Appl. 2014;44:380–385. doi:10.1016/j.msec.2014.08.045
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.