Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 17
Differential Outcomes Following 4 Weeks of Aclidinium/Formoterol in Patients with COPD: A Reanalysis of the ACTIVATE Study
Authors Koopman M , Franssen FME , Gaffron S , Watz H, Troosters T, Garcia-Aymerich J , Paggiaro P, Molins E, Moya M, van Burk L, Maier D, Garcia Gil E, Wouters EFM, Vanfleteren LEGW , Spruit MA
Received 1 June 2021
Accepted for publication 21 February 2022
Published 8 March 2022 Volume 2022:17 Pages 517—533
DOI https://doi.org/10.2147/COPD.S308600
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Maud Koopman,1– 3 Frits ME Franssen,1– 3 Swetlana Gaffron,4 Henrik Watz,5 Thierry Troosters,6,7 Judith Garcia-Aymerich,8– 10 Pierluigi Paggiaro,11 Eduard Molins,12 Miguel Moya,12 Lindy van Burk,13 Dieter Maier,14 Esther Garcia Gil,12 Emiel FM Wouters,1,3,15 Lowie EGW Vanfleteren,16 Martijn A Spruit1– 3
1Department of Research and Development, CIRO+, Center of Expertise for Chronic Organ Failure, Horn, the Netherlands; 2NUTRIM, School of Nutrition and Translational Research in Metabolism, Faculty of Health, Medicine and Life Sciences, Maastricht, the Netherlands; 3Department of Respiratory Medicine, Maastricht University Medical Center (MUMC+), Maastricht, the Netherlands; 4Viscovery Software GmbH, Vienna, Austria; 5Pulmonary Research Institute at LungenClinic Grosshansdorf, Airway Research Center North (ARCN), Member of the German Center for Lung Research (DZL), Grosshansdorf, Germany; 6Department of Rehabilitation Sciences, KU Leuven – University of Leuven, Leuven, Belgium; 7Department of Respiratory Diseases, University Hospitals Leuven, Leuven, Belgium; 8Barcelona Institute for Global Health (ISGlobal), Barcelona, Spain; 9Universitat Pompeu Fabra (UPF), Barcelona, Spain; 10CIBER Epidemiología y Salud Publica (CIBERESP), Madrid, Spain; 11Department of Surgery, Medicine, Molecular Biology and Critical Care, University of Pisa, Pisa, Italy; 12AstraZeneca, Barcelona, Spain; 13AstraZeneca, Den Haag, the Netherlands; 14Biomax Informatics AB, Planegg, 82152, Germany; 15Ludwig Boltzmann Institute for Lung Health, Vienna, Austria; 16COPD Center, Sahlgrenska University Hospital, Institute of Medicine, University of Gothenburg, Gothenburg, Sweden
Correspondence: Maud Koopman, CIRO+, Center of Expertise for Chronic Organ Failure, Hornerheide 1, Horn, 6085 NM, the Netherlands, Email [email protected]
Rationale: It is difficult to predict the effects of long-acting bronchodilators (LABD) on lung function, exercise capacity and physical activity in patients with chronic obstructive pulmonary disease (COPD). Therefore, the multidimensional response to LABD was profiled in COPD patients participating in the ACTIVATE study and randomized to LABD.
Methods: In the ACTIVATE study, patients were randomized to aclidinium bromide/formoterol fumarate (AB/FF) or placebo for four weeks. The primary outcomes included (1) lung function as measured by functional residual capacity (FRC), residual volume (RV), and spirometric outcomes; (2) exercise performance as measured by a constant work rate cycle ergometry test (CWRT); and (3) physical activity (PA) using an activity monitor. Self-organizing maps (SOMs) were used to create an ordered representation of the patients who were randomly assigned to four weeks of AB/FF and cluster them into different outcome groups.
Results: A total of 250 patients were randomized to AB/FF (n = 126) or placebo (n = 124). Patients in the AB/FF group (39.6% women) had moderate-to-severe COPD, static hyperinflation (FRC: 151.4 (27.7)% predicted) and preserved exercise capacity. Six clusters with differential outcomes were identified. Patients in clusters 1 and 2 had significant improvements in lung function compared to the remaining AB/FF-treated patients. Patients in clusters 1 and 3 had significant improvements in CWRT time, and patients in clusters 2, 3 and 6 had significant improvements in PA compared to the remaining AB/FF-treated patients.
Conclusion: Individual responses to 4 weeks of AB/FF-treatment in COPD are differential and the degree of change differs across domains of lung function, exercise capacity and PA. These results indicate that clinical response to LABD therapy is difficult to predict and is non-linear, and show doctors that it is important to look at multiple outcomes simultaneously when evaluating the clinical response to LABD therapy.
Clinical Trial Registration: The original ACTIVATE study was registered on ClinicalTrials.gov, registration number NCT02424344.
Keywords: COPD, hyperinflation, physical activity
Introduction
Long-acting bronchodilators (LABDs) are the cornerstone of pharmacological therapy in patients with mild to very severe chronic obstructive pulmonary disease (COPD),1 as they generally increase the forced expiratory volume in the first second (FEV1),2,3 lower the degree of breathlessness,4,5 improve exercise performance6–9 and health status,3,5 and reduce exacerbation rates.3,10 The effects of LABDs on lung hyperinflation and objectively measured physical activity (PA) remain indecisive.2–4,9,11–14
The benefits of LABD therapy can vary to a great extent between patients with COPD.5 To date, it is difficult to truly understand which patients do or do not benefit from LABDs, as studies only show aggregated data from individuals. Individual patients may respond differentially regarding a variety of clinically relevant outcomes, with variable degree and direction of outcome effect, suggesting non-linearity. Statistical methods, such as self-organizing maps (SOMs), can be used to create an ordered representation of multi-dimensional data, to simplify complexity and to reveal meaningful associations.15–17 The use of composite endpoints (eg, change in lung function (≥100 mL in FEV1), Health status (≥4-units in St George’s Respiratory Questionnaire [SGRQ]), and the incidence of moderate/severe exacerbations) for the assessment of clinically important deterioration in COPD was recently proposed.18 However, the response to LABD therapy on multiple outcomes simultaneously has scarcely been investigated in COPD.19 This, however, will provide a greater insight in the response to LABDs in COPD, and will advance the field by identifying the appropriate subgroup of patients for the appropriate treatment. In turn, this might help clinicians provide their patients with the right treatment, by evaluating the treatment response through various outcomes. Using the data from the ACTIVATE study,9 where the effect of LABD therapy on lung hyperinflation, exercise endurance and PA was studied, we aimed to identify multidimensional outcome patterns for LABD therapy in patients with COPD, including static and dynamic lung function measures, exercise performance and PA by performing a cluster analysis. Moreover, we aimed to determine the initial characteristics associated with these patterns. Based on the multiple pulmonary (ie airflow limitation, lung hyperinflation, and impaired diffusion capacity) and extra-pulmonary components (ie lower-limb muscle weakness, fatigue, depression, and anxiety) that contribute to the pathophysiology of the disease and the clinical presentation of patients with COPD, a priori, we expected a heterogenous and non-linear response to LABDs.
Methods
Study Design
The ACTIVATE study was an 8-week randomized, double-blind, placebo-controlled study investigating the effects of aclidinium bromide/formoterol fumarate (AB/FF) 400/12 µg twice daily on lung hyperinflation, exercise endurance time (EET) and PA in patients with moderate-to-severe COPD.9 The study objectives of the original manuscript were to study the effect of AB/FF versus placebo on lung hyperinflation, exercise endurance time, and physical activity in patients with moderate-to-severe COPD.
Patients who fulfilled the inclusion/exclusion criteria were randomized according to a 1:1 randomization ratio to one of the two treatment arms to randomly receive either AB/FF 400/12 µg twice daily or placebo BID for 8 weeks double-blind treatment phase. The block size of the randomization scheme was not communicated to the investigators. The length of this block was 4 to make it impossible to guess for any allocation. Randomization data were kept strictly confidential, and randomization codes were only available for data analysis just after the unblinding of the allocated treatment, ie after locking the database upon termination of the trial.
Patients received either AB/FF 400/12 µg or placebo twice daily via a multidose dry powder inhaler (Genuair™/Pressair®) (ClinicalTrials.gov number: NCT02424344). During the first four weeks of the study, patients received pharmacotherapy alone (AB/FF or placebo) and, during the second four weeks, behavioral intervention was added to both treatment arms. AB/FF demonstrated improvements in static and dynamic hyperinflation, exercise capacity and physical activity versus placebo after four weeks of treatment. We only analyzed the outcomes after the first four weeks to solely focus on the pharmacotherapy effects of AB/FF.
The original ACTIVATE trial was approved by local ethics committees (Table S1) and performed in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines.9 All patients provided written informed consent prior to conducting any study-specific procedures.
Patients
The key inclusion criteria for the ACTIVATE study were COPD patients aged ≥40 years with a functional residual capacity (FRC) ≥120%, post-bronchodilator FEV1 ≥40% and <80% of the predicted value, FEV1/FVC <70%, and a modified Medical Research Council (mMRC) dyspnea grade ≥2. Detailed inclusion and exclusion criteria are available in the primary publication.9 Two hundred and fifty patients who completed the study were randomized to AB/FF (n = 126) or placebo (n = 124), based on the actual treatment assignment. Patients who completed week four entered the analyses.
Measurements
Lung function was assessed using whole-body plethysmography (FRC, residual volume20 and specific airway conductance [sGAW]) and spirometry (forced vital capacity [FVC] and FEV1) before exercise, at baseline (pre-dose at day one on treatment), and at week four, as described previously4 in accordance with current recommendations.21–23 EET was measured using a constant work rate cycle ergometry test (CWRT) to symptom limitation at 75% of the pre-determined peak work rate (Wmax) at baseline and at 3 h post-dose at week four.4 Inspiratory capacity (IC) was measured using a spirometer at rest, during, and at the end of the CWRT. PA levels were measured at baseline and at week four using an activity monitor (DynaPort MoveMonitor, McRoberts B.V., the Hague, the Netherlands) for a period of one week, worn by patients 24 hours per day.24,25 The average number of steps taken per day, the time in moderate-intensity activities per day (>3 metabolic equivalents [MET] as estimated by the accelerometer), and the activity-related energy expenditure were recorded.4 PA experience was assessed using the Daily PROactive Physical Activity in COPD (D-PPAC) instrument, a daily recall, electronic, hybrid (patient-reported outcome (PRO) and activity monitor) tool developed by the Innovative Medicines Initiative PROactive project.24 The seven-item D-PPAC PRO was filled out by patients every evening for a week, over baseline week and over week four. The D-PPAC consists of three scores: amount domain (combining two PRO items plus two activity monitor outputs (steps per day and vector magnitude units [VMU] per minute)); the difficulty domain (five PRO items) and a total score (calculated as the average of both domain scores). Each score ranges from 0 to 100 points, where higher numbers indicate a better physical activity experience.9
Statistics
Comparison of outcomes at week four within the AB/FF treated group and between this and the placebo group included assessment of change from baseline in lung hyperinflation (FRC and RV), airflow limitation (FEV1, FVC and sGAW), EET and IC during CWRT, D-PPAC scores (total, amount and difficulty), percentage of inactive patients (<6000 steps per day) and PA time spent at different intensities (<1.5, 1.5–3.0 and >3.0 METS).9
Statistical analyses were performed using Viscovery SOMine 7.2 build 6719 by Viscovery Software GmbH (www.viscovery.net). Self-organizing maps (SOMs, also referred to as Kohonen maps)26,27 were used to create an ordered representation of the patients, who were randomly assigned to four weeks of AB/FF and cluster them into outcome groups. The SOM method can be viewed as a non-parametric regression technique that converts multi-dimensional data spaces into lower dimensional abstractions. SOM generates a non-linear representation of the data distribution and allows the user to identify homogenous data groups visually. The attributes that drive the ordering process of the patients in the map were the differences in the outcome indicators: change in pre-dose FRC (expressed in % of baseline), change in pre-dose FEV1 (expressed in % of baseline), change in isotime IC (expressed in % of baseline), change in CWRT endurance time in seconds (expressed as change from baseline), change in D-PPAC amount in arbitrary units (expressed as change from baseline), the change in number of steps/day (expressed as change from baseline) and the change in PA time spent >3 METS (expressed as change from baseline). Patients with similar outcome profiles were placed close to each other on the map. Based on the ordering in the map, the hierarchical SOM-Ward Cluster algorithm was applied, to group the patients into outcome clusters. Data records were ordered by Viscovery based on the similarity of the existing attributes. If attribute values were missing for a specific patient, this patient was matched to the SOM based on all other attributes whose values were known. Indeed, data records with missing values or invalid entries are recognized by Viscovery and treated appropriately in the analysis. No imputation of missing values is necessary when creating of an SOM model. The basic operation with an SOM is to look up the best-matching node. If an input data record is not complete (has missing values), then the look-up is limited to the available values. That is, the SOM is treated as if the nodes were shorter vectors (in math speak: the SOM is projected into the data space that consists of the available values), and then the lookup is conducted on this reduced map. This happens for each individual record. In this way, missing values are so to speak “substituted” with the lookup values of the matching nodes.
The ordering of our model was based on 7 attributes of which 1 up to 20 of 126 values were missing, ie the percentual changes in FEV1 (1 missing), FRC (5 missing), and IC isotime (12 missing), as well as the absolute changes in CWRT times (1 missing), D-PPAC amount (20 missing), Steps/day (12 missing), and Time >3 METS, [%of day] (12 missing). The SOM-Ward Cluster algorithm of Viscovery is a hybrid algorithm that applies the classical hierarchical method of Ward on top of the SOM topology. Summary variables in clinical characteristics for the study sample and for each cluster were presented as mean (standard deviation28) for quantitative variables, and percentage for discrete variables. Data of the lung function attributes, exercise and PA assessments were sigma-transformed to mitigate the effects of extreme values. The mean change per cluster from the intervention group was compared with the mean change from the whole placebo group using an unpaired t-test. Viscovery identified for each cluster the clinical characteristics and demographical factors that differ significantly from the average of the whole AB/FF-treated sample or the placebo-treated sample using the integrated two-sided t-test with a confidence of 95%.
Results
Baseline Characteristics
Two hundred and fifty patients who completed the study were randomized to AB/FF (n = 126) or placebo (n = 124), based on the actual treatment assignment. At baseline, the 126 patients in the AB/FF group (38% women) had moderate-to-severe COPD (mean (SD) post-bronchodilator FEV1: 60.3 (10.7)% predicted), severe static hyperinflation (mean (SD) FRC: 151.4 (27.7)% predicted) and were using multiple pulmonary medications before the study. Fifty-five percent of the patients were physically inactive (<6000 steps per day); and patients generally had a rather preserved exercise capacity (mean (SD) CWRT time: 452 (184) seconds). There were no differences at baseline as compared to the placebo group (Table 1).
 |
Table 1 Baseline Characteristics |
Mean Improvements Following 4 Weeks AB/FF
Compared to placebo, significant improvements in AB/FF were found for FEV1, FVC and sGaw, with a significant reduction in RV (Table 2). With AB/FF, EET improved significantly, as well as percentage of IC during exercise at isotime and at the end of exercise, indicating improvement in dynamic hyperinflation (Table 3). Moreover, significant improvements were found for D-PPAC questionnaire scores, VMU, steps per day and time spent >3 METs (Table 3). These results have been reported in more detail in the original ACTIVATE study.9
 |
Table 2 Change from Baseline (L) and % of Improvement in Pre-Dose Spirometry and Whole Body Plethysmography After 4 Weeks of Treatment |
 |
Table 3 Change from Baseline in Exercise-Related Attributes After Four Weeks of Treatment |
Multidimensional Profiling of AB/FF Group
The 126 patients in the AB/FF group were clustered into six groups with distinct multidimensional outcome profiles (Figure 1).
- Patients in Cluster 1 (n = 19) had significantly larger mean improvements in FEV1, FVC, FRC, RV, sGAW (Table 2), CWRT time, resting IC, IC isotime, post-exercise IC, resting SpO2, resting dyspnea (Table 3), and D-PPAC difficulty (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvement in D-PPAC amount was significantly smaller (Table 4). Overall, this cluster improved the most after treatment with AB/FF compared to the remaining AB/FF-treated patients (Figure 2).
- Patients in Cluster 2 (n = 17) had significantly larger mean improvements in FEV1, FRC, RV (Table 2), and D-PPAC amount (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvement in CWRT time was significantly smaller (Table 3).
- Patients in Cluster 3 (n = 25) had significantly larger mean improvements in CWRT time (Table 3), D-PPAC amount, VMU, steps per day, and time spent >3 METs (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvement in time spent <1.5 METs were significantly smaller (Table 4).
- Patients in Cluster 4 (n = 21) had significantly smaller mean improvements in FEV1 (Table 2), resting IC, IC isotime, post-exercise IC (Table 3), D-PPAC amount, VMU, steps per day, time spent 1.5–3 METs, and time spent >3 METs (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvements in FRC (Table 2) and time spent <1.5 METs (Table 4) were larger.
- Patients in Cluster 5 (n = 24) had significantly smaller mean improvements in FEV1, FVC, FRC, RV (Table 2), CWRT time (Table 3), and time spent <1.5 METs (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvements in D-PPAC amount, VMU, steps per day, time spent 1.5–3 METs, and time spent >3 METs (Table 4) were significantly larger.
- Patients in Cluster 6 (n = 20) had significantly smaller mean improvements in FRC, RV (Table 2), CWRT time, IC isotime, post-exercise IC (Table 3), D-PPAC amount, VMU, steps per day, time spent 1.5–3 METs, and time spent >3 METs (Table 4) compared to the remaining AB/FF-treated patients; while the mean improvement in time spent <1.5 METs were significantly larger (Table 4). Overall, this cluster improved the least after treatment with AB/FF compared to the remaining AB/FF-treated patients (Figure 1).
 |
Table 4 Changes in Physical Activity-Related Attributes |
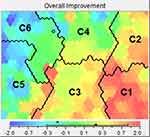 |
Figure 2 Overall improvement following 4 weeks of AB/FF. The overall improvement of the AB/FF-treated patients. The overall improvement is based on the weighted standardized improvements between initial and 4-week assessment of the seven outcome indicators, as shown in Figure 1. Using the colour scale shown below the overall improvement picture, colours can be matched to the corresponding values. Values below 0 (green towards blue) indicate a deterioration, whereas values above 0 (green towards red) indicate an overall improvement in the outcomes. |
Individual AB/FF Clusters versus Placebo
- Patients in Cluster 1 had significantly larger mean improvements in FEV1, FVC, FRC, RV, sGAW (Table 2), CWRT time, resting IC, IC isotime, post-exercise IC, resting SpO2, resting dyspnea, dyspnea isotime (Table 3), and D-PPAC difficulty (Table 4) compared to placebo.
- Patients in Cluster 2 had significantly larger mean improvements in FEV1, FVC, FRC, RV, sGAW (Table 2), resting IC, IC isotime, post-exercise IC (Table 3), D-PPAC amount, VMU, steps per day, and time spent >3 METs (Table 4) compared to placebo; while the mean improvements in CWRT time (Table 3) and time spent <1.5 METs (Table 4) were significantly smaller.
- Patients in Cluster 3 had significantly larger mean improvements in FEV1, FVC, sGAW (Table 2), CWRT time, resting IC, IC isotime, post-exercise IC, dyspnea isotime (Table 3), D-PPAC amount, VMU, steps per day, and time spent >3 METs (Table 4) compared to placebo; while the mean improvement in time spent <1.5 METs was significantly smaller (Table 4).
- Patients in Cluster 4 had significantly larger mean improvements in FEV1, FRC, RV, and sGAW compared to placebo (Table 2).
- Patients in Cluster 5 had significantly smaller mean improvements in FRC (Table 2), CWRT time (Table 3), and time spent <1.5 METs (Table 4) compared to placebo; while the mean improvements in sGAW (Table 2), resting IC, IC isotime, post-exercise IC (Table 3), D-PPAC amount, VMU, steps per day, time spent 1.5–3 METs, and time spent >3 METs (Table 4) were significantly larger.
- Patients in Cluster 6 had significantly larger mean improvements in FEV1, FVC, sGAW (Table 2), and time spent <1.5 METs (Table 4) compared to placebo; while the mean improvement in FRC was significantly smaller (Table 2).
Cluster Characteristics
Baseline characteristics of the six AB/FF-treated clusters are outlined in Table 5. There were differences in baseline characteristics between the clusters. Patients in Cluster 5 were somewhat older, had the highest proportion of physically inactive patients (<6000 steps per day), and used more medication during or 15 days prior to the study. Patients in Clusters 1 and 2 had the worst baseline degree of airflow limitation. Patients in Cluster 1 also had more static hyperinflation and the lowest CWRT time compared to the remaining AB/FF-treated patients. Patients in Cluster 4 had the highest CWRT time and spent most time of the day in 1.5–3.0 METs. There were no clinically relevant differences between the patients in Cluster 3 or 6 versus the patients in the remaining clusters.
 |
Table 5 Baseline Characteristics by Cluster |
Discussion
This is the first study to cluster patients with COPD based on multiple outcomes following four weeks of dual-bronchodilation therapy. It shows that there are differential outcomes across the domains of lung function, exercise capacity and PA following 4 weeks of AB/FF-treatment (summarizing in Table 6). Current baseline characteristics cannot confidently explain these responses to AB/FF-treatment. These results indicate that response to dual-bronchodilation therapy is difficult to predict and outcomes are differential in COPD patients with static lung hyperinflation at the start of the study. Based on the current findings, clinicians do need to consider multiple outcomes simultaneously when evaluating LABD therapy in patients with COPD.
 |
Table 6 Summarizing Changes in Outcome Indicators per Cluster |
Generally, LABD leads to an improved lung function and exercise capacity in patients with COPD,6,7,29 although the outcome can vary considerably between patients.30–32 The current results corroborate these findings (Figure 1; Table 6).
The current analyses show that specific subgroups of patients (eg, Clusters 1, 2 and 4) have a significant reduction in FRC after 4 weeks of treatment, which was greater compared to placebo. Clinicians should be aware of this important finding, as in the original ACTIVATE study, there was no significant effect of AB/FF-treatment on FRC compared to placebo.9 In contrast, patients in Clusters 5 and 6 had a worse change in FRC after four weeks of treatment compared to placebo. In a large observational study, Bhatt et al found that 9% of the COPD patients had a paradoxical lung function (decrease in FEV1 and/or FVC) response to β2-agonists.33 This was associated with respiratory morbidity, current smoking and was more common in African-Americans.33 The mechanisms underlying a paradoxical outcome remain currently unknown.
Patients in Cluster 4 showed significant improvement in static hyperinflation, but smaller mean improvements in IC at rest, isotime and post-exercise. The IC is the difference between TLC and FRC. Since it is assumed that TLC does not change in reaction to bronchodilator therapy,34 it is remarkable that IC at rest did not change particularly in these patients. The IC maneuver during exercise might be challenging for COPD patients; however, patients in Clusters 1, 2 and 3 did show significant improvements in IC at isotime. It is unclear why this was not shown in patients in Cluster 4.
Patients in almost all clusters, except for Cluster 5, showed improvement of FEV1; however, this does not always seem to correlate with reduction of lung volume at rest (Table 2; Cluster 3, 6). It has been described that the volume response of short-acting bronchodilators is reduced in patients with mild/moderate COPD, compared to patients with more severe airflow limitation,35,36 while absolute FEV1 outcome is larger in those with mild/moderate disease.36 However, baseline FEV1 in Clusters 3 and 6 was comparable to other clusters (Table 5), and thus difference in the degree of airflow limitation cannot explain the differential lung volume outcome. Moreover, the differential outcome between flow and volume also cannot be explained by a history of asthma since this was an exclusion criterion of the original study, or differences in medication since subjects in this study all used the same LABD. All studies that investigated these differential outcomes in the lung function domain used short-acting bronchodilation, while long-acting bronchodilation was used in the ACTIVATE study.
Increasing PA in patients with COPD has been challenging,20 as multiple factors influence PA, including but not limited to fatigue, mobility problems, fear, lack of drive, weather and the impact on breathing, and underestimation of the importance of physical activity.37 Interestingly, PA did not change in Cluster 1, even though these patients generally had the greatest improvements in lung function and exercise capacity. These patients did report less difficulty, which also seems to be a clinically relevant finding for patients with COPD. It provides further validation of the use of both amount and difficulty when addressing PA experience in patients with COPD. Finally, these data also show that providing optimal pharmacotherapy to patients with COPD will in most cases not be sufficient to also improve exercise capacity and/or physical activity. This is most likely due to extra-pulmonary and behavioral traits that partially determine PA and exercise capacity, which are not treated by respiratory pharmacotherapy.
Patients in Clusters 2 and 4 showed significant improvements in dynamic and static lung function, which did not transfer to a greater EET. Then again, patients in Cluster 2 were able to increase their amount of PA. This was also true for patients in Cluster 5, who also increased the amount of PA, while the change in lung function, and EET was worse compared to placebo. Patients in Cluster 3 had significant improvements in the dynamic lung function, but not in FRC, which did result in an increased EET and amount of PA. A non-linear outcome has been reported previously in patients with COPD following pulmonary rehabilitation.17
This is the first study to report a detailed non-linear outcome following LABD in patients with COPD. The non-linear outcome of LABD seems unpredictable, as the currently available clinical characteristics shown at baseline overlap between the six Clusters to a great extent. These findings emphasize the clinical complexity of the usual care for patients with COPD.38 This study shows a differential multidimensional response to LABD in a well-characterized, homogeneous subgroup of patients with COPD and hyperinflation. This study also has limitations. First, because this is a well-characterized subgroup of patients and a specific bronchodilator is used, the generalizability of the results in the general COPD population and other bronchodilators is limited. There was also a lower limit to the FEV1 in the inclusion, which left out more severe COPD patients, since the researchers did not want to give subjects with very severe COPD a placebo inhaler. However, other inclusion criteria were a notable level of dyspnea (mMRC 2) and hyperinflation (FRC > 120% of predicted), which have been shown to be important factors limiting exercise capacity and PA in patients with COPD. Second, this study showed short-term effects only, and thus exacerbations, possibly the most important outcome, are not assessed, along with other clinically relevant outcomes including levels of dyspnea and quality of life. Third, the sample size is limited, and larger studies assessing multiple outcomes regarding this topic are needed to further establish the variable response of COPD patients to LABDs. Also, the limited sample size withheld us from comparing the clusters head to head. Fourth, these exploratory analyses are hypothesis-generating only, as they were not formulated a priori as an aim of the ACTIVATE study. Lastly, inhaler use errors were not assessed, which obviously do occur in patients with COPD and may, at least partially, explain the current between-group differences.39 However, the differential outcomes within clusters cannot be explained by this.
In conclusion, the current study profiled the multidimensional outcomes (including lung function, exercise capacity and physical activity) for four weeks of LABD. Patients, healthcare professionals and payers need to start realising that individual patients with COPD respond differently to LABD. To truly understand the impact of LABD in patients with COPD, multiple outcomes have to be considered simultaneously. By assessing just one or two outcomes, clinicians might miss critical information about the LABD therapy response and withhold patients from optimal therapy. Indeed, outcomes may even differ between dynamic and static lung function attributes. Moreover, a significant improvement in lung function attributes does not automatically transfer to an increased exercise capacity and/or physical activity in patients with COPD.
Abbreviation
AB/FF, aclidinium bromide/formoterol fumarate; Bpm, beats per minute; CWRT, Constant Work Rate cycle ergometry Test; COPD, chronic obstructive pulmonary disease; D-PPAC, Daily PROactive Physical Activity in COPD; EET, exercise endurance time; FEV1, forced expiratory volume in the first second; FRC, functional residual capacity; FVC, forced vital capacity; HR, heart rate; IC, inspiratory capacity; kPa, kilopascal; LABDs, long-acting bronchodilators; MET, metabolic equivalents; Min, minute; mMRC, modified Medical Research Council; PA, physical activity; PRO, patient-reported outcome; RV, residual volume; sGaw, specific airway conductance; SOMs, self-organizing maps; SpO2, peripheral capillary oxygen saturation; VMU, vector magnitude units; Wmax, peak work rate.
Data Sharing Statement
Scientists who are interested in doing analyses on the ACTIVATE database should go to https://www.mystudywindow.com/completed.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The ACTIVATE study was sponsored by Menarini Group through its affiliates Berlin-Chemie and AstraZeneca. No specific funding was received regarding this manuscript.
Disclosure
Maud Koopman reports no real or perceived conflict of interest. Frits M.E. Franssen reports grants and personal fees from AstraZeneca and Novartis; personal fees from Boehringer Ingelheim, Chiesi, GlaxoSmithKline, and TEVA, outside the submitted work. Swetlana Gaffron works at Viscovery. Viscovery provides analysis services to Ciro+. Henrik Watz reports personal fees from AZ, during the conduct of the study; grants and/or personal fees from AZ, GSK, Chiesi, Boehringer Ingelheim Novartis, and Menarini, outside the submitted work. Thierry Troosters reports personal fees from AstraZeneca, Boehringer Ingelheim, and Chiesi, outside the submitted work. Judith Garcia-Aymerich reports consulting and lecture fees from AstraZeneca (outside the submitted work) and lecture fees from Esteve and Chiesi (outside the submitted work). Pierluigi Paggiaro reports grants and/or personal fees from ALK, AstraZeneca, Chiesi, GSK, Novartis, Sanofi, Guidotti, Menarini, and Mundipharma, outside the submitted work. Eduard Molins, Miguel Moya, Lindy van Burk, and Esther Garcia Gil are AstraZeneca employees. Dieter Maier works at Biomax. Biomax provides data management and analysis services to CIRO+. Emiel F.M. Wouters reports no real or perceived conflict of interest. Lowie E.G.W. Vanfleteren reports grants and/or personal fees from AstraZeneca, Novartis, GSK, Chiesi, Menarini, Pulmonx, Fisher&Paykel, and Boehringer, outside the submitted work. Martijn A. Spruit reports grants from the Netherlands Lung Foundation, Stichting Astma Bestrijding, GSK, Boehringer Ingelheim, AstraZeneca, TEVA, and Chiesi, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease 2017 Report: GOLD executive summary. Eur Respir J. 2017;49(3):1700214. PubMed PMID: 28182564. doi:10.1183/13993003.00214-2017
2. O’Donnell DE, Casaburi R, Vincken W, et al. Effect of indacaterol on exercise endurance and lung hyperinflation in COPD. Respir Med. 2011;105(7):1030–1036. PubMed PMID: 21498063. doi:10.1016/j.rmed.2011.03.014
3. Troosters T, Sciurba FC, Decramer M, et al. Tiotropium in patients with moderate COPD naive to maintenance therapy: a randomised placebo-controlled trial. NPJ Prim Care Respir Med. 2014;24(1):14003. PubMed PMID: 24841833; PMCID: PMC4373257. doi:10.1038/npjpcrm.2014.3
4. Beeh KM, Watz H, Puente-Maestu L, et al. Aclidinium improves exercise endurance, dyspnea, lung hyperinflation, and physical activity in patients with COPD: a randomized, placebo-controlled, crossover trial. BMC Pulm Med. 2014;14(1):209. PubMed PMID: 25539654; PMCID: PMC4364572. doi:10.1186/1471-2466-14-209
5. Ni H, Moe S, Soe Z, Myint KT, Viswanathan KN. Combined Aclidinium bromide and long-acting beta2-agonist for chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev PubMed PMID: 30536566; PMCID: PMC6517126. 2018;12:CD011594. doi:10.1002/14651858.CD011594.pub2
6. Maltais F, Hamilton A, Marciniuk D, et al. Improvements in symptom-limited exercise performance over 8 h with once-daily tiotropium in patients with COPD. Chest. 2005;128(3):1168–1178. PubMed PMID: 16162703. doi:10.1378/chest.128.3.1168
7. Beeh KM, Singh D, Di Scala L, Drollmann A. Once-daily NVA237 improves exercise tolerance from the first dose in patients with COPD: the GLOW3 trial. Int J Chron Obstruct Pulmon Dis PubMed PMID: 22973092; PMCID: PMC3430121. 2012;7:503–513. doi:10.2147/COPD.S32451
8. Di Marco F, Sotgiu G, Santus P, et al. Long-acting bronchodilators improve exercise capacity in COPD patients: a systematic review and meta-analysis. Respir Res. 2018;19(1):18. PubMed PMID: 29368604; PMCID: PMC5784692. doi:10.1186/s12931-018-0721-3
9. Watz H, Troosters T, Beeh KM, et al. ACTIVATE: the effect of Aclidinium/formoterol on hyperinflation, exercise capacity, and physical activity in patients with COPD. Int J Chron Obstruct Pulmon Dis PubMed PMID: 28883722; PMCID: PMC5574699. 2017;12:2545–2558. doi:10.2147/COPD.S143488
10. Wedzicha JA, Banerji D, Chapman KR, et al. Indacaterol-Glycopyrronium versus Salmeterol-Fluticasone for COPD. N Engl J Med. 2016;374(23):2222–2234. PubMed PMID: 27181606. doi:10.1056/NEJMoa1516385
11. Watz H, Krippner F, Kirsten A, Magnussen H, Vogelmeier C. Indacaterol improves lung hyperinflation and physical activity in patients with moderate chronic obstructive pulmonary disease–a randomized, multicenter, double-blind, placebo-controlled study. BMC Pulm Med. 2014;14(1):158. PubMed PMID: 25280934; PMCID: PMC4197315. doi:10.1186/1471-2466-14-158
12. Watz H, Mailander C, Baier M, Kirsten A. Effects of indacaterol/glycopyrronium (QVA149) on lung hyperinflation and physical activity in patients with moderate to severe COPD: a randomised, placebo-controlled, crossover study (The MOVE Study). BMC Pulm Med. 2016;16(1):95. PubMed PMID: 27301417; PMCID: PMC4908762. doi:10.1186/s12890-016-0256-7
13. O’Donnell DE, Fluge T, Gerken F, et al. Effects of tiotropium on lung hyperinflation, dyspnoea and exercise tolerance in COPD. Eur Respir J. 2004;23(6):832–840. PubMed PMID: 15218994. doi:10.1183/09031936.04.00116004
14. Troosters T, Maltais F, Leidy N, et al. Effect of bronchodilation, exercise training, and behavior modification on symptoms and physical activity in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(8):1021–1032. PubMed PMID: 29664681. doi:10.1164/rccm.201706-1288OC
15. Augustin IML, Wouters EFM, Houben-Wilke S, et al. Comprehensive lung function assessment does not allow to infer response to pulmonary rehabilitation in patients with COPD. J Clin Med. 2018;8(1):27. PubMed PMID: 30591662. doi:10.3390/jcm8010027
16. Augustin IML, Spruit MA, Houben-Wilke S, et al. The respiratory physiome: clustering based on a comprehensive lung function assessment in patients with COPD. PLoS One. 2018;13(9):e0201593. PubMed PMID: 30208035; PMCID: PMC6135389 (SCO115406). doi:10.1371/journal.pone.0201593
17. Spruit MA, Augustin IM, Vanfleteren LE, et al. Differential response to pulmonary rehabilitation in COPD: multidimensional profiling. Eur Respir J. 2015;46(6):1625–1635. PubMed PMID: 26453626. doi:10.1183/13993003.00350-2015
18. Naya IP, Tombs L, Muellerova H, Compton C, Jones PW. Long-term outcomes following first short-term clinically important deterioration in COPD. Respir Res. 2018;19(1):222. PubMed PMID: 30453972; PMCID: PMC6245880. doi:10.1186/s12931-018-0928-3
19. Kostikas K, Greulich T, Mackay AJ, et al. Treatment response in COPD: does FEV1 say it all? A post hoc analysis of the CRYSTAL study. ERJ Open Res. 2019;5(1):00243–2018. PubMed PMID: 30815470; PMCID: PMC6387992. doi:10.1183/23120541.00243-2018
20. Spruit MA, Singh SJ, Garvey C, et al. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188(8):e13–64. PubMed PMID: 24127811. doi:10.1164/rccm.201309-1634ST
21. Wanger J, Clausen JL, Coates A, et al. Standardisation of the measurement of lung volumes. Eur Respir J. 2005;26(3):511–522. PubMed PMID: 16135736. doi:10.1183/09031936.05.00035005
22. Coates AL, Peslin R, Rodenstein D, Stocks J. Measurement of lung volumes by plethysmography. Eur Respir J. 1997;10(6):1415–1427. PubMed PMID: 9192953. doi:10.1183/09031936.97.10061415
23. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. PubMed PMID: 16055882. doi:10.1183/09031936.05.00034805
24. Gimeno-Santos E, Raste Y, Demeyer H, et al. The PROactive instruments to measure physical activity in patients with chronic obstructive pulmonary disease. Eur Respir J. 2015;46(4):988–1000. PubMed PMID: 26022965; PMCID: PMC4589432. doi:10.1183/09031936.00183014
25. Watz H, Waschki B, Meyer T, Magnussen H. Physical activity in patients with COPD. Eur Respir J. 2009;33(2):262–272. PubMed PMID: 19010994. doi:10.1183/09031936.00024608
26. Kohonen T. 3.1 A Qualitative Introduction to the SOM and 3.2 The Original Incremental SOM Algorithm. In: Self-Organizing Maps. Springer; 2001: 106–115.
27. Oja E, Kaski S, Eds. Kohonen Maps. Elsevier Science B.V.; 1999.
28. Bhatt SP, Soler X, Wang X, et al. Association between functional small airway disease and FEV1 decline in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2016;194(2):178–184. PubMed PMID: 26808615; PMCID: PMC5003216. doi:10.1164/rccm.201511-2219OC
29. Maltais F, Celli B, Casaburi R, et al. Aclidinium bromide improves exercise endurance and lung hyperinflation in patients with moderate to severe COPD. Respir Med. 2011;105(4):580–587. PubMed PMID: 21183326. doi:10.1016/j.rmed.2010.11.019
30. Jones PW, Rennard SI, Agusti A, et al. Efficacy and safety of once-daily Aclidinium in chronic obstructive pulmonary disease. Respir Res. 2011;12(1):55. PubMed PMID: 21518460; PMCID: PMC3098801. doi:10.1186/1465-9921-12-55
31. Burge PS, Calverley PM, Jones PW, Spencer S, Anderson JA. Prednisolone response in patients with chronic obstructive pulmonary disease: results from the ISOLDE study. Thorax. 2003;58(8):654–658. PubMed PMID: 12885977; PMCID: PMC1746769. doi:10.1136/thorax.58.8.654
32. Jones PW, Donohue JF, Nedelman J, Pascoe S, Pinault G, Lassen C. Correlating changes in lung function with patient outcomes in chronic obstructive pulmonary disease: a pooled analysis. Respir Res. 2011;12(1):161. PubMed PMID: 22206353; PMCID: PMC3287973. doi:10.1186/1465-9921-12-161
33. Bhatt SP, Wells JM, Kim V, et al. Radiological correlates and clinical implications of the paradoxical lung function response to beta(2) agonists: an observational study. Lancet Respir Med. 2014;2(11):911–918. PubMed PMID: 25217076; PMCID: PMC4306040. doi:10.1016/S2213-2600(14)70185-7
34. Rossi A, Aisanov Z, Avdeev S, et al. Mechanisms, assessment and therapeutic implications of lung hyperinflation in COPD. Respir Med. 2015;109(7):785–802. PubMed PMID: 25892293. doi:10.1016/j.rmed.2015.03.010
35. O’Donnell DE, Forkert L, Webb KA. Evaluation of bronchodilator responses in patients with “irreversible” emphysema. Eur Respir J. 2001;18(6):914–920. PubMed PMID: 11829096. doi:10.1183/09031936.01.00216501
36. Schermer T, Heijdra Y, Zadel S, et al. Flow and volume responses after routine salbutamol reversibility testing in mild to very severe COPD. Respir Med. 2007;101(6):1355–1362. PubMed PMID: 17098407. doi:10.1016/j.rmed.2006.09.024
37. Kosteli MC, Heneghan NR, Roskell C, et al. Barriers and enablers of physical activity engagement for patients with COPD in primary care. Int J Chron Obstruct Pulmon Dis. 2017;12:1019–1031. PubMed PMID: 28405162; PMCID: PMC5378459. doi:10.2147/COPD.S119806
38. Wilson T, Holt T, Greenhalgh T. Complexity science: complexity and clinical care. BMJ. 2001;323(7314):685–688. PubMed PMID: 11566836; PMCID: PMC1121241. doi:10.1136/bmj.323.7314.685
39. Sulaiman I, Cushen B, Greene G, et al. Objective assessment of adherence to inhalers by patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2017;195(10):1333–1343. PubMed PMID: 27409253. doi:10.1164/rccm.201604-0733OC
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

