Back to Journals » Infection and Drug Resistance » Volume 11
Application of tuf gene sequence analysis for the identification of species of coagulase-negative staphylococci in clinical samples and evaluation of their antimicrobial resistance pattern
Authors Khosravi AD , Roointan M, Abbasi Montazeri E, Aslani S , Hashemzadeh M , Taheri Soodejani M
Received 24 April 2018
Accepted for publication 25 June 2018
Published 22 August 2018 Volume 2018:11 Pages 1275—1282
DOI https://doi.org/10.2147/IDR.S172144
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Suresh Antony
Azar Dokht Khosravi,1,2 Mitra Roointan,2 Effat Abbasi Montazeri,2 Sajad Aslani,3,4 Mohammad Hashemzadeh,1,2 Moslem Taheri Soodejani5
1Infectious and Tropical Diseases Research Center, Health Research Institute, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran; 2Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran; 3Department of Microbiology, School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 4Student Research Committee, Kerman University of Medical Sciences, Kerman, Iran; 5Department of Biostatistics and Epidemiology, School of Health, Kerman University of Medical Sciences, Kerman, Iran
Introduction: Coagulase-negative staphylococci (CoNS) are normal inhabitants of human skin and mucous membranes. However, CoNS represent one of the major nosocomial pathogens, especially in immunocompromised patients. The increasing incidence of CoNS and mainly methicillin-resistant strains underlines the need for an accurate identification of Staphylococcus isolates at the species level. Analysis of the tuf gene proved to be an accurate tool for the species identification of CoNS. The aims of this study were to identify the CoNS species by tuf gene-based polymerase chain reaction method and sequencing, and to determine the frequency of CoNS clinical isolates resistant to methicillin (MRCoNS) and other antibiotics.
Methods: A total of 200 staphylococci isolates were collected from various clinical samples. Phenotyping methods were used for initial identification followed by polymerase chain reaction amplification of tuf gene with subsequent sequencing. The phylogenetic relationships among species were analyzed using the neighbor-joining method based on the partial gene sequence of tuf. Microbroth dilution test was used for screening methicillin resistance, and disk diffusion susceptibility testing was performed for evaluation of antibiotic resistance among the isolates.
Results: In the present study, 125 isolates were identified as CoNS; among them, Staphylococcus epidermidis 54(43.2%) and Staphylococcus haemolyticus 50 (40.0%) were demonstrated as the most prevalent species. Resistance to methicillin was detected in 54.4% of the CoNS based on microbroth dilution method. In disk diffusion susceptibility testing, the greatest resistance of CoNS was demonstrated for cefoxitin (65.4%), cotrimethoxazole (54.4%), and clindamycin (49.6%), while daptomycin (87.2%) and linezolid (83.2%) showed the greatest effectiveness for CoNS isolates.
Conclusion: Our results confirmed the predominance of S. epidermidis and S. haemolyticus among CoNS isolates. The high prevalence of MRCoNS strains is a serious concern and strongly suggests the need for control program measures in our hospitals in order to reduce MRCoNS infections, especially in immunocompromised patients.
Keywords: coagulase-negative staphylococci, tuf gene, antimicrobial resistance, PCR, susceptibility testing
Introduction
The pathogenicity of coagulase-negative staphylococci (CoNS) was not accepted until 1980, but later investigations demonstrated that CoNS are common cause of a vast range of important infections and diseases, and in general, newborns, individuals with neutropenia and artificial organs are at highest risk of infection.1–4 The most commonly observed species of CoNS are Staphylococcus epidermidis, Staphylococcus haemolyticus, Staphylococcus saprophyticus, and Staphylococcus lugdunensis.1,2 Among the CoNS, Staphylococcus hominis appears to be the most prevalent cause of septicemia, bacteremia, and endocarditis in immunocompromised patients and infants,5 whereas S. saprophyticus and S. lugdunensis have been reported as the common causes of urinary tract infection in young females and infective endocarditis, respectively.6 There are overwhelming evidences and reports of increasing methicillin resistance among CoNS, and this phenomenon creates new complications for infected patients such as patients with artificial heart valves.7,8 Vancomycin is a drug of choice for the treatment of methicillin-resistant CoNS associated with neonatal infections.9 However, in recent years, there are increasing reports of vancomycin resistance among staphylococci isolates, and the exact mechanism of resistance has not yet been completely understood.10
The tuf gene cluster, which is located in the short tandem repeat region on bacterial chromosome, shows a significant diversity among members of Staphylococci. The small size and its conserved location in bacterial chromosome insist on its superiority in DNA sequencing compared with 16SrRNA for construction of phylogenetic tree on species and genus level in Staphylococci, Enterococci, and Streptococci isolates.11,12 Bergeron et al emphasized on the value of the partial tuf gene sequence for the identification of all staphylococcal species.13 Analysis of the tuf gene proved to be more discriminative for CoNS clinical isolates, and provides a reference method with high accuracy for recognizing hospital infections related to S. epidermidis and S. haemolyticus in critical care units.14,15
Due to the increase in infections caused by CoNS in our settings, the present study was aimed to identify the CoNS species based on tuf gene by polymerase chain reaction (PCR) sequencing method in clinical isolates and to determine the frequency of CoNS resistance to methicillin (MRCoNS) and other antibiotics. We expect that our findings would help us have a better understanding of the frequency of CoNS clinical isolates and their antibiotic resistance pattern in Ahvaz, Southwestern Iran.
Methods
Bacterial isolates
A total of 200 staphylococci spp. isolates belonging to patients admitted to Golestan teaching hospital, Ahvaz, Iran, were collected during 1-year period, from January to December 2015. The study was approved by the joined institutional review board and ethics committee of the Ahvaz Jundishapur University of Medical Sciences, after submission of preliminary proposal and necessary permission for sample collection was granted. As part of the university’s policy, referred patients were requested to provide written informed consent and were asked to sign the informed consent in case that their sample was used for research purposes apart from routine clinical investigation. The staphylococci were isolated from various clinical samples including blood, urine, deep wound, ear discharge, cerebrospinal fluid, and endotracheal secretion. The isolates were cultured on appropriate culture media of blood agar and mannitol salt agar (Merck, Darmstadt, Germany), and were identified as CoNS according to standard morphological criteria and biochemical tests including catalase, coagulase, and DNase.16 CoNS isolates were inoculated in Trypticase soy broth with 20% glycerol and were kept at –70°C until use.
Antimicrobial susceptibility testing
The resistance patterns of CoNS isolates were evaluated by disk diffusion method according to Clinical and Laboratory Standards Institute (CLSI) guidelines,17 using following antibiotic disks: linezolid 30 µg, quinupristin/dalfopristin15 µg, ticarcillin 75 µg, cotrimoxazole 25 µg, clindamycin 2 µg, daptomycin 30 µg, and cefoxitin 30 µg (MAST Co., Berkshire, UK).
Microbroth dilution method
The evaluation of minimal inhibitory concentration for oxacillin was performed by microbroth dilution according to CLSI guidelines.17 Staphylococcus aureus ATCC29213 and ATCC43300 were used as sensitive and resistant controls for Oxacillin, respectively. The Fisher’s exact test was used to evaluate the statistically significant relationship between methicillin-sensitive CoNS (MSCoNS) and MRCoNS isolates.
DNA extraction and PCR amplification
DNA was extracted from bacterial colonies by using simple boiling method as previously described.18 The concentration of extracted DNA was measured by biophotometer (Eppendorf, Hamburg, Germany) at 260–280 nm and was stored at –20°C until use.
PCR amplification was performed on an Eppendorf thermocycler (Roche Co., Mannheim, Germany) by application of a set of primers of 5′-GCCAGTTGAGGACGTATTCT-3′ and 5′- CCATTTCAGTACCTTCTGGTAA-3′, which can amplify a 412 bp fragment of tuf gene,19 as previously described.20 The S. epidermidis ATCC 49134 was used as positive control in amplification reaction. The PCR products were loaded on a 1.5% (w/v) agarose gel with 0.5 µg/mL ethidium bromide and were analyzed using gel electrophoresis, and photographed by using the gel documentation system (ProteinSimple, San Jose, CA, USA). The obtained sequences of tuf gene for each isolate were aligned separately By MEGA 5 (Molecular Evolutionary Genetics Analysis) software and compared with all existing sequences of CoNS annotated in GenBank database.
Phylogenetic trees were obtained from DNA sequences using the neighbor-joining method and Kimura’s two-parameter distance correction model with 1,000 bootstrap replications supported by the MEGA 5 software.21
Results
Based on culture and standard biochemical criteria, 125 out of 200 total tested staphylococci isolates were identified as CoNS. These were originated from samples obtained from 60 (48%) male and 65 (52%) female patients, but there was no statistically significant difference in gender distribution. The distribution of CoNS according to the hospital departments is presented in Table 1, and as it shows, nearly more strains were isolated from patients referred to outpatient department (OPD; n=64[51.2%]) in comparison with the rest of the strains that were isolated from hospitalized patients (n=61[48.8%]).
The majority of CoNS were isolated from urine samples (n=82[65.6%]). The rest of isolates were derived from wound, catheter, blood, sputum, and trachea with lower frequency (Table 2). The most prevalent species revealed by conventional methods were S. epidermidis, S. haemolyticus, and S. hominis, respectively.
The frequency of various CoNS species demonstrated by PCR amplification of tuf gene followed by DNA sequencing were S. epidermidis, 54(43.2%), S. haemolyticus, 50 (40.0%), S. hominis, 11 (8.8%), S. saprophyticus, 6 (4.8%), Staphylococcus warneri, 3 (2.4%), and Staphylococcus petrasii, 1 (0.8%). S. epidermidis was the most frequent species isolated from OPD (n=28 [43.75%]) and hospitalized patients (n=26 [42.62%]), followed by S. haemolyticus with rates of 27 (42.18%) and 23 (37.70%) from OPD and hospitalized patients, respectively. S. petrasii comprised the lowest frequency in this study with only one strain isolated from wound sample of a patient in internal department.
Phylogenetic analysis confirmed the PCR amplification results analyzed by Blast. The CoNS type strains were grouped into separate clusters, with high bootstrap percentages, corresponding to distinct species (Figure 1). The reference strains and their corresponding accession numbers are presented in Table 3.
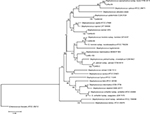  | Figure 1 Phylogenetic tree inferred from the analysis of the partial gene sequence of tuf from CoNS isolates and reference strains (Table 3). Notes: The tree was created using the neighbor-joining method with K2 model based on nucleotide alignment. The support of each branch, as determined from 1,000 bootstrap samples, is indicated by percentages at each node. Bar: 0.02 substitutions per nucleotide position. |
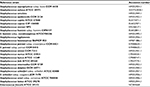  | Table 3 The reference strains used for construction of dendrogram |
Methicillin (oxacillin) resistance was demonstrated in 68 (54.4%) of CoNS based on microbroth dilution method (Table 2). Among the MRCoNS isolates, 41 (60.30%) were isolated from hospitalized patients, which was statistically significant (P<0.005). S. epidermidis, S. hominis, and S. haemolyticus showed the highest methicillin resistance rates of 64.8%, 63.6%, and 46%, respectively.
According to the results of disk diffusion susceptibility testing, the greatest resistance of CoNS was demonstrated for cefoxitin (65.6%), cotrimethoxazole (54.4%), and clindamycin (49.6%). Thirty-five CoNS strains (54.6%) in OPD patients and 47 CoNS strains (77%) in hospitalized patients were resistant to cefoxitin, but we could not find any significant difference between them. S. hominis, S. epidermidis, and S. saprophyticus were again accounted as the most resistant strains for cefoxitin with resistance rates of 72.7%, 72.2%, and 66.7%, respectively. Daptomycin (87.2%) and linezolid (83.2%) showed the greatest effectiveness against CoNS isolates. The only isolate, S. petrasii, was the most sensitive strain in the present study and did not show any resistance to all tested antibiotics (Table 4).
There were statistically significant correlations between MSCoNS and MRCoNS with regard to antibiotic resistance to clindamycin, ticarcillin, cefoxitin, and quinupristin/dalfopristin (P<0.001; Table 5). However, the correlation was not significant for daptomycin, linezolid, and cotrimoxazole (P>0.05).
Discussion
CoNS can colonize on any open surface of body and they are considered as nonpathogenic or opportunistic agents, but they are among the most common causes of secondary infections.22 These bacteria are capable of attaching themselves to prosthesis and catheter; they have the ability to form biofilms which are strong virulence factors.1,23 Because of the increase in the clinical significance of CoNS, there is a need for a more accurate and sensitive method to identify CoNS species in clinical samples.11 Correct species identification is essential to provide an accurate understanding of pathogenic capacity of diverse CoNS species and could help the clinicians for an appropriate treatment strategy.22
In the present study, the frequency of CoNS isolates from OPD and hospitalized patients were demonstrated as 51.2% and 48.8%, respectively. S. epidermidis and S. haemolyticus were the two most prevalent isolates representing skin flora, which under certain circumstances could act as a potential source of infection.24,25
Our findings also indicated that in outpatients, 92% of urinary tract colonization was due to CoNS; however, this study could not emphasize on the role of these bacteria in urinary infection. Moreover, S. haemolyticus and S. epidermidis were responsible for 42.9% and 28.6% of bacteremia, respectively. This finding was in concordant with a similar report from Turkey showing the involvement of S. haemolyticus (43%), and S. epidermidis (11.5%) in bacteremia cases.25
In a review of global studies conducted between 1983 and 2007 on the frequencies of isolated CoNS from patients, S. epidermidis was found to be the leading cause of infections followed by S. haemolyticus with a significant distance,22,25 whereas, we found these prevalent isolates in closer numbers compared with other species of CoNS. In other studies also, S. epidermidis and S. haemolyticus were reported as the most common CoNS isolated from clinical specimens.26–28 In a latter study conducted by Barros et al, however, compared with our findings, a lower prevalence for these species was reported.
The antibiotic resistance among CoNS has created a major problem for health care sectors worldwide.22 The mecA gene is responsible for resistance against methicillin, which is located on staphylococcal chromosomal cassette elements.29,30 It has been proven that this element has the ability of horizontal transfer among species of staphylococci especially S. aureus.
In the present study, the prevalence of MRCoNS was 54.4%, out of which 60.30% belonged to patients from OPD and 39.7% related to hospitalized patients and the statistical analysis demonstrated a significant difference between them. In a report from European countries on the prevalence of methicillin-resistant staphylococci, extremely high rates were demonstrated, which varied from 61.9% in Spain to 83.7% in Greece.31 In a similar study from China, the prevalence of MRCoNS between 2004 and 2009 was reported to be 79.1%, which compared with our results was higher.26 Furthermore, recent studies from Iran and Saudi Arabia reported rates of 81.1% and 63%, respectively.32,33 This study demonstrated the highest prevalence of antibiotic resistance against cefoxitin (n=82 [65.6%]) among the tested CoNS. Fifty-six (68.3%) out of 82 cefoxitin-resistant strains also presented oxacillin resistance, and this relationship has proven to be statistically significant (P=0.0001). In a similar study from Brazil, only 32% of their CoNS strains were reported to be cefoxitin resistant, which was lower than our findings. Moreover, in their study, S. epidermidis and Staphylococcus simolance showed the highest resistance to cefoxitin (41.5%),34 while our study presented S. hominis (72.7%) followed by S. epidermidis (72.2%) as the most resistant species of CoNS to cefoxitin.
To overcome the antibiotic resistance, currently new drugs including linezolid, daptomycin, tigecycline, and quinupristin/dalfopristin have been administered and shown to be useful.31,35 Investigations are still emphasized on the continuous susceptibility of CoNS clinical strains to these antibiotics.35,36
Conclusion
Our results confirmed the predominance of S. epidermidis and S. haemolyticus among CoNS isolates. The accurate identification of CoNS to the species level in reference laboratories is important to establish the role of each staphylococcal species as an infectious agent and to conduct epidemiologic investigations.18
The high prevalence of MRCoNS strains is a serious concern and strongly suggests the need for control program measures in our hospitals in order to reduce MRCoNS infections especially in immunocompromised patients. In this regard, molecular epidemiology surveillance could be a useful way to investigate the transmission patterns of CoNS to control nosocomial infections due to these bacteria.
Acknowledgment
This work was part of MSc thesis of Mitra Roointan, which was approved in Infectious and Tropical Diseases Research Center, and was financially supported by a grant (no. OG-94116) from Research affairs, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
Disclosure
The authors report no conflicts of interest in this work.
References
Becker K, Heilmann C, Peters G. Coagulase-negative staphylococci. Clin Microbiol Rev. 2014;27(4):870–926. | ||
Widerström M, Wiström J, Sjöstedt A, Monsen T. Coagulase-negative staphylococci: update on the molecular epidemiology and clinical presentation, with a focus on Staphylococcus epidermidis and Staphylococcus saprophyticus. Eur J Clin Microbiol Infect Dis. 2012;31(1):7–20. | ||
Rogers KL, Fey PD, Rupp ME. Coagulase-negative staphylococcal infections. Infect Dis Clin North Am. 2009;23(1):73–98. | ||
Dubois D, Leyssene D, Chacornac JP, et al. Identification of a variety of Staphylococcus species by matrix-assisted laser desorption ionization-time of flight mass spectrometry. J Clin Microbiol. 2010;48(3):941–945. | ||
Al Wohoush I, Rivera J, Cairo J, Hachem R, Raad I. Comparing clinical and microbiological methods for the diagnosis of true bacteraemia among patients with multiple blood cultures positive for coagulase-negative staphylococci. Clin Microbiol Infect. 2011;17(4):569–571. | ||
Poutanen SM, Baron EJ. Staphylococcus lugdunensis: a notably distinct coagulase-negative staphylococcus. Clin Microbiol Newsl. 2001;23(19):147–150. | ||
Wang Z, Cao B, Liu YM, Gu L, Wang C. Investigation of the prevalence of patients co-colonized or infected with methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci in China: a hospital-based study. Chin Med J. 2009;122(11):1283–1288. | ||
Widerström M, Monsen T, Karlsson C, Wiström J. Molecular epidemiology of methicillin-resistant coagulase-negative staphylococci in a Swedish county hospital: evidence of intra- and interhospital clonal spread. J Hosp Infect. 2006;64(2):177–183. | ||
Sánchez PJ, Moallem M, Cantey JB, Milton A, Michelow IC. Empiric therapy with vancomycin in the neonatal intensive care unit: let’s “get smart” globally! J Pediatr. 2016;92(5):432–435. | ||
Tarai B, das P, Kumar D. Recurrent challenges for clinicians: emergence of methicillin-resistant Staphylococcus aureus, vancomycin resistance, and current treatment options. J Lab Physicians. 2013;5(2):71. | ||
Hwang SM, Kim MS, Park KU, Song J, Kim EC. Tuf gene sequence analysis has greater discriminatory power than 16S rRNA sequence analysis in identification of clinical isolates of coagulase-negative staphylococci. J Clin Microbiol. 2011;49(12):4142–4149. | ||
Shin JH, Kim SH, Jeong HS, et al. Identification of coagulase-negative staphylococci isolated from continuous ambulatory peritoneal dialysis fluid using 16S ribosomal RNA, tuf, and SodA gene sequencing. Perit Dial Int. 2011;31(3):340–346. | ||
Bergeron M, Dauwalder O, Gouy M, et al. Species identification of staphylococci by amplification and sequencing of the tuf gene compared to the gap gene and by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Eur J Clin Microbiol Infect Dis. 2011;30(3):343–354. | ||
Capurro A, Artursson K, Waller KP, Bengtsson B, Ericsson-Unnerstad H, Aspán A. Comparison of a commercialized phenotyping system, antimicrobial susceptibility testing, and tuf gene sequence-based genotyping for species-level identification of coagulase-negative staphylococci isolated from cases of bovine mastitis. Vet Microbiol. 2009;134(3–4):327–333. | ||
Alexopoulou K, Foka A, Petinaki E, Jelastopulu E, Dimitracopoulos G, Spiliopoulou I. Comparison of two commercial methods with PCR restriction fragment length polymorphism of the tuf gene in the identification of coagulase-negative staphylococci. Lett Appl Microbiol. 2006;43(4):450–454. | ||
Mahon CR, Lehman DC, Manuselis Jr G. Textbook of Diagnostic Microbiology. Amsterdam: Elsevier Health Sciences; 2014. | ||
Patel J, Cockerill F, Alder J, Bradford P, Eliopoulos G, Hardy D. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Fourth Informational Supplement. Vol. 34: CLSI Standards for Antimicrobial Susceptibility Testing. Washington: AACC; 2014:1–226. | ||
Heikens E, Fleer A, Paauw A, Florijn A, Fluit AC. Comparison of genotypic and phenotypic methods for species-level identification of clinical isolates of coagulase-negative staphylococci. J Clin Microbiol. 2005;43(5):2286–2290. | ||
Li X, Xing J, Li B, Wang P, Liu J. Use of tuf as a target for sequence-based identification of Gram-positive cocci of the genus Enterococcus, Streptococcus, coagulase-negative Staphylococcus, and Lactococcus. Ann Clin Microbiol Antimicrob. 2012;11(1):31. | ||
Hendolin PH, Paulin L, Ylikoski J. Clinically applicable multiplex PCR for four middle ear pathogens. J Clin Microbiol. 2000;38(1):125–132. | ||
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28(10):2731–2739. | ||
Piette A, Verschraegen G. Role of coagulase-negative staphylococci in human disease. Vet Microbiol. 2009;134(1–2):45–54. | ||
Vuong C, Otto M. Staphylococcus epidermidis infections. Microbes Infect. 2002;4(4):481–489. | ||
Garza-González E, Morfín-Otero R, Llaca-Díaz JM, Rodriguez-Noriega E. Staphylococcal cassette chromosome mec (SCCmec) in methicillin-resistant coagulase-negative staphylococci. A review and the experience in a tertiary-care setting. Epidemiol Infect. 2010;138(5):645–654. | ||
Koksal F, Yasar H, Samasti M. Antibiotic resistance patterns of coagulase-negative staphylococcus strains isolated from blood cultures of septicemic patients in Turkey. Microbiol Res. 2009;164(4):404–410. | ||
Ma XX, Wang EH, Liu Y, Luo EJ. Antibiotic susceptibility of coagulase-negative staphylococci (CoNS): emergence of teicoplanin-non-susceptible CoNS strains with inducible resistance to vancomycin. J Med Microbiol. 2011;60(Pt 11):1661–1668. | ||
Sheikh AF, Mehdinejad M. Identification and determination of coagulase-negative Staphylococci species and antimicrobial susceptibility pattern of isolates from clinical specimens. Afr J Microbiol Res. 2012;6(8):1669–1674. | ||
Barros EM, Ceotto H, Bastos MC, dos Santos KR, Giambiagi-Demarval M. Staphylococcus haemolyticus as an important hospital pathogen and carrier of methicillin resistance genes. J Clin Microbiol. 2012;50(1):166–168. | ||
Zong Z, Peng C, Lü X. Diversity of SCCmec elements in methicillin-resistant coagulase-negative staphylococci clinical isolates. PLoS One. 2011;6(5):e20191. | ||
Hanssen AM, Kjeldsen G, Sollid JU. Local variants of Staphylococcal cassette chromosome mec in sporadic methicillin-resistant Staphylococcus aureus and methicillin-resistant coagulase-negative Staphylococci: evidence of horizontal gene transfer? Antimicrob Agents Chemother. 2004;48(1):285–296. | ||
Sader HS, Watters AA, Fritsche TR, Jones RN. Daptomycin antimicrobial activity tested against methicillin-resistant staphylococci and vancomycin-resistant enterococci isolated in European medical centers (2005). BMC Infect Dis. 2007;7(1):29. | ||
Nahaei MR, Shahmohammadi MR, Ebrahimi S, Milani M. Detection of methicillin-resistant coagulase-negative staphylococci and surveillance of antibacterial resistance in a multi-center study from Iran. Jundishapur J Microbiol. 2015;8(8):e19945. | ||
Shibl AM, Memish ZA, Kambal AM, et al. National surveillance of antimicrobial resistance among Gram-positive bacteria in Saudi Arabia. J Chemother. 2014;26(1):13–18. | ||
Rosa JO, de Moura JP, Palos MA, et al. Detection of mecA gene in oxacillin-resistant coagulase-negative staphylococci isolated from the saliva of nursing professionals. Rev Soc Bras Med Trop. 2009;42(4):398–403. | ||
Jones JW, Scott RJ, Morgan J, Pether JV. A study of coagulase-negative staphylococci with reference to slime production, adherence, antibiotic resistance patterns and clinical significance. J Hosp Infect. 1992;22(3):217–227. | ||
Flamm RK, Mendes RE, Hogan PA, Streit JM, Ross JE, Jones RN. Linezolid Surveillance Results for the United States (LEADER Surveillance Program 2014). Antimicrob Agents Chemother. 2016;60(4):2273–2280. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




