Back to Journals » Cancer Management and Research » Volume 11
Apolipoprotein A1 and B as risk factors for development of intraocular metastasis in patients with breast cancer
Authors Liu JX , Yuan Q , Min YL, He Y, Xu QH , Li B, Shi WQ , Lin Q , Li QH, Zhu PW , Shao Y
Received 19 October 2018
Accepted for publication 17 January 2019
Published 9 April 2019 Volume 2019:11 Pages 2881—2888
DOI https://doi.org/10.2147/CMAR.S191352
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Jia-Xiang Liu, Qing Yuan, You-Lan Min, Ying He, Qian-Hui Xu, Biao Li, Wen-Qing Shi, Qi Lin, Qing-Hai Li, Pei-Wen Zhu, Yi Shao
Department of Ophthalmology, The First Affiliated Hospital of Nanchang University, Jiangxi Province Clinical Ophthalmology Institute, Nanchang, Jiangxi 330006, People’s Republic of China
Objective: Breast cancer is the most common primary lesion resulting in intraocular metastasis (IOM). In this study, we investigated the differences between breast cancer patients with and without IOM, and clarified the risk factors for IOM in patients with breast cancer.
Methods: A total of 2,381 patients with breast cancer were included in this study from January 2005 to December 2017. The chi-square test and Student’s t-test were applied to evaluate differences between the IOM and non-IOM (NIOM) groups. Risk factors were calculated using binary logistic regression analysis. Receiver operating curve (ROC) analysis was used to assess the diagnostic value of IOM in patients with breast cancer.
Results: The IOM incidence in patients with breast cancer was 1.35%. No significant differences were detected in age, gender, menopausal status, or histopathology between the IOM and NIOM groups. The IOM group had more axillary lymph node metastases, lower ApoA1 and higher ApoB, compared with the NIOM group. Binary logistic regression indicated that ApoA1 and ApoB were risk factors for IOM in breast cancer patients (P-values and P-values=0.005, respectively). ROC curve analysis revealed area under the curve values for ApoA1 and ApoB of 0.871 and 0.633, using cutoff values of 1.165 and 0.835 g/L, respectively. The sensitivity and specificity values for ApoA1 were 0.813 and 0.849, respectively, while those for ApoB were 0.813 and 0.481.
Conclusion: Our data indicate that ApoA1 and ApoB are risk factors for IOM in patients with breast cancer and that ApoA1 is more reliable than ApoB at distinguishing IOM from NIOM in patients with breast cancer.
Keywords: breast cancer, intraocular metastases, apolipoprotein A1, apolipoprotein B
Introduction
Breast cancer has become the most common malignant tumor in women1 and, as a multifactorial disease, is closely associated with both genetic and environmental factors.2,3 Due to their specific molecular characteristics and clinical subtypes, breast cancers are highly malignant tumors prone to forming metastases in distant organs, including lung, bone, liver, and brain.4–7
Although the eye is an uncommon site of metastasis, ocular metastases are closely associated with breast cancer, which accounts for the largest proportion among all primary tumors known to develop ocular metastases.8 Moreover, intraocular metastasis (IOM) represents an advanced stage of breast cancer and can cause a series of clinical symptoms, including ocular pain, foreign body sensation, blurred vision, and visual field defects, severely affecting the quality of the patient’s life.9 Consequently, early detection, diagnosis, and treatment of IOM is of great consequence in patients with breast cancer.
Currently, computed tomography (CT), magnetic resonance imaging (MRI), and ultrasound diagnosis (UD) are the techniques frequently used for the diagnosis of breast cancer;10–12 however, they have clear limitations, including high economic costs and exposure to heavy doses of radiation, caused by repeated use. To improve predictions, it is important to develop methods that are both convenient and reliable. Serum tests are ideal, as they are repeatable, low-cost, and non-invasive. Previous studies have reported that expression of programmed death 1 and the neutrophil-to-lymphocyte ratio in peripheral blood could be diagnostic and prognostic indicators, respectively, in breast cancer.13,14
Blood lipids and relevant proteins have long been of interest because of their close relationship with cardiovascular diseases; however, recent studies have also reported strong correlations of these factors with the development of cancer. Several animal experiments have demonstrated that specific apolipoproteins can affect tumor growth via modulation of immune cell function.15 Moreover, levels of some apolipoproteins are also used to assess prognosis in several cancers;16,17 however, the predictive values of blood lipids and relevant apolipoproteins for IOM in patients with breast cancer are unknown.
In this retrospective study, we aimed to investigate the association between blood lipid-relevant parameters and IOM, and to determine the risk factors for IOM in patients with breast cancer.
Materials and methods
Study design
This study was conducted in accordance with the Declaration of Helsinki and approved by the medical research ethics committee of the First Affiliated Hospital of Nanchang University. All the methods used in this study were conducted under the relevant guidelines and regulations. A series of consecutive patients diagnosed with breast cancer at our hospital between January 2005 and December 2017 were included in this study. The diagnosis was made based on pathological specimens obtained by surgical resection or biopsy. IOM diagnosis was by CT and MRI. Patients with primary ocular malignant tumors, ocular benign tumors, and secondary breast cancer were excluded from the study. All participants were given details of the study design and provided signed informed consent.
Data collection
Clinical data relevant to this study were collected retrospectively from patient medical records, including: age, sex, menopausal status, histopathological subtype, total cholesterol (TC), triglycerides (TG), high density lipoprotein (HDL), low density lipoprotein (LDL), apolipoprotein A1 (ApoA1), apolipoprotein B (ApoB), and lipoprotein A (Lp(A)). All clinical parameters were collected at the time of initial diagnosis of breast cancer. Blood lipids were tested after fasting for at least 12 h.
Measurement of ApoA1 and ApoB levels
ApoA1 and ApoB were tested using the immune turbidimetric method. ApoA1 in reagents (Ruiyuan Biotechnology, Ningbo, Zhejiang) bound to specific antibodies in serum to form antigen-antibody complexes, resulting in turbidity, levels of which were directly proportional to those of ApoA1 in serum. Absorbance was measuredand the ApoA1 content in the serum calculated, with reference to a calibration curve. ApoB was tested using the same method.
Statistical analysis
The Student’s t-test and Chi-square test were used to evaluate whether differences in clinical features between IOM and NIOM patients were significant. Then, binary logistic regression models were established to identify independent risk factors for ocular metastasis. Receiver operating characteristics (ROC) curves were constructed and areas under the curve (AUC) values calculated, to estimate the accuracy of variables for prediction of IOM. P-values<0.05 was considered statistically significant. FDR correction was used to select significant features. All the statistical analyses were conducted using SPSS17.0 (SPSS, IBM Corp, USA) and Excel 2010 (Excel, Microsoft Corp, USA) software. Continuous data are displayed as means ± standard deviation.
Results
Demographic and clinical characteristics
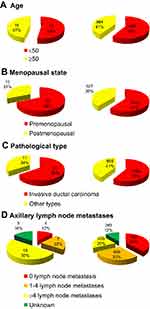
A total of 2,381 patients (7 men and 2,374 women) were recruited to this study, of which 32 were IOM and 2,349 NIOM cases. Average ages of IOM and NIOM patients were 46.59±7.78 and 48.21±10.41 years, respectively. Among women, 1,444 were premenopausal and 937 postmenopausal. Regarding histopathological subtype, invasive ductal carcinoma accounted for the largest proportion of tumors (59.0%). In the IOM group, 71.9% of patients had developed axillary lymph node metastases, while in the NIOM group the proportion was 50.1%. The detailed clinical characteristics of the patients are presented in Table 1 and Figure 1.
 | Table 1 The clinical characteristics of patients with breast cancer |
Evaluation of clinical features as risk factors for IOM
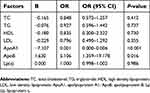
There were no differences in the levels of TC, TG, HDL, LDL and Lp(A) between IOM and NIOM patients; however, levels of ApoA1 were lower (1.01 mmol/L) in the IOM group than those in the NIOM group (1.49 mmol/L). Moreover, ApoB levels were higher (0.91 g/L) in the IOM group than the NIOM group (0.86 g/L) (Table 2). Further analysis using a binary logistic regression model showed that ApoA1 and ApoB levels were independent risk factors for IOM (Table 3).
 | Table 2 The differences of clinical lipids-relevant parameters between patients with and without IOM |
 | Table 3 The binary logistic regression results |
Cut-off, AUC, sensitivity, and specificity values of ApoA1 and ApoB for diagnosis of intraocular metastasis
As shown in Figure 2 and Table 4, the AUC value for ApoA1 was 0.871, while its sensitivity and specificity for prediction of IOM were 0.813 and 0.849, respectively. The AUC value for ApoB was 0.633, with sensitivity and specificity values of 0.813 and 0.481, respectively. These data were based on cut-off values of 1.165 g/L and 0.835 g/L for ApoA1 and ApoB, respectively. All of these results were statistically significant.
 | Table 4 The ROC results of risk factors for predicting IOM in breast cancer patients |
Discussion
The incidence of breast cancer is currently rising rapidly; however, the survival times of patients with breast cancer are also increasing, which can be attributed to the use of mammography for early tumor detection, adjuvant chemotherapy, hormonal therapy, and targeted therapies.18–21
Nevertheless, many patients with breast cancer still develop distant metastases. The incidence of IOM among patients with breast cancer reported by different studies varies from 5% to 30%.22,23 Moreover, IOM represents a marker of poor prognosis in various types of cancer. In 1959, Garrett reported a 79-year-old man with IOM from seminoma.24 To date, IOM has been reported in patients with malignant melanoma,25 non-small cell lung cancer,26 esophageal carcinoma,27 head and neck cancer,28 gastric adenocarcinoma,29 renal cell carcinoma,30 choriocarcinoma,31 and colorectal cancer32 (Table 5). Unfortunately, IOM is difficult to detect at an early stage; hence, it is essential to improve rates of early diagnosis. Since apolipoproteins are reported to influence tumor metabolism,15 and tests on lipids and relevant apolipoproteins are repeatable, low-cost, and do not expose patients to radiation risks, we investigated lipid-relevant parameters as potential predictors of IOM, by comparing blood their levels among breast cancer patients with and without IOM.
 | Table 5 Studies on the IOM from different cancers |
The incidence of IOM in patients with breast cancer in this study was 1.35%, which is lower than the rates reported by Nelson et al and Kreusel et al22,23. This may be attributable to the fact that CT and MRI were not widely used in the past, because of their high cost, hence many patients with breast cancer and IOM did not choose to undergo these examinations, resulting in a lower reported incidence of IOM. Moreover, patients in the IOM group had more axillary lymph nodes metastases, revealing that IOM is associated with advanced stage breast cancer. Additionally, we identified ApoA1 and ApoB as risk factors for IOM in patients with breast cancer.
ApoA1, an essential component of high density lipoprotein (HDL), plays indispensable roles in transporting peripheral lipids to the liver and preventing extrahepatic cells from absorbing excessive lipid.33 Recently, ApoA1 has attracted increasing attention in ophthalmology, as levels of this factor are closely related to dry eye, diabetic retinopathy, age-related macular degeneration, and other eye diseases.34–36 More importantly, ApoA1 has an anti-tumor function. In an animal experiment conducted by Zamaniandaryoush et al15 mice expressing a human ApoA1 transgene had increased numbers of M1 macrophages, an anti-tumor phenotype, which reduced tumor burden and metastasis. In contrast, ApoA1-deficient (A1KO) mice exhibited enhanced tumor growth and reduced survival, while injecting human ApoA1 into A1KO mice significantly reduced tumor growth and metastasis. Researchers also found that ApoA1 did not directly influence the tumor, rather it promoted the anti-tumor functions of macrophages by altering their phenotypes from M2 to M1. Moreover, a correlation between ApoA1 and patient prognosis has been reported for several cancers, including colorectal cancer, gastric cancer, hepatocellular carcinoma, ovarian cancer, and renal cancer.16,17,37–39 Patients with lower levels of ApoA1 and higher levels of circulating tumor cells had elevated recurrence rates and shorter survival times. These data suggest that ApoA1 has an important role in inhibiting tumor growth and invasion. To date, many studies have demonstrated that decreased ApoA1 levels are closely associated with the occurrence and development of breast cancer. His et al40 reported that low serum ApoA1 levels are a high-risk factor for breast cancer, while Lane et al41 demonstrated that women with decreased levels of ApoA1 are more likely to develop early recurrence of breast cancer. These studies suggest that low ApoA1 levels are related to the occurrence and recurrence of breast cancer. However, Martin et al42 reached the controversial conclusion that levels of serum ApoA1 are positively correlated with the risk of breast cancer, based on data from a nested case-control study of 4,690 women who had high mammographic density.
Overall, we hypothesized that levels of ApoA1 are related to IOM in breast cancer patients and we explored this correlation in our retrospective study. The results of our study demonstrate that low ApoA1 levels were associated with IOM of breast cancer, consistent with previous findings. The cut-off value was 1.165 g/L in our study, where patients with breast cancer whose ApoA1 levels were <1.165 g/L were more likely to develop IOM. Moreover, the AUC of the ROC curve showed a relatively high accuracy for distinguishing patients with breast cancer with IOM from those without, revealing the excellent diagnostic value of ApoA1 for prediction of IOM.
ApoB is a type of apolipoprotein present on the surface of LDLs. Unlike ApoA1, the function of ApoB is transporting lipids to cells within the human body.43 ApoB levels are positively correlated with the risk of cardiovascular diseases. As the blood vessels of the eye are dense and tiny, and can easily be covered by lipids, there is also a relationship between ApoB and ophthalmological diseases. Levels of ApoB are associated with elevated intraocular pressure, lens opacity, retinopathy, and primary open angle glaucoma.44–47 Apart from its essential role in lipid transport, ApoB is also associated with cancer genome mutations. Apolipoprotein B mRNA editing enzyme catalytic polypeptide-like (APOBEC) participates in ApoB RNA editing, and abnormal expression of APOBEC can cause mutations in tumor suppressor genes and proto oncogenes.48–50 Although ApoB is not directly relevant to tumors, it has been used to predict cancer development and recurrence. Ma et al37 reported that the ApoB/ApoA1 ratio can be used as a prognostic indicator in patients with gastric cancer (GC), where those with high ApoB/ApoA1 ratios had shorter survival times. In our study, we found that ApoB was also a risk factor for IOM in patients with breast cancer. Breast cancer patients with ApoB levels >0.835 g/L were prone to IOM; however, ROC curve analysis indicated a relatively low accuracy. Consequently, ApoB should be applied cautiously for the prediction of IOM in patients with breast cancer, and we consider ApoA1 a more reliable indicator of IOM diagnosis in these patients.
This study has some limitations. First, data were collected over a long period time and, despite our best efforts, some data were missing, and differences among individuals conducting the tests may have contributed to minor discrepancies; however, these will not have influenced the results. Second, all the records used in this experiment were from the same medical institution, which could potentially have introduced confounding factors. Experiments from additional medical institutions are expected to be conducted. Third, this study merely demonstrates correlations between altered ApoA1 and ApoB levels and IOM in patients with breast cancer; we were unable to determine whether IOM caused the changes in ApoA1 and ApoB or vice versa. Finally, we only investigated altered levels of ApoA1 and ApoB in patients with IOM from breast cancer; hence, whether these two parameters exhibit differences associated with other distant metastases from breast cancer, and how they are altered, remain unknown.
In conclusion, this study is the first to demonstrate that ApoA1 and ApoB are risk factors for IOM in patients with breast cancer. These two parameters are potential targets for the therapy of IOM in patients with breast cancer. At present, many scientists are focused on the anti-tumor effects of chemical reagents and their mechanisms.51,52 We hope that our results will encourage relevant experiments investigating the potential molecular mechanisms underlying the antineoplastic effects of ApoA1 and ApoB.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No: 81660158,81460092, 81400372); Natural Science Key Project of Jiangxi Province (No: 20161ACB21017); Health Development Planning Commission Science Foundation of Jiangxi Province (No: 20175116).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Torre LA, Bray F, Siegel R, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi:10.3322/caac.21262
2. Karami F, Mehdipour P. A comprehensive focus on global spectrum of BRCA1 and BRCA2 mutations in breast cancer. Biomed Res Int. 2013;2013(2):1–21. doi:10.1155/2013/928562
3. Gray JM, Rasanayagam S, Engel C, et al. State of the evidence 2017: an update on the connection between breast cancer and the environment. Environ Health. 2017;16(1):1–61. doi:10.1186/s12940-016-0208-y
4. Minn AJ, Gupta GP, Siegel PM, et al. Genes that mediate breast cancer metastasis to lung. Nature. 2005;436(7050):518–524. doi:10.1038/nature03799
5. Kang Y, Siegel PM, Shu W, et al. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell. 2003;3(6):537–549.
6. Hoshino A, Costa-Silva B, Shen T, et al. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527(7578):329–335. doi:10.1038/nature15756
7. Bos PD, Zhang XHF, Nadal C, et al. Genes that mediate breast cancer metastasis to the brain. Nature. 2009;459(7249):1005–1009. doi:10.1038/nature08021
8. Shields CL, Shields JA, Gross NE, Schwartz GP, Lally SE. Survey of 520 eyes with uveal metastases. Ophthalmology. 1997;104(8):1265–1276.
9. Georgalas I, Paraskevopoulos T, Koutsandrea C, et al. Ophthalmic metastasis of breast cancer and ocular side effects from breast cancer treatment and management: mini review. Biomed Res Int. 2015;2015:1–8. doi:10.1155/2015/574086
10. Reddy S, Kurli M, Tena LB, et al. PET/CT imaging: detection of choroidal melanoma. Brit J Ophthalmol. 2005;89(10):1265–1269. doi:10.1136/bjo.2005.066399
11. Razek AA, Elkhamary S. MRI of retinoblastoma. Brit J Radiol. 2011;84(1005):775–784. doi:10.1259/bjr/32022497
12. Rykun VS. Computerized ultrasonic diagnosis of voluminous intraocular neoplasms. Vestn Oftalmol. 2003;119(3):29–31.
13. Fang J, Shao Y, Su J, et al. Diagnostic value of PD-1 mRNA expression combined with breast ultrasound in breast cancer patients. Ther Clin Risk Manag. 2018;14:1527–1535. doi:10.2147/TCRM.S168531
14. Li Y, Shao Y, Bai L, Zhou X. Increased derived neutrophil-to-lymphocyte ratio and breast imaging-reporting and data system classification predict poor survival in patients with non-distant metastatic HER2+ breast cancer treated with neoadjuvant chemotherapy. Cancer Manag Res. 2018;10:3841–3847. doi:10.2147/CMAR.S174537
15. Zamanian-Daryoush M, Lindner D, Tallant TC, et al. The cardioprotective protein apolipoprotein A1 promotes potent anti-tumorigenic effects. J Biol Chem. 2013;288(29):21237–21252. doi:10.1074/jbc.M113.468967
16. Ma XL, Gao XH, Gong ZJ, et al. Apolipoprotein A1: a novel serum biomarker for predicting the prognosis of hepatocellular carcinoma after curative resection. Oncotarget. 2016;7(43):70654–70668.
17. Sirniö P, Väyrynen JP, Klintrup K, et al. Decreased serum apolipoprotein A1 levels are associated with poor survival and systemic inflammatory response in colorectal cancer. Sci Rep. 2017;7(1):1–8. doi:10.1038/s41598-016-0028-x
18. Bodai B. Breast cancer survivorship: a comprehensive review of long-term medical issues and lifestyle recommendations. The Permanente J. 2015;19(2):48–79. doi:10.7812/TPP/14-241
19. Anampa J, Makower D, Sparano JA. Progress in adjuvant chemotherapy for breast cancer: an overview. BMC Med. 2015;13(1):195. doi:10.1186/s12916-015-0439-8
20. Puhalla S, Bhattacharya S, Davidson N E. Hormonal therapy in breast cancer: a model disease for the personalization of cancer care. Mol Oncol. 2012;6(2):222–236. doi:10.1016/j.molonc.2012.02.003
21. Masoud V, Pagès G. Targeted therapies in breast cancer: new challenges to fight against resistance. World J Clin Oncol. 2017;8(2):120. doi:10.5306/wjco.v8.i2.120
22. Nelson CC, Hertzberg BS, Klintworth GK, et al. A histopathologic study of 716 unselected eyes in patients with cancer at the time of death. Am J Ophthalmol. 1983;95(6):788–793.
23. Kreusel KM, Wiegel T, Stange M, et al. Intraocular metastases of metastatic breast carcinoma in the woman. Incidence, risk factors and therapy. Ophthalmologe. 2000;97(5):342–346.
24. Garrett M. Ocular metastasis from seminoma. Br J Ophthalmol. 1959;43(12):759. doi:10.1136/bjo.43.12.759
25. Kelmenson AT, Grover S. Multiple bilateral ocular metastases from malignant melanoma associated with blue nevi. Eur J Ophthalmol. 2011;21(5):671–673. doi:10.5301/EJO.2011.6375
26. Niu F, Zhou Q, Yang J, et al. Distribution and prognosis of uncommon metastases from non-small cell lung cancer. BMC Cancer. 2016;16(1):149. doi:10.1186/s12885-016-2169-5
27. Mcdonald JW, Proia AD. Ocular metastasis as the initial presentation of esophageal carcinoma. Can J Ophthalmol. 1997;32(1):50–52.
28. Fang KH, Chen CK, Hao SP. Acute visual loss in a head and neck cancer patient with ocular metastasis and sphenoid pyocele. Auris Nasus Larynx. 2007;34(4):569–571. doi:10.1016/j.anl.2007.02.008
29. Sitaula R, Shrestha GB, Paudel N, Shrestha JK. Ocular and orbital metastases presenting as a first sign of gastric adenocarcinoma. BMJ Case Rep. 2011;2011(dec02 1):422–425. doi:10.1136/bcr.10.2011.4927
30. Pompeu AC, Arap S, Silva MN, et al. Ocular metastasis as first presentation of renal cell carcinoma: report of 2 cases. Clinics. 2005;60(1):75–78. doi:10.1590/S1807-59322005000100014
31. Gangadharan VP, Prakash NP, Chitrathara K, Sasidharan K, Ramachandran K. Ocular metastasis of choriocarcinoma. Br J Radiol. 1999;72(864):1216–1217. doi:10.1259/bjr.72.864.10703482
32. Khawaja MR, Minturn JT, Spittler AJ, Chiorean EG. Ocular metastasis of colorectal cancer: an uncommon presentation of a common malignancy. Hematol Oncol Stem Cell Ther. 2015;8(4):176–180. doi:10.1016/j.hemonc.2015.02.002
33. Weers PMM, Patel AB, Wan LC, et al. Novel N-terminal mutation of human apolipoprotein A-I reduces self-association and impairs LCAT activation. J Lipid Res. 2010;52(1):35–44. doi:10.1194/jlr.M007500
34. Nyunt AK, Ishida Y, Yu Y, Shimada S. Topical apolipoprotein A-1 may have a beneficial effect on the corneal epithelium in a mouse model of dry eye: a pilot study. Eye Contact Lens. 2008;34(5):287–292. doi:10.1097/ICL.0b013e318184bdb6
35. Kawai S, Nakajima T, Hokari S, Komoda T, Kawai K. Apolipoprotein A-I concentration in tears in diabetic retinopathy. Ann Clin Biochem. 2002;39(Pt 1):56–61. doi:10.1258/0004563021901748
36. Buch H, Vinding T, La Cour M, Jensen GB, Prause JU, Nielsen NV. Risk factors for age-related maculopathy in a 14-year follow-up study: the copenhagen city eye study. Acta Ophthalmol Scand. 2005;83(4):409–418. doi:10.1111/j.1600-0420.2005.00492.x
37. Ma M, Yuan S, Chen Y, Zhou Z-W. Preoperative apolipoprotein B/apolipoprotein A1 ratio: a novel prognostic factor for gastric cancer. Oncotargets Ther. 2018;11:2169–2176. doi:10.2147/OTT
38. Clarke CH, Yip C, Badgwell D, et al. Proteomic biomarkers apolipoprotein A1, truncated transthyretin and connective tissue activating protein III enhance the sensitivity of CA125 for detecting early stage epithelial ovarian cancer. Gynecol Oncol. 2011;122(3):548–553. doi:10.1016/j.ygyno.2011.06.002
39. Liu Z, Xiao Y, Tang L, et al. Apolipoprotein A1-75 G/A and +83 C/T polymorphisms and renal cancer risk. Lipids in Health Dis. 2015;14(1):1–6. doi:10.1186/s12944-015-0132-0
40. His M, Zelek L, Deschasaux M, et al. Prospective associations between serum biomarkers of lipid metabolism and overall, breast and prostate cancer risk. Eur J Epidemiol. 2014;29(2):119–132. doi:10.1007/s10654-014-9884-5
41. Lane DM, Boatman KK, Mcconathy WJ. Serum lipids and apolipoproteins in women with breast masses. Breast Cancer Res Tr. 1995;34(2):161–169. doi:10.1007/BF00665788
42. Martin LJ, Melnichouk O, Huszti E, et al. Serum lipids, lipoproteins, and risk of breast cancer: a nested case-control study using multiple time points. J Natl Cancer I. 2015;107(5):djv032. doi:10.1093/jnci/djv032
43. Yu Q, Zhang Y, Xu CB. Apolipoprotein B, the villain in the drama? Eur J Pharmacol. 2015;748:166–169. doi:10.1016/j.ejphar.2014.08.037
44. Son JH, Chung YK, Son JS. Apolipoprotein B: novel indicator of elevated intraocular pressure. Eye. 2015;29(10):1315–1320. doi:10.1038/eye.2015.37
45. Modrzejewska A. Relationship between initial lens transparency and ocular circulation in adolescents with Type-1 diabetes mellitus, unstable glycaemia and lipid parameters. Polish Journal of Radiology. 2014;79(1):472–478. doi:10.12659/PJR.890852
46. Kinnunen K, Heinonen SE, Kalesnykas G, et al. LDLR-/-ApoB100/100 mice with insulin-like growth factor II overexpression reveal a novel form of retinopathy with photoreceptor atrophy and altered morphology of the retina. Mol Vis. 2013;19(19):1723–1733.
47. Sacca SC, Centofanti M, Izzotti A. New proteins as vascular biomarkers in primary open angle glaucomatous aqueous humor. Invest Ophthalmol Vis Sci. 2012;53(7):4242–4253. doi:10.1167/iovs.11-8902
48. Blanc V, Davidson NO. C-to-U RNA editing. Mechanisms leading to genetic diversity. J Biol Chem. 2003;278(3):1395–1398. doi:10.1074/jbc.R200024200
49. Matsumoto Y, Marusawa H, Kinoshita K, et al. Helicobacter pylori infection triggers aberrant expression of activation-induced cytidine deaminase in gastric epithelium. Nat Med. 2007;13(4):470–476. doi:10.1038/nm1566
50. Henderson S, Chakravarthy A, Su X, et al. APOBEC-mediated cytosine deamination links PIK3CA helical domain mutations to human papillomavirus-driven tumor development. Cell Rep. 2014;7(6):1833–1841. doi:10.1016/j.celrep.2014.05.012
51. Yu Y, Cai W, Pei CG, et al. Rhamnazin, a novel inhibitor of VEGFR2 signaling with potent antiangiogenic activity and antitumor efficacy. Biochem Bioph Res Co. 2015;458(4):913–919. doi:10.1016/j.bbrc.2015.02.059
52. Yu Y, Yu J, Pei CG, et al. Xanthatin, a novel potent inhibitor of VEGFR2 signaling, inhibits angiogenesis and tumor growth in breast cancer cells. Int J Clin Exp Pathol. 2015;8(9):10355–10364.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.