Back to Journals » Infection and Drug Resistance » Volume 13
Antimicrobial Susceptibility of Mycobacterium abscessus Complex Clinical Isolates from a Chinese Tertiary Hospital
Authors Guo Y, Cao X, Yu J, Zhan Q, Yang J, Wu X, Wan B, Liu Y, Yu F
Received 4 March 2020
Accepted for publication 9 June 2020
Published 26 June 2020 Volume 2020:13 Pages 2001—2010
DOI https://doi.org/10.2147/IDR.S252485
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Yinjuan Guo,1,2 Xingwei Cao,3 Jingyi Yu,4 Qing Zhan,3 Jinghui Yang,1 Xiaocui Wu,1 Baoshan Wan,1 Yin Liu,1 Fangyou Yu1,2
1Department of Laboratory Medicine, Shanghai Pulmonary Hospital, Tongji University School of Medicine, Shanghai 200082, People’s Republic of China; 2Shanghai Key Laboratory of Tuberculosis, Shanghai Pulmonary Hospital, Tongji University School of Medicine, Shanghai 200082, People’s Republic of China; 3Jiangxi Provincial Key Laboratory of Medicine, Clinical Laboratory of the Second Affiliated Hospital of Nanchang University, Nanchang 330000, People’s Republic of China; 4Department of Laboratory Medicine, Wenzhou Medical University, Wenzhou 325000, People’s Republic of China
Correspondence: Fangyou Yu Email [email protected]
Introduction: Mycobacterium abscessus complex (MABC) is a group of important infectious agents that are highly associated with drug resistance, and antibiotic treatment is usually ineffective. This study investigated the characteristics of antimicrobial susceptibility of MABC isolates and the synergy between certain β-lactam combinations against MABC infection.
Methods: We collected 129 MABC isolates from patients with lower respiratory tract infections and categorized them into three subspecies. The minimum inhibitory concentrations (MICs) of 15 antimicrobials for the MABC isolates were determined using commercial Sensititre RAPMYCOI MIC plates and the broth microdilution method, as recommended in the CLSI (M24-A2). In addition, the MICs of imipenem, alone and with ceftazidime and/or avibactam, were assessed in vitro for all isolates. The erm(41) and rrl genes were also sequenced.
Results: The MABC isolates exhibited > 80% resistance to 11 of the 15 antimicrobials. Regarding the remaining four antimicrobials, the isolates were least resistant to tigecycline (12.4%) and amikacin (3.9%), and only partially resistant to two cefoxitin (39.5%) and imipenem (40.3%). Compared with M. massiliense isolates, M. abscessus and M. bolletii isolates were more resistant to amikacin and imipenem, whereas M. abscessus was significantly less resistant to tigecycline relative to M. massiliense and M. bolletii isolates. The clarithromycin inducible resistance rate was 68.4% and 74.3% among M. bolletii and M. abscessus isolates. Furthermore, 88.7% of the M. abscessus isolates carried a T at position 28 of erm(41), which is associated with inducible clarithromycin resistance. In addition, compared to imipenem with avibactam only, the MIC50 and MIC90values of imipenem after adding ceftazidime plus avibactam were decreased fourfold.
Conclusion: The antimicrobial resistance rates and the characteristics of the erm(41) gene associated with inducible clarithromycin resistance were different among the three MABC subspecies. There was also synergy between imipenem and 100μg/mL ceftazidime against MABC isolates.
Keywords: Mycobacterium abscessus complex, resistance, erm(41), synergy, dual β-lactam therapy
Introduction
Mycobacterium abscessus complex (MABC) is a group of pathogens that account for 80% of rapidly growing mycobacteria (RGM) isolates. MABC strains are ubiquitous environmental bacteria often found in dust, soil, and water.1 They often cause pulmonary infections (which can occur in patients with cystic fibrosis), complicated infections of the skin and soft tissues, and disseminated infections, leading to high mortality rates.2 MABC strains can be classified into three clearly divergent subspecies: M. abscessus subsp. Abscessus(M. abscessus), M. abscessus subsp. Massiliense(M. massiliense), and M. abscessus subsp. bolletii (M. bolletii) based on genomic analysis, which has dramatically increased our knowledge regarding MABC.3 The high rates of antimicrobial resistance of MABC strains can render even combination antibiotic treatment ineffective.4 Consequently, identifying novel treatment approaches is imperative.
Combination drug regimens, which can involve clarithromycin (CLA), amikacin (AMK), azithromycin (AZM), the cephalosporin (a type of β-lactam) cefoxitin (FOX), and the carbapenem (another type of β-lactam) imipenem (IPM), are recommended by the American Thoracic Society and the Infectious Disease Society of America, but less than half of patients with MABC infection can be cured with these treatments.5,6 One report indicated that M. abscessus and M. bolletii isolates often exhibit inducible resistance to CLA, while M. massiliense isolates were mostly susceptible.7 However, another study reported that M. abscessus is less responsive to CLA.8 Because of the high rates of resistance, treatment for MABC infections is often time-consuming and costly.
Recently, several molecular mechanisms underlying CLA resistance were identified. The primary innate mechanism underlying CLA resistance in MABC involves an increase in expression of the erythromycin ribosomal methylase gene, erm(41). Induction of erm(41) is mediated by the transcription factor whiB7,9 and rifabutin suppresses inducible CLA resistance by preventing induction of whiB7 and erm(41) expression.10 Inducible resistance to CLA was identified in a sequevar with an intact erm(41) but with a single-nucleotide polymorphism (C to T) at position 2811 (C at position 28 in M. abscessus results in CLA susceptibility). Notably, M. massiliense strains with a CLA susceptible phenotype have a nonfunctional erm(41) gene.7 Acquired high-level resistance to CLA can be caused by a point mutation (A2058G or A2059G) in the rrl gene (which encodes domain V of 23S rRNA).11
In addition, there are some mechanisms of resistance of MABC to carbapenems. Resistance to β-lactams (such as the carbapenem IPM) was found to be related to the initial characterization of the MABC β-lactamase (BlaMab) and transpeptidases.12,13 The development of new antimicrobials is important to overcome the emergence of carbapenem resistance;14 currently, there are several active pharmaceutical programs developing β-lactamase inhibitors. Previous studies reported that the β-lactamase inhibitor avibactam (AVI) could effectively inhibit BlaMab and thus improve the effects of β-lactams in MABC infection.13,15 Recent evidence suggests that dual β-lactam therapy may be more effective than using a single β-lactam.16
In this study, MABC isolates were assessed for susceptibility to various antimicrobials, and the relationship between the erm(41) gene and inducible CLA resistance in each subspecies of MABC was explored. The results provide guidance for the empirical therapy of RGM infections. Additionally, we assessed the individual and combined effects of two β-lactams (imipenem [IPM] and ceftazidime [CAZ]) and a β-lactamase inhibitor (avibactam [AVI]), and the results provide further insights into the treatment options.
Materials and Methods
Collection of MABC Isolates, Culture Conditions, and Informed Consent
Between 2014 and 2018, 129 MABC isolates were randomly isolated from the sputum and bronchoalveolar fluids of patients with clinical signs of lower respiratory tract infections in three tertiary hospitals (Shanghai Pulmonary Hospital, the Second Affiliated Hospital of Nanchang University, and the Second Affiliated Hospital of Suzhou University). All isolates were cultured in Middlebrook 7H10broth (BD, France) supplemented with 10% (vol/vol) oleic acid-albumin-dextrose-catalase (Thermo Fisher Scientific, USA). The cultures were incubated at 37°C for 7 days.
The Ethics Committee of Shanghai Pulmonary Hospital, Tongji University School of Medicine, exempted this study from ethical review because the assessment of the bacteria was part of routine hospital laboratory procedures. Verbal informed consent was obtained from all participants.
Identification of Subspecies and Detection of erm(41) and rrl Mutations
Genomic DNA was extracted from the MABC cultures for identification based on the sequences of the genes 16S rRNA, rpoB, and hsp65. The 16S rRNA gene was amplified using primers F (5ʹ-AGAGTTTGATCCTGGCTCAG-3ʹ) and R (5ʹ-ACGGGCGGTGTCTACAA-3ʹ), as previously described.17 A 723-bp fragment of the rpoB gene was amplified using primers rpoBF (5ʹ-GGCAAGGTCACCCCGAAGGG-3ʹ) and rpoBR (5ʹ-AGCGGCTGCTGGGTGATCATC-3ʹ), as previously described.18 The hsp65 gene was amplified using primers hsp65F (5ʹ-ACCAACGATGGTGTGTCCAT-3ʹ) and hsp65R (5ʹ-CTTGTCGAACCGCATACCCT-3ʹ), as previously described.19 The erm(41) gene (F:5ʹ-TGGTATCCGCTCACTGATGA-3ʹ and R:5ʹ-GCGGTGGATGATGGAAAG-3ʹ) and rrl gene (F: 5ʹ–CCTGCACGAATGGCGTAACG-3ʹ and R: 5ʹ-CACCAGAGGTTCGTCCGTC-3ʹ) were amplified as described by Maurer et al.20 The Basic Local Alignment Search Tool (BLAST) program (https://blast.ncbi.nlm.nih.gov) was used for gene sequence comparisons.
Antibiotics
The carbapenem β-lactam imipenem (IPM; Sigma), the cephalosporin β-lactam ceftazidime (CAZ; Sigma), and the β-lactamase inhibitor avibactam (AVI; IHMA Inc.) were used alone or in combination. AVI was kindly provided by Prof. Chen Liang from Newark University (USA). Stock solutions of IPM and AVI were prepared in sterile water, while a stock solution of CAZ was prepared in dimethyl sulfoxide (DMSO). The stock concentrations of AVI, IPM, and CAZ were 1, 10, and 10 mg/mL, respectively. These stock solutions were diluted to the desired working concentrations with 7H9 medium.
Antimicrobial Susceptibility Tests
The susceptibility of all isolates to the following 15 antimicrobials (Thermo Fisher Scientific) were assessed according to the manufacturer’s instructions: amikacin (AMK), ciprofloxacin (CIP), moxifloxacin (MXF), trimethoprim–sulfamethoxazole (SXT), linezolid (LZD), ceftriaxone (CRO), cefepime (FEP), cefoxitin (FOX), tobramycin (TOB), tigecycline (TGC), minocycline (MIN), doxycycline (DOX), amoxicillin/clavulanic acid (AMC), imipenem (IPM) and clarithromycin (CLA). The minimum inhibitory concentrations (MICs) of the 15 antimicrobials for the 129 MABC isolates were determined using Sensititre RAPMYCOI MIC plates (Thermo Fisher Scientific) and the broth microdilution method, as recommended in the Clinical and Laboratory Standards Institute (CLSI)(M24-A2). The MIC ranges tested and CLSI M24-A221 breakpoints for the 15 antimicrobials are listed in Table 2. We assessed the MICs of the 15 antimicrobials after 72 h of incubation, while we assessed the MICs of CLA after 3 and 14 days of incubation, in order to assess inducible CLA resistance. M. abscessus ATCC19,977 was used as the reference strain to compare the MICs.
 |
Table 1 Antimicrobial Susceptibility of 129 Mycobacterium abscessus Complex (MABC) Isolates |
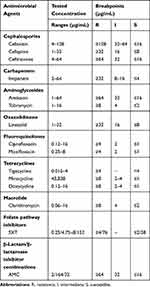 |
Table 2 Antimicrobial Concentration Ranges for Drug Susceptibility Testing and MIC Breakpoints of Antimicrobial Agents |
Synergistic Antimicrobial Susceptibility Tests
The MIC values of IPM, CAZ, and AVI were also assessed, alone and in combination (involving 100μg/mL CAZ and 4μg/mL AVI). The MICs were determined for each of the 129 MABC isolates using the broth microdilution method in 96-well plates after incubation at 37°C for 48 h, according to CLSI (M24-A2) and Chen et al.22
Statistical Analysis
Differences between groups were compared using Pearson’sχ2 test. A two-tailed P<0.05 was considered statistically significant. All computations were performed using Graph Pad Prism (version 7.0, La Jolla, CA, USA).
Results
MABC Subspecies Identification
M. abscessus, M. bolletii and M. massiliense accounted for 75.2% (97/129), 14.7% (19/129), and 10.1% (13/129) of the MABC isolates.
Antimicrobial Susceptibility Profiles
The antimicrobial susceptibilities of the 129 MABC isolates of the three subspecies are summarized in Table 1. The drug sensitivity results of each isolates were provided in the supplementary data (the file called 16_Apr_2020_16_Apr_2020_data.xlsx). In general, the antimicrobial resistance rates were very high. The MABC isolates exhibited >90% resistance to nine of the 15 antimicrobials tested (FEP, CRO AMC, IMI, MIN, CIP, MXF, SXT, and TOB), and the rates of resistance to DOX and LZD were >70%. But the isolates were most susceptible to TGC (87.6%) and AMK (65.1%).
The rate of FOX resistance was significantly lower than the rates of resistance to the two other cephalosporins (FEP and CRO) (P<0.05). The rates of FOX resistance for M. abscessus, M. massiliense, and M. bolletii were 34.0%, 46.2%, and 63.2%, respectively (P<0.05). Additionally, the rate of IPM resistance was significantly lower for M. massiliense (23.1%) than M. abscessus (41.2%) and M. bolletii (47.4%) (P<0.05).
CLA Susceptibility Testing and erm Genotyping in MABC Isolates
Out of all the MABC isolates, 72 (74.2%) were susceptible to CLA on day 3 but resistant on day 14 (indicating inducible resistance). Although the CLA resistance rate was higher on day 14 than on day 3 for M. abscessus (88.7% versus 14.4%), the corresponding rates were similar for M. massiliense (7.7% versus 7.7%), with none of the M. massiliense isolates exhibiting inducible resistance. Of the 19 M. bolletii isolates studied, 68.4% had inducible resistance, which could not be attributed to the presence of the erm(41) T28 polymorphism. In contrast, the majority of M. abscessus isolates (86 isolates) belonged to the erm(41) T28 sequevar. Of the 129 isolates, 24 (18.6%) were susceptible to CLA on day 14. Among these, 14 (10.9%) had an erm(41) deletion (comprising all 13 M. massiliense isolates and one M. abscessus isolates) and the remaining 10 (7.8%) were M. abscessus isolates with the erm(41) C28 mutation. Among the 129 MABC isolates, an rrl point mutation was found in only one isolate (an M. massiliense isolate). There were no erm(41) or rrl mutations in the M. bolletii isolates.
Susceptibility of MABC Isolates to β-Lactams (IPM and CAZ) and a β-Lactamase Inhibitor (AVI)
All 129 MABC isolates underwent susceptibility testing regarding IPM, CAZ, and AVI used alone or in combination. The average MIC50 and MIC90values are shown in Table 3. The drug sensitivity results of each isolates were provided in the supplementary data (the file called 16_Apr_2020_16_Apr_2020_data.xlsx). The MIC values for CAZ alone ranged from 128 to >1024 μg/mL, with average MIC90 and MIC50values of 1024 and 512 μg/mL, respectively; after adding AVI, the MIC values for CAZdid not change. The MIC values for IPM alone ranged from 2 to >64 μg/mL, with average MIC90 and MIC50 values of 32 and 16 μg/mL, respectively; there were also no significant changes when IPM was combined with AVI. However, after adding CAZ (100 μg/mL) only to IPM, there was a 4-fold reduction in the MIC50 and MIC90 values for IPM compared to when IPM was used alone. Additionally, after adding both CAZ (100 μg/mL) and AVI (4 μg/mL), the MIC range of IPM for the MABC isolates decreased from 2–>64 to 0.5–16 μg/mL; there was a 4-fold reduction in the MIC50 and MIC90 values compared to when IPM was used with AVI only, which also occurred in the reference strain (M. abscessus ATCC 19977).
 |
Table 3 Antimicrobial Activities Imipenem (IPM), Ceftazidime (CAZ), and Avibactam (AVI) Alone or in Combination, Against 129 Mycobacterium abscessus Complex (MABC) Isolates |
Discussion
In this study, we assessed the antimicrobial susceptibility of 129 MABC strains belonging to three subspecies and examined the association between the erm(41) gene and inducible CLA resistance. The MABC subspecies exhibited varied resistance rates to antimicrobials. When the antimicrobial resistance rates obtained in the present study were compared with the results of previous studies, we found that there were high rates of resistance to multiple antimicrobials among the three MABC subspecies. The resistance rates regarding fluoroquinolones (CIP and MXF) and other broad-spectrum antibiotics (such as DOX, SXT, and MIN) were similar (all >70%) to those previously reported for MABC isolates in Taiwan Province of China,23 as well as in Japan,24 Thailand,25 the USA,26 and France.27 In contrast, the rate of IPM resistance varied significantly among different studies. It was 40.9% in our study, which was in agreement with the rates reported in Beijing28 and Japan.24 However, studies conducted in 2017 in Taiwan (61%),23 2015 in Australia (68%),29 and 2018 in Shanghai (65%)30 reported higher rates of IPM resistance. Notably, the M. abscessus isolates in our study had a higher resistance rate to FOX (34.02%) than the M. abscessus strains in a study in Japan (16.7%).31 In Korea32 and Japan,33 the TOB resistance rate was 30–32%, while it was 90% in our study. These variations may be due to differences among the diverse studies in patient treatment histories or in the isolates.
In the early 2000s, when LZD was first used clinically, it was reported to be active against many species of RGM.34 However, we found that the rate of resistance to LZD was high, similar to rates reported in Taiwan (70%)23 and the UK (96%),35 but higher than the 5% rate reported by a study in Korea.36 Notably, there have been few reports of a rate of LZD resistance as high as that found in M. bolletii in our study (73.7%). With the exception of the resistance rate for LZD, the resistance rates of M. bolletii were higher than those of M. abscessus and M. massiliense. Therefore, precise differentiation between these subspecies is important for clinical purposes.
Bastian et al reported that MABC infections were usually poorly responsive to CLA because of acquired and/or inducible resistance.37 Mutations in the rrl gene confer acquired CLA resistance, while a single-nucleotide polymorphism (T28) in erm(41) at position 28 leads to inducible CLA resistance.7 Notably, the acquired CLA resistance rate (on day 3) in the M. abscessus isolates of 14.4% (14/97) was similar to the rate reported in South Korea (15.84%), higher than those reported in France (9.09%) and the USA (2.51%), and lower than that reported in China (33.95%).27,38,39 We hypothesize that the geographic diversity in the population structure of MABC may be a major reason why researchers from various regions observed contradictory results. In addition, the high rate of inducible resistance that we identified is consistent with those reported by other studies.38,40 This supports the need to modify the CLA susceptibility test recommended by the CLSI,21 by assessing the MIC of CLA after an additional longer period of incubation (eg, 14 days).
In our study, most of the mutations identified involved the erm(41) gene (75.1%) rather than the rrl gene (0.8%). We concluded that the erm(41) mutations may be associated with the treatment failure that occurs in many cases of MABC infection. Nash et al and Kim et al studied the molecular profiles of MABC isolates and found that the erm(41) C28 polymorphism was related to susceptibility.7,41 Of our129 isolates, 23 (17.8%) were susceptible to CLA. Among these, 14 (10.9%) had an erm(41) deletion (comprising all 13 M. massiliense isolates and one M. abscessus isolate) and10 (7.8%) were M. abscessus isolates with the erm(41) C28 mutation. Of the 19 M. bolletii isolates studied, 68.4% exhibited inducible CLA resistance, which could not be attributed to the presence of the erm(41) T28 polymorphism. This indicates that there is another mechanism of resistance to CLA in M. bolletii. In addition, it has been reported that acquisition of CLA resistance is 100% mediated by structural 50S ribosomal subunit mutations for the M. abscessus erm(41) C28 sequevar and M. massiliense, whereas it is less common for the M. abscessus erm(41) T28 sequevar and M. bolletii(other mechanisms may be responsible for CLA resistance in these strains).42 Hence, we hypothesize that detecting an erm(41) deletion may differentiate M. massiliense from M. abscessus and M. bolletii, but more extensive research is needed to accurately define the reliability of using erm(41) deletions to identify M. massiliense.
Given the increasing prevalence of multidrug-resistant MABC infections, the development of novel treatment regimens is imperative. Although knowledge regarding the efficacy of β-lactams and β-lactamase inhibitors in MABC remains limited, several studies have used the available data to develop synergistic treatment regimens for treating MABC infections.22,43 In this study, we evaluated the in vitro susceptibility of the 129 MABC isolates to IPM in the presence of CAZ and/or AVI. The MIC50 and MIC90values of IPM after the addition of CAZ plus AVI, compared to the values after the addition of AVI only, decreased 4-fold to 4 and 8μg/mL, respectively. Similarly, the addition of 100μg/mL CAZ only led to 4-fold decreases in the MIC50 and MIC90values of IPM (while there were no significant changes in the MIC of IPM when IPM was combined with 4 μg/mL AVI only). The initial resistance or intermediate resistance to IPM changed to sensitivity. Interestingly, CAZ alone had poor activity against MABC but combining IPM and CAZ led to effective activity, indicating that the effect of triple therapy may be driven primarily by CAZ rather than AVI. In contrast, Lefebvre et al.13 reported that inhibition of the β-lactamase BlaMab by AVI improves the in vitro effects of IPM against MABC and Pandey et al.22 reported that 4μg/mL AVI enhanced the bactericidal activity of the β-lactam ceftaroline; it can be deduced that the β-lactamase of MABC strains probably hydrolyzes IPM (though this was not confirmed by our study). Future studies will need to identify effective measures for reducing exposure to these difficult-to-treat pathogens.
Conclusion
The MABC isolates exhibited varied resistance rates to antimicrobials. The antimicrobial susceptibility profile and the characteristics of the erm(41) gene associated with inducible CLA resistance were different among the three MABC subspecies. Additionally, there was synergy between the β-lactam IPM and 100μg/mL of the β-lactam CAZ against MABC infection.
Ethics Statement
As the assessment of the Mycobacterium abscessus complex (MABC) isolates in this study was part of routine hospital laboratory procedures, the study was exempted from ethics review by the Ethics Committee of Shanghai Pulmonary Hospital, Tongji University School of Medicine.
Acknowledgments
We thank Prof. Liang Chen (Newark University, USA) for providing the avibactam used in this study.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no conflicts of interest.
References
1. Griffith DE, Girard WM, Wallace RJ
2. Lee MR, et al. Mycobacterium abscessus complex infections in humans. Emerg Infect Dis. 2015;21:1638–1646. doi:10.3201/2109.141634
3. Leao SC, Tortoli E, Euzeby JP, Garcia MJ. Proposal that Mycobacterium massiliense and Mycobacterium bolletii be united and reclassified as Mycobacterium abscessus subsp. bolletii comb. nov., designation of Mycobacterium abscessus subsp. abscessus subsp. nov. and emended description of Mycobacterium abscessus. Int J Syst Evol Microbiol. 2011;61:2311–2313. doi:10.1099/ijs.0.023770-0
4. Jarand J, Levin A, Zhang L, et al. Clinical and microbiologic outcomes in patients receiving treatment for Mycobacterium abscessus pulmonary disease. Clin Infect Dis. 2011;52:565–571. doi:10.1093/cid/ciq237
5. Stout JE, Koh WJ, Yew WW. Update on pulmonary disease due to non-tuberculous mycobacteria. Int J Infect Dis. 2016;45:123–134. doi:10.1016/j.ijid.2016.03.006
6. Griffith DE, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175:367–416. doi:10.1164/rccm.200604-571ST
7. Nash KA, Brown-Elliott BA, Wallace RJ
8. Nessar R, Cambau E, Reyrat JM, Murray A, Gicquel B. Mycobacterium abscessus: a new antibiotic nightmare. J Antimicrob Chemother. 2012;67:810–818. doi:10.1093/jac/dkr578
9.. Richard M, Gutierrez AV, Kremer L. Dissecting erm(41)-mediated macrolide-inducible resistance in Mycobacterium abscessus. Antimicrob Agents Chemother. 2020;64. doi:10.1128/AAC.01879-19.
10. Aziz DB, Go ML, Dick T. Rifabutin suppresses inducible clarithromycin resistance in Mycobacterium abscessus by blocking induction of whiB7 and erm41. Antibiotics. 2020;9. doi:10.3390/antibiotics9020072.
11. Wallace RJ
12. Soroka D, et al. Characterization of broad-spectrum Mycobacterium abscessus class A beta-lactamase. J Antimicrob Chemother. 2014;69:691–696. doi:10.1093/jac/dkt410
13. Lefebvre AL, et al. Inhibition of the beta-Lactamase BlaMab by Avibactam improves the in vitro and in vivo efficacy of imipenem against Mycobacterium abscessus. Antimicrob Agents Chemother. 2017;61. doi:10.1128/AAC.02440-16
14. Rahme C, Butterfield JM, Nicasio AM, Lodise TP. Dual beta-lactam therapy for serious Gram-negative infections: is it time to revisit? Diagn Microbiol Infect Dis. 2014;80:239–259. doi:10.1016/j.diagmicrobio.2014.07.007
15. Soroka D, et al. Inhibition of beta-lactamases of mycobacteria by avibactam and clavulanate. J Antimicrob Chemother. 2017;72:1081–1088. doi:10.1093/jac/dkw546
16. Story-Roller E, Maggioncalda EC, Cohen KA, Lamichhane G. Mycobacterium abscessus and beta-Lactams: emerging insights and potential opportunities. Front Microbiol. 2018;9:2273. doi:10.3389/fmicb.2018.02273
17. Yu XL, et al. Identification and characterization of non-tuberculous mycobacteria isolated from tuberculosis suspects in Southern-central China. PLoS One. 2014;9:e114353. doi:10.1371/journal.pone.0114353
18. Macheras E, et al. Inaccuracy of single-target sequencing for discriminating species of the Mycobacterium abscessus group. J Clin Microbiol. 2009;47:2596–2600. doi:10.1128/JCM.00037-09
19. Nie W, Duan H, Huang H, Lu Y, Chu N. Species identification and clarithromycin susceptibility testing of 278 clinical nontuberculosis Mycobacteria Isolates. Biomed Res Int. 2015;506598. doi:10.1155/2015/506598
20. Maurer FP, Ruegger V, Ritter C, Bloemberg GV, Bottger EC. Acquisition of clarithromycin resistance mutations in the 23S rRNA gene of Mycobacterium abscessus in the presence of inducible erm(41). J Antimicrob Chemother. 2012;67:2606–2611. doi:10.1093/jac/dks279
21. Institute CA, L S. Susceptibility testing of Mycobacteria, Nocardiae, and other aerobic actinomycetes second edition: approved standard M24-A2. CLSI. 2011. Wayne, PA, USA.
22. Pandey R, et al. Dual beta-Lactam combinations highly active against Mycobacterium abscessus Complex In Vitro. mBio. 2019;10. doi:10.1128/mBio.02895-18
23. Lee MC, et al. Antimicrobial resistance in Mycobacterium abscessus complex isolated from patients with skin and soft tissue infections at a tertiary teaching hospital in Taiwan. J Antimicrob Chemother. 2017;72:2782–2786. doi:10.1093/jac/dkx212
24. Kusuki M, et al. Determination of the antimicrobial susceptibility and molecular profile of clarithromycin resistance in the Mycobacterium abscessus complex in Japan by variable number tandem repeat analysis. Diagn Microbiol Infect Dis. 2018;91:256–259. doi:10.1016/j.diagmicrobio.2018.02.008
25. Ananta P, et al. Analysis of drug-susceptibility patterns and gene sequences associated with clarithromycin and amikacin resistance in serial Mycobacterium abscessus isolates from clinical specimens from Northeast Thailand. PLoS One. 2018;13:e0208053. doi:10.1371/journal.pone.0208053
26. Brown-Elliott BA, Wallace R, J
27. Mougari F, et al. Standardized interpretation of antibiotic susceptibility testing and resistance genotyping for Mycobacterium abscessus with regard to subspecies and erm41 sequevar. J Antimicrob Chemother. 2016;71:2208–2212. doi:10.1093/jac/dkw130
28. Li YM, et al. Prevalence and antimicrobial susceptibility of Mycobacterium abscessus in a general hospital, China. Biomed Environ Sci. 2016;29:85–90. doi:10.3967/bes2016.009
29. Chua KY, Bustamante A, Jelfs P, Chen SC, Sintchenko V. Antibiotic susceptibility of diverse Mycobacterium abscessus complex strains in New South Wales, Australia. Pathology. 2015;47:678–682. doi:10.1097/PAT.0000000000000327
30. Shen Y, et al. In vitro susceptibility of Mycobacterium abscessus and Mycobacterium fortuitum isolates to 30 antibiotics. Biomed Res Int. 2018:4902941. doi:10.1155/2018/4902941.
31. Aono A, et al. Antimicrobial susceptibility testing of Mycobacteroides (Mycobacterium) abscessus complex, Mycolicibacterium (Mycobacterium) fortuitum, and Mycobacteroides (Mycobacterium) chelonae. J Infect Chemother. 2019;25:117–123. doi:10.1016/j.jiac.2018.10.010
32. Park S, et al. In vitro antimicrobial susceptibility of Mycobacterium abscessus in Korea. J Korean Med Sci. 2008;23:49–52. doi:10.3346/jkms.2008.23.1.49
33. Yoshida S, et al. Further isolation of Mycobacterium abscessus subsp. abscessus and subsp. bolletii in different regions of Japan and susceptibility of these isolates to antimicrobial agents. Int J Antimicrob Agents. 2013;42:226–231. doi:10.1016/j.ijantimicag.2013.04.029
34. Wallace RJ
35. Cowman S, Burns K, Benson S, Wilson R, Loebinger MR. The antimicrobial susceptibility of non-tuberculous mycobacteria. J Infect. 2016;72:324–331. doi:10.1016/j.jinf.2015.12.007
36. Park J, Cho J, Lee CH, Han SK, Yim JJ. Progression and Treatment outcomes of lung disease caused by mycobacterium abscessus and Mycobacterium massiliense. Clin Infect Dis. 2017;64:301–308. doi:10.1093/cid/ciw723
37. Bastian S, et al. Assessment of clarithromycin susceptibility in strains belonging to the Mycobacterium abscessus group by erm(41) and rrl sequencing. Antimicrob Agents Chemother. 2011;55:775–781. doi:10.1128/AAC.00861-10
38. Lee SH, et al. The drug resistance profile of Mycobacterium abscessus group strains from Korea. Ann Lab Med. 2014;34:31–37. doi:10.3343/alm.2014.34.1.31
39. Li B, et al. Relationship between antibiotic susceptibility and genotype in Mycobacterium abscessus clinical isolates. Front Microbiol. 2017;8:1739. doi:10.3389/fmicb.2017.01739
40. Hanson KE, Slechta ES, Muir H, Barker AP. Rapid molecular detection of inducible macrolide resistance in Mycobacterium chelonae and M. abscessus strains: a replacement for 14-day susceptibility testing? J Clin Microbiol. 2014;52:1705–1707. doi:10.1128/JCM.03464-13
41. Kim HY, et al. Mycobacterium massiliense is differentiated from Mycobacterium abscessus and Mycobacterium bolletii by erythromycin ribosome methyltransferase gene (erm) and clarithromycin susceptibility patterns. Microbiol Immunol. 2010;54:347–353. doi:10.1111/j.1348-0421.2010.00221.x
42. Mougari F, et al. Selection of resistance to clarithromycin in Mycobacterium abscessus subspecies. Antimicrob Agents Chemother. 2017;61. doi:10.1128/AAC.00943-16
43. Kaushik A, Ammerman NC, Parrish NM, Nuermberger EL. New beta-Lactamase inhibitors nacubactam and zidebactam improve the in vitro activity of beta-Lactam antibiotics against Mycobacterium abscessus complex clinical isolates. Antimicrob Agents Chemother. 2019;63. doi:10.1128/AAC.00733-19.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
