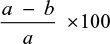Back to Journals » Biologics: Targets and Therapy » Volume 11
Anticholinesterase constituents from the leaves of Spondias mombin L. (Anacardiaceae)
Authors Elufioye TO , Obuotor EM, Agbedahunsi JM , Adesanya SA
Received 2 March 2017
Accepted for publication 10 May 2017
Published 2 August 2017 Volume 2017:11 Pages 107—114
DOI https://doi.org/10.2147/BTT.S136011
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Doris Benbrook
Taiwo Olayemi Elufioye,1 Efere M Obuotor,2 Joseph M Agbedahunsi,3 Saburi A Adesanya4
1Department of Pharmacognosy, Faculty of Pharmacy, University of Ibadan, Ibadan, 2Department of Biochemistry, 3Drug Research and Production Unit, Faculty of Pharmacy, 4Department of Pharmacognosy, Faculty of Pharmacy, Obafemi Awolowo University, Ile Ife, Osun, Nigeria
Abstract: Spondias mombin has been used in traditional medicine for the management of several diseases, including memory loss. This study aimed to evaluate the cholinesterase inhibitory activity of the methanol extract of the leaves and its derived fractions, as well as carry out detailed phytochemical investigations leading to the isolation and characterization of bioactive compounds from the plant. The acetyl cholinesterase (AChE) and butyryl cholinesterase (BUChE) inhibitory activities were evaluated by colorimetric and thin-layer chromatography bioautographic assay techniques. The ethyl acetate fraction was most active against both enzymes, with percentage inhibition of 58.10 ± 1.08% and 52.66 ± 1.34% against AChE and BUChE, respectively. Three compounds, namely, botulin, campesterol and phytol, with IC50 of 0.88 μg/mL (AChE), 4.67 μg/mL (BuChE); 1.89 μg/mL (AChE), 4.08 μg/mL (BuChE) and 12.51 μg/mL (AChE), 23.89 μg/mL (BuChE), respectively, were isolated from the supernatant of the ethyl acetate fraction. The isolated cholinesterase inhibitory compounds correlate with the known memory-enhancing property of the plant and thus support one of its uses in ethnomedicine.
Keywords: Alzheimer’s disease, acetylcholinesterase, butyryl cholinesterase, neurodegenerative, Spondias mombin
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disease characterized by low levels of acetylcholine (ACh) in the brain, with associated cognitive deficit and memory impairment.1 Acetylcholinesterase (AChE) and butyrylcholinesterase (BUChE) are enzymes that catalyze hydrolysis of ACh and butyrylcholine, respectively. The activities of these enzymes increase progressively in AD patients as the severity of dementia progresses,2 and their inhibition results in an increase in the levels of ACh and butyrylcholine in the brain, as well as a corresponding increase in cholinergic functions in AD patients.3 Thus, most drugs available today for the management of AD are cholinesterase inhibitors, such as galantamine, rivastigmine and donepezil4 even though their effectiveness in long-term treatment is debatable.5 Moreover, the primary targets recommended for AD treatment are both AChE and BuChE, but some inhibitors are more selective than others.6
There is still the need to search for newer anticholinesterase treatment for AD due to the serious side effects associated with available drugs. Natural products, especially medicinal plants, have been considered valuable sources of drugs for many conditions, including AD, especially since plants have been used to enhance memory traditionally.7
Spondias mombin, a medium-sized, occasionally large deciduous tree with long compound leaves, large panicles of small white flowers and yellow plumb-like fruits,8 is a common inclusion in remedies used in ethnomedicine in southwestern Nigeria as a memory enhancer and/or an antiaging agent.9 All parts of the S. mombin tree are of medicinal importance in traditional medicine. The leaves have been used as an oxytocic agent,10 particularly for the expulsion of placenta in goats and women when normal delivery of such is delayed or impossible and as an ingredient in postpartum medication.11,12 It is useful as an antidiarrheal agent for the treatment of wounds and as an astringent.13–15 It is also used in treating inflammatory and arthritic conditions.16 In Nigeria, it is used in treating intestinal disorders, particularly those associated with typhoid, diarrhea and dysentery.17 It is also a component of traditional antituberculosis recipes.15 The fruit decoction is used as a diuretic and febrifuge. The bark and leaves are used as an emetic and for hemorrhoids, gonorrhea and leucorrhea.18 A decoction of the leaves and flower is taken as a relief for stomach ache, various inflammatory conditions and wound healings.19 In southwestern Nigeria, the leaves are used traditionally for the treatment of psychiatric disorders.20
Several biological activities of the plant have been reported, including antiviral,21–23 antibacterial and molluscicidal,15,23 β-lactamase inhibitory,24 anti-inflammatory,16 wound healing,19 antipsychotic, anticonvulsant and sedative,18,20 abortifacient,11 oxytocic,25 antimicrobial,26 antifertility,27 antigonadotrophic,28 hematinic,29 antioxidant,30 antidiabetic31 and anticholinesterase activities.32
The compounds isolated from this plant include caryophyllene, myrcene, hexanal, 3-hexenol and (e)-2-hexenal from the fruits,33 cinnamic acid, 4-hydroxycinnamic acid, 3-methoxy-4-hydrocinnamic acid, 3-methoxy-4-hydroxycinnamic acid, benzaldehyde, linalool, hexanoic acid, alpha-terpineol, palmitic acid and octanoic acid,34 as well as anacardic acid.24 The leaves and stems contain two ellagitannins galloylgeraniin and geraniin and two caffeoyl esters 5-O-caffeoylquinic acid and 2-O-caffeoyl hyroxycitric acid, which have also been reported.21,22 Pelandjuaic acid and 6-(81Z,111Z-heptadecadienyl) salicyclic acid have been reported from the ethanolic extract of the leaves and stems of S. mombin.23 Two new phytosterols (mombintanes I and II),35 one new coumarin and three new flavonoids (mombinrin, mombincone, mombinoate and mombinol, respectively) have also been reported.36 The plant also contains estra-17-propoxy, 3,4–dimethoxy–3yl benzoate.37
This study assessed the cholinesterase inhibitory activity of S. mombin leaves, and isolated and characterized its anticholinesterase compounds.
Materials and methods
Chemicals
The chemicals used were as follows: acetylthiocholine iodide (ATChI), butyrylcholine chloride (BuChCl), 5,5′-dithio-bis-(2-nitrobenzoic acid) (DTNB), physostigmine (eserine) salicylate (Sigma-Aldrich, St Louis, MO, USA); and electric eel AChE (EC 3.1.1.7, type VI-s) and horse butyxylcholinesterase (EC 3.1.1.8) (Fluka Co, Germany). The other reagents and buffers, which include disodium hydrogen orthophosphate dihydrate (Na2HPO4∙2H2O) and sodium dihydrogen phosphate (NaH2PO4∙12H2O), were of analytical grade. Silica gel for vacuum liquid chromatography (VLC) (American Society for Testing and Materials [ASTM]) and precoated thin-layer chromatography (TLC) plates with silica gel G60 PF254 (EMD Millipore, Billerica, MA, USA).
Plant material collection and authentication
S. mombin was identified by Mr Oladele of the Department of Pharmacognosy, Faculty of Pharmacy, and was authenticated by Dr H Illoh of the Botany Department, Obafemi Awolowo University, Ile Ife, where herbarium specimen with herbarium number IFE 9572 was deposited. The leaves were collected from the Medicinal farm of the Obafemi Awolowo University Campus in August 2005.
Preparation of extract and fractions
The powdered leaves were extracted with 80% methanol by maceration for 72 hours, and the extract was concentrated to dryness at 40°C on a rotary evaporator. The crude extract was partitioned into n-hexane, ethyl acetate and water. Both the extract and the fractions were screened for their AChE and BuChE inhibitory activities.
Bulk extraction with ethyl acetate and precipitation studies
The powdered leaves of the plant were extracted with 100% ethyl acetate. Nonpolar lipid components were precipitated out by gradual addition of methanol. Both the filtered precipitate and the supernatants were then assessed for their cholinesterase inhibitory activities.
Phytochemical and cholinesterase analyses
TLC of both precipitates and supernatant was done with chloroform–hexane (7:3, v/v) as the solvent system. Some of the developed plates were sprayed with different phytochemical screening reagents, such as vanillin/sulfuric acid, antimony trichloride, Dragendorff’s reagent and anisaldehyde spray reagents. The other plates were used for the TLC bioautographic enzyme assay.
Cholinesterase inhibition assay
Cholinesterase inhibitory activities of the crude extract, fractions, precipitate, supernatant and isolated compounds were analyzed in a 96-well microplate reader according to the modified method of Ellman.38,39
The reaction mixture was made up of 2000 mL 100 mM phosphate buffer (pH 8.0), 100 mL of test sample stock solution in methanol (at 42.5 µg/mL final concentration), 100 mL enzyme, either AChE or BuChE at a final concentration of 0.003 µ/mL or 0.001 µ/mL, respectively, and 100 µL of DTNB (0.3 mM) prepared in 100 M phosphate buffer pH 7.0 containing 120 mM sodium bicarbonate. Preincubation of the assay mixture was done on a water bath at 37°C for 30 minutes following proper mixing, and the reaction started by the addition of 100 µL of ATChI or butyrylthiocholine chloride (BTChCI) at a final concentration of 0.5 mM. Methanol was used as the negative control, while eserin ((–) physostigmine) was used as the positive control. Change in absorbance at λmax 412 was recorded at ambient temperature every 30 seconds for 5 minutes. All determinations were done in triplicate, and percentage inhibition was calculated as follows:
|
where a is the ΔA/min of control, b is the ΔA/min of test sample and ΔA is the change in absorbance.
TLC bioautographic assay method was also used to monitor active spots.40 The various samples were spotted on precoated aluminum TLC plates (G60 PF254) and developed in appropriate solvent systems. The developed plates were air-dried, sprayed with 2.55 × 10–3 units/mL of the cholinesterase enzyme till saturation and then incubated at 37°C for at least 20 minutes before spraying with 0.5 mM of the substrate (ATChI or BTChCI, respectively) and DTNB. Positive result was indicated by white spots on a yellow background.
Isolation of bioactive components
VLC of S. mombin supernatant (19.20 g) was done on silica gel 60 (Sigma-Aldrich), using n-hexane, dichloromethane and methanol as solvents. Fractions were monitored using TLC on precoated G60 PF254 (0.25 mm) plates with vanillin/sulfuric acid reagent and heating at 100°C for a few minutes. A total of 103 subfractions collected were bulked into six based on their TLC patterns. The six bulked samples were tested for their AChE inhibitory activity using TLC bioautographic method. Active subfractions were bulked together and purified further using VLC, leading to the isolation of three bioactive compounds through preparative TLC (PTLC).
Analysis of bioactive compounds
The isolated compounds were subjected to a number of analyses, including different spectroscopic analyses such as 1H-nuclear magnetic resonance (NMR) and 13C-NMR (CDCl3, 300 Hz), TLC analysis in different solvent systems, solubility in water and determination of IC50.
Results
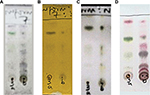
Medicinal plants are known to contain different classes of chemical compounds called secondary metabolites, which are responsible for their various biological activities. Phytochemical analysis of S. mombin was carried out on developed TLC plates. Partial purification of the methanol extract was done by precipitation. Thus, spraying the developed TLC plates of precipitate and supernatant of S. mombin with different phytochemical reagents is shown in Figure 1A–D for vanillin/H2SO4, Dragendorff’s reagent, antimony trichloride and anisaldehyde spray, respectively. Various colors were observed for the spots with the different reagents, indicating the possible nature of these chemical constituents. Organic compounds generally show color reactions to concentrated sulfuric acid41 and could be indicative for detecting steroidal and terpenoidal compounds.42 Figure 1A gave colors with vanillin/H2SO4, which are more prominent in the supernatant than in the precipitate. Alkaloids are detected with Dragendorff’s reagent as an orange–brown zone against a yellow background.42 This seems to be absent in the spotted samples, as seen in Figure 1B. Cardiac glycosides, saponins, terpenoids and flavonoids give colored spots with antimony trichloride, and this can be seen in this plant (Figure IC), while terpenoids can also be detected with anisaldehyde spray giving purple, blue or red spots.42 Again, more colors were detected in the supernatant with anisaldehyde when compared with the precipitate (Figure 1D). Several bioactive constituents belonging to various classes have been previously reported in S. mombin. These include coumarins, flavonoids,36 sterols,35 phenols33,34 and tannins.21,43
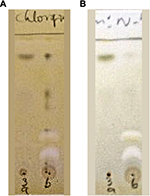
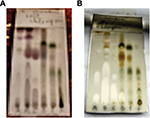
The precipitate and the supernatant were subjected to cholinesterase inhibitory analysis. Qualitative TLC bioautographic (Figure 2) as well as quantitative Ellman39 colorimetric assays (Table 1) showed that cholinesterase inhibitory activity of the supernatant was better than that of the precipitate. The phytochemical investigations, which showed more constituents in the supernatant when compared with the precipitate, corroborate the observed better cholinesterase inhibitory activity of the supernatant. Thus, activity-directed fractionation using VLC with TLC bioautography of the supernatant was carried out. Figure 3A shows the spots of the various bulked fractions after VLC, while Figure 3B shows the AChE assay results, from which it was observed that subfractions A–C were active. These were subjected to a combination of repetitive VLC and PTLC to isolate the active compounds, which were then identified through spectroscopic analysis.
Compound 1
Spectral data
The 1H-NMR spectrum (CDCl3, 300 Hz) showed signals at δ7.8 (m), δ7.75 (m), δ5.45 (t), δ4.6 (s) and δ4.5 (d). The 13C-NMR spectrum (CDCl3, 300 Hz) showed signals at 38.71 (C-1), 20.90 (C-2), 78.83 (C-3), 35.57 (C-4), 55.24 (C-5), 18.30 (C-6), 34.06 (C-7), 39.35 (C-8), 54.96 (C-9), 37.34 (C-10), 27.22 (C-11), 24.92 (C-12), 37.83 (C-13), 39.99 (C-14), 27.19 (C-15), 29.48 (C-16), 47.08 (C-17), 50.22 (C-18), 48.97 (C-19), 150.8 (C-20), 29.66 (C-21), 36.65 (C-22), 27.92 (C-23), 15.96 (C-24), 15.46 (C-25), 16.64 (C-26), 14.33 (C-27), 59.41 (C-28), 109.40 (C-29) and 19.70 (C-30).
Structure elucidation
Compound 1 (35 mg) was isolated as a white powder with retardation factor (Rf) 0.46 in hexane:chloroform 3:7 and Rf 0.35 in 100% chloroform (Table 2). It gave purple color with both vanillin/H2SO4 and anisaldehyde spray reagent, indicating its steroidal nature,44 and had a melting point range of 256°C–258°C (Table 3).
  | Table 2 TLC profiles of the isolated compounds Abbreviations: Rf, retardation factor; TLC, thin-layer chromatography. |
  | Table 3 Analysis of isolated compounds Abbreviations: AChE, acetyl cholinesterase; BUChE, butyryl cholinesterase. |
The 13C-NMR spectrum in the distortionless enhancement by polarization transfer experiment showed that there were 6CH3, 11CH2, 6CH and 7C. Thus, Compound 1 is a C-30 carbon compound.
The 1H-NMR showed a proton at δ4.5 (d) germinal to the hydroxyl group and had a corresponding carbon chemical shift at δ59.41. There was also an olefinic proton at δ4.6, which resided on the carbon at δ109.40. This proton was assigned to C-22, which is a terminal CH2 (Figure 4).
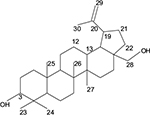  | Figure 4 Compound 1: betulin. |
Compound 2
Spectral data
The 13C-NMR data are as follows: 36.92 (C-1), 34.35 (C-2), 72.22 (C-3), 42.73 (C-4), 141.17 (C-5), 122.14 (C-6), 28.67 (C-7), 32.80 (C-8), 50.53 (C-9), 32.33 (C-10), 21.50 (C-11), 37.66 (C-12), 40.18 (C-13), 57.17 (C-14), 23.42 (C-15), 26.45 (C-16), 56.45 (C-17), 12.26 (C-18), 19.82 (C-19), 36.56 (C-20), 19.44 (C-21), 32.31 (C-22), 24.72 (C-23), 46.23 (C-24), 29.54 (C-25), 20.25 (C-26), 19.20 (C-27) and 12.40 (C-28).
Structure elucidation
Compound 2 (20 mg) had Rf values of 0.2 and 0.27 in hexane:chloroform 2:8 and 100% chloroform, respectively, with purple color in both vanillin/H2SO4 and anisaldehyde spray reagent (Table 2).
The 13C-NMR spectrum of Compound 2 showed that it is a C-28 compound. The attached proton test (APT) revealed 3 quaternary (3 C), 10 methylene (10 CH2), 6 methyl (6 CH3) and 9 methine (9 CH) carbons. The 1H-NMR showed one olefinic proton at δ5.40, with a corresponding carbon chemical shift of δ121.14 in the heteronuclear multiple quantum coherence spectrum. It also revealed the presence of one oxygenated methylene proton at δ3.5, as confirmed by the downfield chemical shift at δ72.22. In the heteronuclear multiple bond coherence spectrum, the diagnostic olefinic proton and the proton germinal to the OH showed connectivity with the quaternary carbon resonating at 141.17. From the combined 1H-NMR, 13C-NMR and APT experiments, as well as comparison with literature data, Compound 2 was identified to be campesterol (Figure 5).45,46
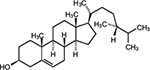  | Figure 5 Compound 2: campesterol. |
Compound 3
Spectral data
13C-NMR spectra of Compound 3 showed signals at 59.85 (C-1), 123.48 (C-2), 130.92 (C-3), 40.29 (C-4), 25.55 (C-5) 33.21 (C-6) 30.13 (C-7), 37.78 (C-8), 24.89 (C-9), 37.08 (C-10), 33.11 (C-11), 37.70 (C-12), 25.22 (C-13), 39.79 (C-14), 28.40 (C-15), 23.15 (C-16), 23.05 (C-17), 20.17 (C-18), 20.14 (C-19) and 16.86 (C-20).
Structure elucidation
Compound 3 (19 mg) was isolated as a yellowish liquid with Rf of 0.64 in hexane:chloroform 1:1 and 0.51 in 100% chloroform. It gave a purple color with vanillin/H2SO4 and a pink color with anisaldehyde spray reagent (Table 2).
The 13C-NMR spectrum revealed 5CH3, 10CH2, 4CH and 1C=C, indicating a C-20 compound. The 1H-NMR spectrum showed a signal at δ 5.4 (t), representing an olefinic proton, assigned to C-2. The signal at δ 4.1 (d) is an alcoholic proton assigned to the proton residing on C-1. A triplet at δ1.98 was assigned to the proton on C-4, while the multiplets at δ 1.44 and δ1.35 are the methine protons on C-7 and C-11. The other methine proton on C-15 had its signal at δ1.52. In addition, the multiplets at δ1.30–δ1.03 were assigned to the protons on C-6, C-8, C-9, C-10, C-12 and C-13, while the signal at δ1.65 (s) was assigned to the methyl proton on C-20. The OH group had a signal at δ1.66. Analysis of the spectra and comparison with literature values showed that Compound 3 is phytol (Figure 6).47
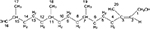  | Figure 6 Compound 3: 3,7,11,15-tetramethyl-2-hexadecen-1-ol. |
Discussion
Three compounds with cholinesterase inhibitory activity were successfully isolated from S. mombin and identified in this study. S. mombin exhibited potent cholinesterase inhibitory activity attributable to the presence of the various isolated compounds and of significance in the management of neurodegenerative disorders such as Alzheimer’s disease. This plant has been used for enhancement of memory in traditional medicine,9 and plants with such history have been previously reported by various researchers to possess cholinesterase inhibitors.48–50
On comparison with data in literature,51–53 Compound 1 was identified as betulin. Betulin has been previously reported in several plants for its different biological activities.51 However, it is being linked with the cholinesterase inhibitory activity for the first time, with an IC50 of 0.88 μg/mL against AChE and 4.67 μg/mL against BuChE.
Compound 3, which is phytol, has been previously reported by us for its cholinesterase inhibitory activity.54 Phytol is a diterpene alcohol, while betulin is a lupane-type triterpene. Several reports have implicated terpenoids as good cholinesterase inhibitors.55–57 In 2004, dihydrotanshinone, cryptotanshinone, tanshinone I and tanshinone IIA were identified as the first example of diterpenoids that inhibit AChE.58 Several others have since been reported. Triterpenes have also been documented as potent cholinesterase inhibitors.59–61 Some other oleanane triterpene saponin compounds have also been implicated in the treatment of dementia and mild cognitive impairment by previous researchers who are already seeking patency in the USA.62
Compound 2 identified as campesterol has been previously reported in several plant species, including rapeseed oil (Brassica napa),63 soybean oil (Glycine max)64 and wheat germ oil (Triticum spp.).65 Campesterol, though not new, is being reported for cholinesterase inhibitory activity for the first time, with an IC50 of 1.89 μg/mL (AChE) and 4.08 μg/mL (BuChE). It is a phytosterol, and several phytosterols have been reported to possess cholinesterase inhibitory activity to varying extents.66–68 However, others such as stigmasterol and β stigmasterol were reported in a molecular docking experiment to have weak bonding with AChE proteins when compared with Aricept®–AChE complex, even though they had comparable Glide score.69
Conclusion
Studies relating to the identification of naturally occurring secondary metabolites from medicinal plants have allowed the discovery of important drugs, including inhibitors of several enzymes such as AChE. This has been useful in the development of new drugs for clinical use. We isolated and identified three compounds from S. mombin with good cholinesterase inhibitory ability. These compounds could be candidates for further studies in the development of new drugs for the treatment of disorders such as AD.
Disclosure
The authors report no conflicts of interest in this work.
References
Murraya PA, Faraonia MB, Castroa MJ, Alzaa NP, Cavallaro V. Natural AChE inhibitors from plants and their contribution to Alzheimer’s disease therapy. Curr Neuropharmacol. 2013;11:388–413. | ||
Khaled NZR, Ana CCS, Paulo MPF, Chistiane MF. Phytoconstituents and evaluation of acetylcholinesterase inhibition by methanol extract of Liquidambar styraciflua (L.) aerial parts. J Appl Pharm. 2014;2(6):143–152. | ||
Čolović MB, Krstić DZ, Lazarević-Pašti TD, Bondžić AM, Vasić VM. Acetylcholinesterase inhibitors: pharmacology and toxicology. Curr Neuropharmacol. 2013;11(3):315. | ||
Chopra K, Misra S, Kuhad A. Current perspectives on pharmacotherapy of Alzheimer’s disease. Expert Opin Pharmacother. 2011;12(3):335–350. | ||
Sangnoi Y, Sakulkeo O, Yuenyongsawad S, et al. Acetylcholinesterase-inhibiting activity of pyrrole derivatives from a novel marine gliding bacterium, Rapidithrix thailandica. Mar Drugs. 2008;6(4):578–586. | ||
Mehta N, Adem A, Sabbagh M. New acetylcholinesterase inhibitors for Alzheimer’s disease. Int J Alzheimers Dis. 2012;2012:1–8. | ||
Houghton PJ, Howes MJ. Natural products and derivatives affecting neurotransmission relevant to Alzheimer’s and Parkinson’s disease. Neurosignals. 2005;14:2–22. | ||
Lawrence GHM. Taxonomy of Vascular Plants. Indianapolis, IN: Macmillan; 1971:545–574. | ||
Elufioye TO, Oladele AT, Cyril-Olutayo CM, Agbedahunsi JM, Adesanya SA. Ethnomedicinal study and screening of plants used for memory enhancement and antiaging in Sagamu, Nigeria. Eur J Med Plants. 2012;2(3):262–275. | ||
Ayensu ES. Mechanical Plants of West Africa. Algonac, MI: Reference Publications Inc.; 1978:282. | ||
Offiah VN, Anyanwu II. Abortifacient activity of an aqueous extract of Spondias mombin leaves. Journal of Ethnopharmacol. 1989;26:317–320. | ||
Kramer A, Mosquera E, Ruiz J, Rodrinquez E. Ethnobotany and biological activity of plants utilized during pregnancy and childbirth in the Peruvian Amazon. In: Herforth AW, Gorospe K, Kheel J, Fraissinet P, Rosane D, Rodriguez E, editors. Emanations from the Rainforests and the Carribean. Vol. 4. Ithaca, NY: Cornell University; 2002:10. | ||
Oliver-Bever B. Medicinal Plants in Nigeria. Ibadan: The Nigerian College of Arts, Science and Technology; 1960:760. Being a Course of Four Lectures Delivered in April 1959 in the Pharmacy Department. | ||
Kokwaro JO. Medicinal Plants of East Africa. Nairobi, Kanpala: East Africa Literature Bureau; 1976:384. | ||
Abo KA, Ogunleye VO, Ashidi JS. Antimicrobial potential of Spondias mombin, Croton zambesicus and Zygotritonia crocea. Phytother Res. 1999;13(6):494–497. | ||
Abad MJ, Bermejo A, Carretero E, Martinez-Acitores C, Noguera B, Villar A. Anti-inflammatory activity of some medicinal plant extracts from Venezuela. J Ethnopharmacol. 1996;55(1):63–68. | ||
Burkil HM. The Useful Plants of West Tropical Africa. Vol. 3. 2nd ed. Kew, London: Royal Botanic Gardens; 1995:857. | ||
Ayoka AO, Akomolafe RO, Iwalewa OE, Upkonmwan EO. Studies on the anxiolytic effect of Spondias mombin L. (Anacardiaceae) extracts. Afr J Tradit Complement Altern Med. 2005;2(2):153–165. | ||
Villegas LF, Fernandez ID, Maldonado H, et al. Evaluation of the wound-healing activity of selected traditional medicinal plants from Peru. J Ethmopharmacol. 1997;55(3):193–200. | ||
Ayoka AO, Akomolafe RO, Iwalewa OE, Akanmu AM, Upkonmwan EO. Sedative, anti-epileptic and anti-psychotic effects of Spondias mombin L (Anacardiaceae) in mice and rats. J Ethnopharmacol. 2005;103(2):166–175. | ||
Corthout J, Pieters LA, Claeys M, Vanden Berghe DA, Vlietinck JA. Antiviral ellagitanins from Spondias mombin. Phytochemistry. 1991;30(4):1129–1130. | ||
Corthout J, Pieters LA, Claeys M, Vanden Berghe DA, Vlietinck JA. Antiviral caffeoyl esters from Spondias mombin. Phytochemistry. 1992;31(6):1979–1981. | ||
Corthout J, Pieters LA, Claeys M, Geerts S, Vanden Berghe DA, Vlietinck JA. Antibacterial and molluscicidal phenolic acids from Spondias mombin. Planta Med. 1994;60(5):460–463. | ||
Coates NJ, Gilpin ML, Gwynn MN, et al. SB-202742, a novel b-lactamase inhibitor isolated from Spondias mombin. J Nat Prod. 1994;57(5):654–657. | ||
Nworu CS, Akah PA, Okoli CO, Okoye TC. Oxytocic activity of leaf extract of Spondias mombin. Pharm Biol. 2007;45(5):366–371. | ||
Amadi ES, Oyeka A, Onyeagba RA, Okoli I, Ugbogu OC. Studies on the antimicrobial effects of Spondias mombin and Baphia nittida on dental caries organism. Pak J Biol Sci. 2007;10(3):393–397. | ||
Uchendu CN, Isek T. Antifertility activity of aqueous ethanolic leaf extract of Spondias mombin (Anacardiaceae) in rats. Afr Health Sci. 2008;8(3):163–167. | ||
Asuquo OR, Ekanem TB, Udoh PB, Eluwa MA, Mesembe OE. Antigonadotrophic effect of Spondias mombin leaf extract in male Wistar rats. J Biol Agric Healthc. 2012;2(7):14–17. | ||
Asuquo RO, Ekanem BT, Udoh BP, Mesembe EO, Ebong EP. Haematinic potential of Spondias mombin leaf extract in Wistar rats. Adv Biores. 2013;4(2):53–56. | ||
Maduka HCC, Okpogba AN, Ugwu CE, et al. Phytochemical, antioxidant and microbial inhibitory effects of Spondias mombin leaf and stem bark extracts. J Pharm Biol Sci. 2014;9(2):14–17. | ||
Moke EG, Ilodigwe EE, Okonta JM, et al. Antidiabetic activity and toxicity evaluation of aqueous extracts of Spondias mombin and Costus afer on Wistar rats. Br J Pharm Res. 2015;6(5):333–342. | ||
Elufioye TO, Obuotor EM, Sennuga AT, Agbedahunsi JM, Adesanya SA. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of some selected Nigerian medicinal plants. Braz J Pharm. 2010;20(4):472–477. | ||
Ceva-Antunes PM, Bizzo HR, Alves SM, Antunes OA. Analysis of volatile compounds of tapereba (Spondias mombin L.) and caga (Spondias mombin L) by simultaneous distillation and extraction (SDE) and solid phase micro extraction (SPME). J Agric Food Chem. 2003;57(5):1387–1392. | ||
Adedeji J, Hartman G, Rosin RT, Chi Tang H. Free and glycosidically bound aroma compounds in hog plum (Spondias mombin L.). J Agric Food Chem. 1991;39(8):1494–1497. | ||
Olugbuyiro JAO, Moody JO, Hamann MT. Phytosterols from Spondias mombin Linn with antimycobacterial activities. Afr J Biomed Res. 2013;16(1):19–24. | ||
Olugbuyiro JAO, Moody JO. Anti-tubercular compounds from Spondias mombin. Int J Pure Appl Sci Technol. 2013;19(2):76–87. | ||
Echeme JO, Ahuchogu AA, Uchegbu RI. Isolation and characterization of estra-17-propoxy, 3/, 4/–dimethoxy – 3yl benzoate from the leaves of Spondias mombin Linn. J Nat Sci Res. 2014;4(19):172–177. | ||
Houghton PJ, Agbedahunsi JM, Adegbulugbe A. Choline esterase inhibitory properties of alkaloids from two Nigerian crinum species. Phytochemistry. 2004;65(21):2893–2896. | ||
Ellman GL, Courtney KD, Andres VJR, Feather-stone RM. A new and rapid colorimetic determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7:88–95. | ||
Rhee IK, van de Meent M, Ingkaninan K, Verpoorte R. Screening for acetylcholinesterase inhibitors from Amaryllidaceae using silica gel thin-layer chromatography in combination with bioactivity staining. J Chromatogr A. 2001;915:217–223. | ||
Harborne JB. Phytochemical Methods. A Guide to Modern Techniques of Plant Analysis. London: Chapman & Hall; 1973:11. | ||
Pothier J. Natural Products: Thin Layer (Planar) Chromatography. Academic Press, UK; 2000:3459–3475. | ||
Njoku PC, Akumefula MI. Phytochemical and nutrient evaluation of Spondias mombin leaves. Pak J Nutr. 2007;6(6):613–615. | ||
Osman SM, Khalek SMA, Koheil MA, El-Haddad AE, Wink M. A new steroidal compound (β-sitosterol-3-O-butyl) isolated from Caesalpinia gilliesii flowers. Int J Appl Res Nat Prod. 2015;8(2):14–19. | ||
Jaju SB, Indurwade NH, Sakarkar DM, Fuloria NK, Ali MD, Basu SP. Isolation of sitosterol diglucosyl caprate from Alpinia galanga. Pharmacognosy Res. 2010;2:264–266. | ||
Jain PS, Bari SB. Isolation of lupeol, stigmasterol and campesterol from petroleum ether extract of woody stem of Wrightia tinctoria. Asian J Plant Sci. 2010;9:163–167. | ||
Arigoni DW, Eisenreich C, Latzel S, et al. Dimethylallyl pyrophosphate is not the committed precursor of isopentenyl pyrophosphate during terpenoid biosynthesis from 1-deoxyxylulose in higher plants. Proc Natl Acad Sci U S A. 1999;96(4):1309–1314. | ||
Howes MJ, Perry NS, Houghton PJ. Plants with traditional uses and activities, relevant to the management of Alzheimer’s disease and other cognitive disorders. Phytother Res. 2003;17(1):1–8. | ||
Kennedy DO, Scholey AB. The psychopharmacology of European herbs with cognition-enhancing properties. Curr Pharm Des. 2006;12(35):4613–4623. | ||
Baradaran A, Rabiei Z, Rafieian M, Shirzad H. A review study on medicinal plants affecting amnesia through cholinergic system. J Herbmed Pharmacol. 2012;1(1):3–9. | ||
Tolstikov GA, Flekhter OB, Shultz EE, Baltina LA, Tolstikov AG. Betulin and its derivatives: chemistry and biological activity. Chem Sustain Dev. 2005;13:1–29. | ||
Sharma PP, Roy RK, Anurag B, Gupta D. Pentacyclic triterpinoids from Betula utilis and Hyptis suaveolens. Int J PharmTech Res. 2010;2(2):1532–1558. | ||
Uddin G, Waliullah BS, Siddiqui M, Alam A, Sadat A, Uddin AA. Chemical constituents and phytotoxicity of solvent extracted fractions of stem bark of Grewia optiva Drummond ex Burret. Middle East J Sci Res. 2011;18(1):85–91. | ||
Elufioye TO, Obuotor EM, Agbedahunsi JM, Adesanya SA. Acetyl and butyrylcholinesterase inhibiting constituent from Morinda lucida Benth (Rubiaceae). Br J Pharm Res. 2015;6(5):358–365. | ||
Perry NS, Houghton PJ, Theobald A, Jenner P, Perry EK. In-vitro inhibition of human erythrocyte acetylcholinesterase by Salvia lavandulaefolia essential oil and constituent terpenes. J Pharm Pharmacol. 2000;52(7):895–902. | ||
Miyazawa M, Yamafuji C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J Agric Food Chem. 2005;53(5):1765–1768. | ||
Öztürk M. Anticholinesterase and antioxidant activities of Savoury (Satureja thymbra L.) with identified major terpenes of the essential oil. Food Chem. 2012;134(1):48–54. | ||
Ren Y, Houghton PJ, Hider RC, Howes MJ. Novel diterpenoid acetylcholinesterase inhibitors from Salvia miltiorhiza. Planta Med. 2004;70(3):201–204. | ||
Barbosa Filho JM, Medeiros KC, Diniz MD, et al. Natural products inhibitors of the enzyme acetylcholinesterase. Revista Brasileira de Farmacognosia. 2006;16(2):258–285. | ||
Mukherjee PK, Kumar V, Houghton PJ. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytother Res. 2007;21(12):1142–1145. | ||
Ercetin T, Senol FS, Orhan IE, Toker G. Comparative assessment of antioxidant and cholinesterase inhibitory properties of the marigold extracts from Calendula arvensis L. and Calendula officinalis L. Ind Crops Prod. 2012;36(1):203–208. | ||
Kim B, Choi W, Lee S, et al. United States Patient Application Number 20100190968. 2006. | ||
Amar S, Ecke W, Becker HC, Möllers C. QTL for phytosterol and sinapate ester content in Brassica napus L. collocate with the two erucic acid genes. Theor Appl Genet. 2008;116(8):1051–1061. | ||
Shi H, Nam PK, Ma Y. Comprehensive profiling of isoflavones, phytosterols, tocopherols, minerals, crude protein, lipid, and sugar during soybean (Glycine max) germination. J Agric Food Chem. 2010;58(8):4970–4976. | ||
Ruibal-Mendieta NL, Rozenberg R, Delacroix DL, et al. Spelt (Triticum spelta L.) and winter wheat (Triticum aestivum L.) whole meals have similar sterol profiles, as determined by quantitative liquid chromatography and mass spectrometry analysis. J Agric Food Chem. 2004;52(15):4802–4807. | ||
Oliveira AP, Valentão P, Pereira JA, Silva BM, Tavares F, Andrade PB. Ficus carica L.: metabolic and biological screening. Food Chem Toxicol. 2009;47(11):2841–2846. | ||
Park SJ, Kim DH, Jung JM, et al. The ameliorating effects of stigmasterol on scopolamine-induced memory impairments in mice. Eur J Pharmacol. 2012;676(1):64–70. | ||
Pereira VV, Silva RR, Duarte LP, Takahashi JA. Chemical constituents of Jacaranda oxyphylla and their acetylcholinesterase inhibitory and antimicrobial activities. Rec Nat Prod. 2016;10(3):392. | ||
Rohit M, Ashok T, Vijaykumar R, Kashniyal K. Molecular docking study of Cassia tora, Brassica campestris and Calotropis procera as acetylcholinesterase inhibitor. Indian J Pharm Educ Res. 2016;50(1):116–122. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.