Back to Journals » Journal of Inflammation Research » Volume 13
Antibiotics, Inflammation, and Preterm Labor: A Missed Conclusion
Authors Hantoushzadeh S, Anvari Aliabad R, Norooznezhad AH
Received 4 February 2020
Accepted for publication 14 April 2020
Published 25 May 2020 Volume 2020:13 Pages 245—254
DOI https://doi.org/10.2147/JIR.S248382
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ning Quan
Sedigheh Hantoushzadeh,1 Roghayeh Anvari Aliabad,2 Amir Hossein Norooznezhad3
1Maternal, Fetal and Neonatal Research Center, Vali-Asr Hospital, Imam Khomeini Hospital Complexes, Tehran University of Medical Sciences, Tehran, Iran; 2Department of Gynecology, School of Medicine, Hamadan University of Medical Sciences, Hamadan, Iran; 3Medical Biology Research Center, Health Technology Institute, Kermanshah University of Medical Sciences, Kermanshah, Iran
Correspondence: Amir Hossein Norooznezhad Email [email protected]
Abstract: Regarding the risk of antibiotic therapy during pregnancy, any medication given to the mother should be according to the indications due to the risk of possible side effects. Antibiotics are one of the most important groups of these medications to be considered. Along with direct antibiotic-induced side effects, indirect pathways also affect the fetus through the maternal changes. According to the data, different cytokines including interleukin-1β (IL-1β), IL-6, and tumor necrosis factor-α (TNF-α) are involved in both term and preterm parturition. These cytokines could trigger expression of different substances such as prostaglandins (PGs), their receptors, and PGs synthetizing molecules with already proven roles in parturition. Moreover, IL-1, IL-6, and TNF-α knocked-out mice have delayed parturition and lower levels of PGs compared to the wild types. The earlier-mentioned cytokines are able to induce matrix metalloproteinases and are also involved in parturition. Certain antibiotics have been shown capable of inducing inflammation cascade directly. Both in-vivo and in-vitro studies in human have also demonstrated this inflammation as elevated levels of inflammatory cytokines especially IL-1, IL-6, and TNF-α. This increase has been observed both in the presence and the absence of lipopolysaccharide (LPS). Moreover, antibiotics can induce endotoxemia in healthy cases which finally leads to the pro-inflammatory cytokine release. Regarding the role of mentioned pro-inflammatory cytokines in both term and preterm parturition, it seems that non-indicated use of antibiotics during pregnancy may increase the risk of preterm labor.
Keywords: preterm labor, antibiotic, inflammation, interleukin-1, interleukin-6, tumor necrosis factor-α
Introduction
Preterm labor, which has been defined as birth in <37 weeks of gestation, categorizes into four major types: late preterm (34 weeks≤birth<37 weeks of gestation), moderate preterm (32 weeks≤birth<34 weeks of gestation), very preterm (28 weeks ≤birth<32 weeks of gestation), and extremely preterm (birth<28 weeks of gestation).1 This phenomenon has been known as the most prevalent cause of prenatal morbidity and mortality in developed countries. However, not only developed countries but also the world is struggling with this issue, with 15 million preterm births in which, unfortunately, 1 million of them lead to death.2–4 With the prevalence of 5–18% in pregnancies3 (as well as morbidities and mortalities), preterm labor forces a lot of expense on the healthcare system beginning from neonatal intensive care unit (NICU) bed requirements.1,5,6 Data have shown that preterm labor induces costs equal to $26.2 billion/year in the United States that increases each year.5 So far, different etiologies such as diabetes, low weight, obesity, increased blood pressure, tobacco, and intrauterine infection have been reported to cause preterm labor. Intrauterine infection has been proven to be related to the preterm rupture of the membranes and therefore preterm labor. According to the data, infection-induced inflammation which could cause chorioamnionitis is one the most important leading causes of preterm labor.2,7 However, not only microbial-induced inflammation but also non-infectious inflammation (also known as sterile inflammation) could cause preterm labor. In a multicenter study it was shown that in patients with preterm labor (with intra-amniotic inflammation and intact membrane) sterile inflammation was significantly higher than microbial-associated inflammation (26% vs 11% with P-value=0.005) while other possible risk factors such as maternal age, body mass index (BMI), race, and cervical dilation at admission were not different between both groups.8
Treating infectious diseases such as pyelonephritis, sexually transmitted diseases, and upper respiratory tract infections during pregnancy has always been given extra attention in the clinic. Despite some possible side effects, the most effective treatment for these diseases is still antibiotic therapy. Unfortunately, due to ethical considerations, the side effects of antibiotics on pregnant women have not been thoroughly evaluated in clinical trials and no exact data are available on this subject. Considering that antibiotics are noticeably, and sometimes blindly, prescribed during pregnancy, it seems very crucial to avoid any non-indicated treatment with antibiotics.9,10 There are reports indicating that consuming antibiotics during pregnancy has adverse effects on the fetus through direct and indirect pathways. In the indirect pathway, the mother is affected by antibiotics which consequently affects the fetus. For example, an antibiotic-induced anaphylactic reaction in the mother11,12 reduces blood flow to the fetus which may lead to hypoxia and even cause fetal mortality.13,14 Through the direct path, on the other hand, the immune system of the fetus is targeted15 where the antibiotics may lead to the development of allergic conditions,16,17 higher risk of necrotizing enterocolitis,18,19 and cerebral palsy.20 Also, it has been shown that non-indicated intrapartum antibiotic administration seems to increase the risk of antibiotic-resistant neonatal sepsis if an infection occurs.21 Also, as we have stated before, consuming antibiotics in pregnant patients without bacterial infection may cause different mental diseases in their offspring.22
As has been reported once, pregnant women receiving empirically antibiotic treatment without any evidence of bacterial infection affirmed by high-sensitive polymerase chain reaction (PCR) had a significantly higher preterm labor risk in comparison to patients with positive infection.23 Thus, to clear up this crucial issue, this review aimed to investigate any possible relation between antibiotic consumption and preterm labor as well as possible pathways and pathogeneses, especially in pregnant women who do not meet the indications for antibiotic treatment.
General Concept
It has been shown that different pro-inflammatory cytokines such as interleukin-1β (IL-1β), IL-6, and tumor necrosis factor-α (TNF-α) are involved in both term and preterm labor. The mentioned cytokines trigger the production of uterine activation proteins and chemokines, and therefore cause preterm parturition.24,25 As would be discussed, using some antibiotics in the absence of bacterial infection increases release of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α, all being capable of inducing preterm parturition through different pathways. Moreover, the cytokine release could be amplified by antibiotic-induced endotoxemia through activation of Toll-like receptors (TLRs). Thus, consumption of non-indicated antibiotics for non-infected pregnant women, especially those more prone to preterm parturition, may increase the risk of preterm parturition.
Pro-Inflammatory Cytokines and Preterm Labor
Interleukin-1
IL-1β is one of the most important members of the IL-1 family26 with affinity to its receptors; IL-1R1 and IL-1R2.26,27 As a major mediator of inflammation, IL-1β is involved in different disorders such as infections, autoimmune, and degenerative diseases which affect a wide variety of organs and cells. IL-1β expresses from hematopoietic cells such as macrophages, monocytes, microglia, and dendritic cells. This pro-inflammatory cytokine produces from the mentioned resources through different pathways such as TLR activation and pro-inflammatory cytokine activities (such as TNF-α and even IL-1β itself).27 IL-1β is the first known pro-inflammatory cytokine involved in preterm parturition produced following the release of different other cytokines including TNF-α, IL-1α, and IL-1β itself.25 The role of IL-1β during parturition has been proved by being detected in the myometrium, cervix, and fetal membranes,28 especially in non-infective preterm parturition.29 In an invivo study in mice, it was shown that IL-1β administration induces premature contractility of the myometrium.30 Moreover, in-vivo studies have determined that the administration of IL-1β could induce preterm labor which, by the way, could be prevented using the antagonist of IL-1 receptor.31,32 Beside term parturition, studies have evaluated the role of IL-1β in preterm labor. According to the data, the IL-1β concentration was increased in amniotic fluid (AF)33,34 and plasma35 of women with preterm labor. Also, systemic administration of IL-1β in pregnant rhesus monkeys was also proven to stimulate the uterine contractility as well as significantly increase the production of other pro-inflammatory cytokines such as IL-6 and TNF-α along with different prostaglandins (PGs) including PGF2α and PGE2.36 PGF2α, on the other hand, has been introduced as the most potent contractile prostanoid in parturition which acts through its receptor, PGF receptor (PTGFR).37 Furthermore, IL-1β is able to activate prostaglandin H synthase 2 (PGHS-2; also known as cyclooxygenase-2 or COX-2) which is responsible for the synthesis of prostaglandins.38–40 As it has been shown, IL-1β (targeting amnion epithelial cells) is able to increase matrix metalloproteinase-9 (MMP-9) expression which leads to degradation of the amniochorionic membrane as well as cervical ripening.41
Tumor Necrosis Factor-α
TNF-α is a pro-inflammatory cytokine belonging to the TNF superfamily which couples to its specific receptors: TNF receptor-1 (TNFR1 or p55) and TNFR2 (p75). This cytokine has a wide range of activity in different inflammatory and non-inflammatory situations such as cachexia, septic shock, inflammatory disorders, and autoimmune diseases. The same as IL-1β, TNF-α is expressed by different cells such as macrophages, monocytes, dendritic cells, B and T cells, and activated natural killer cells.42,43 Similar to IL-1β, TNF-α is another pro-inflammatory cytokine elevated in AF of women with preterm parturition.33 This cytokine is also involved in the regulation and stimulation of PG synthesis during preterm parturition, such as COX-2. Also, TNF-α and IL-1β could decrease levels of chorionic prostaglandin 15-hydroxy dehydrogenase (PGDH) in cultured chorion and placental trophoblast cells. PGDH could inactivate the PGs through catalyzing them to natural substances.24 Moreover, it has been shown that topical administration of TNF-α could stimulate cervical ripening.25 According to the data, knockout mice lacking receptors for both IL-1β and TNF-α had a significantly lower rate of preterm labor following intrauterine inoculation of killed Escherichia coli (E.coli) in comparison to the wild types. This result was accompanied by lower levels of COX-2 in the myometrial tissue in the knockout group in comparison to the controls.44 As it has been studied, inhibiting COX-2 in mice is associated with the prevention of inflammation-induced preterm parturition.45 Furthermore, in a study on rhesus monkeys, the release of bacterial-induced TNF-α and IL-1β led to more uterine contractility.46 Like IL-1β, TNF-α could increase MMP-9 activity, expression, and activity and secretion in trophoblast, chorion cells, and amnion cells, respectively.41 Moreover, it has been shown that TNF-α could induce mRNA expression of MMP-1, MMP-3, and MMP-9 in cervical smooth muscle cells after 24–48 h of administration. This pattern is also seen in treatment with IL-1β.47
Interleukin-6
The IL-6 cytokine family consists of different cytokines such as IL-6, IL-11, IL-27, and leukemia inhibitory factor.48 IL-6, which is the most important pro-inflammatory cytokine in this family, plays a critical role in the inflammation cascade. This pro-inflammatory cytokine is involved in the pathogenesis of different autoimmune and inflammation-dependent diseases and mostly acts through binding to its receptor, IL-6R.49 This pro-inflammatory cytokine has shown to be increased in preterm parturition even in asymptomatic women.50,51 Pregnant IL-6 knocked-out mice have been demonstrated to have a delay in delivery compared to the wild types. This delay is proved to be accelerated through the administration of exogenous IL-6. Moreover, after lipopolysaccharide (LPS) treatment, the IL-6 knocked-out mice group showed lower prevalence of preterm parturition in comparison to the wild types. In the IL-6 knocked-out mice, oxytocin receptor(Oxtr) mRNA expression was 73% lower than the wild type on day 19.5 post coitus, which reached acomparable level with wild type with a 1-day delay. Also, in the wild type group during day 19.5 post coitum to the labor time, PTGFR and prostaglandin-endoperoxide synthase 2(PTGS2) showed a 3.9 and 2.6 (respectively) fold increase. On the other hand, the knocked-out mice showed a similar increase in the expression of PTGFR and PTGS2 from day 20.5 post coitum.52 More interestingly, the amount of PTGFR, as the receptor of most important uterine contractile prostanoid (PGF-2α), was shown to increase in both term and preterm parturition.24 Also, it has been demonstrated that IL-6 is able to induce PGE2 production in amnion and decidual cells.53 According to the data, IL-6 is also responsible for an increase in MMP-9 and MMP-2 activity through targeting trophoblasts.41 Furthermore, data have cleared that IL-1β and IL-6 are able to induce oxytocin secretion in human uterine smooth muscle cells.54 Considering the affirmed role of oxytocin and PGs in term and preterm parturition, it seems that they could be involved in the possible hypothesized molecular pathways caused by IL-6 and IL-1β in preterm parturition.55 Furthermore, IL-1β seems to be capable of increasing PGE2 production from human amnion cells.56 Some of the most important roles of pro-inflammatory cytokines in term/preterm labor have been provided in Table 1.
 |
Table 1 Expression Status and Role of Pro-Inflammatory Cytokines in Preterm Labor |
Toll-Like Receptors, Inflammation, and Preterm Labor
TLRs are a major group of pattern recognition receptors (PRRs). These transmembrane receptors are responsible for recognition of pathogen-associated molecular patterns (PAMPs).57 The involved cells in the innate immune system such as neutrophils, macrophages, monocytes, dendritic cells, and natural killer cells use TLRs for PAMP recognition. These type I transmembrane glycoproteins consist of three major parts: (1) leucine-rich repeats as the ectodomain; (2) the transmembrane domain; and (3) the intracellular Toll-interleukin domain which is responsible for signal transduction.57 For activation of TLRs, two different types of molecules are involved by their origin: endogenous and exogenous. Most endogenous agents such as heat-shock protein 60 (Hsp60), Hsp70, Hsp90, high-mobility group box protein 1 (HMGB1), fibrinogen, and fibronectin could activate the TLR4, while the rest of them which are not notable in count bind to the other TLRs. On the other hand, the exogenous molecules by type have their specific TLR(s). TLR1 (activation by triacetylated lipoproteins), TLR2 (activation by peptidoglycan, lipopeptides, lipoteichoic acids, and zymosan), TLR3 (activation by double-stranded RNA), TLR4 (activation by LPS and paclitaxel), TLR5 (activation by flagellin), and TLR6 (activation by diacylated lipoprotein (with TLR2)) are among the TLRs activated by exogenous factors.58 Activation of TLRs causes a downstream pathway led by nuclear factor-κB (NF-κB) which finally causes pro-inflammatory cytokines, chemokines, proteases, and prostaglandins, which are all role players in preterm labor.59–63 Also, it has been mentioned that HMGB1 seems to be one of the most important factors playing a role in the sterile inflammation during preterm labor. Data have shown that pregnant women with higher amniotic fluid levels of HMGB1 (>8.5 ng/mL) had a higher risk of preterm labor than those with <8.5 ng/mL.8 Other than cells involved in the innate immune response, it has been shown that all TLRs are expressed in the human placenta as well as some co-receptors and accessory proteins such as CD14. Also, in a time-dependent manner, the presence of different TLRs has been shown in the decidua and amnion. As it has been shown, human decidual cells (third trimester) expressed TLR1–6. Moreover, the presence of TLR4 has been approved in the apical side of human amnion64 as well as mesenchymal and epithelial cells of the uterus and cervix.65,66
It has been shown that pregnant mice which received uterus injection of heat-killed E. coli experienced significantly higher risk of preterm labor than the similar TLR4-knockout ones.67,68 In a similar study on the TLR4-knockout mice, it was shown that pro-inflammatory cytokines including IL-1β, IL-6, and TNF-α were decreased as well as CC chemokine ligand 2 (CCL2) which is a chemokine involved in parturition. Also, as it is expected, the knockout group had a longer period of gestation in comparison to the wild types.69 Moreover, in an in-vivo study, (+)-naloxone was used as a TLR4 antagonist for prevention of preterm labor. In the mentioned study, treatment with (+)-naloxone significantly decreased LPS-induced preterm labor in the animals. Also, after intrauterine E. coli instillation, the (+)-naloxone-treated group showed a significant decrease in pro-inflammatory cytokines including IL-1β, IL-6, and TNF-α in comparison to their matched controls.70 In a cross-sectional study it was shown that pregnant women with preterm labor had increased levels of TLR4 expression in the neutrophils obtained from peripheral blood in comparison to the control group.71
Antibiotics in Non-Infectious Situations and Inflammation Cascade
All of the mentioned data were in favor of supporting a proven role of inflammation in preterm labor. In this section, the role of antibiotics in induction of inflammatory cascade in non-infectious situations will be reviewed. In an in vitro study on monocytes and lymphocytes, it was revealed that these cells could be triggered by antibiotics in order to release different cytokines. Beta-lactams (mostly cephalosporins) were also proven to increase the secretion of IL-4 and INF-γ from lymphocytes as well as IL-1, IL-6, and TNF-α in monocytes. Moreover, in monocytes, clindamycin increased the secretion of TNF-α and IL-6 accompanied by an increase in IL-1, IL-6, and TNF-α by teicoplanin.72
As it has been studied in mice, oral neomycin sulfate and streptomycin could significantly increase gut endotoxin content lasting 3 days after antibiotic withdrawal.73 Moreover, another study evaluated endotoxemia after oral antibiotic therapy for gut decontamination in E. coli- challenged rats. As it was shown, all of the groups receiving antibiotics (including tobramycin plus polymyxin and ciprofloxacin) experienced endotoxemia which shows a translocation of endotoxins from the gut to blood circulation after antibiotic therapy in comparison to the control group.74 Similarly, it has been revealed that antibiotic therapy is able to induce bacterial translocation into the mesenteric lymph nodes in mice.75 The antibiotic-induced endotoxemia is quite challenging since endotoxemia can trigger the release of pro-inflammatory cytokines. In an ex-vivo study on incubated rat blood with E. coli, different antibiotics including imipenem, meropenem, and ceftazidime were studied for their effect on cytokine levels in comparison to the control (pyrogen-free saline 0.9%). It was shown that ceftazidime and meropenem induced a higher amount of endotoxin release than the control group. Also, plasma levels of IL-6 and TNF-α in antibiotic-treated groups were higher than the controls. Moreover, a correlation was found between endotoxin levels and both TNF-α and IL-6 levels.76 Furthermore, in a human study on healthy volunteers, the effect of endotoxin on the release of IL-6 and TNF-α was studied. The intravenous injection of 2 ng/kg of endotoxin caused increased serum levels of IL-6 and TNF-α.77
Another study on monocytes indicated that antibiotics are able to increase the expression of IL-1, IL-6, TLR1, TLR2, TLR4, and TLR6 with and without the presence of LPS. In detail, it was shown that the representative of the beta-lactam group (piperacillin) co-stimulated with LPS increased expression of IL-1β, IL-6, TLR1, TLR2, TLR4, and TLR6. Also, piperacillin alone was able to increase TLR1, TLR2, TLR4, and TLR6 in the monocytes. Doxycycline, the representative of the tetracycline group, increased IL-1, IL-6, TLR2, TLR4, and TLR6 co-cultured with LPS in human monocytes. Moreover, in the non-LPS-stimulated monocytes treated with doxycycline, increased expression of IL-1, TLR1, TLR2, TLR4, and TLR6 was observed. Human monocytes treated with the representative member of the macrolide family, erythromycin, showed an increase in expression of IL-6, TLR1, TLR2, TLR4, and TLR6 with and of IL-1, TLR1, TLR2, TLR4, and TLR6 without LPS co-stimulation. Furthermore, the effect of the mentioned antibiotics was assessed on purified human peripheral blood mononuclear cells (PBMCs) as well. It was revealed that gentamycin, doxycycline, and moxifloxacin were able to increase IL-1 expression in PBMCs in the absence of LPS.78 The importance of TLR overexpression by antibiotics is the linking of two mentioned pathways: (1) overexpressed TLRs (especially TLR4); and (2) endotoxemia and LPS release in the non-infectious situations. It could be concluded that both of these outcomes after antibiotic consumption are synchronized with each other which leads to the cytokine overexpression by LPS release as well as TLR overexpression. In a study on pregnant mice it was shown that LPS during the induced endotoxemia could activate TLR4 and therefore increase the expression of pro-inflammatory cytokines including IL-β1 and TNF-α.79 Moreover, in a study on mice, it was determined that after LPS injection leading to induced preterm labor, cytokine expression was increased in different sites. It was also established that serum levels of IL-6 and TNF-α were increased after LPS injection. This increase was accompanied by upregulating IL-1β, IL-6, IL-10, and TNF-α in the uterus and IL-1β and IL-6 in the placenta. As well as the mentioned cytokines, TLR2 and TLR4 were overexpressed in uterus tissue.80 Also, it has been shown that antibiotics are able to induce cytokine production in the absence of sepsis or LPS-mediated pathways in vivo. Healthy rats which received 30 mg/kg (single intravenous dose) ceftazidime had a significant increase in IL-6 and TNF-α levels compared with controls.81 Figure 1 shows the direct and indirect effects of antibiotics in the production of pro-inflammatory cytokines.
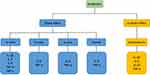 |
Figure 1 Current available data regarding the effects of some antibiotics on cytokine production. Abbreviations: IL, interleukin; TNF-α, tumor necrosis factor-α; INF-γ, interferon-γ. |
Interestingly, as mentioned before, the output of the current hypothesis has been shown in a 104-case study. The mentioned study was performed on pregnant women receiving empirically antibiotic treatment. In patients without any evidence of bacterial infection (affirmed by high-sensitive polymerase chain reaction (PCR)), the preterm labor risk was significantly higher than patients with positive infection.23 Figure 2 shows possible pathways associated with pro-inflammatory cytokine release after antibiotic consumption in the non-infectious situation.
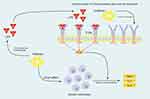 |
Figure 2 Possible pathways involved in sterile inflammation caused by some antibiotics in the non-infectious situation.Notes: Modified from Archives of Medical Research. Hantoushzadeh S, Norooznezhad AH. Inappropriate Antibiotic Consumption as a Possible Cause of Inflammatory Storm and Septic Shock in Patients Diagnosed with Coronavirus Disease 2019 (COVID-19). Epub 2020 Apr 4. Copyright 2020, with permission from Elsevier.86Abbreviations: IL, interleukin; TNF-α, tumor necrosis factor-α; TLR, Toll-like receptor; LPS, lipopolysaccharide; NF-κB, nuclear factor-κB. |
Role of Antibiotics in the Management of Preterm Labor
The main purpose of this study was to review the potential of antibiotics in induction of preterm labor via an inflammatory storm in a non-bacterial infection setting. As has been stated by The American College of Obstetricians and Gynecologists, intrauterine infections are important causes of preterm labor. However, they have relied on a systematic review and meta-analysis which found no statistically significant difference between antibiotic-treated pregnant women and the same placebo-treated group regarding duration prevention of preterm delivery and increasing gestation duration (with intact membrane). Although, they stated that this suggestion is not applied to group B streptococci carriers and pregnant women with preterm premature rupture of membranes (PPROM).82
As has been stated in a guideline by the World Health Organization (WHO), during preterm labor, antibiotic administration (as a routine action) is not recommended when the amniotic membrane is intact and no sign of infection has been found clinically. On the other hand, antibiotic administration is recommended in those pregnant women with PPROM (quality of evidence: strong recommendation).83
According to the National Institute for Health and Care Excellence (UK) guideline (last update: August 2019), when PPROM has not been confirmed, clinicians should avoid administration of prophylactic antibiotics. On the other hand, pregnant women with PPROM should receive antibiotic treatment.84
A recent systematic review has evaluated 16 guidelines related to preterm labor without any language restriction. According to their findings, most of the guidelines are against prophylactic antibiotic administration for a pregnant woman without PPROM.85
Conclusion
Antibiotic therapy is a sensitive area in pregnant women with infection. It has been proven that treatment of these patients with proper antibiotic(s) increases the gestational period. However, it seems that in patients with no bacterial infection, antibiotic treatment may cause preterm parturition through direct (direct cytokine release) and indirect (induced endotoxemia) inflammatory reactions. The authors suggest complementary studies on this issue in order to prevent preterm parturition, one of the most important issues in gynecology and neonatology. Due to the difference between antibiotics, it is necessary to evaluate a single one of them for better insight regarding the already mentioned side effect.
Acknowledgment
The authors are grateful for the kind help of Mrs. Fatemeh Norooznezhad, Tehran University of Medical Sciences.
Disclosure
The authors report no funding and no conflicts of interest for this work.
References
1. Zainal H, Dahlui M, Soelar SA, Su TT, Isangula KG. Cost of preterm birth during initial hospitalization: a care provider’s perspective. PLoS One. 2019;14(6):e0211997. doi:10.1371/journal.pone.0211997
2. Gilman-Sachs A, Dambaeva S, Garcia MDS, Hussein Y, Kwak-Kim J, Beaman K. Inflammation induced preterm labor and birth. J Reprod Immunol. 2018;129:53–58. doi:10.1016/j.jri.2018.06.029
3. Romero R, Dey SK, Fisher SJ. Preterm labor: one syndrome, many causes. Science. 2014;345(6198):760–765. doi:10.1126/science.1251816
4. Cappelletti M, Della Bella S, Ferrazzi E, Mavilio D, Divanovic S. Inflammation and preterm birth. J Leukoc Biol. 2016;99(1):67–78. doi:10.1189/jlb.3MR0615-272RR
5. Frey HA, Klebanoff MA. The epidemiology, etiology, and costs of preterm birth. Semin Fetal Neonatal Med. 2016;21(2):68–73. doi:10.1016/j.siny.2015.12.011
6. Jacob J, Lehne M, Mischker A, Klinger N, Zickermann C, Walker J. Cost effects of preterm birth: a comparison of health care costs associated with early preterm, late preterm, and full-term birth in the first 3 years after birth. Eur J Health Econ. 2017;18(8):1041–1046. doi:10.1007/s10198-016-0850-x
7. Jiang M, Mishu MM, Lu D, Yin X. A case control study of risk factors and neonatal outcomes of preterm birth. Taiwan J Obstet Gynecol. 2018;57(6):814–818. doi:10.1016/j.tjog.2018.10.008
8. Romero R, Miranda J, Chaiworapongsa T, et al. Prevalence and clinical significance of sterile intra‐amniotic inflammation in patients with preterm labor and intact membranes. Am J Reprod Immunol. 2014;72(5):458–474. doi:10.1111/aji.12296
9. Bookstaver PB, Bland CM, Griffin B, Stover KR, Eiland LS, McLaughlin M. A review of antibiotic use in pregnancy. Pharmacotherapy. 2015;35(11):1052–1062. doi:10.1002/phar.1649
10. Norwitz ER, Greenberg JA. Antibiotics in pregnancy: are they safe? Rev Obstet Gynecol. 2009;2(3):135–136.
11. Borahay MA, Harirah HM, Olson G, Kilic GS, Karipcin S, Hankins GD. Disseminated intravascular coagulation, hemoperitoneum, and reversible ischemic neurological deficit complicating anaphylaxis to prophylactic antibiotics during cesarean delivery: a case report and review of literature. AJP Rep. 2011;1(1):15–20. doi:10.1055/s-0030-1271219
12. Sengupta A, Kohli JK. Antibiotic prophylaxis in cesarean section causing anaphylaxis and intrauterine fetal death. J Obstet Gynaecol Res. 2008;34(2):252–254. doi:10.1111/j.1447-0756.2008.00775.x
13. Berenguer A, Couto A, Brites V, Fernandes R, Berenguer A. Case report: anaphylaxis in pregnancy: a rare cause of neonatal mortality. BMJ Case Rep. 2013;2013(jan11 1):bcr2012007055. doi:10.1136/bcr-2012-007055
14. Simons FER, Schatz M. Anaphylaxis during pregnancy. J Allergy Clin Immunol. 2012;130(3):597–606. doi:10.1016/j.jaci.2012.06.035
15. Bizzarro MJ, Dembry L-M, Baltimore RS, Gallagher PG. Changing patterns in neonatal escherichia coli sepsis and ampicillin resistance in the era of intrapartum antibiotic prophylaxis. Pediatrics. 2008;121(4):689–696. doi:10.1542/peds.2007-2171
16. Jakobsson HE, Jernberg C, Andersson AF, Sjölund-Karlsson M, Jansson JK, Engstrand L. Short-term antibiotic treatment has differing long-term impacts on the human throat and gut microbiome. PLoS One. 2010;5(3):e9836. doi:10.1371/journal.pone.0009836
17. Bedford Russell A, Murch SH. Could peripartum antibiotics have delayed health consequences for the infant? BJOG. 2006;113(7):758–765. doi:10.1111/j.1471-0528.2006.00952.x
18. Kenyon S, Taylor D, Tarnow-Mordi W. Broad-spectrum antibiotics for preterm, prelabour rupture of fetal membranes: the ORACLE I randomised trial. Lancet. 2001;357(9261):979–988. doi:10.1016/S0140-6736(00)04233-1
19. Kenyon S, Boulvain M, Neilson JP. Antibiotics for preterm rupture of membranes. Cochrane Database Syst Rev. 2003;2:CD001058.
20. Flenady V, Hawley G, Stock OM, Kenyon S, Badawi N. Prophylactic antibiotics for inhibiting preterm labour with intact membrane. Cochrane Database Syst Rev. 2013;(12):CD000246.
21. Stetzer BP, Mercer BM. Antibiotics and preterm labor. Clin Obstet Gynecol. 2000;43(4):809–817. doi:10.1097/00003081-200012000-00011
22. Hantoushzadeh S, Anvari Aliabad R, Norooznezhad AH. Antibiotics, pregnancy, and fetal mental illnesses: where is the link? Am J Obstet Gynecol. 2020. doi:10.1016/j.ajog.2020.01.050
23. Yoneda S, Shiozaki A, Yoneda N, et al. Antibiotic therapy increases the risk of preterm birth in preterm labor without intra‐amniotic microbes, but may prolong the gestation period in preterm labor with microbes, evaluated by rapid and high‐sensitive PCR system. Am J Reprod Immunol. 2016;75(4):440–450. doi:10.1111/aji.12484
24. Christiaens I, Zaragoza DB, Guilbert L, Robertson SA, Mitchell BF, Olson DM. Inflammatory processes in preterm and term parturition. J Reprod Immunol. 2008;79(1):50–57. doi:10.1016/j.jri.2008.04.002
25. Romero R, Espinoza J, Kusanovic JP, et al. The preterm parturition syndrome. BJOG. 2006;113(S3):17–42.
26. Dinarello CA. Overview of the interleukin-1 family of ligands and receptors. Semin Immun. 2013;25(6):389–393. doi:10.1016/j.smim.2013.10.001
27. Garlanda C, Dinarello C, Mantovani A. The interleukin-1 family: back to the future. Immunity. 2013;39(6):1003–1018. doi:10.1016/j.immuni.2013.11.010
28. Osman I, Young A, Ledingham MA, et al. Leukocyte density and pro‐inflammatory cytokine expression in human fetal membranes, decidua, cervix and myometrium before and during labour at term. Mol Hum Reprod. 2003;9(1):41–45. doi:10.1093/molehr/gag001
29. Elliott C, Loudon J, Brown N, Slater D, Bennett P, Sullivan M. IL‐1β and IL‐8 in human fetal membranes: changes with gestational age, labor, and culture conditions. Am J Reprod Immunol. 2001;46(4):260–267. doi:10.1034/j.1600-0897.2001.d01-11.x
30. Nadeau-Vallée M, Quiniou C, Palacios J, et al. Novel noncompetitive IL-1 receptor-biased ligand prevents infection-and inflammation-induced preterm birth. J Immun. 2015;195(7):3402–3415. doi:10.4049/jimmunol.1500758
31. Romero R, Tartakovsky B. The natural interleukin-1 receptor antagonist prevents interleukin-l-induced preterm delivery in mice. Am J Obstet Gynecol. 1992;167(4):1041–1045. doi:10.1016/S0002-9378(12)80035-4
32. Romero R, Mazor M, Tartakovsky B. Systemic administration of interleukin-1 induces preterm parturition in mice. Am J Obstet Gynecol. 1991;165(4):969–971. doi:10.1016/0002-9378(91)90450-6
33. Lyon D, Cheng C-Y, Howland L, et al. Integrated review of cytokines in maternal, cord, and newborn blood: part I—associations with preterm birth. Biol Res Nurs. 2010;11(4):371–376. doi:10.1177/1099800409344620
34. Romero R, Brody DT, Oyarzun E, et al. Infection and labor: III. interleukin-1: a signal for the onset of parturition. Am J Obstet Gynecol. 1989;160(5):1117–1123. doi:10.1016/0002-9378(89)90172-5
35. Vitoratos N, Mastorakos G, Kountouris A, Papadias K, Creatsas G. Positive association of serum interleukin-1β and CRH levels in women with pre-term labor. J Endocrinol Invest. 2007;30(1):35–40. doi:10.1007/BF03347393
36. Sadowsky DW, Novy MJ, Witkin SS, Gravett MG. Dexamethasone or interleukin-10 blocks interleukin-1β-induced uterine contractions in pregnant rhesus monkeys. Am J Obstet Gynecol. 2003;188(1):252–263. doi:10.1067/mob.2003.70
37. Olson DM. The promise of prostaglandins: have they fulfilled their potential as therapeutic targets for the delay of preterm birth? J Soc Gynecol Investig. 2005;12(7):466–478. doi:10.1016/j.jsgi.2005.06.004
38. Romero R, Durum S, Dinarello C, Oyarzun E, Hobbins J, Mitchell M. Interleukin-1 stimulates prostaglandin biosynthesis by human amnion. Prostaglandins. 1989;37(1):13–22. doi:10.1016/0090-6980(89)90028-2
39. Brown N, Alvi S, Elder M, Bennett P, Sullivan M. A spontaneous induction of fetal membrane prostaglandin production precedes clinical labour. J Endocrinol. 1998;157(2):R1–R6. doi:10.1677/joe.0.157r001
40. Bartlett SR, Sawdy R, Mann GE. Induction of cyclooxygenase‐2 expression in human myometrial smooth muscle cells by interleukin‐1β: involvement of p38 mitogen‐activated protein kinase. J Physiol. 1999;520(2):399–406. doi:10.1111/j.1469-7793.1999.00399.x
41. Vadillo‐Ortega F, Estrada‐Gutiérrez G. Role of matrix metalloproteinases in preterm labour. BJOG. 2005;112(S1):19–22. doi:10.1111/j.1471-0528.2005.00579.x
42. Jung MK, Lee JS, Kwak J-E, Shin E-C. Tumor necrosis factor and regulatory T cells. Yonsei Med J. 2019;60(2):126–131. doi:10.3349/ymj.2019.60.2.126
43. Ciebiera M, Włodarczyk M, Zgliczyńska M, et al. The role of tumor necrosis factor α in the biology of uterine fibroids and the related symptoms. Int J Mol Sci. 2018;19(12):pii3869. doi:10.3390/ijms19123869
44. Hirsch E, Filipovich Y, Mahendroo M. Signaling via the type I IL-1 and TNF receptors is necessary for bacterially induced preterm labor in a murine model. Am J Obstet Gynecol. 2006;194(5):1334–1340. doi:10.1016/j.ajog.2005.11.004
45. Gross G, Imamura T, Vogt SK, et al. Inhibition of cyclooxygenase-2 prevents inflammation-mediated preterm labor in the mouse. Am J Physiol Regul Integr Comp Physiol. 2000;278(6):R1415–R1423. doi:10.1152/ajpregu.2000.278.6.R1415
46. Gravett MG, Witkin SS, Haluska GJ, Edwards JL, Cook MJ, Novy MJ. An experimental model for intraamniotic infection and preterm labor in rhesus monkeys. Am J Obstet Gynecol. 1994;171(6):1660–1667. doi:10.1016/0002-9378(94)90418-9
47. Watari M, Watari H, DiSanto ME, Chacko S, Shi G-P, Strauss III JF. Pro-inflammatory cytokines induce expression of matrix-metabolizing enzymes in human cervical smooth muscle cells. Am J Pathol. 1999;154(6):1755–1762. doi:10.1016/S0002-9440(10)65431-4
48. Rose-John S. Interleukin-6 family cytokines. Cold Spring Harb Perspect Biol. 2018;10(2):a028415. doi:10.1101/cshperspect.a028415
49. Kang S, Tanaka T, Narazaki M, Kishimoto T. Targeting interleukin-6 signaling in clinic. Immunity. 2019;50(4):1007–1023. doi:10.1016/j.immuni.2019.03.026
50. Chiesa C, Pacifico L, Natale F, Hofer N, Osborn JF, Resch B. Fetal and early neonatal interleukin-6 response. Cytokine. 2015;76(1):1–12. doi:10.1016/j.cyto.2015.03.015
51. Wei S-Q, Fraser W, Luo Z-C. Inflammatory cytokines and spontaneous preterm birth in asymptomatic women: a systematic review. Obstet Gynecol. 2010;116(2, Part 1):393–401. doi:10.1097/AOG.0b013e3181e6dbc0
52. Robertson SA, Christiaens I, Dorian CL, et al. Interleukin-6 is an essential determinant of on-time parturition in the mouse. Endocrinology. 2010;151(8):3996–4006. doi:10.1210/en.2010-0063
53. Mitchell MD, Dudley DJ, Edwin SS, Schiller SL. Interleukin-6 stimulates prostaglandin production by human amnion and decidual cells. Eur J Pharmacol. 1991;192(1):189–191. doi:10.1016/0014-2999(91)90090-D
54. Friebe‐Hoffmann U, CHIAO JP, RAUK PN. Effect of IL‐1β and IL‐6 on oxytocin secretion in human uterine smooth muscle cells. Am J Reprod Immunol. 2001;46(3):226–231. doi:10.1034/j.1600-0897.2001.d01-6.x
55. Ivanišević M, Djelmiš J, Buković D. Review on prostaglandin and oxytocin activity in preterm labor. Coll Antropol. 2001;25(2):687–694.
56. Mitchell M, Edwin S, Lundin-Schiller S, Silver R, Smotkin D, Trautman M. Mechanism of interleukin-1β stimulation of human amnion prostaglandin biosynthesis: mediation via a novel inducible cyclooxygenase. Placenta. 1993;14(6):615–625. doi:10.1016/S0143-4004(05)80379-0
57. Dajon M, Iribarren K, Cremer I. Toll-like receptor stimulation in cancer: a pro-and anti-tumor double-edged sword. Immunobiology. 2017;222(1):89–100. doi:10.1016/j.imbio.2016.06.009
58. Koga K, Izumi G, Mor G, Fujii T, Osuga Y. Toll‐like receptors at the maternal‐fetal interface in normal pregnancy and pregnancy complications. Am J Reprod Immunol. 2014;72(2):192–205. doi:10.1111/aji.12258
59. Liu T, Zhang L, Joo D, Sun S-C. NF-κB signaling in inflammation. Signal Transduct Target Ther. 2017;2(1):17023. doi:10.1038/sigtrans.2017.23
60. Kawasaki T, Kawai T. Toll-like receptor signaling pathways. Front Immunol. 2014;25(5):461.
61. Romero R, Espinoza J, Gonçalves LF, Kusanovic JP, Friel L, Hassan S. The role of inflammation and infection in preterm birth. Semin Reprod Med. 2007;25(1):21–39. doi:10.1055/s-2006-956773
62. Challis JR, Sloboda DM, Alfaidy N, et al. Prostaglandins and mechanisms of preterm birth. Reprod. 2002;124(1):1–17. doi:10.1530/rep.0.1240001
63. Deguine J, Barton GM. MyD88: a central player in innate immune signaling. F1000Prime Rep. 2014;6:97. doi:10.12703/P6-97
64. Adams KM, Lucas J, Kapur RP, Stevens AM. LPS induces translocation of TLR4 in amniotic epithelium. Placenta. 2007;28(5–6):477–481. doi:10.1016/j.placenta.2006.08.004
65. Gonzalez JM, Xu H, Ofori E, Elovitz MA. Toll-like receptors in the uterus, cervix, and placenta: is pregnancy an immunosuppressed state? Am J Obstet Gynecol. 2007;197(3):
66. Sheldon IM, Roberts MH, Chabalgoity JA. Toll-like receptor 4 mediates the response of epithelial and stromal cells to lipopolysaccharide in the endometrium. PLoS One. 2010;5(9):e12906. doi:10.1371/journal.pone.0012906
67. Wang H, Hirsch E. Bacterially-induced preterm labor and regulation of prostaglandin-metabolizing enzyme expression in mice: the role of toll-like receptor 4. Biol Reprod. 2003;69(6):1957–1963. doi:10.1095/biolreprod.103.019620
68. Elovitz MA, Wang Z, Chien EK, Rychlik DF, Phillippe M. A new model for inflammation-induced preterm birth: the role of platelet-activating factor and toll-like receptor-4. Am J Pathol. 2003;163(5):2103–2111. doi:10.1016/S0002-9440(10)63567-5
69. Chen Z, Liu Q, Zhu Z, Xiang F, Wu R, Kang X. Toll‐like receptor 4 contributes to uterine activation by upregulating pro‐inflammatory cytokine and CAP expression via the NF‐κB/P38MAPK signaling pathway during pregnancy. J Cell Physiol. 2020;235(1):513–525. doi:10.1002/jcp.28991
70. Chin PY, Dorian CL, Hutchinson MR, et al. Novel toll-like receptor-4 antagonist (+)-naloxone protects mice from inflammation-induced preterm birth. Sci Rep. 2016;6(1):36112. doi:10.1038/srep36112
71. Moço NP, Batista RAC, Martin LF, et al. Toll-like receptor-2 and-4 expression by maternal neutrophils in preterm labor. Gynecol Obstet Invest. 2018;83(1):1–8. doi:10.1159/000468930
72. Tufano M, l’Ero G, Ianniello R, Baroni A, Galdlero F. Antimicrobial agents induce monocytes to release IL-1α, IL-6, and Tnf and induce lymphocytes to release IL-4 and TNFτ. Immunopharmacol Immunotoxicol. 1992;14(4):769–782. doi:10.3109/08923979209009234
73. Rogers M, Moore R, Cohen J. The relationship between faecal endotoxin and faecal microflora of the C57BL mouse. Epidemiol Infect. 1985;95(2):397–402.
74. Schulze C, Oesser S, Hein H, Seifert J. Risk of endotoxemia during the initial phase of gut decontamination with antimicrobial agents. Res Exp Med (Berl). 2001;200(3):169–174.
75. Knoop KA, McDonald KG, Kulkarni DH, Newberry RD. Antibiotics promote inflammation through the translocation of native commensal colonic bacteria. Gut. 2016;65(7):1100–1109. doi:10.1136/gutjnl-2014-309059
76. Norimatsu M, Morrison DC. Correlation of antibiotic-induced endotoxin release and cytokine production in Escherichia coli-inoculated mouse whole blood ex vivo. J Infect Dis. 1998;177(5):1302–1307. doi:10.1086/515291
77. Van Deventer S, Buller HR, Ten Cate J, Aarden L, Hack CE, Sturk A. Experimental endotoxemia in humans: analysis of cytokine release and coagulation, fibrinolytic, and complement pathways. Blood. 1990;76(12):2520–2526. doi:10.1182/blood.V76.12.2520.2520
78. Bode C, Diedrich B, Muenster S, et al. Antibiotics regulate the immune response in both presence and absence of lipopolysaccharide through modulation of Toll-like receptors, cytokine production and phagocytosis in vitro. Int Immunopharmacol. 2014;18(1):27–34. doi:10.1016/j.intimp.2013.10.025
79. Harju K, Ojaniemi M, Rounioja S, et al. Expression of toll-like receptor 4 and endotoxin responsiveness in mice during perinatal period. Pediatr Res. 2005;57(5Part 1):644. doi:10.1203/01.PDR.0000156212.03459.A9
80. Salminen A, Paananen R, Vuolteenaho R, et al. Maternal endotoxin-induced preterm birth in mice: fetal responses in toll-like receptors, collectins, and cytokines. Pediatr Res. 2008;63(3):280–286. doi:10.1203/PDR.0b013e318163a8b2
81. Alkharfy KM, Kellum JA, Frye RF, Matzke GR. Effect of ceftazidime on systemic cytokine concentrations in rats. Antimicrob Agents Chemother. 2000;44(11):3217–3219. doi:10.1128/AAC.44.11.3217-3219.2000
82. The American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 169: interim update multifetal gestations: twin, triplet and higher-order multifetal pregnancies. Obstet Gynecol. 2016;128(4):e155–64. doi:10.1097/AOG.0000000000001711
83. World Health Organization. WHO recommendations on interventions to improve preterm birth outcomes; 2015. Available from: https://apps.who.int/iris/bitstream/handle/10665/183037/9789241508988_eng.pdf;jsessionid=68AF40687305B371ECF183FDC9498404?sequence=1.
84. National Institute for Health and Care Excellence. Preterm labour and birth NG25; 2015. Available from: https://www.nice.org.uk/guidance/ng25.
85. Medley N, Poljak B, Mammarella S, Alfirevic Z. Clinical guidelines for prevention and management of preterm birth: a systematic review. BJOG. 2018;125(11):1361–1369. doi:10.1111/1471-0528.15173
86. Hantoushzadeh S, Norooznezhad AH. Inappropriate Antibiotic Consumption as a Possible Cause of Inflammatory Storm and Septic Shock in Patients Diagnosed with Coronavirus Disease 2019 (COVID-19). Arch Med Res. Epub 2020 Apr 4. doi:10.1016/j.arcmed.2020.03.015
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
