Back to Journals » OncoTargets and Therapy » Volume 13
Anlotinib as Exploratory Therapy for Platinum-Resistant Ovarian Cancer: A Retrospective Study on Efficacy and Safety
Authors Ni J , Cheng X , Chen J , Guo W, Dai Z
Received 20 June 2020
Accepted for publication 12 August 2020
Published 5 October 2020 Volume 2020:13 Pages 9857—9863
DOI https://doi.org/10.2147/OTT.S268613
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nicola Silvestris
Jing Ni,1 Xianzhong Cheng,1 Jin Chen,1 Wenwen Guo,2 Zhiqin Dai1
1Department of Gynecologic Oncology, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, The Affiliated Cancer Hospital of Nanjing Medical University, Nanjing, Jiangsu 210009, People’s Republic of China; 2Department of Pathology, The Second Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu 210009, People’s Republic of China
Correspondence: Zhiqin Dai
Department of Gynecologic Oncology, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, The Affiliated Cancer Hospital of Nanjing Medical University, 42# Baiziting Street, Nanjing, Jiangsu 210009, People’s Republic of China
Tel +86-25-83284675
Email [email protected]
Purpose: Survival of platinum-resistant ovarian cancer (PROC) patients is significantly shortened to around 12 months. Anlotinib is a new multi-target tyrosine kinase inhibitor. The goal of this study is to evaluate the efficacy and safety of anlotinib in PROC patients.
Patients and Methods: PROC patients treated with anlotinib in Jiangsu Cancer Hospital between June 2018 to September 2019 were recruited. Most patients received an initial bolus of 12mg orally once daily on days 1– 14 of a 21-day cycle (except one received a dose of 10mg and another one received a dose of 8mg orally once a day). The adverse events (AEs) and efficacy were analyzed by CTCAE 4.0 and RECIST 1.1.
Results: Of all 15 enrolled patients, 12 patients received anlotinib as multi-line therapy and 3 patients received it as maintenance therapy. In the multi-line therapy group, eight patients received anlotinib monotherapy and four patients received anlotinib combined with chemotherapy. Ultimately, evaluation showed that one patient achieved partial response (PR), five patients achieved stable disease (SD) and one patient had progressive disease (PD) with monotherapy, yielding objective response rate (ORR) of 14.3% (95% CI=0.01– 0.58) and disease control rate (DCR) of 85.7% (95% CI=0.42– 0.99). One patient achieved PR, two patients achieved SD with combination therapy, yielding ORR of 33.3% (95% CI=0.02– 0.87) and DCR of 100% (95% CI=0.31– 1.00). Three patients with maintenance therapy were followed up for 5, 8, and 11 months, respectively. The most grade 1– 2 AEs were hand-foot syndrome, nausea, and hypertension. Serious AEs (SAEs) (Grade 3– 4) were observed in one patient with oral ulcer and another patient with hand-foot syndrome that were managed by dose reduction.
Conclusion: Anlotinib was of promising efficacy and well tolerated in PROC patients. This is the first retrospective study about exploratory therapy for ovarian cancer patients with anlotinib.
Keywords: platinum-resistant ovarian cancer, anlotinib, efficacy, safety
Introduction
Ovarian cancer is the leading cause of death for patients with gynecologic malignancies, and the standard first-line treatment is platinum-based chemotherapy and tumor cytoreductive surgery.1 Although most ovarian cancer patients are sensitive to platinum-based chemotherapy, about 80% patients relapse within 1 to 2 years after initial treatment and gradually progress to platinum-resistance ovarian cancer, accompanied by significantly shortened survival.2 Recently, patients can have remarkable benefits from poly ADP-ribose polymerase inhibitor (PARPi) or PARPi/bevacizumab as first-line or second-line maintenance therapy.3–7 However, the efficacy of posterior-line treatment for ovarian cancer remains limited.8,9 Therefore, it is important to seek an alternative agent that can improve the outcome of platinum-resistance ovarian cancer.
Bevacizumab, a monoclonal antibody targets all isoforms of vascular endothelial growth factor (VEGF)-A and inhibits tumor angiogenesis. GOG218 and ICON7 trials showed that front-line chemotherapy with bevacizumab throughout improved progression-free survival (PFS) and prolonged overall survival (OS).10,11 Also, the reductions in the risk of progression or death by adding bevacizumab to chemotherapy were observed both in platinum-sensitive and platinum-resistant recurrent ovarian cancer.12,13
Anlotinib is a novel tyrosine kinase inhibitor targeting multiple receptor kinases involved in tumor proliferation, vasculature, and tumor microenvironment. Anlotinib inhibits VEGF/VEGFR signaling by selectively targeting VEGFR2/3 and fibroblast growth factor receptor (FGFR)1–4 with high affinity and suppresses the activity of platelet-derived growth factor receptor (PDGFR) α and β, stem cell factor receptor (c-Kit), and Ret.14 In vivo, anlotinib has shown broad activity against human tumor xenograft models of the colon (SW-620), ovarian (SKOV-3), liver (SMMC-7721), renal (Caki-1), glioma (U87MG), and non-small cell lung (Calu-3) during the dosing period.15 A phase Ib/IIa study assessing the safety and efficacy of adding anlotinib to standard platinum-based chemotherapy in subjects with recurrent or metastatic endometrial, ovarian or cervical carcinoma presented at the 2019 EMSO meeting demonstrated that both endometrial and ovarian cancer patients could get better synergistic profit from anlotinib combined standard platinum-based plus/or paclitaxel chemotherapy.16 Recently another prospective, single-arm, single-center, Phase II clinical study showed that patients with recurrent platinum-resistant or refractory ovarian carcinoma who used anlotinib single agent yielded ORR 35.7% and DCR 85.7% in 2020 ASCO meeting.17
At present, there are no detailed data about the efficacy and safety of anlotinib in ovarian cancer as multi-line therapy in the real world. We conducted a retrospective evaluation of anlotinib as exploratory therapy to investigate its efficacy and toxicity in patients with platinum-resistant ovarian cancer.
Patients and Methods
Study Population
All platinum-resistant ovarian cancer patients treated with anlotinib between June 2018 and September 2019 in Jiangsu Cancer Hospital were included. We reviewed the basic characteristics of these patients, including the age, Eastern Cooperative Oncology Group performance status (ECOG PS) before the start of the treatment, histological type, clinical stage on the basis of Federation International of Gynecology and Obstetrics (FIGO), prior therapy before and after the using of anlotinib and the follow-up.
Patients recurred within 6 months after platinum-based chemotherapy were defined as platinum-resistant ovarian cancer. We excluded patients who had been on treatment for less than two cycles. The study was approved by the ethics committee of Jiangsu Cancer Hospital. Informed consent was obtained from all involved participants.
Dosing Regimen
The dosing of the regimen was as follows: anlotinib (12mg, 10 mg, 8 mg) orally once daily on days 1–14. Each cycle was 21 days long. The initial dose was judged by the clinicians based on the patient’s conditions. Dose reduction (12 to 10mg or 8mg; 10 to 8mg) or interruption for drug-related AEs was allowed. Serum CA125 levels were assessed at baseline and every month thereafter until progression. Each patient received radiological assessment of treatment efficacy every 2 cycles.
Patient Demographics
Basic demographics, tumor characteristics, prior treatment details were summarized and analyzed. Safety was evaluated throughout the study. AEs were graded according to the NCI Common Terminology Criteria for AEs (CTCAE), version 4.0. The efficacy was estimated by RECIST 1.1.
Statistical Analysis
The efficacy was classified as complete response (CR), PR, SD and PD by RECIST 1.1. ORR was defined as the proportion of patients who had a partial or complete response. DCR was defined as the proportion of patients who achieved CR, PR and SD for at least 8 weeks. The 95% confidence interval was calculated using the Wilson procedure with a correction for continuity. Data were statistically analyzed using SPSS version 19.0 professional statistical software and all the count data were expressed as a percentage (%).
Results
Patients’ Characteristics
Fifteen patients with platinum-resistant ovarian cancer treated with anlotinib were recruited. The median age at onset of treatment was 60 years (range, 30–70 years). The clinical and pathological characteristics are listed in Table 1. Twelve (80%) patients received anlotinib after multi-line chemotherapy, and three patients (20%) used it as maintenance therapy. In the multi-line therapy group, eight patients received anlotinib monotherapy and four patients received anlotinib combined with chemotherapy. Median number of the previous regimen was four (range=3-7) before anlotinib was administered. Stage FIGO III or IV affected 10 (66.7%) and 3 (20%) of patients, respectively. Thirteen patients received an initial bolus of anlotinib 12mg orally once daily, the rest of patients received lower initial treatment due to relatively poor state of the bone marrow. One patient received a dose of 10mg and another one received a dose of 8mg orally once a day (Table 1).
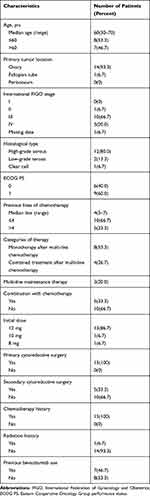 |
Table 1 Baseline Characteristics in 15 Patients. Values Were Reported as Frequency (n [%]) or as Mean (Range) |
Efficacy
In the multi-line therapy group, ultimately therapeutic evaluation showed that one patient achieved partial response (PR), five patients achieved stable disease (SD) and one patient had progressive disease (PD), yielding the objective response rate (ORR) of 14.3% (95% CI=0.01–0.58) and the disease control rate (DCR) of 85.7% (95% CI=0.42–0.99) in patients with monotherapy (Table 2). The patient who had PD was clear cell carcinoma and the remaining patients were all high-grade serous carcinomas.
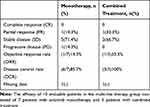 |
Table 2 Summary of Tumor Response to the Multi-line Therapy Group |
According to the different therapeutic strategy, one patient with anlotinib combined with chemotherapy achieved PR, two patients achieved SD with anlotinib combined with chemotherapy, yielding the ORR of 33.3% (95% CI=0.02–0.87) and the DCR of 100% (95% CI=0.31–1.00) (Table 2). Waterfall plot for the best percentage change in target lesion size of 10 patients was shown in Figure 1. Two patients (one patient used as monotherapy and another one used as combination therapy) strongly refused to perform imaging examination and were merely followed up with CA125 (Figure 2). Serum CA125 decreased after two patients took anlotinib.
In the maintenance therapy group, three patients were followed up for 5, 8, and 11 months, respectively. Furthermore, for the analysis of efficacy based on the history of bevacizumab, one patient with the longest Duration of Response (DOR) (11 months) in the maintenance therapy group received bevacizumab at her latest chemotherapy. Another patient with anlotinib combined with oral etoposide after the usage of bevacizumab at her last intravenous chemotherapy achieved PR.
Safety
The most common grade 1–2 AEs were hand-foot syndrome (53.3%), nausea (33.3%), hypertension (26.7%), fatigue (6.7%), voice alteration (6.7%), leukopenia (6.7%), thrombocytopenia (6.7%), decreased appetite (6.7%), upper abdominal pain (6.7%) and vomiting (6.7%). Serious AEs (SAEs) were observed in one patient with oral ulcer and another patient with hand-foot syndrome. Both of them were managed by dose reduction. AEs were usually managed by dose reduction, rather than interruption and discontinuation. Two patients received reduced treatment, and none of the patients interrupted treatment after the alleviation of AEs (Table 3).
 |
Table 3 Adverse Events Observed in Anlotinib-Treated Patients |
Discussion
Although PARP inhibitor is a major advance in the treatment of ovarian cancer, chemotherapy is still the preferred treatment for patients with advanced ovarian cancer. Most patients tend to develop into platinum-resistance ovarian cancer during the progression of the disease.2 The response rates (RR) of platinum-based chemotherapy in platinum-resistance ovarian cancer patients are only 10%. Single-cytotoxic drugs without cross-resistance to platinum are recommended, such as docetaxel, oral cyclophosphamide, gemcitabine, topotecan, liposome doxorubicin, oral etoposide, and paclitaxel. The RR of these agents is in the 10–15% range and overall survival of platinum-resistance ovarian cancer patients is around 12 months.18 PARP inhibitors as maintenance therapy can improve the prognosis of patients, but the efficacy of monotherapy for platinum-resistance ovarian cancer is still limited.8,9 It is urgent to explore new treatment patterns and methods for platinum-resistant ovarian cancer patients.
Tumor angiogenesis is an important factor in the occurrence and development of most solid tumors which is closely related to VEGF. The family members mainly include VEGFR-1, VEGFR-2, and VEGFR-3 . VEGFR-2 is a major receptor for angiogenesis in solid malignancies and its high expression induces lymph node and peritoneal metastases.19 The AURELIA is a randomized, open, Phase III trial in which bevacizumab combined with standard single-drug chemotherapy was used for platinum-resistant ovarian cancer patients. The results showed that bevacizumab combined with standard single-drug chemotherapy could significantly improve PFS (3.4 months vs 6.7 months, HR = 0.48, 95% CI 0.38–0.60, P < 0.01) and ORR (11.8% vs 27.3%, P < 0.01); OS also had an extended trend (13.3 months vs 16.6 months, HR = 0.85, P = 0.174).13
Anlotinib is a new oral multi-target tyrosine kinase inhibitor and its anti-tumor vascular targets include VEGFR, PDGFR and FGFR. A prospective, single-arm, single-center, phase II clinical study from 2020 ASCO meeting showed that 14 recurrent platinum-resistant or refractory ovarian carcinoma patients were evaluable. 1 of complete response (CR), 4 of partial response (PR), 7 of stable disease (SD), and 2 of progressive disease (PD), yielding the ORR 35.7% and DCR 85.7%.17 In our study, the ORR and DCR were 14.3% and 85.7%, respectively. We consider the differences may be due to the small number of patients enrolled in our study and the criteria for enrollment in our real-world study.
It has been confirmed that adding bevacizumab to chemotherapy statistically significantly improved PFS and ORR in recurrence ovarian cancer.12,13 In our study, three patients were all controlled by anlotinib combined with chemotherapy. The AEROC study found that the combination of apatinib with oral etoposide shows promising efficacy and manageable toxicities in patients with platinum-resistant or platinum-refractory ovarian cancer.20 Interestingly, we also observed that one patient who was on anlotinib with oral etoposide combination therapy for 19 months and was still achieved PR.
Two phase III trials (GOG218, ICON7) showed that the PFS of paclitaxel and carboplatin chemotherapy plus bevacizumab and maintenance with bevacizumab was significantly longer than chemotherapy alone.10,11 One patient with the longest Duration of Response (DOR) (11 months) in the multi-line maintenance therapy group may be related to the previous treatment with bevacizumab in the present study. And this patient received an initial bolus of anlotinib 10mg orally once daily for 11 months. Further prospective studies are needed to observe whether maintenance therapy is administered at lower dosage.
Hypertension, hand-foot syndrome, and fatigue were the most common AEs of anlotinib.21 The incidence of hand-foot syndrome and hypertension was highest in our observation which was similar to other studies and the higher incidence of nausea than other studies which might be associated with concurrent chemotherapy. Two patients with SAEs were alleviated by dosage reduction, and the remaining patients with grade 1–2 adverse reactions were tolerable.
Conclusion
In conclusion, this is the first real-world study on the efficacy and safety of the anlotinib as multi-line therapy and maintenance therapy in patients with platinum-resistant ovarian cancer. The results of this study demonstrate that anlotinib is an exceptionally effective and well-tolerated regimen in platinum-resistant ovarian cancer patients with heavily previously treated. However, our data are limited representative due to the lower number of cases. The definite efficacy of anlotinib warrants further evaluation involving large-scale clinical prospective and retrospective research.
Data Sharing Statement
We would not share the data and material used in this manuscript, because we need them for further research.
Ethics Approval and Informed Consent
This retrospective study was approved by the institutional review board of Jiangsu Cancer Hospital, Nanjing Medical University, China. Informed consent was obtained from all involved participants.
Acknowledgments
We sincerely thank Dr Fei Deng, Hong Pei, Yuzhong Wu and Xuehui Zhou. We list the name in alphabetical order of their family name.
Authors’ Information
Jing Ni, Department of Gynecologic Oncology, Jiangsu Cancer Hospital, Nanjing, Jiangsu 210,009, PR China, [email protected]; Xianzhong Cheng, Department of Gynecologic Oncology, Jiangsu Cancer Hospital, Nanjing, Jiangsu 210,009, PR China, [email protected]; Jin Chen, Department of Gynecologic Oncology, Jiangsu Cancer Hospital, Nanjing, Jiangsu 210,009, PR China, [email protected]; Wenwen Guo, Department of Pathology, The Second Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu, 210,009, PR China, [email protected].
Funding
This study was supported by grants from the National Natural Science Foundation of China (No. 81501205) and Institute level project of Jiangsu Cancer Hospital(No. ZM201804).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Coleridge SL, Bryant A, Lyons TJ, et al. Chemotherapy versus surgery for initial treatment in advanced ovarian epithelial cancer. Cochrane Database Syst Rev. 2019;10:CD005343. doi:10.1002/14651858.ED000142
2. Guan LY, Lu Y. New developments in molecular targeted therapy of ovarian cancer. Discov Med. 2018;26(144):219–229.
3. Moore K, Colombo N, Scambia G, et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379(26):2495–2505. doi:10.1056/NEJMoa1810858
4. Pujade-Lauraine E, Ledermann JA, Selle F, et al. Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21): a double-blind, randomised, placebo-controlled, Phase 3 trial. Lancet Oncol. 2017;18(9):1274–1284. doi:10.1016/S1470-2045(17)30469-2
5. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375(22):2154–2164. doi:10.1056/NEJMoa1611310
6. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 Part 1): an international, multicentre, open-label, Phase 2 trial. Lancet Oncol. 2017;18(1):75–87. doi:10.1016/S1470-2045(16)30559-9
7. Ray-Coquard I, Pautier P, Pignata S, et al. Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N Engl J Med. 2019;381(25):2416–2428. doi:10.1056/NEJMoa1911361
8. Moore KN, Secord AA, Geller MA, et al. Niraparib monotherapy for late-line treatment of ovarian cancer (QUADRA): a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):636–648. doi:10.1016/S1470-2045(19)30029-4
9. Kaufman B, Shapira-Frommer R, Schmutzler RK, et al. Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation. J Clin Oncol. 2015;33(3):244–250. doi:10.1200/JCO.2014.56.2728
10. Oza AM, Cook AD, Pfisterer J, et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): overall survival results of a phase 3 randomised trial. Lancet Oncol. 2015;16(8):928–936. doi:10.1016/S1470-2045(15)00086-8
11. Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011;365(26):2473–2483. doi:10.1056/NEJMoa1104390
12. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: a randomized, double-blind, placebo-controlled phase III trial of chemotherapy with or without bevacizumab in patients with platinum-sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer. J Clin Oncol. 2012;30(17):2039–2045. doi:10.1200/JCO.2012.42.0505
13. Pujade-Lauraine E, Hilpert F, Weber B, et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: the AURELIA open-label randomized phase III trial. J Clin Oncol. 2014;32(13):1302–1308. doi:10.1200/JCO.2013.51.4489
14. Shen GS, Zheng FC, Ren DF, et al. Anlotinib: a novel multi-targeting tyrosine kinase inhibitor in clinical development. J Hematol Oncol. 2018;11(1):120. doi:10.1186/s13045-018-0664-7
15. Sun Y, Niu W, Du F, et al. Safety, pharmacokinetics, and antitumor properties of anlotinib, an oral multi-target tyrosine kinase inhibitor, in patients with advanced refractory solid tumors. J Hematol Oncol. 2016;9(1):105. doi:10.1186/s13045-016-0332-8
16. Miller D, Cheung E, Huang M, et al. Phase 1b/2a study assessing the safety and efficacy of adding AL3818 (Anlotinib) to standard platinum-based chemotherapy in subjects with recurrent or metastatic endometrial, ovarian or cervical carcinoma. Poster- ESMO-2019; 1033NO.
17. Shan BE, Shen WB, Wang HY. Anlotinib in patients with recurrent platinum-resistant or refractory ovarian carcinoma: A prospective, single-arm, single-center, phase II clinical study. Abstract-ASCO-2020;6061NO.
18. Naumann RW, Coleman RL. Management strategies for recurrent platinum-resistant ovarian cancer. Drugs. 2011;71(11):1397–1412. doi:10.2165/11591720-000000000-00000
19. Zhao Y, Adjei AA. Targeting angiogenesis in cancer therapy: moving beyond vascular endothelial growth factor. Oncologist. 2015;20(6):660–673. doi:10.1634/theoncologist.2014-0465
20. Lan CY, Wang Y, Xiong Y, et al. Apatinib combined with oral etoposide in patients with platinum-resistant or platinum-refractory ovarian cancer (AEROC): a phase 2, single-arm, prospective study. Lancet Oncol. 2018;19(9):1239–1246. doi:10.1016/S1470-2045(18)30349-8
21. Han B, Li K, Zhao Y, et al. Anlotinib as a third-line therapy in patients with refractory advanced non-small-cell lung cancer: a multicentre, randomised phase II trial (ALTER0302). Br J Cancer. 2018;118(5):654–661. doi:10.1038/bjc.2017.478
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


