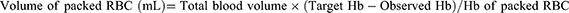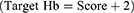Back to Journals » Therapeutics and Clinical Risk Management » Volume 19
An Individualized Red Blood Cell Transfusion Strategy Using Pediatric Perioperative-Transfusion-Trigger Score Reduced Perioperative Blood Exposure for Children: A Randomized Controlled Clinical Trial
Authors Luo Z , Li Y, Li X, Liao R
Received 6 September 2022
Accepted for publication 26 February 2023
Published 11 March 2023 Volume 2023:19 Pages 229—237
DOI https://doi.org/10.2147/TCRM.S388924
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Zhen Luo,1 Yansong Li,2 Xiaoqiang Li,1 Ren Liao1
1Department of Anesthesiology, Sichuan University West China Hospital, Chengdu, People’s Republic of China; 2Department of Anesthesiology, Center for Brain Science, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, People’s Republic of China
Correspondence: Xiaoqiang Li, Email [email protected]
Objective: The optimal red blood cell transfusion strategy for children remains unclear. We developed an individualized red blood cell transfusion strategy for children and tested the hypothesis that transfusion guided by this strategy could reduce blood exposure, without increasing perioperative complications in children.
Methods: In this randomized controlled clinical trial, 99 children undergoing noncardiac surgeries who had blood loss of more than 20% total blood volume were randomly assigned to an individualized-strategy group using Pediatric Perioperative-Transfusion-Trigger Score or a control group. The amount of transfused red blood cell was counted, and patients were followed up for postoperative complications within 30 days.
Results: Twenty-six children (53.1%) in the individualized-strategy group received transfusion perioperatively, as compared with 37 children (74%) in the control group (p < 0.05). During surgery, children in the individualized-strategy group were exposed to fewer transfusions than in the control group (0.87± 1.03 vs 1.33± 1.20 Red-Blood-Cell units per patient, p = 0.02). The incidence of severe complications in the individualized-strategy group had a lower trend compared to the control group (8.2% vs 18%, p = 0.160). No significant difference was found in the other outcomes.
Conclusion: This trial proved that red blood cell transfusion guided by the individualized strategy reduced perioperative blood exposure in children, without increasing the incidence of severe complications. This conclusion needs to be reaffirmed by larger-scale, multicenter clinical trials.
Keywords: red blood cell, transfusion strategy, perioperative red blood cell transfusion, pediatrics, hemoglobin concentration
Introduction
Allogeneic red blood cell (RBC) transfusion is an important therapeutic way in children with substantial amount of blood loss during surgery. Allogeneic transfusion can increase post-surgical risks, which has been reported to be associated with increased perioperative morbidity and mortality in children.1–3 Consequently, the benefits and risks of allogeneic RBC transfusion in children should be assessed carefully.
Current studies have demonstrated significant changes in RBC transfusion practices, among which are its restrictive strategy which has become a worldwide consensus.4–6 However, careful attention must be paid to the use of restrictive strategy in children, for the concern that it may lead to the risk of oxygen deficiency.7–9 This is why an individualized strategy that specifically considers the patients’ medical condition and respective age is necessary.
Evaluating oxygen supplied and consumption balance, we established an individualized transfusion strategy using “Perioperative-Transfusion-Trigger Score” in adults. We conducted a clinical trial of adult patients who underwent spinal surgery and the indicative results revealed this tailored approach was safe and cost-effective.10
By taking account of these children’s age and their developmental stage, we designed a Pediatric Perioperative-Transfusion-Trigger Score (Pediatric-POTTS) (Table 1) that guides the transfusion in children. Based on this score, we designed the trial to test the hypothesis that transfusion guided by tailored approach could decrease blood exposure, without increasing perioperative complications in children.
 |
Table 1 Pediatric Perioperative-Transfusion-Trigger Score |
Methods
Ethics and Consent of Participants
This study complies with the Declaration of Helsinki and the study protocol was registered at http://www.chictr.org.cn/showprojen.aspx?proj=13361 (Registration number: ChiCTR-IRP-16007909). The protocol was reviewed and approved by the ethics committee of West China Hospital, Sichuan University (No. 2013–101). Informed consent in writing was obtained from all the children’s parents or legal guardians.
Study Design and Treatment Protocols
This study was designed as a prospective, open-label, randomized, controlled clinical trial, assessing the safety and efficacy of the individualized strategy in pediatric patients. Participants were randomly assigned to an individualized-strategy or a control group. By following the NIDCR Policy on Data and Safety Monitoring, the statistician and committee members were masked.
From June 2016 to June 2018, we enrolled children between 1 day to 14 years of age who were scheduled to undergo elective non-cardiac surgery with blood loss of more than 20% total blood volume at the West China Hospital of Sichuan University. Premature infants or low-birth-weight newborns (body weight <2500g), those who declined transfusion treatment, patients with chronic conditions such as cyanotic congenital heart disease, severe hematological disorders or hemoglobinopathy, metastasis of tumor or multiple tumors were all excluded from the trial.
The Pediatric Perioperative-Transfusion-Trigger Score
RBC transfusion in individualized-strategy group were guided by the Pediatric Perioperative-Transfusion-Trigger Score (Table 1), which is a modified model of the Perioperative-Transfusion-Trigger Score for adults. It consists of 4 items: 1) Minimum inhale oxygen concentration to keep pulse oxygen saturation ≥95%, which is a key clinical indicator for pulmonary function; 2) Infusion rate of adrenaline required to maintain an adequate cardiac output. Here, adrenaline could be replaced by other inotropic agents with equivalent potent infusion rates as different institutions or medical groups may choose another drug as their first line of inotropic; 3) Core body temperature, a clinical indicator of total body oxygen consumption; and 4) Age of the child.
Treatment Assignment
For all patients, both the hemoglobin (Hb) concentration and the Pediatric-POTTS were measured and taken before surgery, at completion of surgery, on the day of discharge, and whenever RBC transfusion was considered.
In the individualized-strategy group, RBC transfusion triggered when the Hb concentration was below the Pediatric-POTTS. If the point of care Hb concentration was equal to or lower than the Pediatric-POTTS, RBC would be transfused (see Supplementary 1 Appendix to use Pediatric-POTTS).
The amount of RBC transfusion was set according to the formula:
In the control group, when the patient’s Hb concentration was lower than 8g per deciliter (newborn lower than 10g per deciliter), RBC transfusion would be initiated. The target Hb concentration was based on the decision of the physician, which was not subject to special restrictions.
During the 30-day follow-up period, Hb concentration was measured at least once every 6 hours after each transfusion and on hospital discharge. Any postoperative complications were observed and recorded daily. Follow-up after discharge were completed by telephone. Data were collected by trained research personnel and stored in West China Hospital. Statistician and members of the safety and data monitoring committee were unaware of the grouping allocations.
Outcomes
The primary outcomes were the utilization of RBC transfusion and perioperative complications by day-30. The secondary outcomes included ICU occupancy, ICU duration time, rate of mechanical ventilation, hospitalization costs, duration of postoperative care to discharge costs.
Statistical Analysis
We estimate that we need to enroll at least 90 children in order to detect an absolute reduction of 10% in RBC transfusion exposure in the individualized-strategy group compared to the control group, with an overall one-sided alpha of 5% and a power of 90%.
The statistical analysis of this study was performed using the SAS 9.1 statistical software. All statistical tests were performed on a two-sided test with an alpha of 0.05.
Preoperative demographic baseline characteristics were compared between individualized-strategy and the control group. We presented mean ± standard deviation or median [Interquartile range] for continuous variables as appropriate. Frequency (percentage) was used to summarize categorical variables.
The chi-square test was used to study transfusion rate and ICU occupancy rates between two groups. The Student’s t-test was used for comparison of the amount of RBC transfusion, time in ICU, hospitalization time and costs. A rank-sum test was used to compare the incidence of severe complications.
Results
Baseline Information
Two hundred and forty-two patients were eligible for participation in this trial (from June 2016 to June 2018), of whom 142 were excluded. One hundred patients underwent randomization and one patient was withdrawn at a surgeon’s request. Therefore, a total of 99 patients enrolled in the study and were included in the final analysis, of whom 49 were placed in the individualized-strategy group and 50 in the control group (Figure 1).
Patients’ baseline characteristics were similar between the two groups. Hemoglobin concentrations at the time of randomization were similar (Table 2).
 |
Table 2 Baseline Characteristics of the Patients |
RBC Transfusion Rate
Twenty-six patients (53.1%) received RBC transfusions in the individualized-strategy group while 37 patients (74.0%) received RBC transfusions in the control group perioperatively (53.1% vs 74.0%; odds ratio, 0.40; 95% confidence interval [CI] 0.17 to 0.93; p = 0.03). In addition, the hemoglobin concentrations at discharge were 9.87±2.34 g per deciliter in individualized-strategy group as compared to 11.03±1.53 g per deciliter in the control group (Difference in means, −1.16; 95% CI −1.95 to −0.38; p = 0.004). There were no significant differences in the amount of RBC transfusion per patient (162.25±305.25 mL vs 247.40±263.09 mL, p = 0.140).
During surgery, 24 patients (49.0%) received RBC transfusions, as compared with 34 patients (68.0%) in the control group (p = 0.055). Children in the individualized-strategy group were exposed to fewer transfusions than in the control group (0.87±1.03 vs 1.33±1.20 RBC units per patient; difference in means, −0.46; 95% CI −0.91 to −0.016; p = 0.043). A smaller volume of RBC was transfused per patient in individualized group than that in the control group (123.67 ± 186.34 vs 219.60 ± 250.00 mL per patient; difference in means, −95.93; 95% CI −184.02 to −7.83; p = 0.033).
During the period from postoperative to discharge, the number of transfused patients in both groups only had a small cohort of 6. There were no significant differences in the amount of RBC per patient (38.57±160.08 mL vs 27.80±82.23 mL, p = 0.67) postoperatively (Table 3).
 |
Table 3 Red Blood Cell Transfusion |
Postoperative Complications
Of the group, 20 patients developed postoperative complications. According to our complication rating system (Supplementary 2 Appendix), grades 1 and 2 were defined as mild complications, grades 3, 4, and 5 were defined as severe complications. There were 1 patient with severe complication in individualized-strategy group and 3 patients in the control group. The 30-day postoperative mortality was equal in two groups. The prevalence of severe complication in the individualized-strategy group was no more than that in the control group (8.2% vs 18%, p = 0.160) (Table 4).
 |
Table 4 Postoperative Complications |
Secondary Analysis
The number of patients who were admitted to ICU after surgery was 26 (53.1%) in the individualized-strategy group and 28 (56%) in the control group (p = 0.769). There were no significant differences in ICU duration between the two groups (55.69±85.74 h vs 54.14±96.16 h, p = 0.8). There were no statistical differences in mechanical ventilation time after surgery (5.43±17.23 h vs 2.70±4.00 h, p = 0.802). There were no significant differences between two groups in the total hospitalization costs (¥37,863.77±25,960.83 vs ¥41,991.12±24,421.14, p = 0.198), ICU costs (¥5858.00±9341.74 vs ¥6899.45±12,332.26, p = 0.901), and postoperative costs (¥12,453.8±14,989.81 vs ¥11,405.93±11,713.63, p = 0.605) (Table 5).
 |
Table 5 ICU Occupancy and Hospitalization Costs |
Discussion
When compared with the control group, the individualized-strategy group resulted in a 44.6% decrease for the number of patient exposure to transfusion and 35.7% reduction in the amount of RBC transfused. Meanwhile, this study found that there was no difference in the incidence of severe postoperative complications.
Surgeons, anesthesiologists, transfusion specialists, patients highly regard the use of over- and/or under-transfusion during surgery. However, decision-making surrounding pediatric perioperative transfusion, which is a dynamic and complex procedure may be affected by many factors.
There are significant differences in the practice of RBC transfusion in children11 and most of the recommendations for perioperative transfusion are based on hemoglobin or HCT (hematocrit).
Transfusion guidelines provide either trigger hemoglobin (the hemoglobin threshold for transfusion), target hemoglobin (the target hemoglobin level after transfusion) and/or hematocrit levels to guide RBC transfusion in adults.12–15 Apart from this, the guidelines also mentioned that varying factors such as age, bleeding volume, hemodynamic status or organ perfusion should be considered when deciding whether to transfuse or not.12,13,16
The American Society of Anesthesiologists Task Force on Perioperative Blood Management states that a restrictive RBC transfusion strategy may be safe, and further specifies, “the determination of whether hemoglobin concentration between 6.0 and 10.0 g/dL justify or require RBC transfusion should be based on potential or actual ongoing bleeding, intravascular volume status, signs of organ ischemia, and adequacy of cardiopulmonary reserve.16
Therefore, we designed this individual RBC transfusion strategy for children after taking into account all of the above factors. Red blood cells are the main carrier of oxygen in human body. The requirements to transfuse RBC depends on whether it could prevent or reverse tissue hypoxia which is caused by insufficient oxygen transport.17,18
As is known to all, the balance of oxygen supply and consumption is directly proportional to hemoglobin concentration, arterial oxygen saturation, cardiac output and inversely proportional to human oxygen consumption.19 Based on this, we used 1) infusion rate of adrenaline required to maintain an adequate cardiac output; 2) Minimum inhale oxygen concentration to keep pulse oxygen saturation ≥95%; 3) Core body temperature to establish the Perioperative-Transfusion-Trigger Score to guide RBC transfusion in adults. We integrated age in Pediatric Perioperative-Transfusion-Trigger Score because ranges for hemoglobin concentration in children change with age.20,21 More importantly, this semi-quantitative score could be applied by any physician without special equipment and complex measurement.10,22
From our results, it can be concluded that the pediatric individual transfusion strategy did reduce perioperative blood exposure in children. The reason why this strategy was able to reduce the amount of RBC transfusion may be multifactorial. First, this strategy was based on restrictive transfusion strategies, setting a minimum hemoglobin threshold in the best status of children. Second, when the transfusion target was reached, RBC transfusion stopped immediately.
There are individual differences in children’s tolerance to anemia. We were able to identify and treat children of insufficient oxygen supply by the transfusion strategy using Pediatric-POTTS, which guaranteed those children to receive enough oxygen to prevent further damage.
Furthermore, the Pediatric-POTTS is a dynamic measurement. With the change of clinical condition of patients, repeated evaluations could be carried out to determine whether RBC transfusion was needed, which would provide a prompt solution to patients’ needs.
In the control group, we chose the strategy of starting RBC transfusion when Hb concentration was lower than 8g per deciliter (newborn less than 10g per deciliter), which was recommended by the guideline of RBC transfusion from the European Society of Anesthesiology.13,23
There are certain limitations in our study. First of all, this study was completed under the condition of a single-center study with a sample size of 99 patients that included general surgeries, orthopedic surgeries, and a small number of thoracic surgeries. It did not include any cardiac surgery or neurosurgery. In addition, we were unable to detect any meaningful differences in its secondary clinical outcomes. Due to the low incidence of transfusion adverse reactions as a whole in pediatrics,24 there were no reports of pertinent transfusion reactions in the study. Thirdly, when acute massive blood loss occurred, this strategy did not deem to be applicable. Thus, decisions still need to be made quickly based on the experience of anesthesiologists and surgeons.
With the findings of this study, we now plan to conduct a multicenter, randomized, large-sample trial to produce a stronger evidence case. We are eager to observe whether individualized RBC transfusion strategy will affect long-term prognosis and neurodevelopment of children who undergo surgeries.
Conclusion
This trial demonstrated that the individualized RBC transfusion strategy with Pediatric-POTTS can significantly reduce intraoperative blood exposure in children, without increasing the incidence of severe complications. This conclusion needs to be reaffirmed by larger-scale, multicenter clinical trials.
Data Sharing Statement
The data that support the findings of this study will be available from the corresponding author, Xiaoqiang Li, upon reasonable request. All of the individual participant data collected during the trial will be available after deidentification. The data will be made available immediately following article publication, with no end date.
Acknowledgments
We would like to thank all the patients and their families who participated in this study.
Disclosure
The authors have no conflicts of interest to declare for this work.
References
1. Lavoie J. Blood transfusion risks and alternative strategies in pediatric patients. Paediatr Anaesth. 2011;21(1):14–24. doi:10.1111/j.1460-9592.2010.03470.x
2. Muszynski JA, Spinella PC, Cholette JM, et al. Transfusion-related immunomodulation: review of the literature and implications for pediatric critical illness. Transfusion. 2017;57:195–206.
3. Faraoni D, Goobie SM. The efficacy of antifibrinolytic drugs in children undergoing noncardiac surgery: a systematic review of the literature. Anesth Analg. 2014;118:628–636.
4. Jl C, Sj S, Roubinian N, et al. Transfusion thresholds and other strategies for guiding allogeneic red blood cell transfusion. Cochrane Database Syst Rev. 2016;10:CD002042.
5. Patel NN, Avlonitis VS, Jones HE, et al. Indications for red blood cell transfusion in cardiac surgery: a systematic review and meta-analysis. Lancet Haematol. 2015;2:e543–53.
6. Burke M, Sinha P, Luban NLC, et al. Transfusion-associated hyperkalemic cardiac arrest in neonatal, infant, and pediatric patients. Front Pediatr. 2021;29:1.
7. Marik PE, Corwin HL. Efficacy of red blood cell transfusion in the critically ill: a systematic review of the literature. Crit Care Med. 2008;36:2667–2674.
8. Minhas SV, Chow I, Bosco J, et al. Assessing the rates, predictors, and complications of blood transfusion volume in posterior arthrodesis for adolescent idiopathic scoliosis. Spine. 2015;40:1422–1430.
9. Wang Y, Chan O, Chiang M. ScienceDirect’s red blood cell transfusion and clinical outcomes in extremely low birth weight preterm infants. Pediatr Neonatol. 2018;58:216–222.
10. Liao R, Sun H-R, Liu J, et al. Safety and effectiveness of reduced red blood cells consumption in spine surgery under the guidance of West China Perioperative Transfusion Score (WCPTS): a prospective, randomized, controlled trial. J Anesthetic Perioper Med. 2015;2(3):117–125. doi:10.24015/JAPM.2015.0017
11. Mo YD, Delaney M. Transfusion in pediatric patients: review of evidence-based guidelines. Clin Lab Med. 2021;41:1–14.
12. Cholette JM, Willems A, Valentine SL, et al. Recommendations on RBC transfusion in infants and children with acquired and congenital heart disease from the pediatric critical care transfusion and anemia expertise initiative. Pediatr Crit Care Med. 2018;19:S137–48.
13. Kozek-Langenecker SA, Ahmed AB, Afshari A, et al. Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology. First update 2016. Eur J Anaesthesiol. 2017;34(6):332–395.
14. Carson JL, Guyatt G, Heddle NM, et al. Clinical practice guidelines from the AABB: red blood cell transfusion thresholds and storage. JAMA. 2016;316:2025–2035.
15. Villeneuve A, Arsenault V, Lacroix J, et al. Neonatal red blood cell transfusion. Vox Sang. 2021;116:366–378.
16. Khanna P, Bhatt R. Practice guidelines for perioperative blood management an updated report by the American society of anesthesiologists task force on perioperative blood management. Anesthesiology. 2015;122:241–271.
17. Madjdpour C, Spahn DR. Allogeneic red blood cell transfusions: efficacy, risks, alternatives and indications. Br J Anaesth. 2005;95:33–42.
18. Spahn DR, Goodnough LT. Alternatives to blood transfusion. Lancet. 2013;381:1855–1865.
19. Casutt M, Seifert B, Pasch T, et al. Factors influencing the individual effects of blood transfusions on oxygen delivery and oxygen consumption. Crit Care Med. 1999;27:2194–2200.
20. Pérel Y, Runel C, Huguenin Y, et al. Transfusion and its specific problems in pediatrics and neonatology. Transfus Clin Biol. 2017;24:101–105.
21. Marks PW. Approach to anemia in the adult and child. In: In: FH, editor. Basic Principles and Practice.
22. Liao R, Sun H, Liu J. West China Liu’s score for individualized red cells transfusion strategy in non-cardiac surgery: study protocol for a multicenter, randomized, controlled trial. J Anesth Perioper Med. 2016;3:225–232.
23. Kozek-Langenecker SA, Afshari A, Albaladejo P, et al. Management of severe perioperative bleeding: guidelines from the European Society of anaesthesiology. Eur J Anaesthesiol. 2013;30:270–382.
24. Stainsby D, Jones H, Wells AW, et al. Adverse outcomes of blood transfusion in children: analysis of UK reports to the serious hazards of transfusion scheme 1996–2005. Br J Haematol. 2008;141:73–79.
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.